Abstract
Recently, substrate stiffness has been involved in the physiology and pathology of the nervous system. However, the role and function of substrate stiffness remain unclear. Here, we review known effects of substrate stiffness on nerve cell morphology and function in the central and peripheral nervous systems and their involvement in pathology. We hope this review will clarify the research status of substrate stiffness in nerve cells and neurological disorder.
1 Introduction
Substrate stiffness is provided by a microenvironment with certain rigidity comprising the extracellular matrix, adjacent cells, etc., showing different viscoelasticity, and serves as a guidance cue for various biological processes such as embryonic development, tissue formation, and cell migration. In recent years, increasing evidence has demonstrated the role of substrate stiffness in neurodevelopment and disease. Researchers have discovered that neurons can sense and respond to changes in substrate stiffness. For example, tissue stiffness can promote collective migration of Xenopus laevis neural crest cells [1], increasing substrate stiffness correspondingly increases the proliferation rate of glioma cells [2, 3], and the stiffness of rat spinal cord neurons is always harder than that of the substrate [4]. Despite the numerous examples of its relevant biological implications, however, the exact effects of substrate stiffness in the physiology and pathology of nerve cells are less understood.
Current research mainly focuses on the effect of substrate stiffness on neuron morphology. However, new research is expanding the knowledge on the effects of substrate stiffness on neuron function. This review aims to summarize recent knowledge on both these areas, morphology and function.
2 The role of substrate stiffness on neurites
Neurites, dendrites and axons, are elongated extensions of the nerve cell’s body used to transmit information between cells. Neuron development begins with the formation of minor neurites, one of which develops rapidly into an axon while the remaining neurites slowly develop into dendrites. Previous studies have shown that substrate stiffness significantly influences the neurites. The neurites of different nerve cells respond differently to substrate stiffness. Various studies indicate that hippocampal neurons in the central nervous system sense substrate stiffness and regulate neural protrusion development. For instance, rat hippocampal neurons have a higher number of neurite branches [5–7] with increasing substrate stiffness in the range of 300 Pa to 7 kPa. For a substrate stiffness range of 7–20 kPa, the number of branches in mice hippocampal neurons increased with increasing substrate stiffness [8], in the substrate stiffness range of 100 Pa to 4 kPa, the neurite length of mice hippocampal neurons extended with increasing substrate stiffness [9]. However, Kostic et al. demonstrated that mice hippocampal neurons prefer a soft (500 Pa) to a more rigid substrate (7.5 kPa) [10], which was further confirmed by Chen et al. who showed that the development of hippocampal neurons was biased towards a soft substrate (88 kPa) [11].
In summary, hippocampal neuron neurite development is regulated by substrate stiffness. Nevertheless, the regulation law is unclear given the varying stiffness range and conditions reported. Further, a study showed that rat hippocampal neurites do not change under substrate stiffness from 150 Pa to 5 kPa [12]. However, this finding is controversial as some experimental conditions differed from other studies.
Based on all available research, it appears that a specific range of substrate stiffness might promote neurite growth and development in hippocampal neurons; conversely, hippocampal neuron growth and development might be inhibited outside this substrate range.
Neurite development in the dorsal root ganglion (DRG) of the peripheral nervous system is also regulated by substrate stiffness. In one study, the substrate was coated with different collagen to simulate different stiffness environments; under such conditions, neurites from chicken DRG extended the longest at an intermediate concentration. However, this research is limited in that the exact stiffness is not known [13]. Mice DRG cultured on substrates with stiffnesses ranging between 20–200 kPa reached a maximum neurite density at an intermediate elastic modulus of 88 kPa [14]. In contrast, the neurite of rat DRG extends longer, with substrate stiffness increasing in the range of 150 Pa to 5 kPa [11]. However, another study not providing accurate stiffness data showed lower mouse DRG axon extension with increasing substrate stiffness [15]. The above evidence shows that DRG neurite growth is promoted by increasing stiffness within a certain range and neurite development may be inhibited when the stiffness exceeds this range.
In addition, Nichol showed that for the same experimental setting, human motor neurons (HMN) prefer rigid conditions (25 kPa) close to muscle elasticity, whereas human forebrain (hFB) neurons prefer softer substrates close to brain tissue elasticity (0.5 kPa). HMN generate huge neurites on rigid substrates whereas the hFB neurite varies little with changes in substrate stiffness [16].
