Abstract
For decades, intraoperative neurophysiological monitoring (IONM) has been used to guide selective dorsal rhizotomy (SDR) for the treatment of spastic cerebral palsy (CP). Electromyography (EMG) interpretation methods, which are the core of IONM, have never been fully discussed and addressed, and their importance and necessity in SDR have been questioned for years. However, outcomes of CP patients who have undergone IONM-guided SDR have been favorable, and surgery-related complications are extremely minimal. In this paper, we review the history of evolving EMG interpretation methods as well as their neuroelectrophysiological basis.
Keywords
1 Introduction
Selective dorsal rhizotomy (SDR) is an operation in the field of neurosurgery that can effectively reduce spasticity in patients with spastic cerebral palsy (CP) while ensuring the muscle strength and sensory function of the lower extremities [1]. The word “selective” refers to the intraoperative selection and dissection of the afferent nerve rootlets in the spinal cord that are considered to be more involved in spasticity, decreasing the afferent signaling to the motor neurons in the anterior horn of the spinal cord, so as to achieve a reduction in muscle tone after surgery. At present, some centers that perform SDR use intraoperative neurophysiological monitoring (IONM) to assist or even fully guide the operation [2 –13]. Because the role of IONM in SDR has never been fully discussed, its importance and necessity have been long questioned. In the current review, we address those concerns by reviewing the history of IONM and evaluating the evolving electromyography (EMG) interpretation methods as well as their neuroelectrophysiological basis.
2 History of IONM-guided SDR
The selection of dorsal nerve roots (rootlets) in SDR was initially based on the human dermatome. Surgeons were struggling to determine the balance between over- and under-sectioning of afferent spinal nerve fibers. In 1960, Gros et al. introduced intraoperative electrical stimulation into SDR [14]. His initial purpose was mainly to help properly differentiate dorsal roots from ventral ones to avoid motor function damage in these cases. In 1976, Fasano et al. first developed clinically applicable dorsal rootlet selection criteria via interpreting EMG responses using IONM [1]. He classified all EMG responses induced by dorsal rootlet train stimulation as either “normal” or “abnormal” on the basis of Magdelary’s achievement in 1952 and Decandia’s animal experiments in 1967 [15]. By identifying the nerve roots (rootlets) that elicit abnormal EMG responses, it is relatively easier for surgeons to make the decision to selectively cut those rootlets that are considered to be more closely associated with lower limb spasticity (Fig. 1). Fasano’s SDR protocol provided a practical method to achieve the goal of “to maximize the improvement of motor