Abstract
The global economy and public health are currently under enormous pressure since the outbreak of COVID-19. Apart from respiratory discomfort, a subpopulation of COVID-19 patients exhibits neurological symptoms such as headache, myalgia, and loss of smell. Some have even shown encephalitis and necrotizing hemorrhagic encephalopathy. The cytoskeleton of nerve cells changes drastically in these pathologies, indicating that the cytoskeleton and its related proteins are closely related to the pathogenesis of nervous system diseases. In this review, we present the up-to-date association between host cytoskeleton and coronavirus infection in the context of the nervous system. We systematically summarize cytoskeleton-related pathogen-host interactions in both the peripheral and central nervous systems, hoping to contribute to the development of clinical treatment in COVID-19 patients.
Keywords
1 Introduction
Eukaryotes have a complex cytoskeleton system, including actin filaments, microtubules, and intermediate filaments. This filamentous network regulates cellular structural functions, as well as the nervous system, including cell morphogenesis, epithelial–mesenchymal transition, and cell migration. Moreover, the cytoskeleton is also involved in important intracellular biological activities, including vesicle and cargo transportation, immunity against viral infection, and autophagy activation [1–3].
2 Cytoskeleton
2.1 Actin filaments
Actin filaments, also known as microfilaments, have an average diameter of about 7 nm. Actin filaments are composed of globular actin monomers. They are a major component of all cells and are probably the most abundant animal protein in nature. Neuronal actin is a mixture of beta and gamma and differs from muscle actin in some amino acid residues. In terms of the distribution of actin filaments in neurons, β-actin is enriched at the edge of migrating cells, while γ-actin is relatively concentrated in stress fibers and regions close to the nucleus. Actin filaments are always in a dynamic state, undergoing cycles of polymerization and depolymerization. About half of the actin in a neuron can exist as unpolymerized monomers at any one time. The dynamic polymerization and depolymerization of actin are important for short-distance transport and plasma membrane reorganization together with myosin.
Functionally, actin filaments are involved in the development and activity of neurons. For instance, they serve as scaffolds of neurons and growth cones to guide axonal routing and are also involved in neural network assembly and neuronal reprogramming [4]. Some researchers have also found that the actin filament system provides the driving force required for viral assembly, budding, and release after infection, thereby participating in the regulation of the viral life cycle [2]. Clinically, studies have demonstrated the important role of the actin filament system in viral infection-related diseases [5–7].
2.2 Microtubules
Microtubules serve as the skeleton of cells and are polymers of 55kD α- and β-tubulins. Microtubules have hollow tubular structures with a diameter of about 25 nm and extend the entire length of neurons to act as scaffolds. They are involved in the development and maturation of neurons. Furthermore, microtubules are actively involved in neuronal polarization, organization of intracellular compartments, and remodeling of dendritic spines during neuronal development and maintenance [8]. Since tubulin is encoded by a multigene family and undergoes post-translational modifications, there are more than 20 tubulin isoforms in the brain. In an axon, all microtubules are arranged in parallel, with the positive end pointing away from the cell body and the negative end facing the cell body. Thus, through this regular orientation, different molecules and subcellular structures are allowed to move in an orderly manner along the axon, thereby maintaining a specific distribution throughout the cell. Microtubules can also serve as transport tracks for dynein and kinesin, enabling long-distance cargo transportation [9–11].
Studies have confirmed that tubulin is abundantly expressed in various brain regions of the mouse nervous system. Abnormalities in the stability of microtubules can lead to the development and occurrence of neurological diseases, making microtubule stability-targeting drugs potential therapies for neurological diseases and neuronal damage [12]. Therefore, some scholars have confirmed that the microtubule system in highly differentiated neurons, especially in axons, is more stable [11]. This also suggests that axons are more sensitive to changes in tubulin content or function.