In summary, the above nerve cells can respond to substrate stiffness by changing their protrusion development. Therefore, different cell types adapt to different substrate stiffness ranges. However, the specific laws governing their regulation require further research.
All the above were studies testing nerve cells on static substrate stiffness. However, the stiffness environment of cells in vivo changes spatially and temporally. To overcome this deficiency, Jiang et al. mimicked the dynamic substrate stiffness in the brain to observe neurite changes, finding that with decreasing stiffness, the axon shortens and the number of neurites decreases. This is consistent with the static substrate experimental results [17]. Given the challenges of these experiments, there are few studies on dynamic substrates. However, more dynamic substrate studies are expected in the future, which will help us better understand the function of substrate stiffness in neurite development under more realistic physiological conditions.
The neurites of spinal cord neurons are also regulated by substrate stiffness. Studies show that spinal cord neurons on softer substrates have more neurites [18, 19]. With increasing substrate stiffness between 100 Pa to 30 kPa, neurite length shortens [19].
Besides, substrate stiffness has a guidance role in the direction of the neurite extension. Xenopus RGCs sense the stiffness gradients and develop their axons toward soft substrates [20, 21]. Further, NG108-15 (fusion mouse neuroblastoma and rat glioma cells) cells respond to stiffness gradients by changing the number of neurites and direction of development [22]. Growing substrate stiffness promotes directional DRG neurite outgrowth [23], suggesting neurite outgrowth pattern governing by substrate stiffness.
Together, this indicates that substrate stiffness can affect the protrusion development of nerve cells. Different cell types adapt to different substrate stiffness ranges. However, how does substrate stiffness affect neurite development?
The growing movement of neurites is strongly associated with the actin cytoskeleton and adhesion proteins [24–29], as well as numerous mechanosensitive signaling proteins, including integrins, Talin, vinculin, shootin1, and cortactin (Fig. 1).
Tanaka et al. found that neurofilament division of hippocampal neurons was significantly inhibited on a substrate with an elastic modulus higher than the usual brain tissue stiffness. Actin structure depends on substrate stiffness [30, 31], similarly, several adhesion proteins are regulated by substrate stiffness. For example, FAK (focal adhesion kinase, a protein associated with integrin, a cell adhesion molecule that regulates protrusion growth and development) expression in rat spinal cord neurons is higher in softer substrates [17, 19], and HMN have increased Ras homolog gene family member A (RHOA) and myosin activity on stiffer substrates [16]. Under conditions comparable to brain tissue stiffness, cultured mice hippocampal neurons extend longer axons on stiffer substrates. The enhancing interaction between L1 protein and laminin on stiffer substrates promotes force generation for axon growth. In addition, increased substrate stiffness promotes actin adhesion coupling. Stiffness dependent axon growth requires actin adhesion coupling mediated by shootin1a, L1, and laminin on the substrate. Hence, it is likely that substrate stiffness affects neurite growth and development by acting on actin structure as well as the activity and expression of adhesion molecules.
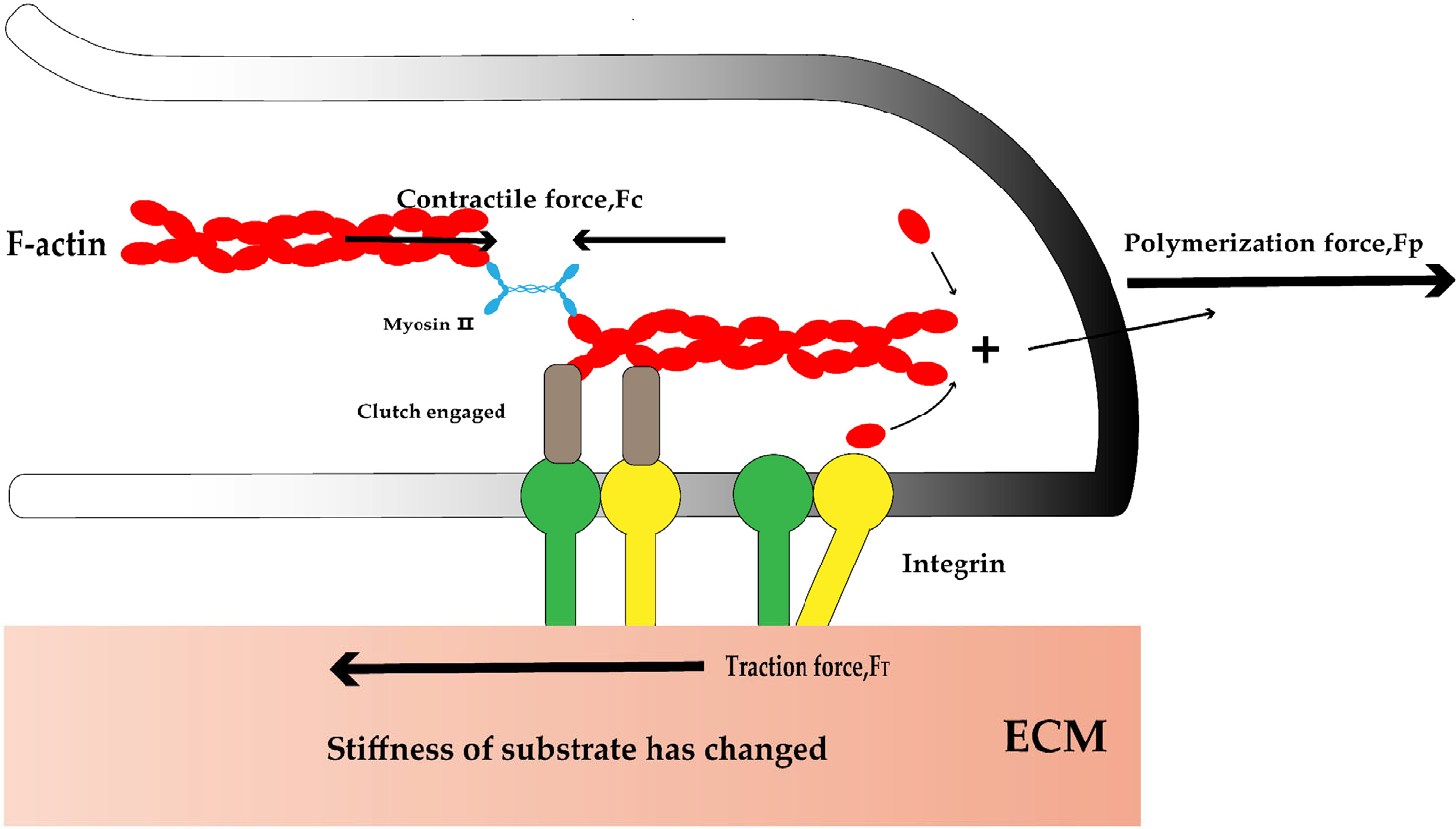
Mechanosensitive signaling proteins involved in neurite development. Mechanosensitive signal molecules (like integrins) in neurons can sense changes in substrate stiffness and bind with F-actin. The actin in the protrusion starts to polymerize, generating a polymerization force (FP) to push the cell membrane forward. The contraction force (FC) generated by myosin pulls the actin in reverse. Then, the internal force is transmitted to the outside through the mechanosensitive signaling protein, generating the traction force (FT) to pull the substrate and move forward. “Clutch engaged” represents a series of proteins connecting cytoskeleton and integrin, such as Talin, vinculin, shootin1, and cortactin.
3 The role of substrate stiffness on synapses
Synapses are key to neuronal connections and information transmission, which play a vital regulatory and control role in the normal activity of the nervous system. Since changes in substrate stiffness have a significant effect on neurites, substrate stiffness may also affect synapses.
Previous research in stiffer substrates (500 kPa) found increased amplitude and frequency of Ca2+ oscillations of mouse hippocampal neurons and more frequent spontaneous neuron excitability compared to 50 kPa [32, 33]. Moreover, the number of synapses on stiffer substrates (500 kPa) was higher than on the soft substrate (50 kPa) based on higher co-localization of the synapse marker synaptotagmin and postsynaptic density protein-95 on stiffer substrates [8, 34].
The influence of substrate stiffness on synapse formation may be mediated by cytokines secreted by astrocytes. As higher hevin and SPARC mRNA transcription and protein expression levels were detected in neurons cultured on the 500 kPa substrate [34], substrate stiffness appears to play a significant role in synapse formation and function.
4 The role of substrate stiffness on glial cells
Glial cells, another class of nerve cells, also have neurites but without distinction between dendrites and axons; they are widely distributed in the central and peripheral nervous systems and include astrocytes, Schwann cells, oligodendrocytes, microglia, etc.