function with minimized side effects”, although initial dorsal root selection in his practice was still based on the dermatome, and such a procedure required a multilevel approach with the opening from L2 to S2. Because the repeatability of the EMG discharge pattern evoked by train stimulation is not very stable during surgery, Fasano’s rhizotomy scheme is difficult to apply widely. Nevertheless, Fasano’s EMG response interpretation criteria were the first attempt to achieve rhizotomy balance in the SDR procedure, and this method as well as other protocols derived from it were practiced often in many medical centers during that time period.
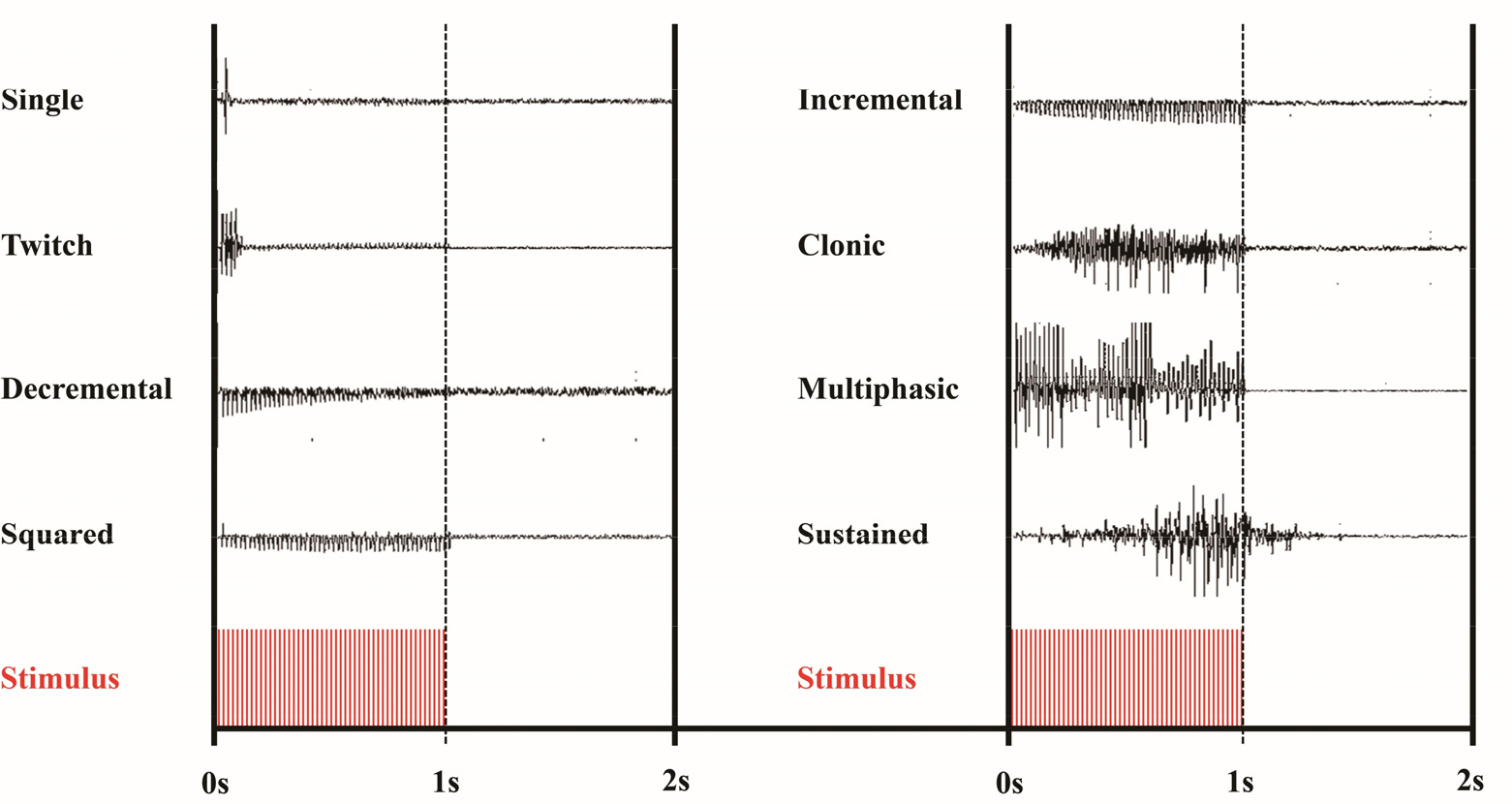
Patterns of EMG discharges from nerve rootlets when a 50 Hz train stimulus is given for 1 s (pulse duration 0.2 ms). Four patterns in the right column are considered to be abnormal.
In 1989, Philips and Park proposed a novel grading system for EMG responses [2]. They refined and classified EMG responses evoked by electrical stimulation (train stimulation) into five grades (the EMG response grading system), which were adopted by many SDR centers globally and have been used until the present (Table 1). Park’s method put more focus on the EMG spreading pattern instead of the EMG discharge pattern, making its repeatability more reliable. In addition, such EMG interpretation could guide the selection of dorsal roots (rootlets) that are thought to be more closely associated with spasticity just beneath the conus level, which means the selection could be fully achieved without the need to identify the level of those nerve roots (rootlets). The use of Park’s method could allow SDR to be performed via a single-level approach, particularly in cases with moderate or severe spasticity [2 –8].
EMG response grading system.

In an attempt to find a universally applicable EMG interpretation scheme that could be applied to all kinds of spastic CP cases (including mild ones), Browd developed a new rhizotomy protocol in 2016 [10]. In addition to applying the EMG response grading system, Browd’s rhizotomy scheme also took preoperative assessment results (identifying the “target muscle” in CP cases before SDR) into account when EMG responses were interpreted during surgery [9, 10]. This protocol was the first to provide the potential to decrease muscle tone in a particular muscle group in affected lower limbs via a single-level laminectomy approach. Because this approach requires a somewhat sophisticated interpretation method, our team in 2019 proposed a simplified EMG interpretation scheme that focuses solely on the particular spastic muscles in the lower extremities of CP cases, by which SDR via a guided single-level approach could be applied to all kinds of spastic CP cases with fewer rootlets being cut. We have achieved favorable outcomes using SDR guided by this simplified rhizotomy protocol (Fig. 2) [11 –13].
