Abstract
Lumbar disc herniation (LDH) refers to a pathological state in which the nucleus pulposus (NP) protrudes, leading to compression or irritation of nerve roots. This condition manifests with clinical symptoms including lower back and leg pain, hyperalgesia, and altered sensory perceptions. Depending upon clinical observations, the administration of centrally acting analgesic has been associated with the alleviation of pain symptoms LDH patients. The central nervous system sensitization performs a crucial role in pain-regulating perception in LDH. Nevertheless, the precise neural circuitry and mechanism of action remain enigmatic. In the present study, we observed the activation of glutamatergic neurons in the Paraventricular nucleus of the hypothalamus (PVN) and Prelimbic cortex (PrL) in LDH rats. Experimental validation using viral tracers confirmed the existence of a projection pathway between the PVN and PrL. Inhibition of the input from PVN glutamatergic neurons to PrL glutamatergic neurons alleviates chronic pain in LDH, whereas activation of the PVNGlu-PrLGlu projection induces chronic pain in rats. These findings imply a pivotal role for the PVNGlu-PrLGlu circuit in the regulation of chronic pain in LDH.
Keywords
Introduction
LDH represents the most commonplace cause of chronic pain in the low back and lower extremities. 1 The prevalence of LDH is predominantly observed among young and middle-aged males, particularly those engaged in strenuous manual labor, with a reported incidence rate of 7.62% in China.2,3 However, due to the rapid transformation of contemporary production and lifestyle, there has been a notable increase in the number of younger LDH patients, as epidemiological studies have indicated that the incidence rate of LDH among young Chinese is approximately 20%.4,5 Clinically, LDH is characterized by the rupture of the disk annulus fibrosus, leading to the protrusion of NP, which subsequently irritates or compresses nerve roots.6,7 The pathological mechanism of LDH is intricate, with prior research suggesting that its inception is due to mechanical compression, chemical inflammatory stimuli, and autoimmune reactions elicited by disk herniation.8–10 Despite relief of nerve root compression, roughly 20% of patients still have pain symptoms despite the administration of oral anti-inflammatory analgesic medications and other therapeutic interventions.11–13 Therefore, it is imperative to study the pathological mechanisms underlying chronic pain associated with LDH, optimize analgesic treatment strategies, and implement targeted intervention measures.
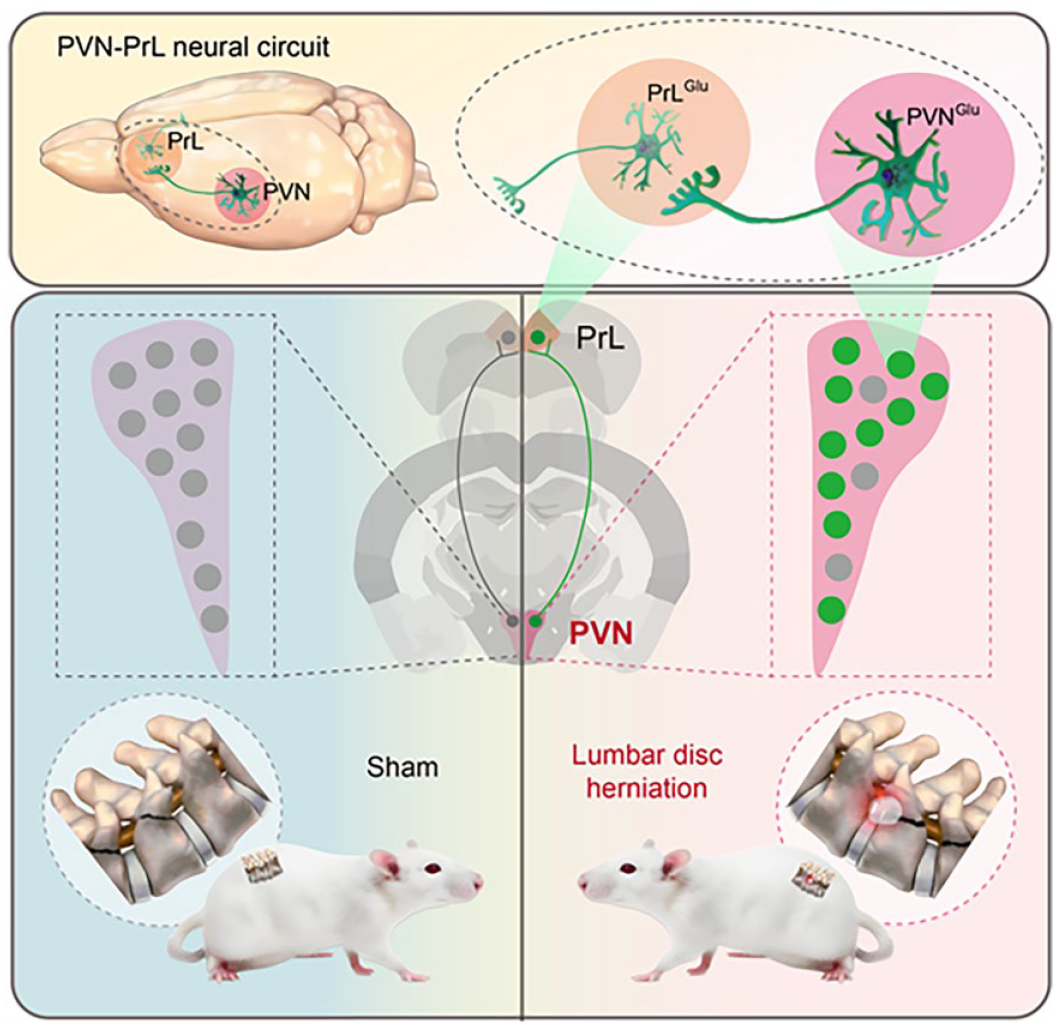
The perception of pain is a complex process from the peripheral nerves to the brain, however, current research on the pain mechanism of LDH mainly focused on the peripheral nerves and the dorsal horn of the spinal cord.14–16 Existing studies on the brain’s role in regulating LDH are extremely limited.17–19 The functional magnetic resonance imaging (fMRI) imaging results of patients with LDH revealed alterations in various brain regions, including the prefrontal cortex, hippocampus, and thalamus.20–22 These findings imply a potential association between these specific brain regions and the phenomenon of central sensitization in LDH patients. 23 Our previous experimental study revealed that following the injection of neurotropic virus (HSV-1H129 strain) into rats’ feet to trace the pain afferent pathway, HSV fluorescence virus was observed in multiple brain regions such as the paraventricular nucleus of hypothalamus (PVN), primary sensory cortex (S1), periaqueductal gray (PAG), anterior cingulate cortex (ACC), and others. The PVN exhibited the most pronounced fluorescence enrichment. 24 The PVN is located in nearby proximity to the third ventricle within the hypothalamus.25,26 The studies have demonstrated the involvement of PVN in regulating a diverse range of physiological activities, including autonomic nervous system function, cardiovascular dynamics, circadian rhythm regulation, energy homeostasis and feeding behavior modulation, social interaction facilitation, as well as anxiety and depression-like behavior.27–30 Recent literature has highlighted the pivotal role of oxytocin neurons in the PVN in the regulation of physical pain, encompassing both inflammatory and neuropathic pain. 31 Therefore, the PVN may be a potential chronic pain regulatory center in LDH. Furthermore, previous studies have demonstrated that the PrL receives anterograde projections from the PVN and plays a crucial role in modulating anxiety, social interaction, and stress-related behaviors.32–34 The medial prefrontal cortex (mPFC), comprising the PrL and infralimbic cortex (IL), is implicated in the perception and integration of pain.35–37 The reorganization of the morphology and function of the mPFC have been demonstrated in chronic pain rats, coupled with a significant elevation in glutamate release within the mPFC.38–40 Although both the PVN and PrL have been implicated in various physiological and pain-related processes, their specific roles in LDH-associated pain remain unclear.
