Abstract
The paraventricular nucleus of the hypothalamus (PVH) acts as a cohesive functional unit that regulates neuroendocrine and autonomic function, complex behavior, and negative emotions after stress. However, how the PVH integrates arousal with these biological functions has only recently been explored. Clinical reports, combined with neurotoxic lesioning, immunochemistry, neuronal activity recordings, and the polysomnographic analyses of genetically modified animals, have revealed that the PVH is important for the control of wakefulness. Here, we review emerging anatomical and neural mechanisms for sleep–wake regulation in the PVH to support its essential role in the promotion and maintenance of wakefulness.
1 Introduction
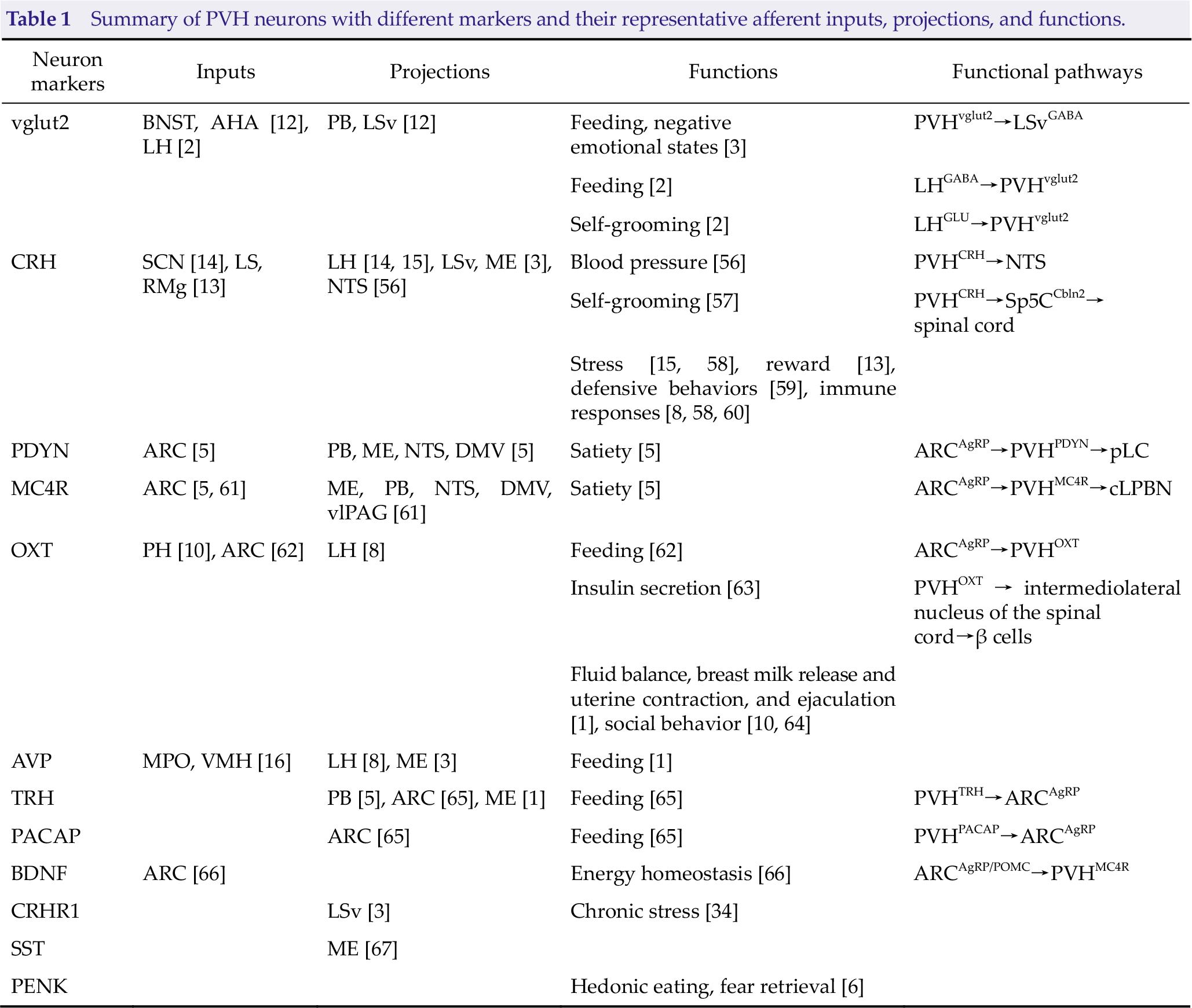
The paraventricular nucleus of the hypothalamus (PVH), located in the ventral diencephalon adjacent to the third ventricle [1], is involved in numerous neurobiological processes, including regulating complex behavior and negative emotions after stress [2, 3], fever response [4], feeding [2, 5, 6], neuroendocrine [7], and autonomic control [8, 9]. The diverse functions of the PVH are executed by various ensembles. The PVH is composed of glutamatergic neurons and lacks inhibitory neurons with vesicular γ-aminobutyric acid (GABA) transporters (Vgat) [6]. The PVH can be divided into three subregions: the anterior PVH (aPVH), middle PVH (mPVH), and posterior PVH (pPVH) [6]. These three subregions have different marker gene enrichment: glutamic acid decarboxylase 2 (GAD2) and netrin G1 (NTNG1) are significantly enriched in the aPVH; the excitatory neuron marker vesicular glutamate transporter type 2 (vglut2), corticotropin-releasing hormone (CRH), and arginine vasopressin (AVP) are significantly expressed in the mPVH; and vglut2, GAD2, neuropeptide Y receptor type 1 (NPY1R), CRH, reelin (RELN), NTNG1, prodynorphin (PDYN), oxytocin (OXT), and AVP are abundantly expressed in the pPVH. Almost 100% of neurons enriched with CRH, PDYN, and OXT have been found to co-express vglut2 in the mPVH and pPVH [6]. Based on their morphological features, neurons in the PVH can also be canonically classified into parvocellular neurons [e.g., somatostatin (SST), thyrotropin-releasing hormone (TRH), OXT [10, 11] and CRH neurons], magnocellular neurons (e.g., OXT [10, 11] and AVP neurons), and long-projecting neurons [e.g., melanocortin-4 receptor (MC4R) neurons] [1].
