Abstract
Astrocytes are promising source cells to replace neurons lost to disease owing to a shared lineage and capacities for dedifferentiation and proliferation under pathological conditions. Reprogramming of astrocytes to neurons has been achieved by transcription factor modulation, but reprogramming in vitro or in vivo using small‐molecule drugs may have several advantages for clinical application. For instance, small molecules can be extensively characterized for efficacy, toxicity, and tumorigenicity in vitro; induce rapid initiation and subsequent reversal of transdifferentiation upon withdrawal, and obviate the need for exogenous gene transfection. Here we report a new astrocyte–neuron reprogramming strategy using a combination of small molecules (0.5 mM valproic acid, 1 μM RepSox, 3 μM CHIR99021, 2 μM I‐BET151, 10 μM ISX‐9, and 10 μM forskolin). Treatment with this drug combination gradually reduced expression levels of astroglial marker proteins (glial fibrillary acidic protein and S100), transiently enhanced expression of the neuronal progenitor marker doublecortin, and subsequently elevated expression of the mature neuronal marker NeuN in primary astrocyte cultures. These changes were accompanied by transition to a neuron‐like morphological phenotype and expression of multiple neuronal transcription factors. Further, this drug combination induced astrocyte‐to‐neuron transdifferentiation in a culture model of intracerebral hemorrhage (ICH) and upregulated many transdifferentiation‐associated signaling molecules in ICH model rats. In culture, the drug combination also reduced ICH model‐associated oxidative stress, apoptosis, and pro‐inflammatory cytokine production. Neurons derived from small‐molecule reprogramming of astrocytes in adult Sprague–Dawley rats demonstrated long‐term survival and maintenance of neuronal phenotype. This small‐molecule‐induced astrocyte‐to‐neuron transdifferentiation method may be a promising strategy for neuronal replacement therapy.
1 Introduction
The death of neurons in the mature mammalian central nervous system (CNS), as may occur due to traumatic injuries, metabolic insults, and neurodegenerative diseases, usually results in permanent loss of function because endogenous regeneration capacity is limited. Therefore, restoration of function requires some form of cell replacement therapy, and indeed numerous such treatments have been developed and demonstrated promise in experimental animals and even some clinical cases [1]. A major challenge for widespread clinical application is obtaining sufficient supplies of replacement cells able to survive, differentiate, and integrate into neural circuits for functional restoration. Studies conducted over the past two decades have suggested that implantation of neurons differentiated from neuron stem cells, other neural cell types, or even somatic cells may be a feasible strategy for neuronal regeneration [2, 3].
Previous researches indicated mammalian astrocytes could be converted into neurons in vitro by application of certain small molecules or by transcription factor (TF)‐mediated neuronal reprogramming, and subsequently implanted into the CNS with good survival and phenotypic stability [4 –7]. The TF‐mediated reprogramming of neuronal lineage cells provides one possible strategy for generating functional neurons, but potential risks such as tumorigenesis, immunological rejection, and difficulties delivering differentiated cells into the brain have hindered clinical application [8, 9]. Alternatively, membrane‐permeable small molecules may allow for rapid and controllable reprogramming without introducing exogenous transgenes [10, 11].
Astrocytes are the most abundant cell type in the mammalian CNS and share a common lineage with neurons. Further, astrocytes are capable of proliferating in response to brain damage and degeneration [12 –14]. Therefore, we speculated that astrocytes could be used as the source cells for small‐molecule‐induced reprogramming. The purpose of this study was to examine the efficiency and safety of an astrocyte‐to‐neuron reprogramming method using a combination of small molecules in culture and adult rats under both control conditions and under conditions resembling intracerebral hemorrhage (ICH).
2 Material and methods
2.1 Animals
Newborn Sprague–Dawley (SD) rats for astrocyte culture (Section 2.2) were obtained from the animal center of Tongji Medical College of Huazhong University of Science and Technology. The male SD rats (8‐ to 10‐week‐old) used for the ICH modeling and in vivo treatment experiments (Sections 2.5 and 2.6) were housed in a controlled environment (55% ± 5% humidity, 22 ± 2°C, 12‐h/12‐h dark/light) with free access to food and water except for an 8‐h fast prior to surgery.
2.2 Primary culture of astrocytes
Newborn SD rats (≤ 24 h after birth) were anesthetized by cooling and soaked in ice‐cold 75% alcohol for 3–5 min. The skin and skull were carefully separated, and the cerebral cortex carefully removed. Cortices were cleared of meninges and blood vessels using curved tweezers and ophthalmic tweezers, washed twice in Hank’s balanced salt solution (Gibco, USA), and cut into small pieces. Tissue sections were transferred to 15‐mL centrifuge tubes with 0.125% trypsin‐containing ethylenediamine tetraacetic acid solution (Gibco, USA) and digested for 15 min in a 37℃ water bath with shaking every 5 min. Digestion was terminated by added a 1.5× volume of complete medium containing 10% fetal bovine serum (FBS; Gibco, USA) and 90% DMEM‐F12 (Gibco, USA) culture medium.
