Abstract
Spasticity can be caused by central nervous system dysfunction, such as cerebral palsy and stroke. The accepted pathogenesis of spasticity is that the muscles are in the state of uninhibited stretch reflex without enough control of central nervous system. So far, there is no ideal way about how to repair central nervous system. However, the uninhibited stretch reflex can be reduced, targeting the posterior root of the spinal cord and peripheral nerves innervating the limbs, which are called selective posterior rhizotomy (SPR) and selective peripheral neurotomy (SPN), respectively. SPN is indicated for focal or multifocal spasticity, which is well accepted due to its low invasiveness and ease of use. How does the operation work? What do we do before and during this operation? Is there any risk to the patients? Our review summarizes the mechanism, indications, preoperative assessments, techniques, and complications of SPN. We hope that the spastic patients, such as pediatric cerebral palsy patients and older stroke patients, will benefit from this surgery.
1 Introduction
Spasticity represents one of the positive signs of upper motor neuron syndrome caused by central nervous system dysfunction [1 –3], including traumatic brain injury, spinal cord injury (SCI), cerebral palsy, stroke, and neurodegenerative illnesses. Spasticity itself is not an indication for surgical management, as it can be useful or harmful. Only the latter will be the target of treatment. Harmful spasticity can impair functionality of a patient, cause pain, make it more difficult to take care of the patient, and lead to irreversible deformities [4]. The understanding of the pathogenesis of spasticity, created in 1932 by Sherrington who won the Nobel prize, is that the muscle spasticity occurs with uninhibited stretch reflex and without control of the central nervous system due to its injury [5, 6]. Some spastic patients may not respond to the conventional treatment and subsequently develop intractable spastic disorder, so surgical procedures will be needed. It is important to differentiate diffuse spasticity from focal spasticity. Selective dorsal rhizotomy (SDR) or selective posterior rhizotomy (SPR), and intrathecal baclofen may be the best surgical treatments for diffuse spasticity, while selective peripheral neurotomy (SPN) can be the surgical treatment of choice for focal or multifocal spasticity. Botulinum toxin injection, a kind of drug therapy, is also widely used for local, harmful spasticity. And it is suggested that SPN should be performed after failure of botulinum toxin [7]. If botulinum toxin injection has little or no effect, or patients refuse to accept this injection either because of the high price or its repeated injection, then SPN can be performed. SPN provides nearly precise degree of spastic release on certain muscle groups. When muscle contractures and irreversible joint deformities are present, orthopedic surgery should be done to improve the outcomes. SPN may have an 80%–85% success rate in reducing focal spasticity. In this way, functional activity may be improved and worsening deformities can be prevented [4].
2 History of SPN
The first peripheral neurotomy was performed by F. Lorenz in 1887 [8] and then by A. Stoffel in 1912 [9]. This procedure recieved no enough attention due to its high complication rate. In 1976, Gross introduced microscopes and intraoperative electrical stimulation into the surgical procedure [7]. From then on, it has been called “selective peripheral neurotomy” and widely accepted in the world. SPN is more invasive than SPR and, therefore, should be performed only after rehabilitation and other conservative treatments fail. It is safe and efficient when performed by experienced neurosurgeons using microscopes and intraoperative electrical stimulation.