Numerous neuroanatomical studies have mapped the afferent input and downstream pathways of the major types of neurons in the PVH. As the largest cluster of PVH neurons, PVHvglut2 neurons receive most inputs from the bed nucleus of the stria terminalis (BNST) and anterior hypothalamic area (AHA), and send glutamatergic projections to the brainstem, thalamus, hypothalamus, and cortex [12]. PVHCRH neurons receive direct long-range GABAergic inputs from a range of brain sites, such as the suprachiasmatic nucleus (SCN), lateral septum (LS), and raphe magnus nucleus (RMg) [13], but only project to limited areas, including the lateral hypothalamus (LH) [14, 15], ventral LS (LSv), median eminence (ME), and fornix [3]. As for the PVHPDYN neurons, they mainly project to three neuroanatomical regions, including the parabrachial complex (PB), ME, and the nucleus tractus solitaries and dorsal motor nucleus of the vagus (NTS and DMV), and receive afference from neurons expressing agouti-related peptide in the arcuate hypothalamus (ARC) [5]. Another well-studied type of neuron, PVHAVP neurons, whose inputs are primarily found in the medial preoptic nucleus (MPO) and ventromedial nucleus of the hypothalamus (VMH) [16], project selectively to the ME [3] and LH [8]. The PVH also sends projections to hypoglossal motor neurons, which innervate tongue muscles and play key roles in obstructive sleep apnea [17]. Details of these neuronal types and their anatomical connections and other executive functions, such as feeding control, are summarized in Table 1.
Summary of PVH neurons with different markers and their representative afferent inputs, projections, and functions.
Given the multiple biological functions of the PVH, how it integrates arousal with these fundamental functions has recently received much attention. As a primary biological need for all complex organisms with a central nervous system, sleep is one of the most essential physiological functions of the brain. Despite this, the mechanisms underlying sleep remain largely obscure, with sleep–wake states resulting from complex interactions of neuronal ensembles and influenced by both environmental and internal factors [18]. Herein, we reviewed evolving anatomical and molecular mechanistic models of sleep–wake regulation in the PVH.