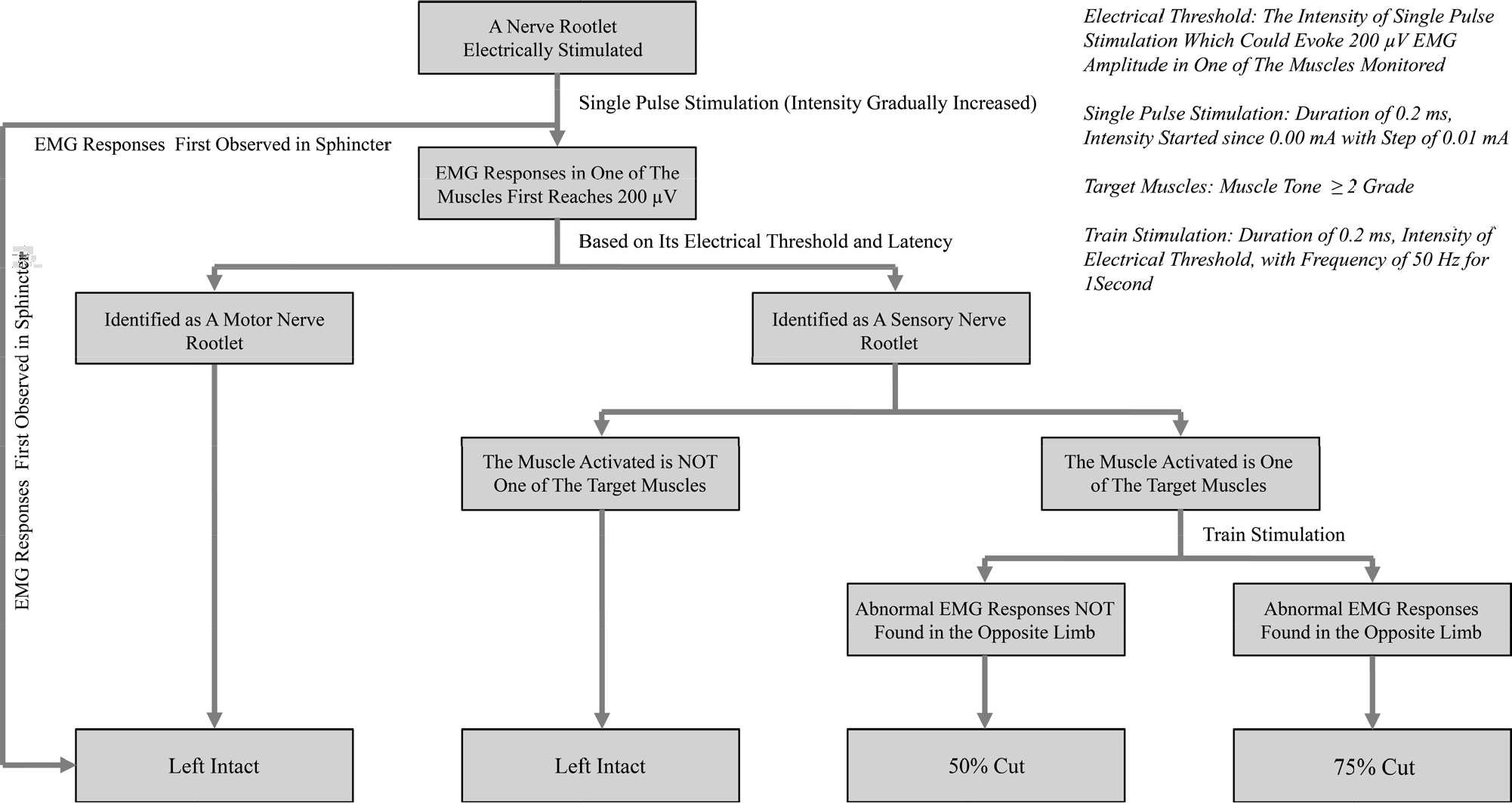
3 Setup of IONM-guided SDR
3.1 Anesthesia
As physicians gained practice through more IONM-guided SDR procedures, they became gradually aware of the importance of anesthesia during surgery when EMG responses are required to be interpreted in a standardized manner. Studies have shown that different anesthetics can affect intraoperative electrophysiological outcomes [16 –18], including changes in threshold, latency to stimulus, and EMG response patterns. Because, to date, there is no proven anesthetic protocol used specifically for SDR, clinical practitioners have either just applied those protocols conducted in other neurosurgical procedures requiring IONM or simply developed their own [2 –13].
3.2 EMG recordings
With the development of IONM technology, the scope of muscles monitored during SDR has gradually expanded from 4 muscle groups to 23 muscle groups (including the anal sphincter) [19]. Over the past 20 years, needle electrodes have been used for recording purposes, whereas bipolar probes are used as stimulation electrodes in almost all medical centers that perform IONM-guided SDR [2 –4]. To avoid recording noise, rootlet stimulation must be elevated in the air using stimulation probes without tension, so as to keep the rootlet away from the cerebrospinal fluid [10].