Digested tissues were centrifuged for 8 min at 100g, resuspended in DMEM‐F12 containing 20% FBS, 1% glutamine, and 1% streptomycin, filtered through a 100 μm nylon sieve (Corning, USA), and seeded at 37°C under a 5% CO2 atmosphere to be selected for rapidly adherent cells (mainly hematopoietic cells). Non‐adherent cells (including astrocytes and precursors) were transferred to culture flasks and incubated at 37°C under 5% CO2. The purity was then confirmed by immunostaining (Fig. 1)
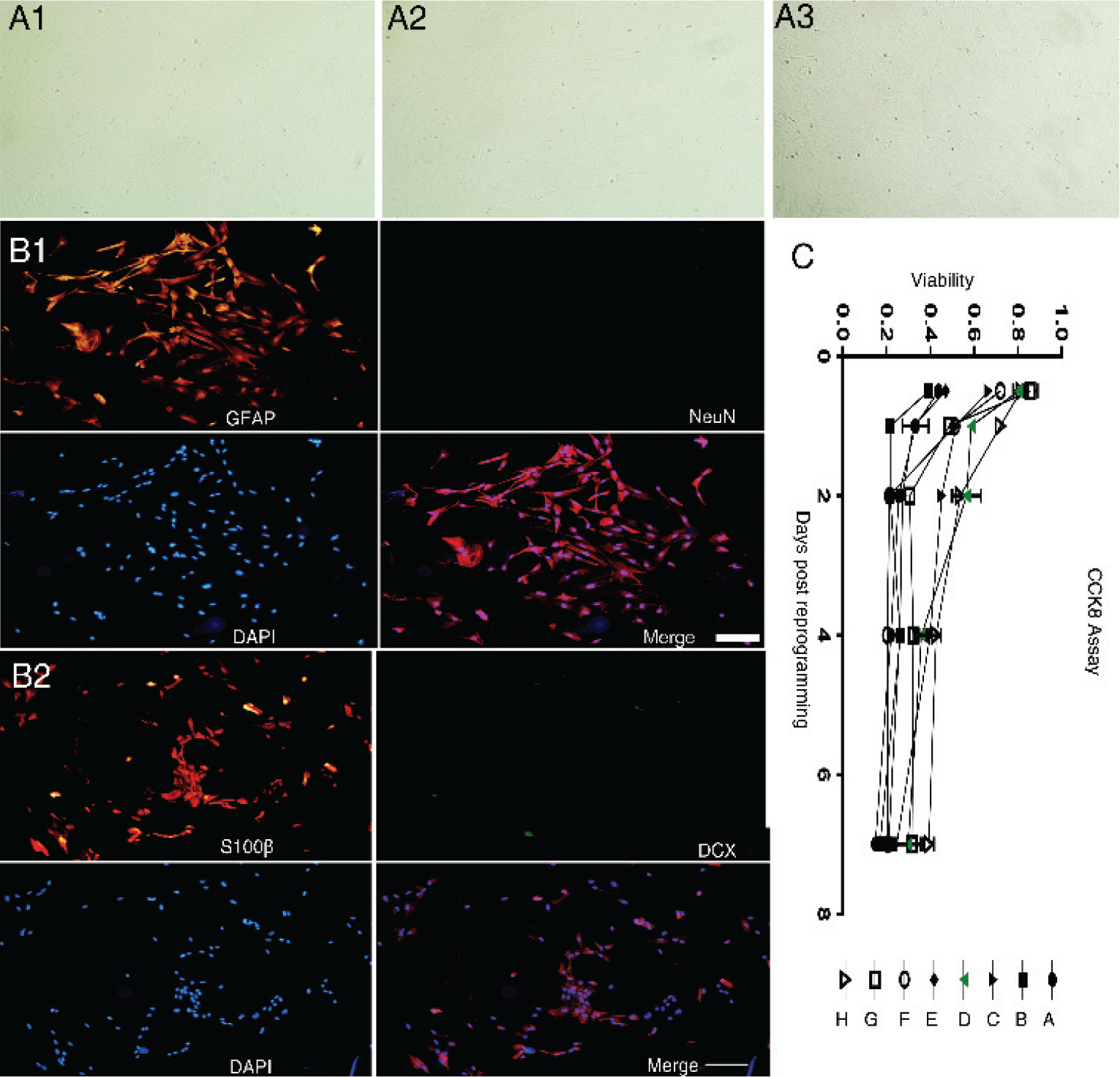
Purity of P3 astrocyte cultures and assessment of transdifferentiation drug toxicity. (A1)–(A3) Morphology of P3 astrocytes (40×). (B1) and (B2) Untreated P3 generation astrocytes were mainly immunopositive for GFAP (red fluorescence) but not for NeuN (bar = 400 μm), as were astrocytes under experimental ICH (40×). (C) Viability of P3 generation astrocytes during treatment with the indicated drug combination (Combination A–H are defined in Table 1) as measured by CCK8 assay.
2.3 Small‐molecular drugs for transdifferentiation test
Valproic acid (VPA; Cat. #HY‐10585), RepSox (Cat. #HY‐13012), CHIR99021 (Cat. #HY‐13012), I‐BET151 (Cat. #HY‐13235), ISX‐9 (Cat. #HY‐12323), and forskolin (Cat. #HY‐15371) were purchased from MedChemExpress (Monmouth Junction, NJ, USA). For cell culture treatment, all compounds were first dissolved in dimethyl sulfoxide (DMSO; Gibco, USA) under sterile conditions and added to complete neurobasal medium. For in vivo treatment, the indicated compounds were dissolved in DMSO without neurobasal medium or neurotrophic factors.
To evaluate the most efficient and safest small‐molecular compounds for astrocyteto‐neuron transformation in cell culture, neuron yield was compared among the drug combinations shown in Table 1, all made in freshly prepared neurobasal medium (Gibco, USA) supplemented with 1 × B27 Plus Supplement (Gibco, USA), 1 × N2 Plus Supplement (Gibco, USA), and 20 ng/mL each of brain‐derived neurotrophic factor (BDNF; Gibco, USA), glial cell line‐derived neurotrophic factor (GDNF; Gibco, USA), insulin‐like growth factor (IGF)‐1 (Gibco, USA), basic fibroblast growth factor (bFGF; Gibco, USA), and epidermal growth factor (EGF; Gibco, USA).
Combinations of small molecules tested for transdifferentiaiton efficacy.
2.4 Intraventricular injection
Male rats (8‐ to 10‐week‐old) treated as indicated were infected with glial fibrillary acidic protein (GFAP):: green fluorescent protein (GFP) lentivirus by encephalic stereotactic injection, followed 48 h later by infusion of the reprogramming drug combination through the same injection tube. Repeated encephalic injections were administered every second day for two months following the protocol of our previous study [15]. Briefly, the microsyringe was slowly advanced into the lateral ventricles, and virus or drug was administered at 0.2 μL/min.