2 Evidence for the roles of the PVH in the sleep–wake cycle
2.1 Correlation studies
There is emerging evidence showing that medial parvocellular subdivision of the PVH mediates yawning responses by its oxygen sensor [19], and microinjection of orexin/hypocretin-A into the PVH can also elicit yawning responses [20]. In addition to these correlation studies, the PVH has also been found to receive projections from sleep-active neurons in the preoptic area, and these lamina terminalis neurons projecting to the PVH have been shown to have more prevalent c-fos protein immunoreactivity in awake rats [21]. Furthermore, we visualized the unbiased c-fos expression in the PVH and found a 306% increase in c-fos-positive neurons in the PVH from mice sacrificed in the active period compared to those sacrificed in the inactive period [12]. Additionally, at the onset of behavioral arousal from non-rapid eye movement (NREM) sleep, the mean firing rate of PVH neurons increased from 4.83 Hz to 13.52 Hz, which is indicative of a faster rate in wakefulness than during sleep [12]. Through in vivo multi-unit activity recordings, a significant and sustained up-regulation of neuronal activity in the PVH was observed after 6 h of sleep deprivation (SD) in rats [22]. Focusing on the major population in the PVH, we found that PVHvglut2 neurons were preferentially active during wakefulness. In vivo fiber photometry data from freely moving mice during spontaneous sleep– wake cycles demonstrated that the PVHvglut2 neuronal activity during wakefulness was higher than that during NREM sleep and rapid eye movement (REM) sleep [12]. This evidence points to a high possibility for PVH neuronal activity-dependent engagement in sleep–wake control. Another study found that the PVH was activated when melanopsin retinal ganglion cells were chemogenetically activated to simulate the excitatory effects of bright light, which induces danger context-dependent increases of arousal in dark-housed mice [23]. White light induced higher phosphorylated extracellular signal-regulated kinase expression in the PVH compared to blue light [24]. Taken together, these observations indicate that the PVH is active under light exposure, which be related to the PVH’s regulation of stress and negative emotional behavior [25]. More interestingly, the PVH also serves as the interconnectivity of sympathetic and sleep networks through its GABAA receptor [26], which provides more support for the PVH as an essential node for sleep–wake control.
2.2 Lesion studies
In 1985, Piepenbrock et al. destroyed the hypothalamic paraventricular tissue by electrolytic coagulation in rats and found that the amount of paradoxical sleep significantly decreased along with abolished circadian sleep–wake cycles of paradoxical and slow-wave sleep [27]. Conversely, another group conducted similar experiments on cats, but found that the PVH had little influence on sleep regulation [28]. Recently, we found that specific ablation of PVHvglut2 neurons by bilaterally microinjecting AAV-EF1a-DIO-taCasp3-TEVp into the PVH region of vglut2-Cre mice also induced hypersomnia-like behavior, resulting in a 28.6% reduction in wakefulness and a 74.7% increase in NREM sleep during the dark period [12]. These findings suggest that PVHvglut2 neurons play an important role in the maintenance of wakefulness.
Consistent with the results of animal studies, human patients with lesions around the PVH area exhibited excessive daytime sleepiness and prolonged nocturnal sleep lasting more than 20 h per day [Figs. 1(a) and (b)]. After treatment of primary disorders, these patients recovered with decreased sleep and PVH lesion areas, suggesting that the PVH plays an essential role in the occurrence of hypersomnia [29]. Notably, NREM sleep stage 2 was detected as the dominant stage for these three reported cases, and little REM sleep was observed [Fig. 1(c)] [29], indicating disrupted sleep structures following lesions. Another clinical observation reported that there was a substantial loss (97%) of hypocretin-1 immunoreactive and CRH neurons (88%) in the PVH, along with markedly less CRH-positive fibers in the ME, in narcolepsy type 1 postmortem brains [30]. In contrast, other major neuronal types (AVP, orexin/hypocretin, tyrosine hydroxylase, and TRH) in the PVH were unaffected [30]. These two clinical reports and animal experiments all imply that dysfunction of the PVH leads to an overabundance of sleep and provides solid evidence of the necessity of PVH neurons in physiological arousal.