3.3 Parameters of electrical stimulus
Since Fasano’s setup, intraoperative electrical stimulation has typically included both singlepulse stimulation and train stimulation. Singlepulse stimulation, which contains a constant current with a duration that varies from 0.05 to 0.5 ms and an intensity starting at 0.01 mA [2 –13], is used mainly (1) to differentiate dorsal roots from ventral ones, (2) to identify those rootlets associated with the sphincter, and (3) to determine the electrical intensity for the following train stimulation. In both Browd’s and our team’s setups, single-pulse stimulation was also used to determine whether such rootlet stimulation initially evokes EMG responses in those target muscles. Based on Browd’s protocol, the intensity of a single-pulse stimulation that can elicit EMG responses reaching 20 µV (threshold) in any monitored muscle would be used in the following train stimulation [9, 10]. In our institute, we use an EMG response of 200 µV as the threshold to guide train stimulation. We considered elevating the amplitude in Browd’s protocol by 10 times in our practice because the amplitude range of the EMG signal under physiological conditions is mainly between 50 µV and 4 mV.
The purpose of performing train stimulation in SDR is to determine whether the dorsal rootlet stimulated has matched rhizotomy criteria [1 –13]. At present, most centers performing SDR set the train stimulation to a frequency of 50 Hz for 1 s. The reasons for setting this frequency are the following: (1) electrically stimulating a dorsal nerve root at a rate greater than 10 Hz usually leads to the gradual attenuation of EMG responses, and (2) when the frequency of train stimulation is between 30 and 50 Hz, motor neurons in the spinal cord respond only to the first stimulus [2].
4 EMG interpretation
Rootlet selection in the SDR procedure relies on EMG interpretation. The lack of full understanding of neuroelectrophysiological characteristics in the spinal cord in patients with spastic CP makes EMG interpretation difficult and prevents its optimization. Nevertheless, in addition to having the capability of avoiding damage to motor and urinary (bowel) function caused by mistransection, IONM can reveal the variance in neuroelectrophysiological characteristics among those dorsal roots (rootlets) being stimulated, therefore providing surgeons a reference to use in their decision making. In clinical practice, physicians have proposed a series of EMG interpretation methods to guide SDR, with the goal of “to maximize the improvement of motor function with minimized side effects”.
In reviewing the EMG interpretation methods proposed over the past approximately 40 years, we could roughly group those methods into three major categories mainly on the basis of their EMG interpretation patterns: EMG discharge pattern evoked by Spreading pattern of EMG responses evoked by Initial EMG appearance (reaching a certain threshold) elicited by
The method first proposed and practiced by Fasano belonged to Category 1, which simply divided EMG responses into normal and abnormal [1]. His EMG interpretation criteria helped him to decide which rootlet(s) at a certain level needed to be cut. However, in his setup, the dorsal root level that needed be tested was still determined using the reference of the human dermatome. Because of the poor repeatability of EMG discharges, Fasano’s EMG interpretation method as well as those protocols derived from it were difficult to standardize with regard to the intraoperative rootlet selection among centers [1]. Furthermore, normal EMG discharge patterns were hardly observed during the SDR procedure, rendering the selection made using this protocol not that meaningful [2].
In 1989, Philips and Park proposed a novel EMG interpretation scheme that mainly focused the attention on the spreading pattern of the EMG responses instead of the EMG discharge pattern in a particular muscle [2]. They divided EMG responses evoked by train stimulation to dorsal rootlets into five grades (EMG response grading system) and solely cut those in which EMG responses reached 3+ or 4+ (considered to be associated more closely with spasticity). The hypothesis underlying such an EMG interpretation is that the wider the evoked EMG responses spread, the more electrophysiologically abnormal that dorsal rootlet was likely to be (Category 2). The spreading pattern of EMG responses evoked by train stimulation has a more reliable repeatability. In addition, based on Park’s EMG interpretation scheme, the dorsal rootlet rhizotomy ratio was positively correlated with the severity of those CP cases; therefore, for the first time, such a protocol could provide SDR the potential to be performed just beneath the L1 level via a single-level approach, without the need to use the human dermatome as a basic reference [2 –8]. SDR guided by Park’s EMG response interpretation scheme has been practiced for more than 20 years and performed in thousands of cases. The long-term outcome in CP patients who have undergone this type of SDR is quite favorable [7, 8]. With the increased use of the EMG response grading system in practice in SDR, clinicians have gradually revealed that it is hard to select dorsal rootlets for cutting based solely on Park’s rhizotomy scheme, particularly in cases with preoperative Gross Motor Function Classification System (GMFCS) level 1 or 2, because in up to 20% of these cases, none of the dorsal rootlets tested in surgery match the rhizotomy criteria [11].