2.5 ICH cell culture model
When astrocytes reached 50%–60% confluence, the medium was exchanged for the fresh medium containing 30 μM chlorinated hemoxyritin (hemin). After 24 h, the medium was again exchanged for fresh medium without hemin.
2.6 Western blot analysis
Rats were euthanized by deep anesthesia and brain tissues surrounding the injection region were isolated for western blotting of proteins associated with phenotype conversion. Western blotting was performed as previously described [15]. Nitrocellulose membranes were incubated at 4°C overnight with the indicated primary antibodies and immunolabeling detected by Dylight™ 800 4XPEG‐conjugated goat antimouse IgG or goat anti‐rabbit IgG (1:12000, Cell Signaling Technology, Danvers, MA, USA). Target bands were captured using an Odyssey Infrared Imaging System (LI‐COR, Lincoln, NE, USA) and density quantified using ImageJ (NIH,
USA).
2.7 Immunofluorescence
Rats were anesthetized by intraperitoneal (i.p.) injection of 1% sodium pentobarbital at 100 mg/kg, and then transcardially perfused with saline containing 4% paraformaldehyde. Brain samples were removed, dehydrated by sequential incubation in 15% sucrose for 24 h and 30% sucrose for 24 h, and then cut into 10 μm‐thick sections using a cryostat. Alternatively, cell cultures were fixed in 4% paraformaldehyde for 30 min. Fixed preparations were then permeabilized by incubation in Triton‐X100 for 30 min and blocked in 5% bovine serum albumin for 30 min with washes in phosphate buffered saline between each step. Fixed brain sections and cultures were then incubated in the indicated primary antibodies at 4°C for at least 12 h, and then stained with the indicated secondary antibodies. Preparations were examined and photographed under a confocal laser scanning microscope (Olympus, Tokyo, Japan). For statistical analysis, three preparations were selected from each rat and three visual fields were randomly selected. Staining intensity was analyzed using ImageJ.
2.8 Astrocyte labeling by lentivirus: GFP infection
The P3 generation of rat primary astrocytes were infected with lentivirus vector expressing GFP and examined by fluorescence microscopy to assess infection efficiency. Infected cells were grown in the containing medium showed in the Session 2.3, and expression levels of the neuronal markers doublecortin (DCX), neuronal nuclei (NeuN), beta III Tubulin (Tuj1), and microtubule‐associated protein 2 (MAP2) were monitored by immunofluorescence staining for 14 days during transdifferentiation. For in vivo labeling, 2–3 μL GFAP:: GFP virus (multiplicity of infection = 50) was stereotaxically injected into the cortex of 8‐ to 10‐week‐old SD rats. After two days, 5 μL of the optimal smallmolecule combination in DMSO was injected in the same location every 2 days. Expression levels of GFP and the neuronal marker NeuN were examined in brain slices obtained 14, 21, and 30 days post‐transdifferentiation (dpt).
2.9 CCK8 assay, qPCR, cell staining and flow cytometry
The cell counting kit‐8 (CCK8) assay was operated with CCK8 kits (Absin, China). Annexin V‐FITC/propidium iodide (PI) staining were operated with kits (Miltenyi, German) bought from Univ. Quantitative polymerase chain reaction (qPCR) was conducted using the qPCR kit (ThermoFisher Scientific, USA). These experiments were performed as previously described [15].
2.10 Statistical analysis
All results are presented as mean ± standard error of the mean (SEM) and statistically analyzed using GraphPad Prism (GraphPad Software Inc., CA, USA). Datasets were first tested for normality using the Shapiro–Wilk method. Expression measures from multiple groups (more than two) were compared by one‐way analysis of variance (ANOVA) followed by Tukey’s post‐hoc comparisons. P < 0.05 was considered a significant difference for all tests.
3 Results
3.1 Characterization of primary astrocyte cultures
In the P3 generation primary cultures, about 98% of cells were immunopositive for the astrocyte markers GFAP and S100B [Fig. 1(A)], while very few cells expressed the neuronal precursor marker DCX or the mature neuron marker NeuN [Fig. 1(B)].
3.2 Identification of the optimal small‐molecule reprogramming system
To identify the best combination of smallmolecule transdifferentiation inducers, a set of candidate drugs was chosen based on relevant published research and extensive preliminary experiments. These studies yielded 6 candidates: VPA, RepSox, CHIR99021, I‐BET151, ISX‐9, and forskolin. We then tested the 8 combinations (Table 1) for transdifferentiation efficiency and toxicity [Fig. 1(C)].
Through preliminary experiments, it was speculated that reprogramming efficiency would be positively associated with the number of different drugs in the mixture as well as their individual concentrations. However, the high initial concentrations impeded the long‐term survival of reprogrammed cells [Fig. 1(C)], and the combination Scheme D (Table 1) was identified as the most fitting scheme that could induce marked changes in cellular morphology and expression phenotype after 12 h of treatment. With prolonged treatment, cell bodies became smaller and closer together, and grew more complex branches that tended to be stable once formed. This optimal combination consisted of VPA 0.5 mM, forskolin 10 μM, I‐BET151 2 μM, ISX‐9 10 μM, RepSox 1 μM, and CHIR99021 3 μM. In contrast, no relevant morphological changes were observed in control cultures treated with vehicle (DMSO).
3.3 Adult rats’ astrocytes acquired neuronal expression properties during drug treatment in vitro
Immunostaining results revealed expression of the neuronal precursor cell marker DCX by 3 dpt. Expression reached a peak on 7 dpt and then gradually disappeared. Further, immunostaining for the mature neuron marker NeuN emerged by 12 dpt, and peaked on 18 dpt before gradually disappearing. In contrast, immunoexpression of the astrocyte marker GFAP was high initially and reach a peak on 3 dpt before gradually disappearing. The neural stem cell marker nestin was slightly detectable only around 14 dpt [Figs. 2(A)–(C) and (E)]. Collectively, these results indicated that cultured astrocytes attained an immature neuronal phenotype after 7 days and a mature neuronal phenotype after 14 days of drug treatment. We further traced the phenotype changes of astrocytes by infection with a lentivirus expressing GFP driven by the rat GFAP promoter (GFAP:: GFP) [Fig. 2(D)]. Results again suggested that GFAP‐positive astrocytes can be reprogrammed into neurons (expressing neuronal protein markers) by this smallmolecule induction system.