Substrate stiffness has been shown to have a great influence on glial cell morphology. Astrocytes appear round and small on soft substrates (100–200 Pa) but show a rich morphology on stiffer substrates (7–11 kPa) [35–38]. With increasing substrate stiffness, the traction force, strain energy, and intercellular stress of astrocytes increase while cell velocities decrease [35]. In the range of substrate stiffness from 100 Pa to 10 kPa, astrocyte proliferation increases with increasing stiffness, with hyperplasia occurring >2 MPa. Two markers of astrocyte proliferation, glial fibrillary acidic protein (GFAP) and vimentin gene transcription, become significantly elevated with increasing stiffness [36, 37]. However, substrate stiffness has little effect on the structure of actin and the protein Cx43, which is essential for intercellular communication between astrocytes [35].
Between 1–30 kPa, Schwann cell adhesion as well as the percentage of cells with bipolar morphology, and the longitudinal length of cells increased whereas the number of lamellar pseudopods decreased with increasing substrate stiffness. Conversely, the traveled distance and velocity of Schwann cells increased with decreasing substrate stiffness. Moreover, cell stiffness increased with substrate stiffness [39].
Stiffer substrates (30 kPa compared to 1.5 kPa) promote Schwann cell differentiation and prevent Schwann cell dedifferentiation, with increased expression of pro-myelin proteins, Mbp myelin protein [40, 41], and Krox 20 pro-myelin protein [42] and decreased expression of the dedifferentiation marker c-Jun. With increasing substrate stiffness, the migration distance of Schwann progenitor cells from the axon increases [23] as does actin skeleton traction [43].
In contrast, among substrates at 4.42 kPa, 7.45 kPa, 9.10 kPa, and 12.04 kPa, Schwann cells showed the longest cell elongation, highest number of adherent cells, highest cell viability and proliferation, highest mobile distance migration distance, highest expression of neurotrophic molecules ciliary neurotrophic factor, neutrophil cytosolic factor, brain-derived neurotrophic factor, and the highest expression of adhesion-related protein N-calmodulin in at 7.45 kPa [44]. These results differ slightly from those of previous research, probably owing to differences in experimental conditions. Most previous experiments involved laminin while this study did not mention using it.
In studies on oligodendrocytes, substrate stiffness appears to hinder myelin formation, associated with nuclear translocation of Yes-associated protein (YAP). Increased stiffness promotes oligodendrocyte differentiation but not associated with YAP [45]. However, Urbanski et al. showed that substrate stiffness inhibited the formation and differentiation of oligodendrocyte progenitor cell branches; only simple branches were formed on rigid substrates [42]. Extracellular matrix with different substrate stiffness modulates laminin expression. B-type laminin decreases and A-type laminin increases with increasing substrate stiffness in oligodendrocytes [42]. In addition, substrate stiffness also affects the morphological maturation of oligodendrocytes [46].
Increasing substrate stiffness significantly increases the spreading area of primary microglia, showing a rounded state on soft substrates and a complex morphology with longer neurites on stiffer substrates. Microglia traction also increased with increasing substrate stiffness and microglia prefer to migrate to stiffer substrates [47, 48].
In summary, substrate stiffness significantly impacts glial cell morphology and proliferation.
5 The role of substrate stiffness in the pathology of neurological diseases
Substrate stiffness is essential in the pathology of some neurological diseases. Although numerous brain disorders are associated with changes in substrate stiffness, the molecular mechanisms are unclear. Brain stiffness is related to many factors, such as sex, age, brain location, and the presence or absence of brain disease.