In 2016, Browd formulated new rhizotomy criteria for SDR [9, 10]. In addition to using the EMG response grading system to achieve rootlet selection, he introduced preoperative evaluation data into his rhizotomy protocol. His rhizotomy scheme required identification of target muscles which were defined as any major muscle groups with their muscle tone ≥ grade 2 (Modified Ashworth Scale) in the lower limbs of CP cases. In his SDR surgery, he also sectioned those afferent rootlets in which single-pulse stimulation could first evoke EMG spikes reaching a certain threshold (20 µV) in those target muscles, and he disregarded the EMG results from the subsequent train stimulation (combination of Categories 2 and 3). The hypothesis of his EMG interpretation mainly consisted of two parts: (1) any afferent rootlets that could evoke vast spreading of EMG responses (train stimulation) would be taken as abnormal (like Park’s), and (2) afferent rootlets that could first elicit EMG spikes in target muscles when a short monophasic electric pulse is given would be considered to have more weight in the spasticity of that muscle in the physiological condition. For the first time, Browd’s protocol-guided SDR provided the potential to mainly release muscle tone in a particular muscle group in the lower extremities of spastic CP patients in a quantitative manner, and also potentially allowed the application of this method to mild cases, and it achieved good outcomes. The price of this method-guided SDR is that more rootlets are cut than in Park’s selection method-guided SDR [11].
In 2019, our team proposed a simplified rhizotomy scheme based on Browd’s protocol, which places the focus on only those afferent rootlets mainly associated with muscle groups that have muscle tone of grade ≥ 2 (target muscle) in the patient’s lower limbs (Category 3) [11 –13]. This method completely excludes dorsal nerve fibers that evoke spreading EMG responses (train stimulation) but that elicit EMG spikes during single-pulse stimulation first revealed in nontarget muscles, which are cut when the EMG response grading system is applied to guide SDR (grade of 3+ or 4+). The main advantages of this simplified SDR guided scheme are that it can reduce the number of rootlets being transected and also be applied in mild CP cases.
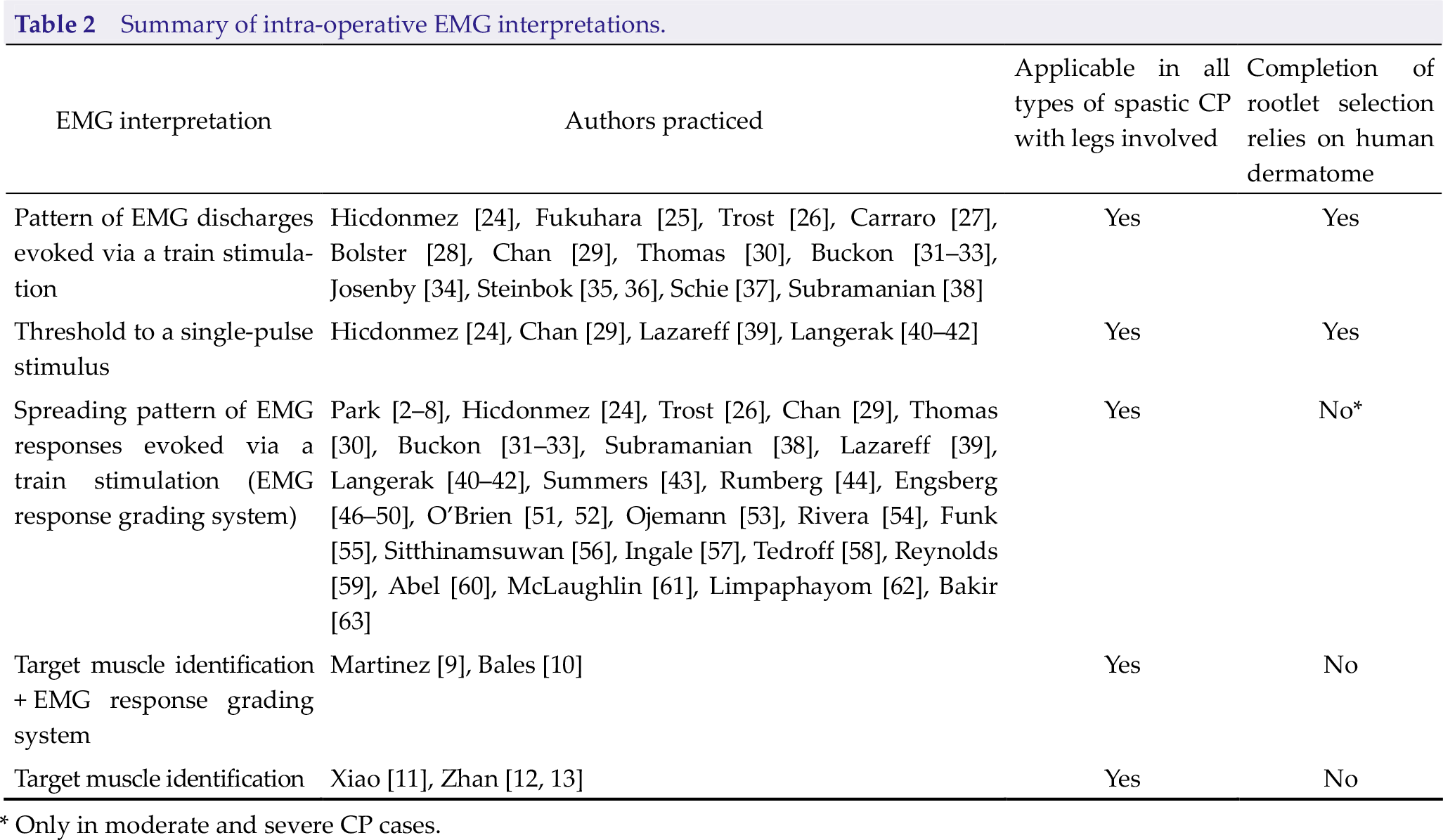
The main intraoperative EMG interpretations are summarized in Table 2, and a comparison of EMG interpretation methods is shown in Fig. 3. The differences in rootlet selection among the rhizotomy schemes proposed by Park, Browd, and our team are depicted in Fig. 4.