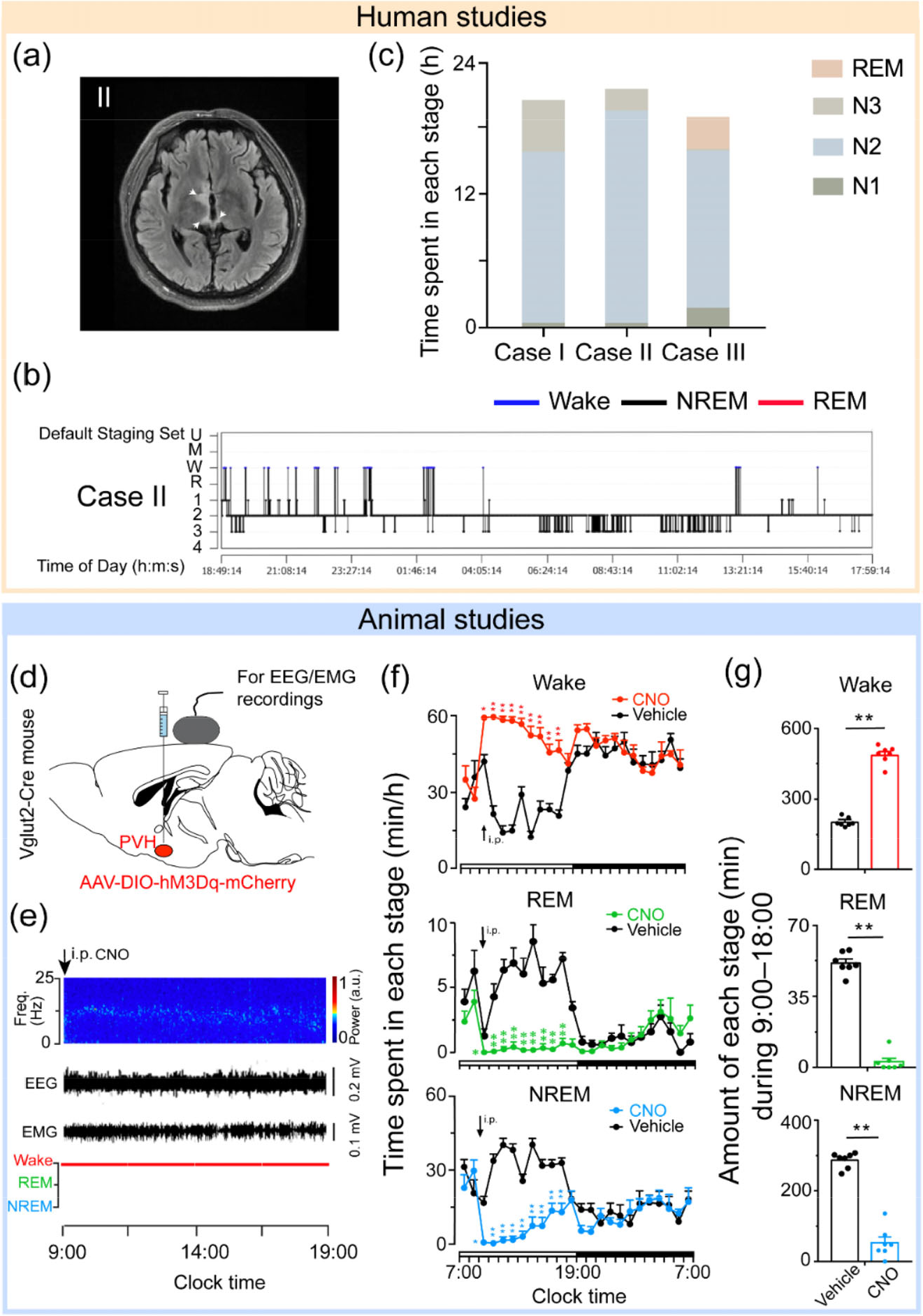
Representative results from human and animal studies supporting the importance of the PVH in sleep–wake regulation. (a) and (b) Three patients with lesions in the PVH areas showed hypersomnia. (a) Diffusion-weighted imaging signals from case II. The injury was in the right hypothalamus and showed a slight hypersignal in the fluid-attenuated inversion recovery image. White arrows indicate the sites of injury. (b) A sleep-structure chart of case II. Blue lines represent wakefulness, red lines represent REM sleep, and black lines represent NREM sleep (including stages N1, N2, and N3). (c) The sleep duration of N1, N2, N3, and REM in all three cases. (d) – (g) Chemogenetic activation of PVHvglut2 neurons increases wakefulness in mice. (d) Expression of the adeno-associated virus (AAV) injection site in the PVH of vglut2-Cre mice. (e) Representative electroencephalogram (EEG)/electromyogram (EMG) traces, and a heatmap of EEG power spectra showing that chemogenetic activation of PVHvglut2 neurons significantly increases wakefulness. (f) Time-course changes in wakefulness, NREM sleep, and REM sleep after administration of vehicle or clozapine-N-oxide (CNO) in mice expressing hM3Dq in PVHvglut2 neurons. (g) Total time spent in each stage after vehicle and CNO injection into vglut2-Cre mice. (a) – (c) are adapted with permission from Ref. [29] © 2022, Wang, Zhong, Jiang, Qu, Huang and Chen; (d) – (g) are adapted with permission from Ref. [12] with permission © 2021, Chen et al.
2.3 Genetic studies
As a result of advances in neuroscience techniques, cutting-edge methods can be applied to selectively manipulate different types of neurons in the PVH. Indeed, four ensembles have been proven to be involved in sleep–wake regulation: PVHvglut2, PVHCRH, PVHOXT, and PVHPDYN neurons (Fig. 2).