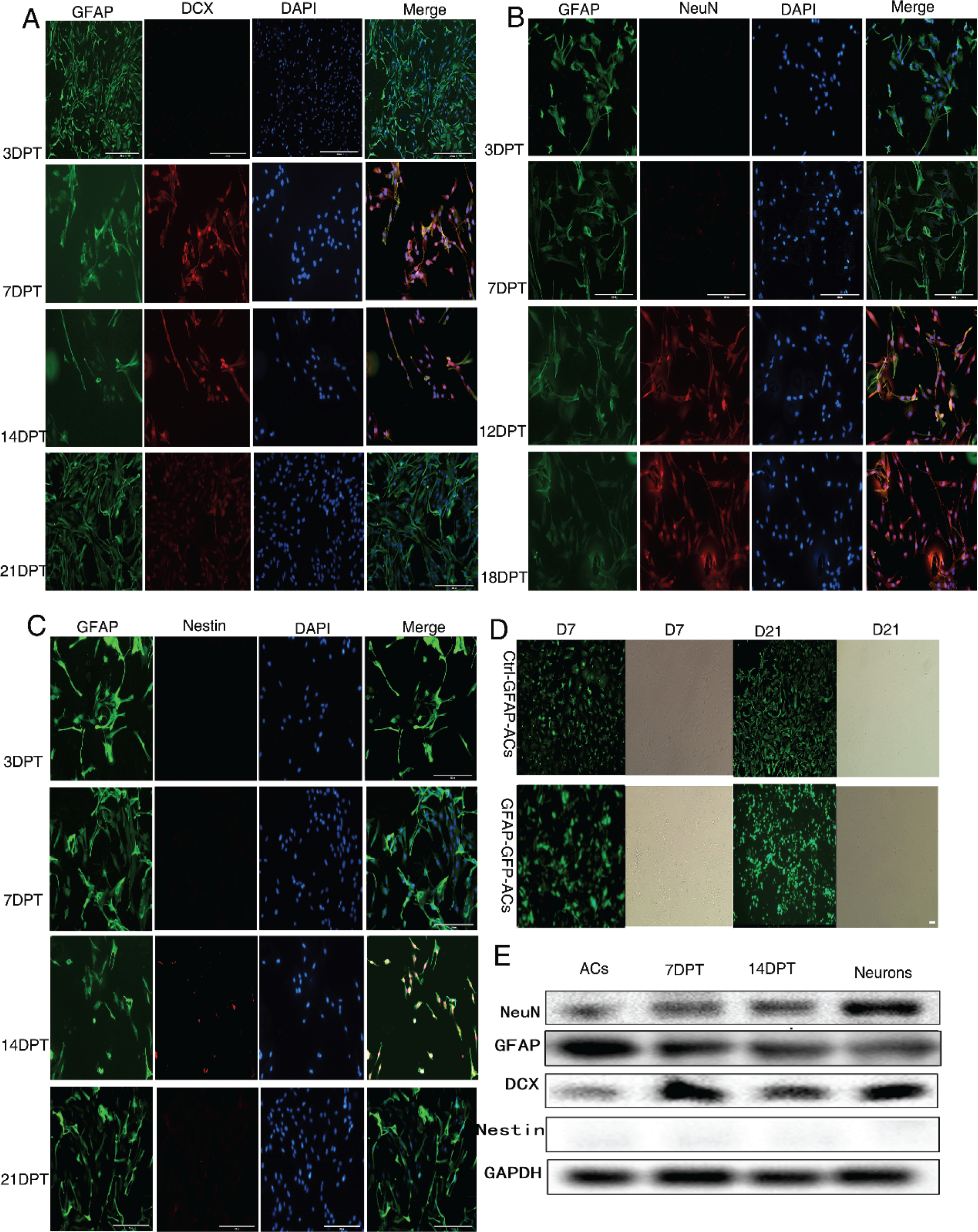
Changes in cell phenotype during treatment of astrocyte cultures with the optimal small‐molecule drug combination. (A)–(C) Changes in the expression levels of the astrocyte marker GFAP, neuronal precursor marker DCX, and mature neuron marker NeuN at 3, 7, 14, and 21 dpt. Bar = 200 μm). (D) Changes in GFP expression driven by the GFAP promoter following GFAP:: GFP lentivirus infection. Bar = 200 μm. (E) Sample western blot to identify protein expression. ACs, the new‐born rat astrocytes cultured in conventional astrocytes culture medium in the period of day 7 to day 14; Neurons, the new‐born rat neurons cultured in conventional neurons culture medium in the period of day 7 to day 14.
3.4 Induction of astrocyte‐to‐neuron transdifferentiaiton by the small‐molecule reprogramming system under in vitro ICH conditions
Cultured astrocytes under the ICH model conditions presented with typical signs of injury and impairment [Fig. 3(A)]. Under light microscopy, ICH model astrocytes showed enhanced light refraction indicative of edema and aberrant morphological changes. Cultures also contained a few floating and dying cells. Immunofluorescence further demonstrated cell body shrinkage, a reduced number of branches, and longer retained branches. However, cells still stably expressed the astrocyte marker proteins GFAP and S100B [Fig. 3(B)]. Further, the small‐molecule reprogramming system still induced astrocyte‐to‐neuron transdifferentiation under ICH model conditions. Briefly, P3 generation astrocytes treated with 30 μM hemin for 24 h to simulate activation still demonstrated an increase in DCX immunostaining by 7 dpt, followed by gradually increasing NeuN expression and a concomitant decrease in GFAP expression as evidenced by immunostaining [Figs. 3(C)–(E)]. In contrast, no nestin expression was detectable at any time. Similar changes in the expression levels of these marker proteins were also observed in cultures under ICH model conditions as evidenced by western blotting [Fig. 3(F)], suggesting potential applicability for neuron replacement therapy following ICH.