Summary of intra-operative EMG interpretations.
* Only in moderate and severe CP cases.
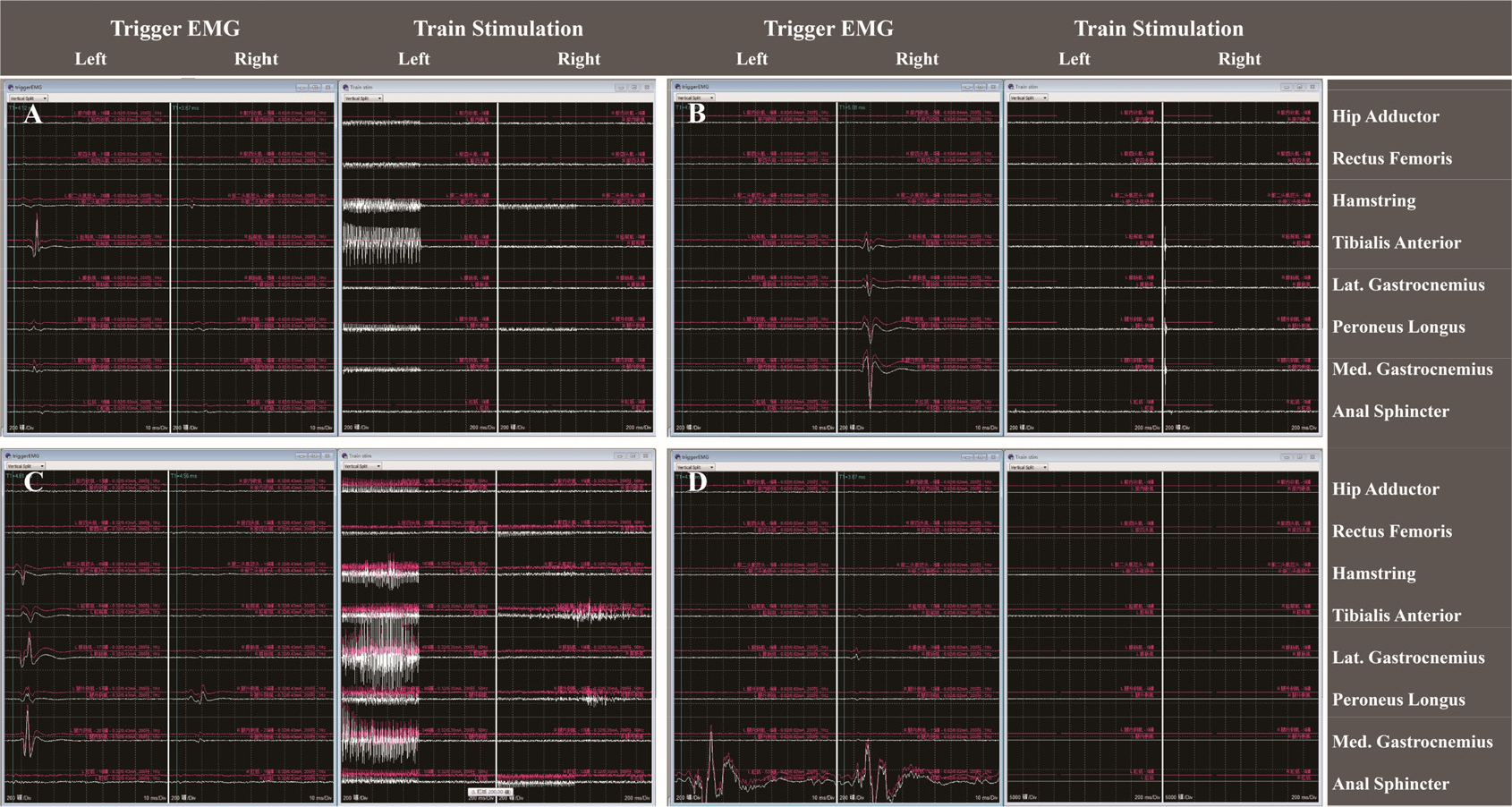
Example of differences in EMG response interpretation (obtained from four different cases). (A) EMG responses reaching the rhizotomy criteria of Park and Browd. EMG responses evoked by single-pulse stimulation were observed mainly in the left (Lt.) tibialis anterior, which was not the target muscle identified using the method of Browd and our team. The EMG responses elicited by train stimulation were shown in the Lt. adductor, Lt. quadriceps, Lt. hamstring, Lt. tibialis anterior, Lt. gastrocnemius, and Lt. peroneus longus, which reached an EMG response grade of 3+. (B) EMG responses reaching the rhizotomy criteria of Browd and our team. Single-pulse stimulation innervated EMG responses in the right (Rt.) medial gastrocnemius, which was identified as the target