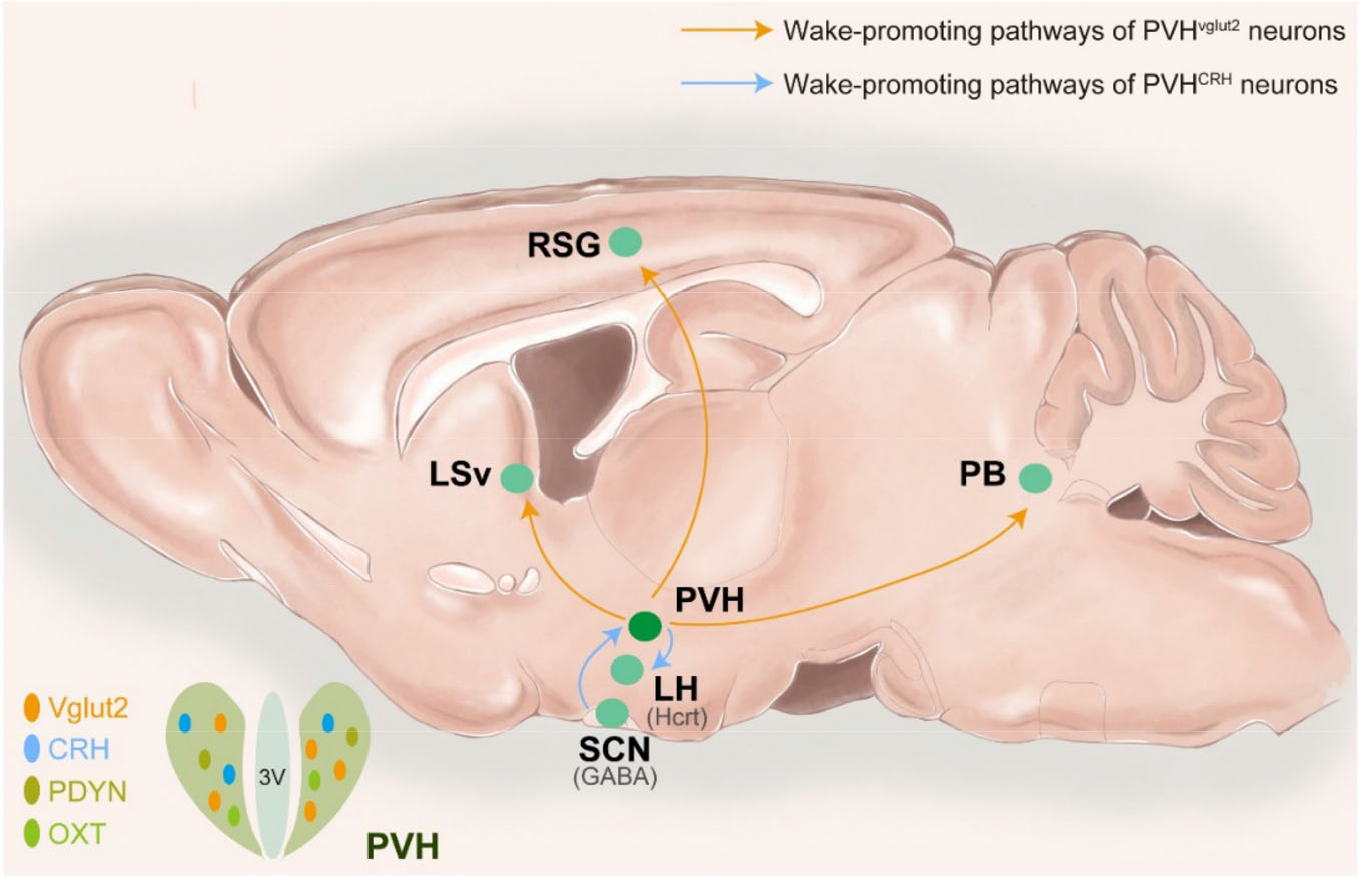
Summary of the roles of the PVH in sleep–wake regulation. Four ensembles were proven to be involved in sleep–wake regulation: PVHvglut2 (orange), PVHCRH (blue), PVHPDYN (brown), and PVHOXT (green) neurons. PVHvglut2 neurons promote wakefulness via PVHvglut2→ LSv/PB pathways, and send direct projections to the retrospleniel granular cortex (RSG) to induce cortical activation. PVHCRH neurons mediate circadian rhythms in the SCN and regulate wakefulness via the SCN→PVHCRH→LH pathway.
With further exploration, we found that chemogenetic activation of PVHvglut2 neurons induced up to 9 h of wakefulness [Figs. 1(d)–(g)]. Moreover, we discovered that it was the PVHvglut2→PB/LSv circuits that drove transitions from sleep to wakefulness immediately by optogenetic approaches [12]. Among these two downstream regions, the PB is also a crucial wake-promoting node, as proven by our previous study showing that activation of PB glutamatergic neurons accelerated transitions from general anesthesia to the arousal state [31–33]. In contrast, chemogenetic inhibition of PVHvglut2 neurons led to a 3-h increase in NREM sleep [12]. These results demonstrate the critical role and underlying mechanisms of PVHvglut2 neurons in arousal promotion and maintenance.