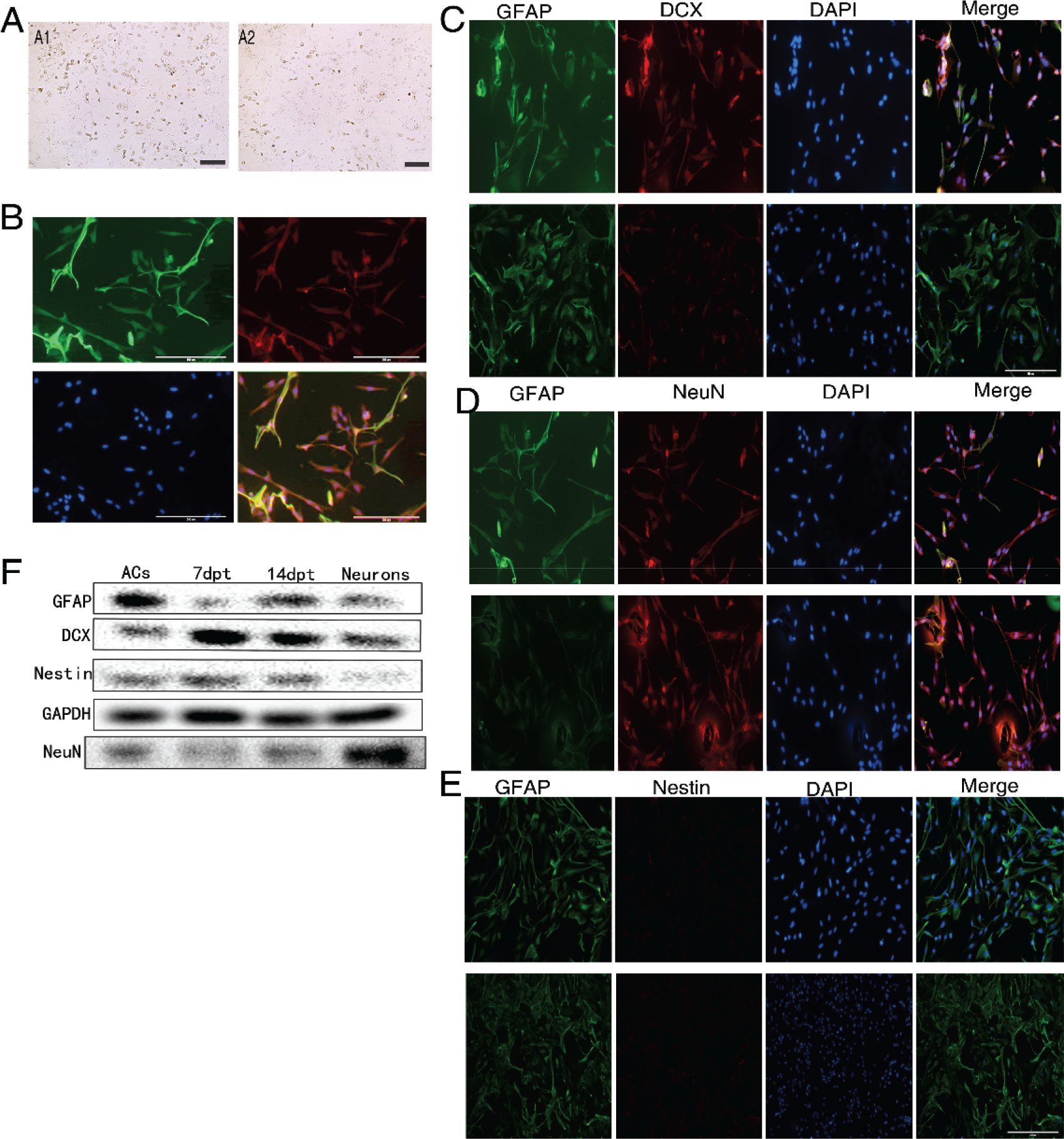
The same small‐molecule combination also induced astrocyte‐to‐neuron transdifferentiation under experiment intracerebral hemorrhage conditions. (A) and (B) Cultured astrocytes under the ICH model conditions presented with typical signs of injury and impairment under light microscopy and fluorescent microscopy. (C)–(E) Changes in cell‐specific marker protein expression revealed by immunofluorescence (100×). (F) Changes in marker protein expression as measured by Western blot.
3.5 The small‐molecule transdifferentiation inducers reduced inflammation, oxidative stress, and cell death in the ICH culture model
As neuroinflammation contributes to neuropathology and limits the survival of transplanted cells, we next examined inflammatory responses and apoptosis of induced cells in ICH model cultures [Figs. 4(A)–(C)]. Treatment with the drug combination reduced reactive oxygen species production (P < 0.0001), lactate dehydrogenase release (from dead cells) (P < 0.01), and the mRNA expression levels of pro‐inflammatory factors interleukin (IL)‐1β, IL‐6, and tumor necrosis factor (TNF)‐α (all P < 0.05) under model ICH conditions, again supporting potential therapeutic efficacy.