In this study, we observed the activation of glutamatergic neurons in the PVN and PrL in LDH rats, highlighting the pivotal role of the PVN-PrL circuit in LDH-associated chronic pain. This elucidates the fundamental mechanism of chronic pain in LDH from a neurocircuitry perspective, thereby presenting novel therapeutic avenues for managing LDH-associated pain.
Material and methods
Experimental animals
Sprague-Dawley (SD; RRID:RGD-70508) were obtained from Animal Center of Soochow University and approved by the Animal Ethics Committee of Soochow University (Permission Number: 202302A0104). The male Sprague-Dawley adult rats (6–8 weeks old), weighing 230 ± 20 g, were selected as experimental animals for this study. The rats were housed under standard conditions with a light cycle (day/night) of 12 h, a room temperature of 24 ± 2°C, and a relative humidity of 40%–60%. All rats were kept four per cage in standard cages, with free access to tap water and pelleted food. All experimental and animal handling procedures according to the guidelines established by the International Association for the Study of Pain (IASP) The number of rats used in the study design is shown in S1. We monitored behavioral changes, physiological indicators, and signs of pain such as abnormal postures, vocalizations, excessive licking, or biting of affected areas. To reduce animal pain, we administered sodium pentobarbital during surgery, provided a warm and comfortable environment post-operatively, and performed euthanasia when necessary. Since the focus of this study was on the pain hypersensitivity mechanisms in the lumbar disc herniation model in rats, we were concerned that administering analgesics post-surgery might affect the model, so no analgesics were given.
Induction of LDH model
The surgeries for the LDH model were conducted via autologous NP transplantation, as previously described in detail. 41 We used 20 mg/kg of 2% pentobarbital sodium (XY-biotechnology, shanghai, China) administered intraperitoneally (i.p.) for anesthesia. A midline incison was performed on the spine under anesthesia. The paraspinous muscles were dissected to expose the left L5-6 transverse processes, freeing them from the left spinous processes. The left articular processes, lamina, and pedicle of L5-6 were carefully removed with surgical instruments. The autologous NP derived from coccygeal intervertebral discs was transplanted onto the lumbar nerve roots. The sham group received the same operation without NP transplantation.
Nociceptive testing
The paw withdrawal threshold (PWT) in response to a static mechanical stimulus was measured as described previously. 42 Behavioral assessments were conducted before modeling and on days 1, 3, 7, 14, 21, and 28 post-modeling. A set of von Frey filaments (Rayward Life Technology, Shenzhen, China) were applied perpendicularly to the surface of the rat’s hind paws with adequate force until either the filament bended or the rat displayed foot lifting, foot licking, or trembling behavior. The 50% paw withdrawal threshold (unit: g) was determined by applying the formula 50% PWT = Power[10(Xf + κ*δ)], as per the established methodology. 43 All behavioral experiments were performed in double-blind conditions.
Immunofluorescence
The expression of c-Fos in the rat brain and the specific types of activated neurons were observed through immunofluorescence staining on the seventh day post-modeling. Prior to c-Fos detection, mechanical stimulation was applied to the rats using a 3.84 g von Frey filament. Each stimulus lasted for 3 s with an inter-stimulus interval of 1 min, and the stimulation was repeated for a total duration of 10 min (i.e. 10 stimulations in total). After stimulation, the rats were left undisturbed for 1.5 h to allow for immune response development, followed by immunofluorescence staining to analyze c-Fos expression. After anesthetizing the rat with an intraperitoneal injection (i.p.) of 20 mg/kg of 2% sodium pentobarbital, secure the limbs and expose the heart. Then, snip the right atrial appendage to allow the blood to drain out. Perfuse approximately 50 mL of 0.9% saline at a steady rate until the rat’s limbs turn pale, the heart becomes white, and no fresh blood is present in the body. Next, immediately perfuse 20 mL of 4% paraformaldehyde at a steady rate until the rat’s body is completely rigid, with the tail raised and limbs stiffened. The rat brain tissue was removed and placed in a 4% paraformaldehyde solution, followed by fixation at 4°C for approximately 4 h. The brain tissue section embedded had a thickness of 30μm. The main primary antibodies used in this study are Anti-cFos Rabbit mAb (1:200, Cell Signaling Technology, USA, cat.no.2250), anti-cFos Mouse mAb (1:200, Santa Cruz Biotechnology, USA, no.sc-271243), anti-Glutamate Rabbit mAb (1:200, Sigma,USA, cat.no. G6642), anti-GAD67 Mouse mAb (1:200, Sigma, USA, RRID:AB_2278725). The main secondary antibodies used in this study are AlexaFluor™488 donkey anti-IgG (H+L) Rabbit (Thermo Fisher Scientific, USA, cat.no.2376850), AlexaFluor™555 Donkey Anti-IgG Mouse (H+L) (Thermo Fisher Scientific, USA, cat.no.2387458). The images were captured by an Axioscope A1 microscope (Zeiss). The number of c-Fos positive cells was analyzed using ImageJ software (RRID: SCR_003070; National Institute of Mental Health). The average from at least three slices per rat in the PVN and PrL regions was calculated and used for statistical analysis.