Serving as the initial nodes of the hypothalamic-pituitary-adrenal axis [34], PVHCRH neurons have received considerably more attention than the major type of neurons in the PVH. Almost simultaneously, Ono et al. [14] and Li et al. [8] both reported the role of PVHCRH neurons from different perspectives. Comparing both Ca2+ transients during natural sleep/wake transitions, Ono et al. found that PVHCRH neurons were preferentially active during wakefulness, while Li et al. reported only minimal changes in the PVHCRH neuronal activity. However, similarly, they both found that optogenetic activation of PVHCRH neurons promoted wakefulness through orexin/hypocretin neurons in the LH, whose glutamatergic neurons we have previously proven to be essential for the initiation and maintenance of wakefulness [35]. Furthermore, Ono et al. chemogenetically suppressed and selectively ablated PVHCRH neurons and found decreased locomotor activity and time in wakefulness [14]. Li et al. disrupted the PVHCRH→LHHcrt (hypocretin neurons in the LH) circuitry and observed compromised restraint stress-induced insomnia/ hyperarousal. In our recent study, we also investigated PVHCRH neurons using chemogenetic approaches and found a potent increase in wakefulness lasting approximately 3 h, along with decreases in both NREM and REM sleep after activation of PVHCRH neurons [12]. Combined data from three groups revealed the indispensable role of PVHCRH neurons in sleep–wake regulation.
PVHOXT and PVHPDYN neurons are also involved in sleep–wake regulation. We found that chemogenetic activation of either PVHOXT and PVHPDYN neurons both induced a 1-h increase in wakefulness with decreased NREM and REM sleep [12]. Taken together, recent advances in genetic research have contributed to the emerging view that PVHvglut2 neurons and their subtypes (PVHCRH, PVHOXT, and PVHPDYN neurons) are critical for wakefulness and that each ensemble may be responsible for arousal in different contexts (e.g., natural or stress contexts).
3 Evidence for roles of the PVH in the circadian rhythm
3.1 PVH and circadian rhythm
The SCN serves as a biological clock [36]. Ono et al. [37] recently proposed the GABAergic mechanisms regulating circadian rhythm in the SCN, by suggesting that GABA might work on downstream neuronal pathways from the SCN to control the temporal order of physiology and behavior. Although the SCN provides few outputs directly to sleep-regulatory systems [38], it uses a dual (neuronal and humoral) mechanism for communication with the PVH [39]. Indeed, our previous study identified that SCN cholecystokinin neurons received inputs from the ipsilateral PVHAVP neurons [40]; thus, it is essential to explore the role of the PVH in circadian processes.
José et al. [41] subjected rats to electrolytic lesion of either the SCN or the subparaventricular zone (SPZ) and found that the SCN lesion abolished the circadian rhythm of all behavioral patterns, while lesion of the SPZ only led to abolished eating and drinking behavior, as well as reduced amplitude of a behavioral item associated with REM sleep. In 2000, Lu et al. conducted experiments in which the lesions were more restricted to the PVH, and surprisingly observed contrasting effects of ibotenate lesions of the PVH on sleep–wake cycle and temperature regulation: lesions of the PVH had no significant effect on any circadian rhythms but markedly decreased the fever response induced by lipopolysaccharide [4]. Similarly, another lesion study also concluded that ablation of the PVH did not affect rhythms of rest-activity [42].
However, there is another important physiological timing cue of the circadian rhythm, hormones. Melatonin secretion is driven by the circadian clock through a multi-synaptic pathway connecting the SCN to the PVH and then to sympathetic fibers innervating the pineal gland [43], and lesions of the PVH abolish the nocturnal rise in pineal melatonin [42]. Additionally, most output of the SCN flows into the dorsomedial nucleus of the hypothalamus (DMH) and the DMN is the origin of projections to the PVHCRH neurons for corticosteroid cycles [44]. These findings provide evidence that the PVH is involved in the circadian rhythms of secretion of these hormones.
Furthermore, PVH neuronal activity exhibits a diurnal pattern because of dynamic changes in synaptic inputs and hormonal actions, as well as cell-autonomous changes [39]. PVH neurons express circadian genes, including per1, per2, and brain and muscle aryl hydrocarbon receptor nuclear translocator-like protein (BMAL), and these three clock genes in the PVH showed a clear rhythmic expression in antiphase with their respective expression in the SCN [45]. Moreover, the deletion of BMAL1 in PVH neurons diminishes diurnal rhythmicity in metabolism and therefore leads to obesity [46]; this reveals the key role of the PVH in mediating the clock gene function in diurnal rhythms of feeding and metabolism.