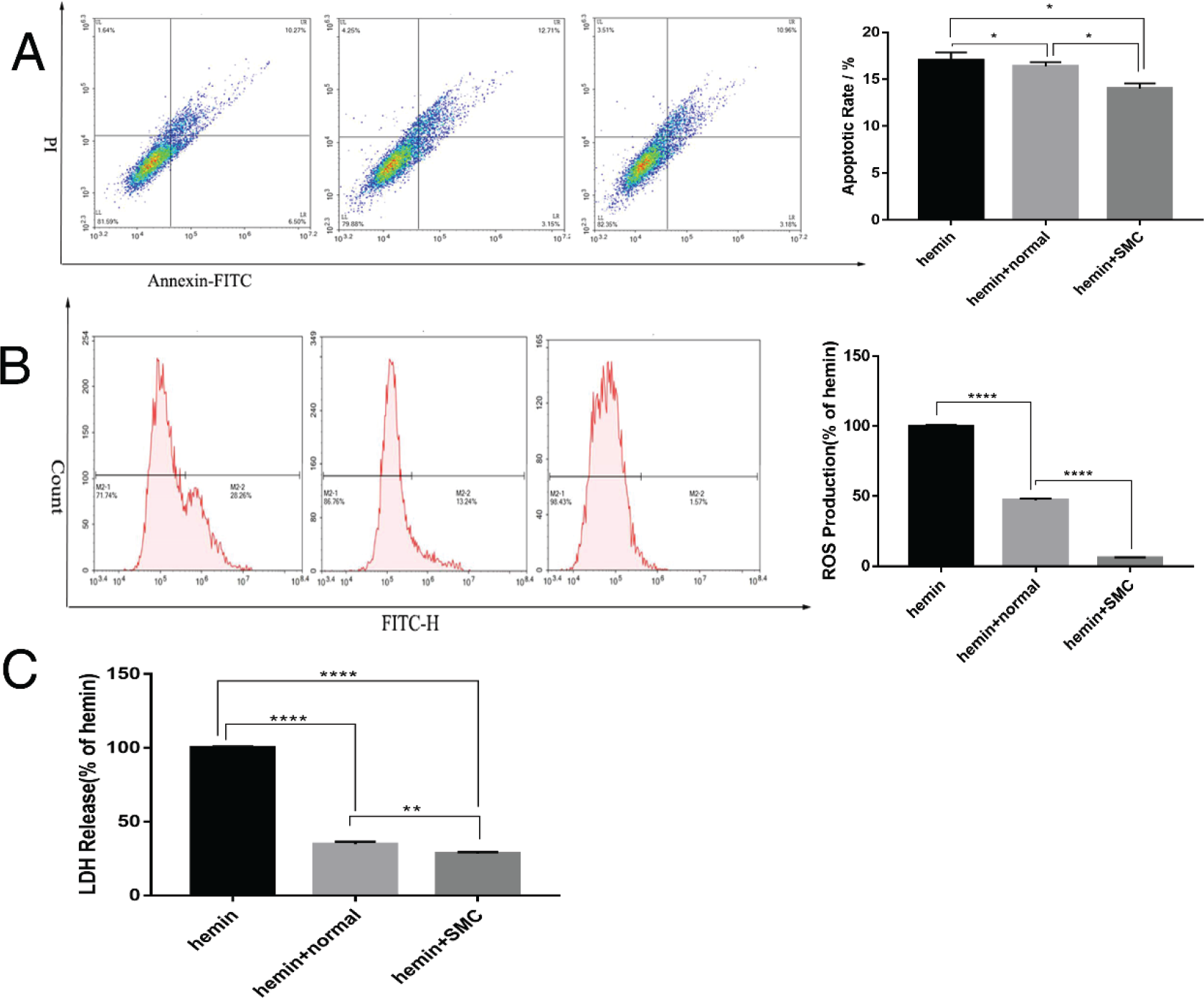
The small‐molecule transdifferentiation compounds reduced inflammation, oxidative stress, and apoptosis of cultured astrocytes under model ICH conditions. (A) Apoptosis rate as measured by Annexin V‐FITC/PI staining and flow cytometry. (B) Oxidative stress as measured by reactive oxygen species accumulation. (C) Lactate dehydrogenase release from dying cells.
3.6 Survival and maturation of induced neurons in vivo
We first confirmed that the small‐molecule combination could also induce transdifferentiation in cultures infected with GFAP:: GFP lentivirus [Fig. 2(D)], then used this method to assess the long‐term changes in cell morphology in vivo. During drug treatment, cultured astrocytes exhibited a gradual reduction in cell body size, and overall morphology changed to resemble neurons. Further, GFAP‐positive cells showed DCX expression at 14 dpt and expression of the mature neuron markers Tuj1, MAP2, and NeuN at 21 dpt [Fig. 5(A)], while no NeuN expression was observed in vivo at 30 dpt [Fig. 5(B)].