Stereotaxic injection and optical fiber implantation
During viral injection, the rats were deeply anesthetized with isoflurane (1.5%–2%) and secured in a stereotaxic apparatus (RWD, 71,000-M, Shenzhen, China). The coordinates are defined as the Dorsoventral (DV) dimension of the brain surface, the Anteroposterior (AP) dimension from the bregmatic point, and the Mediolateral (ML) dimension from the midline, measured in millimeters (mm). The injection was administered in the PVN brain region (AP: −1.56 mm; ML: −0.3 mm; DV: −8.0 mm) and PrL brain area (AP: +3.2 mm; ML: −0.6 mm; DV: −3.6 mm). Subsequent implantation of optical fiber were determined relative to this point. Inject 200 nL or 300 nL of virus at a rate of 20 nL/min per minute using a microinjection pump (Longer Pump, TJ-2A, Baoding, China), and maintain the needle in position for a duration of 10 min following completion of the injection. Rats were warmed with a heat lamp until they regained consciousness and were then moved back to the animal facility. In our experiment, we ensured balance between optogenetic and chemogenetic treatments through a detailed randomization process and inter-group balancing. First, each animal was marked with a unique identification number using non-toxic ink to ensure easy tracking. We employed a block randomization method to allocate the subjects to different experimental groups. A computer-based random number generator (such as the one provided by the software R’s random module) was used to create random sequences. Each animal’s identification number was entered into the software, and the output was a randomized list of numbers corresponding to the treatment groups. This list was then matched with the numbered animals to assign them to either the optogenetic or chemogenetic treatment group. By using block randomization, we ensured that each block contained an equal number of subjects from each treatment group, reducing potential allocation bias. Additionally, we performed inter-group balancing to further minimize any unforeseen discrepancies, thereby enhancing the reliability of our results.
In vivo fiber photometry system
To monitor real-time changes in calcium signaling of glutamatergic neurons in the PVN and PrL, AAV2/9-CaMKIIα-GCaMP6s (Taitool Bioscience, Shanghai, China, titer: 3.6 × 1012 genome copies/mL, speed: 20 nL/min, volume: 300 nL) was stereotactically injected into these regions. To monitor real-time changes in glutamate and oxytocin signaling in glutamatergic neurons of the PrL, AAV2/9-CaMKIIα-iGluSnFR (Taitool Bioscience, Shanghai, China, titer: 5.3 × 1012 genome copies/mL, speed: 20 nL/min, volume: 300 nL) or AAV2/9-CaMKIIα-Oxytocin sensor (Taitool Bioscience, Shanghai, China, titer: 4.6 × 1012 genome copies/mL, speed: 20 nL/min, volume: 300 nL) was stereotactically injected into the PrL region, and an optical fiber was implanted above the injection site for in vivo fiber photometry recording. The local fiber photometry (ThinkerTech Nanjing Bioscience Inc.) was conducted in awake, behaving rats 3 weeks after viral injection. Calcium signaling was recorded in conscious rats, during stimulation from 2 s prior to von Frey stimulation and continuing up to 15 s post-stimulation. The repetition of each stimulus for 8 times was implemented to ensure the precision and authenticity of the collected data. The photometry data were analyzed with custom-written MATLAB codes (MATLABR2021a, MathWorks). Afterwards, the rats were perfused to harvest the target brain region for immunofluorescence experiments.
Chemogenetic manipulations
The following viruses were injected into the PrL or PVN regions of rat: AAV2/9-CaMKIIα-hM4Di-mCherry (BrainVTA, Wuhan, China, titer: 2.44 × 1012 genome copies/mL, speed: 20 nL/min, volume: 200 nL), AAV2/9-CaMKIIα-hM3Dq-mCherry (from BrainVTA, Wuhan, China, titer: 5.40 × 1012 genome copies/mL, speed: 20 nL/min, volume: 200 nL), and AAV2/9-CaMKIIα-mCherry (BrainVTA, Wuhan, China, titer: 5.33 × 1012 genome copies/mL, speed: 20 nL/min, volume: 200 nL. After a period of 21 days, the chemogenetic receptor was activated through intraperitoneal injection of CNO (3 mg/Kg, Clozapine-N-oxide, Apex Bio Technology, USA). The PWT was recorded both before and after intraperitoneal administration of CNO. Subsequently, the rats were perfused and the target brain region was collected for immunofluorescence experiments.
Optical stimulation
Stereotaxic injections were performed into the rat PVN or PrL with the following AAV vectors: AAV2/9-CaMKIIα-GtACR1-EGFP (BrainVTA, Wuhan, China, titer: 5.20 × 1012 genome copies/mL), AAV2/9-CaMKIIα-ChR2-mCherry (titer: 5.20 × 1012 genome copies/mL, speed: 20 nL/min, volume: 200 nL), AAV2/9-CaMKIIα-EGFP (titer: 5.33 × 1012 genome copies/mL, speed: 20 nL/min, volume: 200 nL), AAV2/9-CaMKIIα-eNpHR-EGFP (titer: 6.27 × 1012 genome copies/mL, speed: 20 nL/min, volume: 200 nL), and AAV2/R-hSyn-mCherry (titer: 5.56 × 1012 genome copies/mL, speed: 20 nL/min, volume: 200 nL). The changes in PWT before and after exposure to light were recorded using a optogenetic system (Alpha Omega Engineering, Nazareth, Israel) while the rats were stimulated with blue light pulses (473 nm, 5 mW, 10 ms, 20 Hz) or yellow light pulses (589 nm, 5–8 mW, constant) in the fifth week after virus injection. Subsequently, the rats were perfused and the target brain region was collected for immunofluorescence experiments.
Data analyses and statistics
Summarized data were presented as mean values ± SEM. The normal distribution of the data was preliminary estimated prior to data process and analysis. Statistical analysis was performed with GraphPad Prism 8 (GraphPad Software, Inc). When more than two groups were analyzed, inter-group comparisons were performed using two-way ANOVA followed by Tukey’s multiple comparisons test. A value of p less than 0.05 was considered statistically significant. No tests for outliers were conducted and no data points were excluded. All data were assessed for normality using the Shapiro–Wilk test and for homogeneity of variances using Bartlett test in different groups.
Results
von Frey stimulation significantly induced c-Fos overexpression of PVN glutamatergic neurons in LDH rat
PWT behavioral method was used to detect the pain threshold of mechanical stimulation after NP long-term implantation into the spinal nerve roots of lumbar 5-6. Compared with sham rats, the PWT of ipsilateral hind-paw was significantly reduced 3 days after NP transplantation, and this reduction persisted until the 21 day (Figure 1(a)). These results indicate a persistent hypersensitivity to pain after NP transplantation, which is consistent with our previous studies. 41 To investigate whether the PVN is involved in chronic pain in LDH rats, the c-Fos expression induced by the von Frey was performed (Figure 1(b)). The results demonstrated a significant increase in the number of c-Fos positive neurons within the PVN of rats in the LDH group, compared to those in the sham group, following identical stimulation (Figure 1(c) and (d)). This indicates that PVN neurons in LDH rats were significantly activated and involved in the modulation of pain sensitivity. Immunofluorescence assay showed that c-Fos+ cells were mainly co-expressed with Glutamate in the PVN (Figure 1(e)), a recognized marker for glutamatergic neurons. The ratio of Glu++c-Fos+/c-Fos+ was 75.73% (Figure 1(f)), while only a very small portion of c-Fos was co-labeled with GABA(Figure 1(g) and (h)), suggesting that the PVN neurons activated by LDH are primarily glutamatergic.