Viruses typically rely on an intact microtubule network to complete their replication cycles. Viruses such as Herpesviridae [13], Adenoviridae [14], and Parvoviridae [15] are directly associated with microtubule-dependent motors for the integration of intact virus particles. Capsids, individual viral proteins, or RNA are transported to the replication site and to the plasma membrane [16]. Microtubules are also used for transport within extracellular vesicles or to transport capsid and nucleoprotein particles from the cytoplasm out of the budding compartment during virion assembly and release. In summary, the microtubule system plays a key role in the transport, polarity, and movement of cargo within cells. In the nervous system, the microtubule cytoskeleton can be hijacked by viruses for directed trafficking to support their replication.
2.3 Intermediate filaments
Intermediate filaments, about 10 nm in diameter, are composed of proteins, including vimentin, keratin, desmin, neuronal fibers, and glial fibers. However, the composition varies greatly in different cells. For example, neurofilament is expressed in the nervous system, acidic keratins are specifically expressed in epithelial tissue, and desmin is always expressed in smooth muscle. Neurofilaments are neuron-specific intermediate fibers and are the most stable cytoskeletal component in this cell type. Each filament consists of a heavy chain, medium chain, light chain, and α-inernexin/peripherin protein [17]. Neurofilaments are distributed in the soma and processes of mature neurons and are more abundant in axons and sparser in dendrites.
Early research on intermediate fibers focused on their function in mechanical support. Vimentin is the most important component of the intermediate filament protein family, which is responsible for maintaining cell shape zbeen found that vimentin also serves as a hub that regulates many signaling pathways, even in cancer. For example, vimentin participates in signaling pathways, such as Raf-1 and RhoA, and regulates tumor necrosis factor (TNF)-α-mediated apoptosis [18, 19]. Meanwhile, vimentin also affects protein kinase C, protein kinase A, c-Jun N-terminal kinase, calmodulin-dependent protein kinase-II, Akt, and phosphatase pathways, among others [20–23].
In recent years, more researchers have paid attention to the regulatory mechanisms of the intermediate filament vimentin in viral infection. For example, vimentin acts as a receptor to influence pathogen invasion. Cell surface vimentin in Neuro-2a cells interacts with the Chandipura virus (CHPV) and acts as a receptor to promote CHPV binding to cells [24]. In addition, vimentin is involved in transcellular migration and immune responses [24].
In conclusion, although researchers have identified the importance of the cytoskeletal network for viral infection, the mechanisms by which viruses invade the nervous system cytoskeleton, regulators, and dynein junctions are critical due to the current large-scale COVID-19 infection worldwide. Future studies, especially on host signaling pathways and downstream effector mechanisms, will undoubtedly provide important new ideas for the underlying mechanisms by which cytoskeletal regulation in the nervous system plays a role during viral infection.
3 Nervous system and SARS-CoV-2
3.1 Nervous system
The nervous system can be divided into two parts: the central nervous system (CNS) and the peripheral nervous system (PNS). The CNS includes the brain and spinal cord, and the PNS includes the cranial and spinal nerves. In animals, the nervous system is involved in perceiving, interpreting, and responding to internal and external stimuli. It is organized at the level of cells, tissues, and organs.
Neurons are the most basic structural and functional units of the nervous system. Their highly polarized morphology is divided into the soma and longer protrusions. The cytoskeleton and its related proteins not only participate in the regulation of neuronal development but also support neurons and their soma and long-process structures formed after cell polarization. Most importantly, the cytoskeletal system also acts as an intracellular transport track, maintaining neuronal structure and function.
Nervous system function requires morphological and functional plasticity of neurons and glial cells. The maintenance of myelin is essential for a healthy adult nervous system. Oligodendrocytes are specialized glial cells of the CNS. Oligodendrocytes contact and myelinize multiple axons and extend multiple actin-based protrusions to form a multilayered myelin membrane, increasing conduction velocity and promoting long-term axonal integrity [25, 26]. It is worth mentioning that the cytoskeleton of oligodendrocytes consists of microtubules and actin filaments, with no intermediate filaments present [27–29].
A growing number of scholars have described the neurological symptoms of COVID-19. The presence of SARS-CoV-2 RNA and protein in anatomically distinct regions of the nasopharynx and brain has also been well documented [30]. We will also review in detail the regulation and impact of the cytoskeleton on SARS-CoV-2 in the context of the nervous system.