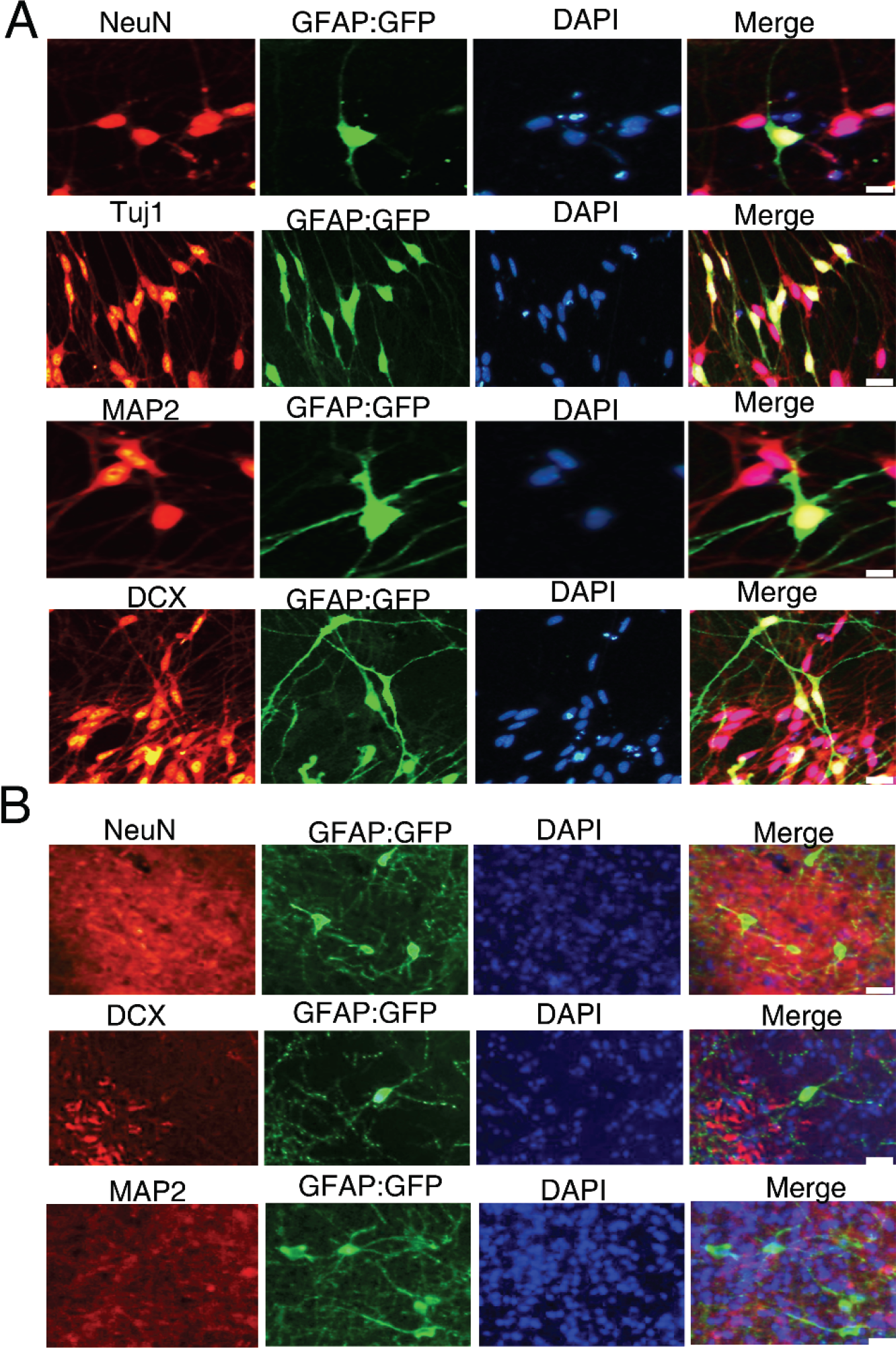
Immunofluorescence staining of astrocyte, neuronal precursor, and neuron markers in astrocytes infected by GFAP:: GFP lentivirus in vitro (bar = 20 μm; A) and in vivo (bar = 50 μm; B). All staining was conducted at 30 dpt.
3.7 Genome‐wide transcriptome analysis of astrocyte‐derived neurons revealed upregulation of multiple reprogramming transcription factors
To examine molecular mechanisms underlying astrocyte‐to‐neuron transdifferentiation, we conducted transcriptome analysis of treated and untreated cultures at 7 and 14 dpt by qPCR. We focused on the basic helix‐loop‐helix (bHLH) family of TF implicated in neuronal differentiation [Figs. 6(A) and (C)] as well as Notch signaling pathway members known to regulate expression of proneural TF genes [Figs. 6(B) and (D)].
We found that mRNA expression levels of the proneural genes NeuroD1, NeuroG2, Sox2, Pax6, and Dlx2 were upregulated significantly during the early stage of transdifferentiation (7 dpt), while the mRNA levels of NeuroD1, NeuroG2, Pax6, and Dlx2 were still significantly upregulated during the intermediate phase (14 dpt) [Fig. 6(A)]. Further, expression levels were also upregulated under ICH conditions [Fig. 6(C)]. Multiple Notch receptor and ligand genes were also significantly upregulated during transdifferentiation (P < 0.05) [Fig. 6(B)], consistent with studies showing that the Notch signaling pathway is critical for neuronal differentiation and nervous system development. However, studies ablating or activating single receptor or ligand genes have yielded disparate results, and there is also uncertainty regarding the downstream targets of Notch signaling during astrocyte activation. The expression levels of Notch signaling pathway receptors and ligands were also markedly upregulated under ICH [Fig. 6(D)].