von Frey stimulation induce higher c-Fos expression in LDH rat. (a) The PWT of the ipsilateral side were decreased at 3 days, 7 days, 14 days, and 21 days after NP transplantation (two-way ANOVA followed by Sidak’s multiple comparison test, 7 days, F(1,60) = 75.86, ****p < 0.0001; n = 6 rat per group). (b) Schematic diagram of c-Fos expression evoked by von Frey filaments. (c) Representative images of c-Fos+ neurons in PVN after von Frey filament stimulation, Scale bar: 150 μm. (d) Total number of c-Fos+ neurons of the PVN after von Frey stimulation (Student’s t-test, t = 8.793, df = 4, ***p < 0.001; n = 3 rat per group). (e) Representative images showing c-Fos (green) and Glutamate (red) co-expression in the PVN. Scale bars: 150 μm (main panels), 50 μm (insets). (f) Percentage of co-expression of Glutamate and c-Fos in PVN (n = 3 brain sections from three rat). (g) Representative images showing c-Fos (green) and GABA (red) co-expression in the PVN. Scale bars: 150 μm (main panels), 50 μm (insets). (h) Percentage of co-expression of GABA and c-Fos in PVN (n = 3 brain sections from three rat).
von Frey stimulation significantly enhanced calcium activity of glutamatergic neurons in the PVN
GCaMP6 is a fluorescent calcium indicator used to detect changes in intracellular calcium ion concentration. To further validate the alterations in calcium dynamics of PVN glutamatergic neurons, an in vivo optical fiber recording system was employed to capture the calcium response upon von Frey stimulation of the ipsilateral hind paw (Figure 2(a)). The results demonstrated that AAV2/9-CaMKIIα-GCaMP6s virus was successfully expressed in the PVN glutaminergic neurons (Figure 2(b)). Under the stimulation of 15 g von Frey filament, the changes in GCaMP6 signal of PVN glutamatergic neurons in both the sham and LDH groups exhibited subtle or pronounced dynamic fluctuations (Figure 2(c)), but the changes in GCaMP6 signal in the LDH group was significantly higher than that in the sham group (Figure 2(d) and (e)). These findings suggest that the glutamatergic neurons in the PVN of the LDH group exhibited heightened sensitivity to mechanical stimulation.

von Frey stimulation affects calcium in PVN glutamatergic neurons. (a) Schematic of the in vivo optical fiber recording system. (b) Representative image of AAV2/9-CaMKIIα-GCaMP6s virus expression in PVN, Scale bar: 150 μm. (c) Heatmap and event correlation map depicting calcium signal changes in glutamatergic neurons of the PVN of rats from the sham or LDH groups subjected to von Frey stimulation (left). Calcium signals (ΔF/F) of PVN glutamatergic neurons in Sham (blue) and LDH (red) rats (right). (d and e) Averaged area under the curve (AUC) and peak ΔF/F of calcium activity of the PVN glutamatergic neurons in sham and LDH rats (Student’s t-test, AUC (t = 3.584, df = 12, **p < 0.01); n = 7 rat per group, Peak (t = 7.158, df = 12, ****p < 0.0001); n = 7 rat per group).
Optogenetic or chemogenetic manipulation of PVN glutaminergic neurons activity affected mechanical hyperalgesia in rats
To further validate the association between excitability of PVN glutamatergic neurons and hyperalgesia, the excitability of these neurons was manipulated using chemogenetics and optogenetics method, and alterations in PWT were observed (Figure 3(a) and (e)). AAV2/9-CaMKIIα-hM4Di-mCherry, AAV2/9-CaMKIIα-mCherry, AAV2/9-CaMKIIα-GtACR1-EGFP, or AAV2/9-CaMKIIα-EGFP were injected into the PVN to suppress glutamatergic neurons (Figure 3(b) left, (f) left). In LDH rats, PWT was increased and hyperalgesia was significantly relieved after intraperitoneal injection of CNO or yellow light (589 nm) irradiation to inhibit PVN glutamatergic neurons (Figure 3(c) and (g)). Conversely, AAV2/9-CaMKIIα-hM3Dq-mCherry, AAV2/9-CaMKIIα-mCherry, AAV2/9-CaMKIIα-ChR2-mCherry, and AAV2/9-CaMKIIα-mCherry were injected into the PVN to activate glutamatergic neurons (Figure 3(b) and (f)). In sham rats, PWT decreased and develop hyperalgesia after intraperitoneal injection of CNO or blue light (473 nm) irradiation to activate PVN glutamatergic neurons (Figure 3(d) and (h)). The findings suggest that PVN glutamatergic neurons play a role in the regulation of pain hypersensitivity in LDH.

PVN glutamatergic neurons affects LDH-induced hyperalgesia. (a) Experimental protocol for chemogenetic manipulation of PVN glutamatergic neurons. (b) Representative image of AAV2/9-CaMKIIα-hM4Di-mCherry (left) and AAV2/9-CaMKIIα-hM3Dq-mCherry (right) expression in PVN glutamatergic neurons, Scale bar: 150 μm. (c) CNO inhibition of PVN glutamatergic neurons significantly increases the PWT in rat injected with AAV2/9-CaMKIIα-hM4Di-mCherry, compared with rat injected with AAV2/9-CaMKIIα-mCherry (two-way ANOVA followed by Tukey’s multiple comparison test, Pre-CNO: hM4Di-mcherry vs Post-CNO: hM4Di-mCherry, F(1,24) = 20.62, ****p < 0.0001, n = 7 rat per group; Post-CNO: mcherry vs Post-CNO: hM4Di-mCherry F(1,24) = 38.70, ****p < 0.0001, n = 7 rat per group). (d) CNO activation of PVN glutamatergic neurons significantly decreases the PWT in rat injected with AAV2/9-CaMKIIα-hM3Dq-mCherry, compared with rat injected with AAV2/9-CaMKIIα-mCherry (two-way ANOVA followed by Tukey’s multiple comparison test, Pre-CNO: hM3Dq-mCherry vs Post-CNO: hM3Dq-mCherry, F(1,20) = 17.80, ***p < 0.001, n = 6 rat per group; Post-CNO: mCherry vs Post-CNO: hM3Dq-mCherry, F(1,20) = 17.28, ***p < 0.001, n = 6 rat per group). (e) Experimental design for optogenetics manipulation of PVN glutamatergic neurons. (f) Representative image of AAV2/9-CaMKIIα-GtACR1-EGFP (left) and AAV2/9-CaMKIIα-ChR2-mCherry (right) expression in PVN glutamatergic neurons, Scale bar: 100 μm. (g) Yellow light inhibition of PVN glutamatergic neurons significantly increases the PWT in rat injected with AAV2/9-CaMKIIα-GtACR1-EGFP, compared with rat injected with AAV2/9-CaMKIIα-EGFP (two-way ANOVA followed by Tukey’s multiple comparison test, Light off: GtACR1-EGFP vs Light on: GtACR1-EGFP, F(1,20) = 13.76, ***p < 0.001, n = 6 rat per group; Light on: EGFP vs Light on: GtACR1-EGFP, F(1,20) = 15.76, ***p < 0.001, n = 6 rat per group). (h) Blue light activation of PVN glutamatergic neurons significantly decreases the PWT in rat injected with AAV2/9-CaMKIIα-ChR2-mCherry, compared with rat injected with AAV2/9-CaMKIIα-mCherry (two-way ANOVA followed by Tukey’s multiple comparison test, Light off: ChR2-mCherry vs Light on: ChR2-mCherry, F(1,24) = 17.43, ***p < 0.001, n = 7 rat per group; Light on: mCherry vs Light on:ChR2-mCherry, F(1,24) = 22.07, ****p < 0.0001, n = 7 rat per group).