3.2 SARS-CoV-2
Human coronaviruses (HCoVs) appear regularly around the world [31] and are a potential threat to humans. SARS-CoV-2 is a large positive-stranded RNA virus belonging to the family Coronaviridae and is a β-CoV. SARS-CoV-2 caused the COVID-19 pandemic that started in December 2019. As of October 2022, over 600 million cases and 6.56 million deaths have been reported. This event led to an unprecedented impact on public health and the economy, triggering an unprecedented response and concern from researchers. Some of the existing studies have focused on elucidating the mechanisms by which SARS-CoV-2 enters host cells. These include the binding of the spike (S) protein to its receptor angiotensin-converting enzyme 2 (ACE2) and subsequent membrane fusion [32]. In addition to the inflammatory response leading to lung injury, SARS-CoV-2 has also been reported to cause long-term respiratory and neurological sequelae [33].
3.3 Harmful effects of SARS-CoV-2 infection in the nervous system
SARS-CoV-2 spreads through contact with respiratory droplets, infecting the respiratory tract, liver, and intestines. In some cases, they can invade the CNS and cause neurological disorders [34]. As with other neurotropic viruses, the pathogenesis of SARS-CoV-2 in the nervous system is related to the life cycle of viral replication in neurons and the host response to nervous system damage [26]. Neurological symptoms, such as headache, dizziness, myalgia, and loss of smell, have been reported in patients with SARS-CoV-2 infection. Some patients exhibit severe pathologies such as encephalopathy, encephalitis, and necrotizing hemorrhagic encephalopathy.
Although a large amount of data now points to SARS-CoV-2 as the cause of neurological lesions in COVID-19 patients, there is currently no data directly indicating that SARS-CoV-2 is neurotoxic. What is certain is that the neuroimmune aspect is caused by viral infection. SARS-CoV-2 binds to toll-like receptors and releases pro-inflammatory cytokines, such as interleukin (IL)-1 and IL-6, which in turn induce immune responses leading to brain tissue damage and stroke. SARS-CoV-2 has been reported to induce ACE2 downregulation in the nervous system and activation of the canonical RAS pathway cascades events, leading to oxidative stress, neuroinflammation, vasodilation, coagulation disorders, and thrombosis. These clinical manifestations are initially caused by innate immune hyperactivity, followed by immunosuppression [35]. In addition, most COVID-19 patients also showed a tendency to increase interferon (IFN) release [36], which can lead to inflammation and immune system suppression to achieve homeostasis. People also found that SARS-CoV-2 unregulated the vascular and immune functions of brain pericytes through the S protein [37, 38]. Numerous cytokines are also involved in response to viral infection to maintain autoimmune homeostasis.
Together, we aim to review the regulation of the cytoskeletal system on the nervous system and elucidate new approaches targeting the cytoskeleton and its related proteins to regulate the nervous system lesions caused by SARS-CoV-2.
4 Cytoskeletal regulations of the nervous system during SARS-CoV-2 infection
Neurological complications in COVID-19 patients have become one of the important causes of morbidity and mortality. Numerous investigators have also provided evidence of SARS-CoV-2 neurotropism [30]. The latest research demonstrates that SARS-CoV-2 viral RNA has also been detected in the brain and cerebrospinal fluid. Moreover, intact virus particles were also detected in the nasopharynx [39]. As CNS and PNS are the main players in regulating the physiological functions of the body, it is particularly important to focus on their regulation by cytoskeletal systems after SARS-CoV-2 infection.
4.1 Cytoskeletal regulations of the CNS during SARS-CoV-2 infection
SARS-CoV-2 can enter the nervous system through three routes. The first is through endocytosis or exocytosis, nerve terminal internalization, and retrograde transport. Viruses can be transported back to neuronal cell bodies along microtubules by vesicular transport (via transsynaptic transfer and axonal transport mechanisms) [40]. Second, from the nasal epithelium to the brain, entry is via a transsynaptic pathway that branches off the trigeminal nerve [41]. The last is through internalization and transendothelial transport across the intact blood–brain barrier (BBB). This is also a common route by which other viruses enter the nervous system.