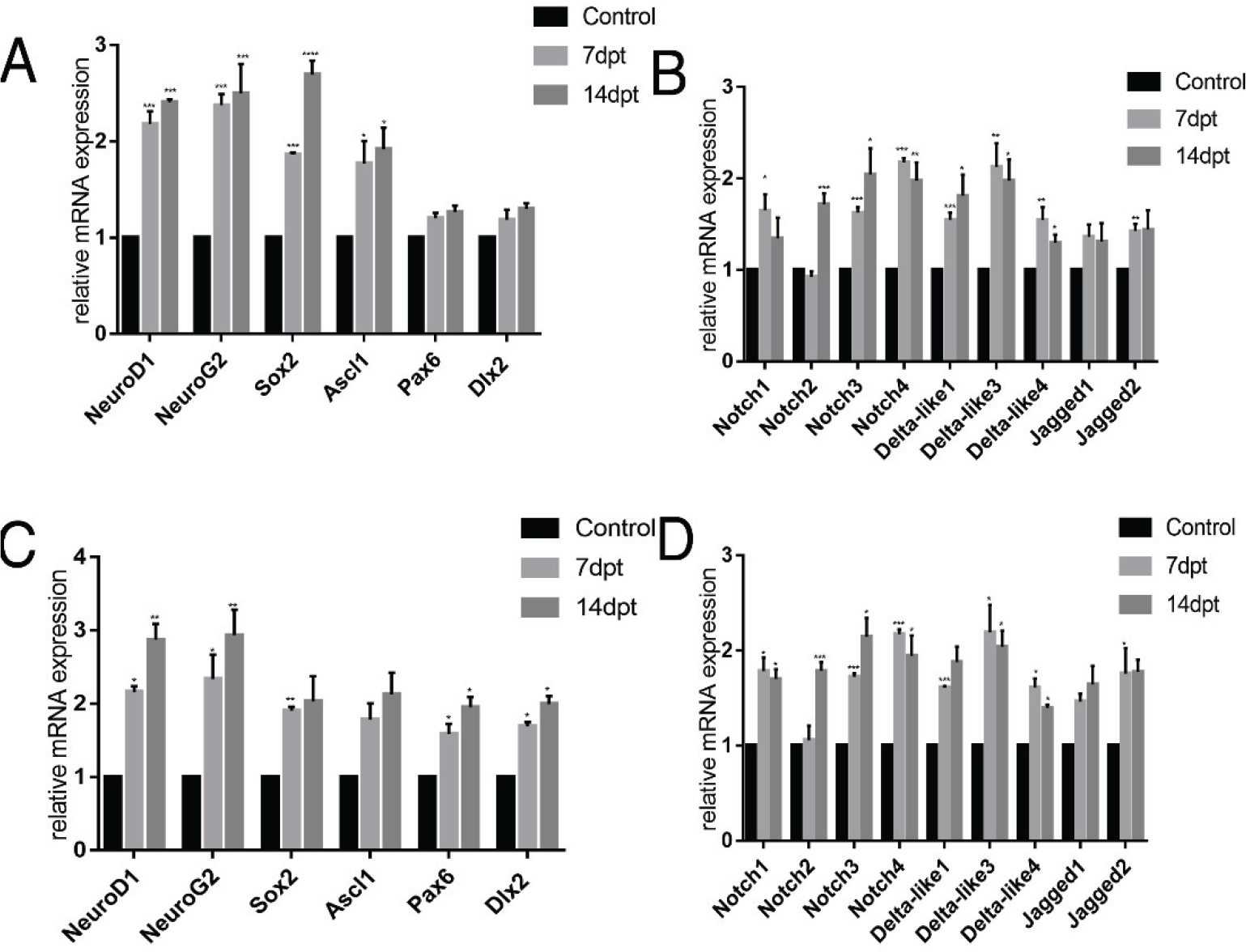
Changes in the relative mRNA expression levels of reprogramming‐associated genes during drug treatment under control (A, B) and ICH (C, D) conditions. (A) mRNA expression levels of key reprogramming‐related genes in the absence of ICH. (B) mRNA expression of Notch signaling pathway members in the absence of ICH. (C) mRNA expression levels of key reprogramming‐related genes under ICH. (D) mRNA expression levels of Notch signaling pathway members under ICH.
4 Discussion
Neural reprogramming using small molecules is a promising strategy for neuron replacement therapy with several distinct advantages compared to gene manipulation or exogenous gene transfection, including greater temporal control of activity and potentially better biosafety [1−3]. However, the molecular mechanisms of chemical reprogramming are still not well described, so the optimal drug targets are generally unclear [1]. Nonetheless, several small molecules with neural reprogramming potential have been identified [1, 12], although none has been thoroughly evaluated for clinical efficacy and safety [9, 11]. In this study, we have developed a drug combination showing a good safety profile and efficacy for inducing astrocyte‐to‐neuron transdifferentiation as evidenced by morphological transformation, altered expression of cell‐specific markers, and upregulation of multiple bHLH family TFs and upstream Notch pathway receptors and ligands. Moreover, this chemical reprogramming still occurred under pathological conditions associated with astroglial activation, and treatment actually improved the viability of source astrocytes by reducing oxidative stress and inflammatory signaling. These findings support the potential future clinical applications of this drug regimen for neuronal replacement in the acute phase following ICH. Further study is required to elucidate the molecular pathways involved, which may in turn provide guidance for improvement of the drug combination.
Previous studies examining small molecules for neural reprogramming in vivo have generally used a two‐step approach involving chemical treatment in vitro, followed by transplantation to assess if the treated cells can survive and restore neuronal function without adverse effects [2, 12]. This study is significant in that we examined the effect of direct drug injection. However, not all in vitro results were replicated in vivo. For instance, injection did not induce expression of neuron marker proteins in astrocytes within 30 days. However, there may be many factors in the brain microenvironment that serve to maintain cell phenotype [16], influences that must be overcome for efficient reprogramming. In addition, the efficiency of astrocyte GFAP:: GFP lentivirus infection in the resting state is not ideal, so additional experiments will focus on improving infection efficiency in vivo for more precise cell tracking. We also need to reduce the influence of injection‐induced changes in intracranial pressure. Subsequent experiments should examine other disease models, such as the needle‐stick injury model as well as various cerebrovascular disease and neurodegenerative disease models [17]. Further, the observation time for induced differentiation may still be too short, so longer observation periods, possibly using additional neuronal markers and electrophysiological methods are needed to examine if induced neurons can survive and integrate into functional circuits.
Despite the above limitation, the current study demonstrates that a specific combination of small molecules can induce astrocyte‐toneuron transdifferentiation under physiological and pathological conditions. Further improvements in the drug combination, individual concentrations, solvents, and delivery methods may allow for clinical application in the near future.
Footnotes
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. The experimental protocol was approved by the Ethics Committee of Huazhong University of Science and Technology and performed according to the guide for the care and use of laboratory animals.
Conflict of interests
The authors declare that they have no conflict of interests.
Funding
This study was funded by the National Natural Science Foundation of China (Grant Nos. 82071330, 81873750), the Science and Technology Plan Project of Wuhan, Hubei Province, China (Grant No. 2018060401011316) and the Natural Science Foundation of Hubei Province, China (Grant No. 2019CFB113).
Author contributions
Zhouping Tang and Na Liu designed the study. Yangyang Feng performed most of the experiments and wrote the manuscript. Hao Nie and Gaigai Li established the animal model. Shiling Chen performed the western blot. Chao Pan performed the immunofluorescent staining. Ping Zhang and Yingxin Tang analyzed the data. All the authors read and approved the final manuscript.