Recently, using selective manipulation techniques, Ono et al. provided direct evidence of how the PVH functions in the circadian rhythm of sleep [14]. A combination of optogenetics and calcium imaging revealed that PVHCRH neurons were negatively regulated by GABAergic neurons in the SCN. In turn, PVHCRH neurons mediate circadian rhythms in the SCN and positively drive LHHcrt neurons to promote wakefulness. These results suggest that the SCNGABA →PVHCRH → LHHcrt pathway is crucial for the circadian regulation of sleep and wakefulness in mammals.
3.2 PVH and ultradian rhythms
As a subset of mammalian circadian rhythms, ultradian rhythms (with periods of 1–6 h, < 24 h), are ubiquitous in various functions of mammalian physiology, including behavioral arousal, NREMREM sleep cycles, hormone release, locomotion, body temperature, and gene expression [47]. The PVH synthesizes and secretes various neurohormones, such as CRH, OXT, and AVP, in an ultradian fashion [48, 49]. Additionally, neurosecretory cells in the PVH producing OXT and AVP display rhythmic patterns of activity [50, 51], indicating that the pulsatile secretion of these hormones in the PVH is regulated by the circadian rhythm. The PVH is also known as an important hub in regulating behaviors exhibiting ultradian rhythms [47], such as feeding and self-grooming (Table 1), providing adaptive value that enables animals to respond appropriately, especially when the nocturnal circadian rhythms are greatly diminished. Although all of these findings are indicative of possible control of the PVH in ultradian rhythms, the direct empirical evidence is still lacking. Of note, we successfully identified ultradian calcium rhythms with a range of 0.5–4.0 h in the SPZ-PVH region [Figs. 3(a)–(c)]. More importantly, we found that this region is the origin of the ultradian calcium rhythms in the SCN because the SPZ-PVN–only slices still displayed the ultradian rhythm [Fig. 3(d)] but the SCN-only slice did not [Fig. 3(e)] [52]. This study first provides clues to unravel the loci and mechanisms of ultradian rhythm in the hypothalamus and underscores the pivotal role of the PVH in ultradian rhythms.
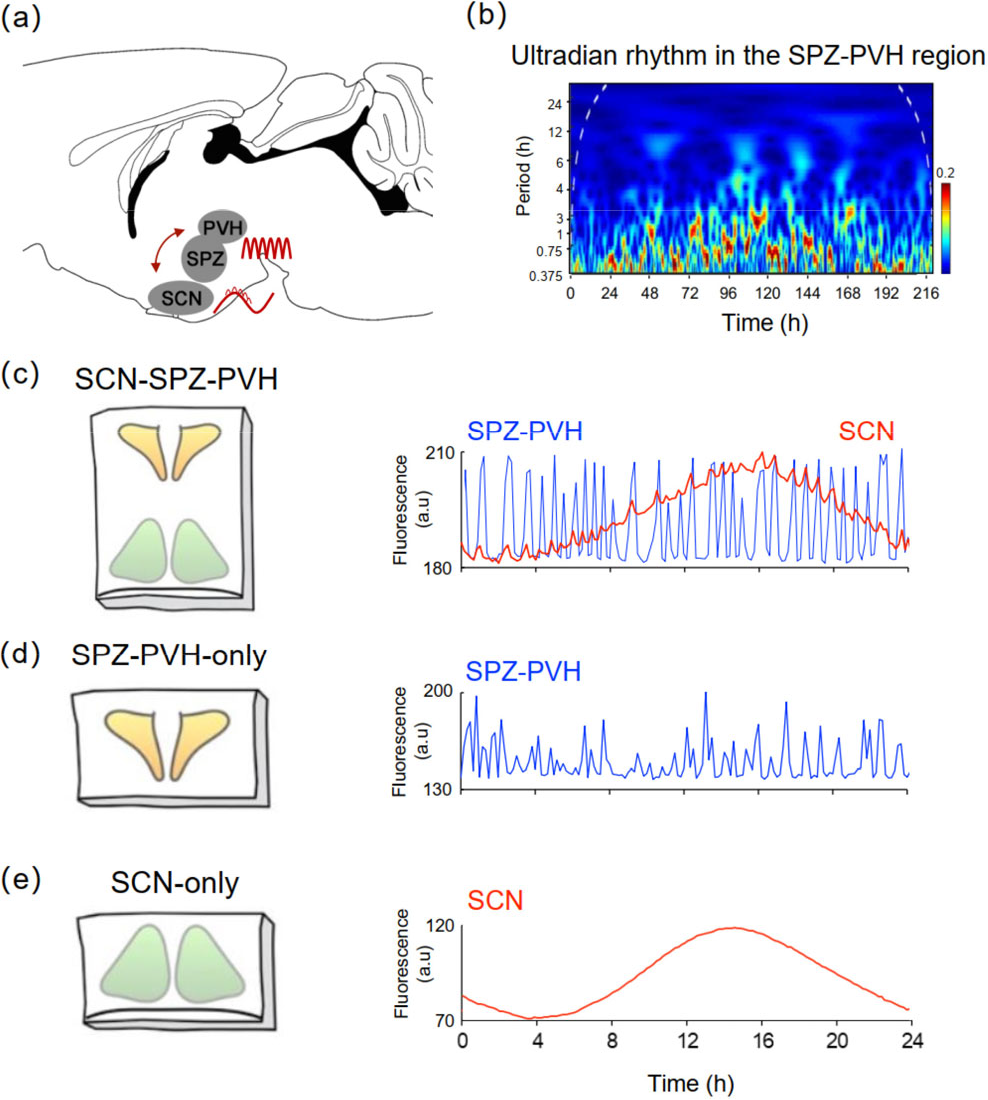
SPZ-PVH region is the origin of ultradian rhythms. (a) The ultradian rhythms originate from the SPZ-PVH region and are transmitted to the SCN, and the SCN controls circadian rhythms of the SPZ-PVH region. (b) Wavelet spectrum of SPZ-PVH calcium rhythms demonstrates ultradian calcium rhythms over a 0.5–4.0-h period. (c) – (e) Time course of the circadian calcium rhythms in the entire SCN region (c), SPZ-PVH-only slice (d), and SCN-only slice (e) during a 24-h recording. The SPZ-PVH ultradian bouts (blue line) were superimposed with the SCN circadian rhythm (red line). None of the SPZ-PVH-only slices exhibited circadian calcium fluctuation, whereas the ultradian rhythms were still detectable (d). In the SCN-only slices, the circadian rhythms were preserved but the ultradian rhythms that were superimposed on the circadian rhythm were abolished (e). Figures are adapted with permission from Ref. [52] © 2018, the Author(s).