After entry of SARS-CoV-2, brain pericytes exposed to S protein undergo morphological changes in elongation and contraction in the dynamic regulation of the nervous system and the virus. This leads to increased expression of cytoskeleton-related proteins such as fibronectin [42]. The clinical symptoms of infected people showed loss of smell and taste, fatigue, nausea, and headache. Clinical reports indicate that coronavirus affecting the CNS can lead to complications, including cerebrovascular disease [43], multifocal cerebral micro-occlusion, and stroke [44, 45].
4.2 Cytoskeletal regulations of the PNS during SARS-CoV-2 infection
SARS-CoV-2 RNA has been shown to be present in the PNS [35, 46, 47]. By infecting olfactory neurons, viral proteins enter the nervous system from PNS terminals and the olfactory epithelium, where they eventually reside in the medulla oblongata of the brain. Infection with SARS-CoV-2 could induce multiple neuropathic pain and skeletal muscle damage, causing severe disease, such as Guillain–Barre syndrome, cranial polyneuritis, neuromuscular junction disease, neuro-ophthalmic disease, and autonomic dysfunction [35].
Upon entry of SARS-CoV-2, PNS myelin sheaths serve to maintain axonal integrity and perform key functions required for PNS homeostasis [48]. Axonal cytoskeletal networks regulate axonal caliber, provide nutritional and potential metabolic support, potentially provide genetic material for translation, and protect axons from viral entry. Although the process between the onset of axonal degeneration and the subsequent neuropathic events induced by SARS-CoV-2 is unknown, the regulation of the cytoskeletal network has been the focus of research. This also demonstrates that the intermediate filament cytoskeleton system is involved in the regulation of viral infection (Fig. 1).
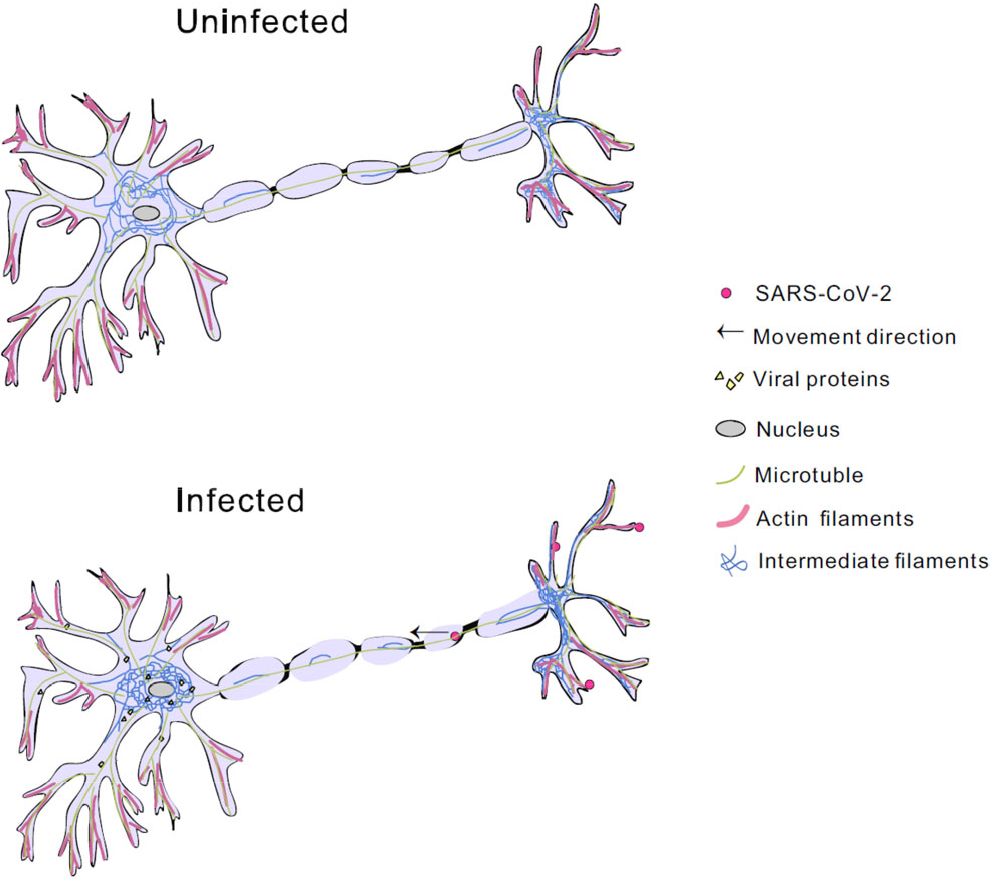
5 Cytoskeleton involved in SARS-CoV-2 induced neurological diseases
As we mentioned above, there are compelling pieces of evidence indicating that SARS-CoV-2 could affect the nervous system, causing severe damage and neurological changes. These neurological disorders are grouped into categories ranging from nonspecific and moderate symptoms (including headache, myalgia, and depression) to severe symptoms (including cerebrovascular disease and intracranial infection). Severe neurological symptoms, such as acute cerebrovascular disease, have been documented only in a minority of patients with conventional risk factors and have been associated with poor outcomes. However, most COVID-19 patients show only mild neurological symptoms. Nonetheless, CNS manifestations have been reported in approximately 25% of COVID-19 patients [49].