PVN glutamatergic neurons project to the PrL, and c-fos expression in the PrL is significantly increased following von Frey stimulation
To elucidate the neural circuits of PVN glutamatergic neurons, an anterograde tracer, AAV-CaMKIIα-ChR2-mCherry virus, was stereotaxically delivered into the PVN of Con rat (Figure 4(a)). After a duration of 3 weeks, a substantial quantity of axonal terminations was observed within the PrL (Figure 4(a)). Stereotactic injection of retrograde tracer virus (AAV-2/R-hSyn-mCherry) into PrL showed that PrL could accept direct projection of PVN (Figure 4(b)). Specific neurons projected retrograde from PrL to PVN are primarily colocalized with glutamate (Figure 4(c)). The findings suggest that PrL may be regulated by glutamatergic neurons in the PVN, and the PVNGlu-PrL neural circuit may mediate the development of LDH. According to the von Frey induced c-Fos test, positive expression of c-Fos was observed in the PrL of rats in both the Sham and LDH groups (Figure 4(d)). Furthermore, there was a significant increase in the number of c-Fos positive neurons in the PrL of rats in the LDH group compared to those in the sham group (Figure 4(e)). Immunofluorescence analysis revealed that the proportion of Glu++c-Fos+/c-Fos+ in the PrL was 74.67% (Figure 4(f) and (g)), while GAD67++c-Fos+/c-Fos+ in the PrL was 15.83% (Figure 4(h) and (i)). The findings demonstrated that LDH exerts a significant activating effect on glutamatergic neurons in the PrL.
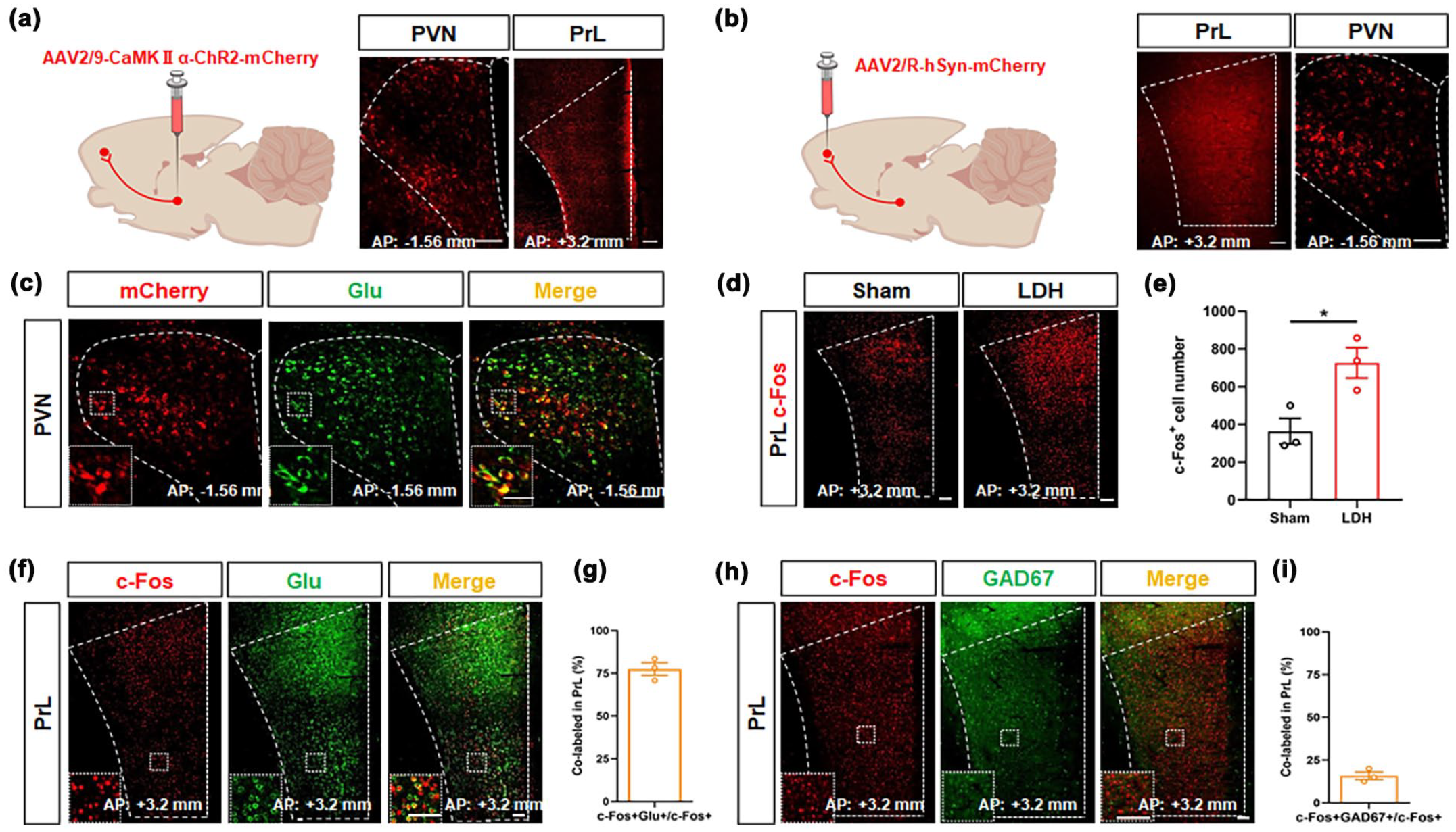
The anterograde projective relationship between PVN glutaminergic neurons with PrL. (a) Representative image of AAV- CaMKIIα-ChR2-mCherry expression in PVN and PrL, Scale bar: 150 μm(left), 200 μm (right). (b) Representative image of AAV-2/R-hSyn-mCherry expression in PVN and PrL, Scale bar: 200 μm (left),150 μm (right). (c) The neurons labeled by mCherry in the PVN co-localize with neurons labeled by Glutamate, Scale bars: 150 μm (main panels), 50 μm (insets). (d) Representative images of c-Fos+ neurons in PrL after von Frey filament stimulation, Scale bar: 200 μm. (e) Total number of c-Fos+ neurons of the PrL after von Frey stimulation (Student’s t-test, t = 3.433, df = 4, *p < 0.05; n = 3 rat per group). (f and h) Representative images of c-Fos (red), Glutamate (green), and GAD67 (green) co-expression in PrL, Scale bars: 150 μm (main panels), 150 μm (insets). (g) Percentage of co-expression of Glutamate and c-Fos in the PrL (n = 3 brain sections from three rat). (i) Percentage of co-expression of GAD67 and c-Fos in the PrL (n = 3 brain sections from three rat).