muscle during the preoperative assessment. Train stimulation evoked limited activities on the Rt. side, which reached grade 2+ of the EMG response grading system. (C) EMG responses matching the rhizotomy criteria of Park, Browd, and our team. The EMG responses were observed mainly in the Lt. medial gastrocnemius, which was the target muscle, when single-pulse stimulation was given. Train stimulation innervated widespread EMG responses on both sides, which met grade 4+ of the EMG response grading system. (D) EMG responses evoked by a single-pulse stimulation were mainly observed in the sphincter; the rootlet stimulated was left intact based on all of these protocols.
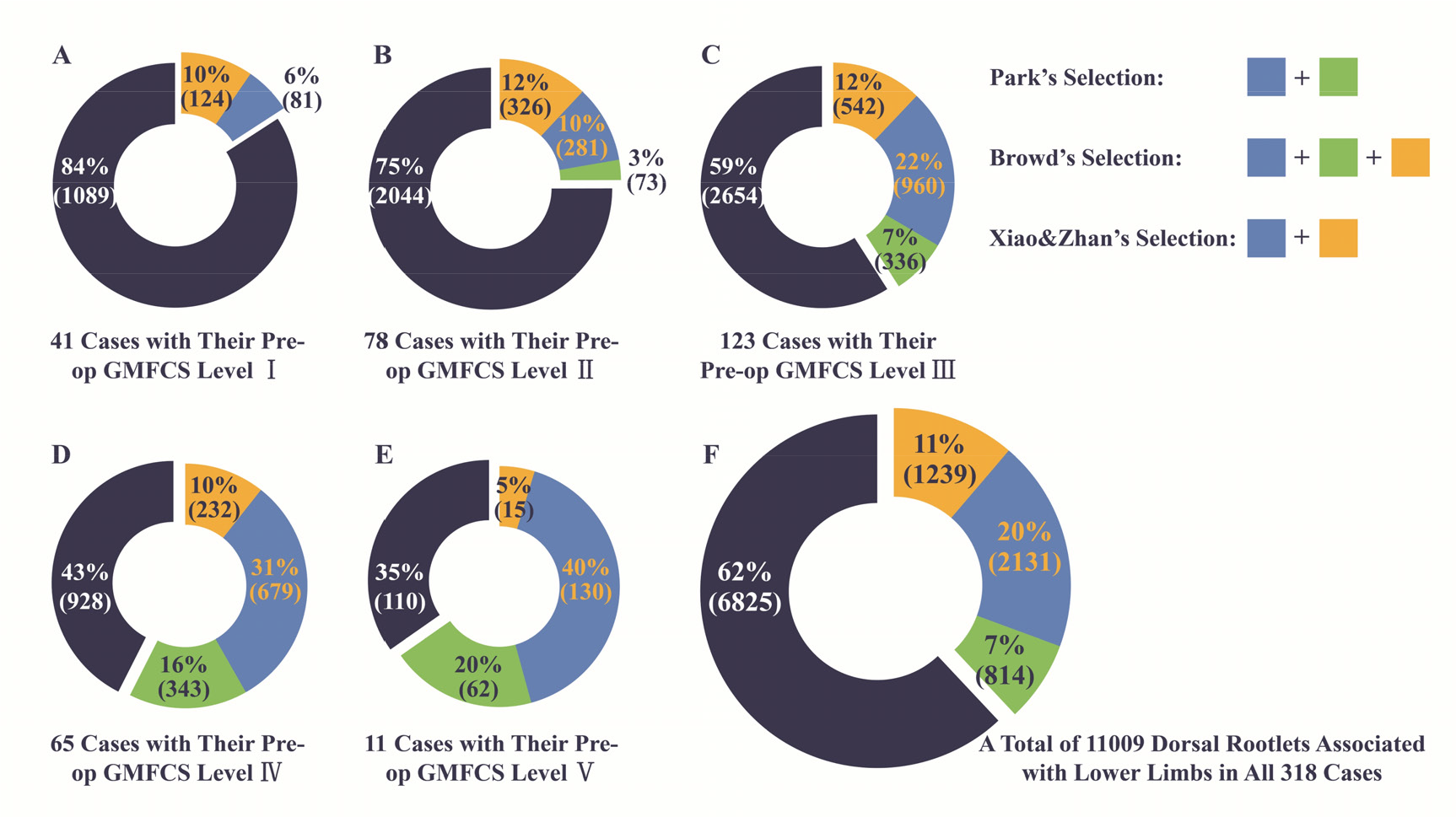
Difference in dorsal rootlet selection among the methods of Park, Browd, and our team (Xiao & Zhan) in 318 cases categorized into five groups based on their preoperative GMFCS level [11].