5.1 Alzheimer’s disease
Among the CNS complications of COVID-19, AD ranks first in incidence. It has been reported that COVID-19 shares pathogenic cofactors with Alzheimer’s disease (AD) [49]. Due to the urgency of COVID-19 drug development ins the current situation, exploring the association between COVID-19 and AD or drugs used in AD treatment will provide ideas for targeted treatment of COVID-19.
Cytoskeleton-associated protein 4 (CKAP4), a type II transmembrane (TM) protein, has been found to regulate the number and survival of neuronal precursor cells (NPCs) [50]. The level of CKAP4 is significantly increased in the early stage of viral infection of neurons, and it is also used as a target for the detection and treatment of AD caused by coronavirus infection. In addition, its role in the embryonic development of the mammalian CNS has also been recognized. This demonstrates the importance of the cytoskeletal system and associated cytoskeletal proteins in regulating and maintaining the homeostasis of the nervous system during viral invasion.
5.2 Blood–brain barrier
The BBB acts as a physical, metabolic, and transport barrier between the peripheral circulation and the CNS. Its function is achieved by brain microvascular endothelial cells and the interaction of these cells with the astrocyte foot processes of the neurovascular unit (NVU) and the extracellular matrix [51, 52]. It is shown that SARS-CoV-2 S protein could disrupt BBB function. ACE2, a viral S protein binding target, is mainly expressed in endothelial cells in the brain [53]. During infection, S protein triggers RhoA activation, resulting in dynamic changes of endothelial cytoskeleton and tight junction complex, finally lending to BBB disruption and affecting its function [42, 54].
6 Summary
The development and function of the nervous system are inseparable from the dynamic regulation of cytoskeletal systems, such as microtubules, actin filaments, and intermediate filaments. Microtubules are involved in ‘cargo’ transport and nervous system development within neuronal axons [55]. Actin filaments are involved in neural network assembly and neuronal reprogramming. Intermediate filaments provide more structural support for neurons. Widespread neurological manifestations have been caused when SARS-CoV-2 infects the nervous system, leading to disruption of the homeostasis in the CNS and PNS. The cytoskeleton in the nervous system changes to defend against or coordinate virus invasion. This may also be related to the high incidence of neurological complications of AD and BBB disruption in COVID-19 patients. The increasing number of confirmed PNS manifestations caused by COVID-19 makes our work urgent and important. In the context of the ongoing COVID-19 pandemic, the existing basic and clinical researches are still insufficient. We will continue to track and analyze new progress.
Footnotes
Conflict of interests
The authors declare that they have no competing interests and agree to publish this manuscript.
Funding
This work was supported by Key Research and Development Program, Ministry of Science and Technology of China (Grant Nos. 2022YFC2303502 and 2021YFC2300204); and CAS-VPST Silk Road Science Fund (Grant No. GJHZ2021138).
Authors’ contribution
QZ drafted the manuscript and figure. YJ polished the text and evaluate the manuscript. Both authors approved the final manuscript.