von Frey stimulation significantly enhances calcium activity in PrL glutamatergic neurons
By stereotactically injecting AAV2/9-CaMKIIα-GCaMP6s virus into PrL, an in vivo fiber-optic recording system was used to capture alterations in intracellular calcium concentration changes in PrL glutamatergic neurons (Figure 5(a) and (b)). Under the stimulation of 15 g von Frey filament, intracellular calcium concentration changes of PrL glutamatergic neurons in both the sham group and LDH groups exhibited dynamic fluctuations (Figure 5(c)). In contrast, the LDH group demonstrated a markedly elevated changes in GCaMP6 signal compared to the sham group (Figure 5(d) and (e)). These findings suggested that glutamatergic neurons in the PrL exhibit heightened sensitivity in the group.

von Frey stimulation elicits a sharp increase in calcium signals within PrL glutamatergic neurons. (a) Schematic of the in vivo optical fiber recording system. (b) Representative image of AAV2/9-CaMKIIα-GCaMP6s virus expression in PrL, Scale bar: 200 μm. (c) Heatmap and event correlation map illustrating changes in calcium signaling within glutamatergic neurons of the PrL in rats from both the sham and LDH groups. Calcium signals (ΔF/F) of PrL glutamatergic neurons in Sham (blue) and LDH (red) rats. (d, e) Averaged area under the curve (AUC) and peak ΔF/F of calcium activity in the PrL glutamatergic neurons between sham and LDH rats (Student’s t-test, AUC (t = 3.960, df = 10, **p < 0.01); n = 6 rat per group, Peak (t = 5.694, df = 10, ***p < 0.001); n = 6 rat per group)).
PrL glutamatergic neurons modulate persistent hypersensitivity to pain after NP transplantation
The excitability of glutamatergic neurons in PrL was regulated by Chemogenetic and optogenetic methods to further verify the relationship between the excitability of glutamatergic neurons in PrL and hyperalgesia in LDH rats (Figure 6(a), (b), (d), and (e)). After the administration of CNO to suppress the activity of glutamatergic neurons in the PrL, which were transduced with AAV2/9-CaMKIIα-hM4Di-mCherry virus, PWT was significantly increased and hyperalgesia rats was markedly alleviated in the LDH group (Figure 6(c)). After the activation of glutamatergic neurons in the PrL using blue light (473 nm) in the Sham group expressing AAV2/9-CaMKIIα-ChR2-mCherry virus, a significant decrease in PWT was observed, indicating the development of hyperalgesia in these rats (Figure 6(f)). The PWT of rats expressing AAV2/9-CaMKIIα-mCherry control virus did not change significantly.
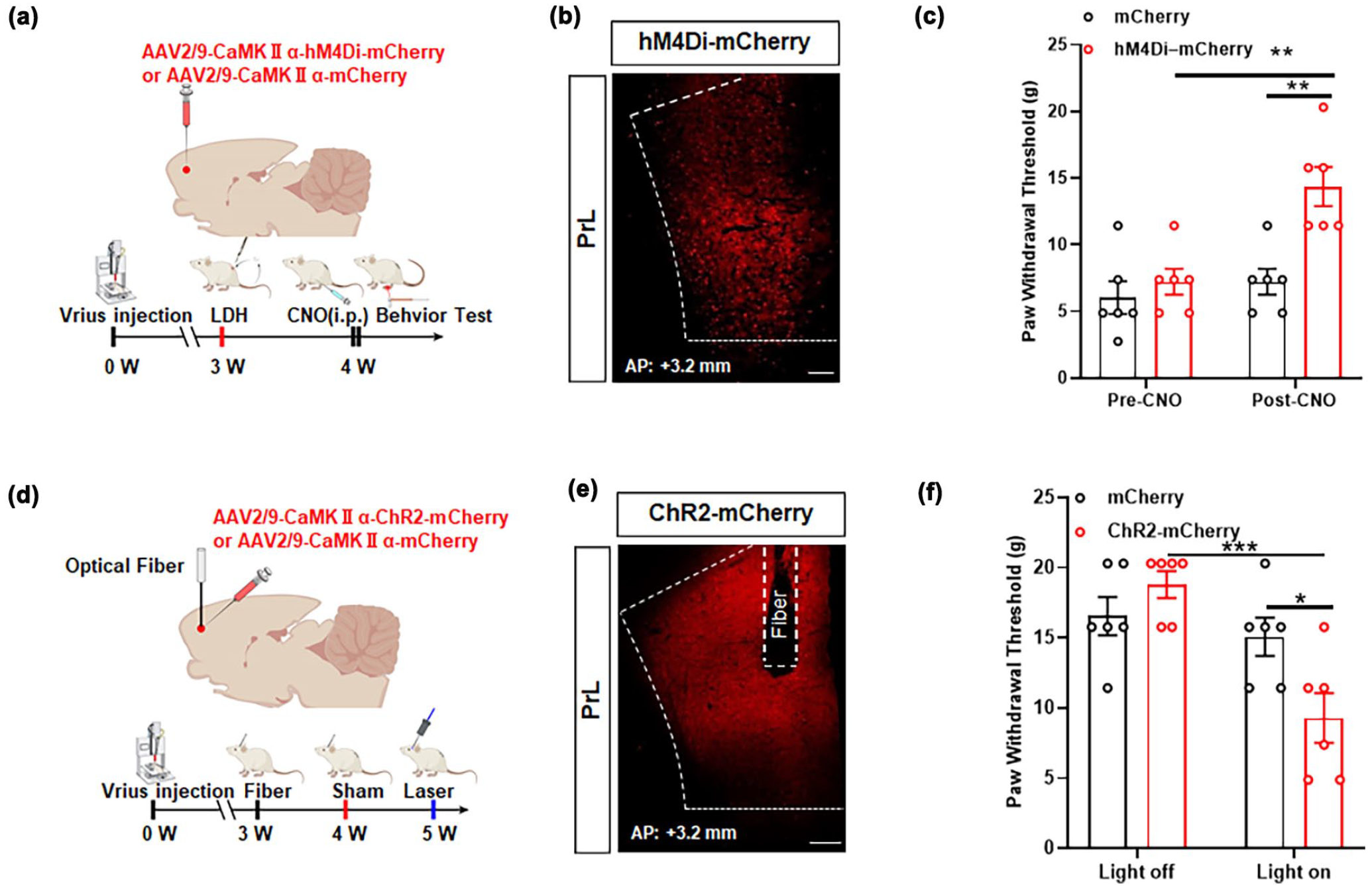
PrL glutamatergic neurons modulate persistent hypersensitivity to pain of LDH. (a, d) Model of virus injection site and timeline of experimental process. (b, e) Representative image of AAV2/9-CaMKIIα-hM4Di-mCherry and AAV2/9-CaMKIIα-ChR2-mCherry expression in PrL glutamatergic neurons, Scale bar: 100 μm. (c) CNO inhibition of PrL glutamatergic neurons significantly increases the PWT in rat injected with AAV2/9-CaMKIIα-hM4Di-mCherry, compared with rat injected with AAV2/9-CaMKIIα-mCherry (two-way ANOVA followed by Tukey’s multiple comparison test, Pre-CNO: hM4Di–mCherry vs Post-CNO: hM4Di–mCherry, F(1,20) = 12.25, **p < 0.01, n = 6 rat per group; Post-CNO: mcherry vs Post-CNO: hM4Di-mCherry F(1,20) = 12.25, **p < 0.01, n = 6 rat per group). (f) Blue light activation of PrL glutamatergic neurons significantly decreases the PWT in rat injected with AAV2/9-CaMKIIα-ChR2-mCherry, compared with rat injected with AAV2/9-CaMKIIα-mCherry (two-way ANOVA followed by Tukey’s multiple comparison test, Light off: ChR2-mCherry vs Light on: ChR2-mCherry, F(1,20) = 15.53, ***p < 0.001, n = 6 rat per group; Post-CNO: mcherry vs Light on: mCherry vs Light on:ChR2-mCherry, F(1,20) = 1.622, *p < 0.05, n = 6 rat per group).