4 Conclusion
Widely accepted concepts hold that sleep–wake states are regulated by homeostatic factors (i.e., sleep pressure), circadian rhythms, and allostatic (i.e., food availability or stress) factors [53]. In the first case, the homeostatic process is regulated by the sleep propensity, which builds during wakefulness and declines during sleep [54], and the fact that the PVH was robustly activated after SD [21, 22] indicates its participance of increased sleep pressure. In contrast, the circadian process is controlled by an internal pacemaker in the SCN and is independent of prior sleep and waking [55]. The PVH is also involved in this process by its functional connections with the SCN [14]. However, strong behavioral arousal is required in stressful situations, and PVHCRH neurons actively mediate stress-induced insomnia via the PVHCRH→LHHcrt pathway [8]. The above studies provide evidence to support the role for the PVH in controlling sleep–wake states from three aspects, as suggested from human clinical observations [29, 30] and animal experiments.
The emerging anatomical and functional diversity of the PVH suggests that both afferent and efferent projections are probably discretely localized; hence, numerous anatomical connections and their corresponding functions remain to be fully described. In future studies, deciphering the precise mechanisms of the PVH in sleep–wake control will provide insights into the treatment of patients with neurologic and sleep disorders.
Footnotes
Abbreviations
Conflict of interests
The authors declare no conflict of interests.
Funding
This study was partly supported by the National Major Project of China Science and Technology Innovation 2030 for Brain Science and Brain-Inspired Technology (Grant No. 2021ZD0203400 to Z.-L.H.), the National Natural Science Foundation of China (Grant Nos. 82020108014 and 32070984 to Z.-L.H., the Shanghai Science and Technology Innovation Action Plan Laboratory Animal Research Project (Grant No. 201409001800 to Z.-L.H.), Program for Shanghai Outstanding Academic Leaders (to Z.-L.H.), the Shanghai Municipal Science and Technology Major Project, and ZJLab (Grant No. 2018SHZDZX01 to Z-.L.H.).
Author contributions
Shan Jiang and Lu Chen drafted the manuscript; Zhi-Li Huang and Chang-Rui Chen edited and revised the manuscript; Zhi-Li Huang and Chang-Rui Chen approved the final version of manuscript.