Stiffness is increased in the developing brain [49] and decreases with age [50–53]. On the other hand, the occipital and temporal lobes of females generally have a higher stiffness than those of males of the same age [50, 53]. In rats, white and grey matter have different stiffness [54]. Patients with Alzheimer’s disease (AD) and frontotemporal dementia have reduced brain stiffness and softening of the parietal and sensorimotor regions [51, 55]. Magnetic resonance elastography showed stiffness changes mainly in the frontal, parietal, and temporal lobes in AD, with decreasing stiffness correlating with increasing disease severity [56–58]. Patients with normal pressure hydrocephalus show sclerosis of the parietal, occipital, and sensorimotor regions and patients with autoimmune encephalomyelitis have reduced brain viscoelasticity, which increases as inflammation subsides [56, 58]. Mice with multiple sclerosis have a softer cerebral cortex in males than females both healthy and disease [59, 60]. In patients with Parkinson’s disease, viscoelastic parameters are elevated in the midbrain, transiently increase in the hippocampus, and decrease in the substantia nigra [61]. Further, ipsilateral brain tissue stiffness decreases and contralateral brain tissue stiffness increases after ischemic stroke [62]. As some diseases have changes in stiffness in some areas of the brain, changes in brain stiffness can be used to predict the associated diseases, such as in AD [63]. With respect to brain cancer, glioma cells proliferate rapidly, spread widely, and migrate rapidly on highly rigid substrates [3]. Moreover, the stiffness of glioma cells increases with increasing substrate stiffness [64]. In addition, the expression of Talin-1 protein changes with the stiffness of glioma cells [65].
The research results from substrate stiffness in disorder is expected to guide the treatment strategy of diseases. For example, the degree of myelin formation is proportional to the stiffness of central nervous system tissue, and myelin-related disorders can be treated with the aid of tissue stiffness [58]. Brain stiffness increases with increasing myelin [62] and decreases with increasing demyelination [66]. Glial cells can proliferate on stiffer substrates and are prone to scar formation [38], inhibiting damaged neuron’s regenerative function. We could potentially upregulate regeneration related proteins in damaged neurons by chondroitinase ABC which can degrade chondroitin sulfate proteoglycans in the scar [67–69]. Protoporphyrin IX, produced after exogenous 5d-aminolevulinic acid (PPIX) administration, can be used for photodynamic therapy and fluorescence-directed resection. PPIX quantification is the basis for accurate calculation of therapeutic dose and identification of resected malignant tissue. The PPIX concentration required for cells cultured under comparable brain tissue stiffness was significantly higher than for those cultured on stiffer substrates [2].
Studying the neurons differentiation of neural stem cells is expected to guide the nerve regeneration and therapeutic strategies for neurodegenerative diseases. Soft substrates favor neuronal differentiation and rigid substrates favor glial cell differentiation [70–76]. When soft substrate stiffness is close to the elastic modulus of the brain, differentiated neurons have relatively long axons and mature presynaptic terminals, in contrast to rigid substrates [75, 76]. That means that a substrate stiffness closely to brain physiology can drive neuron maturation [72]. There are many differentiated oligodendrocytes from neural stem cell on the rigid substrate, more myelinated and mature neurons are produced when the elastic modulus is <1 kPa, which is close to the brain stiffness. Nevertheless, the molecular mechanisms by which substrate stiffness regulates nervous cell differentiation remain unknown and warrant further research [72].
6 Discussion
Substrate stiffness plays a significant role in neurite development, synapse formation, and glial cell morphology. Nerve cell neurites can sense changes in substrate stiffness and respond to them but respond differently. Given the differences in experimental conditions and specific range of substrate stiffness used in available data, further research is needed to dissect the mechanisms involved. Substrate stiffness is also essential in the pathology of some neurological diseases but the underlying mechanisms are unclear. The role of substrate stiffness on synapses has not been investigated much, mainly because synapses are difficult to observe. The current literature shows that a changing substrate stiffness, may alter the structure of the cytoskeleton and the expression of adhesion-related proteins, such as L1, FAK, and shootin1 by so far unknown mechanisms. As mentioned above, kinds of neurological diseases are accompanied by changes of substrate stiffness. It is reasonable to assume that the development of these diseases is likely related to the molecule that response substrate stiffness change, like L1, FAK, and shootin1 as abovementioned. However, the molecular mechanisms involved is still not clear enough. Is substrate stiffness the cause or result of these diseases? If the substrate stiffness is the cause, shall we treat some of these diseases by regulate the substrate stiffness? These conjectures deserve more relevant studies to verify.
Footnotes
Conflict of interests
The authors declare no competing interests.
Funding
This work was supported by the National Natural Science Foundation of China (Grant Nos. 32170699 and 32200560), National Science Foundation of Hubei (Grant Nos. 2020CFA025 and 2022CFB906), and the Fundamental Research Funds for the Central Universities of South-Central Minzu University (Grant No. CZQ22005).
Author’s contribution
Weijin Si drafted the manuscript. Jihong Gong and Xiaofei Yang revised the manuscript. All the authors approved the final manuscript.