The PVNGlu-PrLGlu neural circuit is responsible for the induction of hyperalgesia in LDH rats
Our findings suggested that glutamatergic neurons in the PVN and PrL play a modulatory role in the hypersensitivity to pain of LDH, and that glutamatergic neurons of the PVN have an anterograde projection to the PrL. To understand the role of PVN-PrL pathway in LDH, the AAV2/9-CaMKIIα-eNpHR-EGFP virus and AAV2/9-CaMKIIα-EGFP virus, or AAV2/9-CaMKIIα-ChR2-mCherry virus and AAV2/9-CaMKIIα-mCherry virus was injected to PVN, meanwhile optical fibers were injected into PrL (Figure 7). The synaptic transmission of PVN-PrL circuit was regulated by optogenetics (Figure 7(a)). The findings revealed a significant increase in the PWT of the LDH group subsequent to inhibition of the PVN-PrL circuit using 589 nm yellow light (Figure 7(b)). After optogenetic activation of the PVN-PrL circuit using 473 nm blue light, a significant decrease in PWT was observed in rats from the Sham group (Figure 7(d)). These results indicated that the PVNGlu-PrL neural circuit is involved in regulating hyperalgesia in LDH rats. Subsequently, we further employed a dual virus strategy to further investigate the role of PVN-PrL in LDH-associated chronic pain in rats. AAV2/9-CaMKIIα-eNpHR-EGFP virus and AAV2/9-CaMKIIα-ChR2-mCherry were injected into the PVN, while AAV2/9-CaMKIIα-hM3Dq-mCherry and AAV2/9-CaMKIIα-hM4Di-mCherry were injected into the PrL. Additionally, an optical fiber was embedded within PrL (Figure 7(e) and (g)). After the yellow light silencing PVN glutamatergic neurons projected to PrL axon terminals, a significant increase in the PWT was observed in rats of the LDH group. After intraperitoneal injection of CNO, it was found that the activation of glutamatergic neurons in PrL partially blocked the inhibitory effect of yellow light, resulting in a decrease in PWT compared to yellow light alone (Figure 7(f)). After the blue light activated PVN glutamatergic neurons projected to PrL axon terminals, PWT of rats in sham group was significantly reduced. After intraperitoneal injection of CNO, the suppression of glutamatergic neurons in PrL was found to partially block the activating effect of blue light, resulting in an increase in PWT compared to blue light alone (Figure 7(h)). This result is also consistent with the characteristics of optogenetics, which offers high spatiotemporal resolution, and chemogenetics, which provides sustained and stable effects. Chemogenetic inhibition of the PrL brain region partially blocked the activation effects of blue light, reversing the behavior in rats.The present findings provide preliminary evidence supporting the involvement of the PVNGlu-PrLGlu glutamatergic neural circuit in mediating hyperalgesia in LDH rats.

The PVNGlu-PrLGlu neural circuit is responsible for the induction of hyperalgesia in LDH rats. (a, c, e, g) Model of virus injection site and timeline of experimental process. (b) Impact of optogenetic inhibition of PVN-derived neuron axon terminals in PrL on PWT in rats (two-way ANOVA followed by Tukey’s multiple comparison test, Light off: CaMKIIα-eNpHR-EGFP vs Light on: CaMKIIα-eNpHR-EGFP, F(1,20) = 8.687, **p < 0.01, n = 6 rat per group; Light on: CaMKIIα-EGFP vs Light on: CaMKIIα-eNpHR-EGFP, F(1,20) = 4.149, *p < 0.05, n = 6 rat per group). (d) Impact of optogenetic activation of PVN-derived neuron axon terminals in PrL on PWT in rats (two-way ANOVA followed by Tukey’s multiple comparison test, Light off: ChR2-mCherry vs Light on: ChR2-mCherry, F(1,20) = 14.43, **p < 0.01, n = 6 rat per group; Light on: mCherry vs Light on: ChR2-mCherry, F(1,20) = 5.488, *p < 0.05, n = 6 rat per group). (f) The impact of optogenetic inhibition of PrL derived from PVN neuron axon terminals and chemogenetic activation of glutamatergic neurons in PrL on PWT in rats with LDH group (one-way ANOVA followed by Tukey’s multiple comparison test, F(2,15) = 15.62, Light off vs Light on, ***p < 0.001; Light on vs Light on+CNO, **p < 0.01; n = 6 rat per group). (h) The impact of optogenetic activation of PrL derived from PVN neuron axon terminals and chemogenetic inhibition of glutamatergic neurons in PrL on PWT in rats with LDH group (one-way ANOVA followed by Tukey’s multiple comparison test, F(2,15) = 17.30, Light off vs Light on, ***p < 0.001; Light on vs Light on+CNO, **p < 0.01; n = 6 rat per group).
Discussion
The pathophysiological underpinnings of LDH are multifaceted and the central nervous system has emerged as a promising therapeutic avenue for LDH.44–46 Notably, LDH patients exhibit alterations in brain function and structure, and central analgesics drugs administering mitigate pain symptoms, indicating that elucidating the central mechanisms underpinning chronic pain in LDH is pivotal to its effective management.47–49 This study revealed that glutamatergic neurons in the PVN project to glutamatergic neurons in the PrL, offering novel insights into the regulation of pain hypersensitivity in LDH rats through selective modulation of the PVN-PrL neural circuit. These findings offer a unique perspective on understanding and modulating chronic pain in LDH rats.