For decades, SDR has been applied to patients with spastic CP to decrease muscle tone mainly in the lower limbs, with the goal of improving their motor function. In clinical practice, physicians have found that patients do not experience improved motor function after SDR surgery, as the muscle tone of the spastic lower extremities decreases too much after the procedure, resulting in hypotonia, even with a postoperative intensive rehabilitation program. Therefore, achieving an appropriate decrease in spastic muscles in SDR is crucial. Until now, the definition of “appropriate” has not been determined. The literature suggested that in cases with satisfactory improvement of motor function after SDR, reduction in muscle tone of spastic muscles was usually of an average 1–1.5 degrees when compared with the preoperative condition [20 –23]. The continuing decrease of muscle tone in the lower limbs of patients who have undergone SDR over time was observed, creating a fundamental basis for further improvement of their motor function.
Although currently applied EMG interpretation methods provide the clinician the ability to select dorsal rootlets for sectioning in a quantitative manner, differences in selection criteria exist among those schemes. Based on our data [11], the rhizotomy ratio was roughly 29% : 41% : 34% in cases with preoperative GMFSC level 3, 47% : 57% : 41% in level 4, and 60% : 65% : 45% in level 5, when the same EMG recording data were interpreted using the EMG interpretation scheme of Park, Browd, and ours, respectively. In our previous study, we examined the intraoperative EMG data of 318 cases and found that the rhizotomy ratio was highly correlated with the severity of those cases, with an R of –0.76 and –0.66 when recording data were interpreted by applying the method of Browd and our team, respectively [11].
The EMG interpretation schemes proposed by Park, Browd, and our team have the capability of guiding SDR via a single-level approach without the need to use the human dermatome as a primary reference. Although SDR guided by the EMG response grading system via a singlelevel approach is usually applied to treat patients with moderate to severe spastic CP, Browd’s and our methods could be used universally to treat all kind of spastic CP cases in which the lower extremities are involved. The currently discussed EMG interpretation schemes are based on two hypotheses or their combination. Because of the lack of evidence from experimental studies, it is not clear which hypothesis better fits the neuroelectrophysiological nature of the spinal cord in those spastic CP patients. Nevertheless, in patients who have undergone SDR guided by these schemes, outcomes were favorable, with very minimal surgery-related complications. However, the follow-up of the cases in Browd’s and our reports were still too short.
Besides the ultimate question of “whether a best, universally applicable EMG interpretation method for SDR exists”, many technical questions still need to be answered with regard to IONM current practice: How much does the number of muscles monitored affect the EMG interpretation? To what extent do the threshold settings influence the rootlet selection? Is there a “best” threshold value for identifying target muscles? Because it is still difficult to use animal CP models to clarify the neuroelectrophysiological characteristics of the spinal cord in spastic CP patients, EMG data in IONM-guided SDR combined with postoperative evaluation results have the potential to provide a practical approach to better understanding the properties of neuronal circuits of the spinal cord in these patients, providing clinicians the opportunity to optimize relevant treatments.
5 Conclusion
The selection of dorsal roots (rootlets) for rhizotomy in SDR is crucial and directly correlated with the outcomes of CP patients who undergo the procedure. The best rootlet selection for sectioning should be quantitative, and have the goal of “to maximize motor function improvement by decreasing spasticity at an appropriate degree with minimized surgery-related complications”. Given the fact that the EMG interpretation methods developed over the past 20 years are based mainly on clinical studies, the question of “which method fits more the neuroelectrophysiological nature in spastic CP patients” still remains unanswered, although outcomes of CP patients who have undergone SDR guided by these methods were favorable, and surgery-related complications were extremely minimal. IONM-guided SDR has provided an approach to better under standing of neuronal electrophysiological circuits in spinal cord of spastic CP patients, therefore providing the potential to optimize treatment.
Footnotes
Conflict of interests
The authors declare no conflict of interests in this work.