In recent years, with the continuous deepening of the understanding of neural circuits in central mechanisms, the pivotal roles of multiple brain structures and their circuits in pain regulation have been confirmed.50–52 The PVN, a key neuroendocrine center within the hypothalamus, is intricately involved in pain-related pathophysiological processes, encompassing visceral, inflammatory, and neuropathic pain states.53–55 However, the underlying mechanism of LDH’s regulatory role in pain remains elusive. Here, our study determined that LDH significantly increased c-Fos expression and changes in GCaMP6 signal in the PVN, a region predominantly innervated by glutamatergic neurons. 56 We conducted an experiment to stain the whole brain of a LDH model using c-fos to pinpoint specific brain regions associated with chronic pain in LDH. Immunofluorescence staining revealed that following Von Frey stimulation on the soles of LDH rats, c-fos expression was observed in the PAG, PVN, ACC, among other brain regions. Notably, the paraventricular nucleus exhibited the most pronounced expression and the c-fos neurons with positive expression were co-labeled with glutaminergic neurons. These findings indicate that LDH may induce hyperalgesia in rats through PVN activation. Simultaneously, optogenetic or chemogenetic studies have revealed that hyperalgesia in LDH rats is associated with glutamatergic neurons, aligning with the observation that LDH primarily activates glutamatergic neurons. Although other cell types, representing a smaller fraction in PVN, may contribute to chronic pain, glutamatergic neurons are instrumental in the manifestation of hyperalgesia in LDH.
Growing evidence demonstrates that the PrL modulates pain perception through distinct neural circuits. Specifically, PrL projections to the basolateral amygdala (BLA) are implicated in the affective and anxiety-related components of chronic pain, while PrL–ventrolateral periaqueductal gray (vlPAG) pathways are involved in the regulation of pain hypersensitivity. 57 Both optogenetic and pharmacological studies have shown that activation of the PrL–vlPAG circuit attenuates hyperalgesia, whereas inhibition of the PrL–BLA circuit alleviates pain-related anxiety. Additionally, the PrL projects to the nucleus accumbens (NAc), and strengthening this corticostriatal pathway can reduce both the sensory and aversive dimensions of pain. 58 Collectively, these findings underscore the pivotal role of the PrL in the top-down modulation of pain processing. In our study, we confirmed the direct projection from the PVN to the PrL using viral tracing. Furthermore, through immunofluorescence, in vivo fiber photometry, chemogenetic, and optogenetic approaches, we provided both histological and functional evidence that glutamatergic signaling in the PrL modulates allodynia-like behaviors in the LDH model. As shown in Supplemental Figures 2 and 3, under von Frey stimulation, PrL glutamatergic neurons predominantly receive glutamate, with minimal oxytocin release detected. This suggests that the PVNGlu–PrLGlu circuit is mainly governed by glutamatergic transmission under these conditions. However, our current experiments only measured glutamate and oxytocin, and did not directly assess the involvement of other neurotransmitter systems, such as GABAergic or monoaminergic pathways (including noradrenergic, dopaminergic, and serotonergic systems). Previous studies have indicated that GABAergic and monoaminergic systems are important for prefrontal cortical function and pain regulation.59–62 Therefore, we cannot rule out the potential contribution of these systems in modulating the PVNGlu–PrLGlu circuit. Future investigations employing specific molecular markers, pharmacological interventions, or genetic tools will be necessary to elucidate the roles of these neurotransmitter systems in this pathway.
Utilizing gain- and loss-of-function methods, this study comprehensively demonstrated the crucial role of PVNGlu-PrLGlu circuit in the occurrence and development of chronic pain in LDH rats. The interconnections between distinct brain regions offer novel perspectives for elucidating pathophysiology and therapeutic strategies of chronic pain. Although direct homologs of the PVN-PrL circuit are challenging to identify in humans due to anatomical differences, accumulating clinical and preclinical evidence suggests that neuromodulation targeting prefrontal regions holds significant promise for pain management. For example, repetitive transcranial magnetic stimulation (rTMS) applied to the prefrontal cortex has been shown to markedly reduce pain and associated symptoms in clinical populations, such as patients with failed back surgery syndrome. 63 Similarly, neuromodulation approaches in animal models have demonstrated functional recovery following neural injury, with MRI findings supporting these improvements.64,65 Collectively, these findings highlight the feasibility and therapeutic potential of neuromodulation targeting prefrontal circuits to modulate pain-related neural networks in humans, as supported by both clinical outcomes and neuroimaging data. Therefore, both rTMS and deep brain stimulation (DBS) represent promising strategies for targeting pain-related neural circuits in future clinical applications.
In conclusion, this study reveals that the glutamatergic neural circuit in the PVN-PrL brain region plays a key role in chronic LDH pain. Our work complements the basic research on neural regulation of LDH and provides a new perspective for precision treatment of LDH patients with low back and leg pain by targeting this pathway.
Supplemental Material
sj-pptx-1-mpx-10.1177_17448069251342984 – Supplemental material for Unraveling the PVNGlu-PrLGlu circuit: A new frontier in chronic pain management for lumbar disc herniation
Supplemental material, sj-pptx-1-mpx-10.1177_17448069251342984 for Unraveling the PVNGlu-PrLGlu circuit: A new frontier in chronic pain management for lumbar disc herniation by Qianliang Wang, Jianpeng Chen, Kang Jia, Yujian Peng, Yuanbin Wang, Qianqian Chen and Jun Yan in Molecular Pain
Footnotes
Acknowledgements
We would like to thank all members of our laboratory for their technical assistance and valuable discussions. We also appreciate the support from the animal facility staff at Soochow University. Special thanks to our funding agencies, the National Natural Science Foundation of China (grant nos. 81902239 and 81971036) and the Postdoctoral Fellowship Program of CPSF (grant no. GZC20231896), for their generous support.
Abbreviations
ACC: anterior cingulate cortex
AP: anteroposterior
AUC: averaged area under the curve
BLA: basolateral amygdala
CRH: corticotropin-releasing hormone
DBS: deep brain stimulation
DV: dorsoventral
fMRI: functional magnetic resonance imaging
IL: infralimbic cortex
LDH: lumbar disc herniation
ML: mediolateral
NAc: nucleus accumbens
NP: nucleus pulposus
PAG: periaqueductal gray
PrL: prelimbic cortex
PVN: paraventricular nucleus
PWT: paw withdrawal threshold
rTMS: repetitive transcranial magnetic stimulation
S1: primary sensory cortex
vlPAG: ventrolateral periaqueductal gray
vmPFC: ventromedial prefrontal cortex
Author contributions
All the authors have read and approved the paper.
Data availability statement
Datasets during this current study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the grants from the National Natural Science Foundation of China (grant nos. 81902239 and 81971036) and Postdoctoral Fellowship Program of CPSF (grant nos. GZC20231896).
Ethics approval and consent to participate
All experimental procedures were carried out in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals (NIH Publications No. 8023, revised 1978) and approved by the Animal Ethics Committee of Soochow University (Permission Number: 202302A0104).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