3 Mechanism of SPN
In order to understand SPN, we should first understand spasticity. The stretch reflex is the key in inducing tendon jerk (a type of dynamic stretch reflex) and maintaining appropriate muscle tension (a type of static stretch reflex). In addition, central nervous system gives inhibiting information to this reflex arc, maintains the balance, and allows normal movement for human beings. Injury of central nervous system will lead to upper motor neuron syndrome, which will induce hypermyotonia and tendon hyperreflexia, namely, spasticity. The key elements in the stretch reflex are stimulator, receptor, afferent nerves (sensory nerve fibers), center, efferent nerves (motor nerve fibers), effector, and response. Muscle spindle is the main receptor tissue in limbs, primarily detecting the length of muscles. In dynamic stretch reflex, muscle spindle receives a sudden stretch of muscle and causes a tendon jerk. In static stretch reflex, muscle spindle receives continuous stretching of muscle, leading to prolonged muscle contraction, presenting as muscle tension. Most skeletal muscles are composed of a mixture of rapid, slow, and intermediate fibers in variable proportions. Limb muscles tend to have large and rapid fibers (type Ⅱ fibers), which can mainly cause short-term contractions. In contrast, postural muscles are often composed of small and slow fibers (type Ⅰ fibers), which can stay contracted for a long time. In spastic patients, the mechanical properties of muscles change, with type Ⅱ fibers transforming into type I fibers. As the duration of contraction of type I fibers can be about 20−30 times longer than type Ⅱ fibers, the muscle contraction prolongs abnormally. Not all muscles are involved with the same degree of spasticity in a person. The commonly interfered muscles are soleus, semitendinosus, vastus intermedius of quadriceps, flexor carpi radialis, and brachialis muscles [7]. SPN, which is performed on the peripheral nerves, is an efficient procedure for treating focal or multifocal spasticity [10 –12]. Neurotomy in targeted peripheral nerves can eliminate the efferent information of the spastic muscles, diminishing the reflex arc and reducing spasticity. At the same time, this procedure may induce paralysis to the targeted muscles. SPN aims at restoring the balance between agonist and antagonist muscles to maintain normal movement and posture [13, 14]. Decreasing the uninhibited stretch reflex, at the level of a muscle’s nerve, is SPN’s mechanism of action.
4 Assessments before SPN
Assessments before SPN should include the assessment for spasticity and other evaluations, such as the general state of the patients, musculoskeletal contractures, gross motor function, and cognitive and psychological abilities of patients. The last one depends on whether the patients can accept the treatment and adhere to it. Clinically, full attention is paid to the appearance of limbs, posture and gait, passive range of motion, and the spasticity degree, using Ashworth and Tardieu Scales. With detailed clinical assessments, we can try to make ideal operation planning to determine the degree of resection, thus avoiding excessive loss of motor strength.
4.1 Observation of appearance of limbs and gait
The patients are told to lie on their back, and then the potential asymmetry of the limbs will be identified, which may be caused by high muscle tone or joint deformity. The observation of gait not only provides information about weakened muscle strength but also helps us to recognize spastic muscle groups which disturb the movement. According to the weakness and spasticity of certain muscles, the targeted muscles and selective nerve fibers for treatment are selected.
Winters et al. described four types of gait patterns for hemiplegic patients (Table 1) [15]. Rodda et al. developed the classification of common gait patterns seen in children with spastic diplegia (Table 2) [16, 17].
Gait pattern types described by Winters et al. [15].

Three-dimensional gait analysis (3DGA) may be an excellent tool for appropriate treatment selection in spastic patients, because it does not only describe 3D motion of the body posture but also provides kinetic analysis to measure the forces produced by the body [18]. However, due to the high cost of 3DGA, observational (2D) gait analysis can be used as a substitute. Additionally, it is also easy to manipulate 2D gait analysis. There are various observational gait scales that can be used to assist the analysis of the video recording of gait. These include the Ranchos Los Amigos Observational Gait Assessment [19], Edinburgh Visual Gait Scale [20], Salford Gait Tool [21], and the Physician Rating Scale [22].
4.2 Quantification of spasticity
The spasticity of muscles is evaluated using Ashworth and/or Tardieu Scales (sometimes Modified Ashworth Scale or Modified Tardieu Scale) globally. Compared with the Tardieu Scale, the Ashworth Scale may be a better option for the assessment of global spasticity due to its convenience (Table 3) [23, 24]. However, sometimes, using only the Ashworth Scale may be insufficient, because it does not evaluate the velocity of passive joint movement and the muscle and joint contractures may reduce the scale’s reliability [17, 25].
The Tardieu Scale, which is named after Tardieu’s work from the 1950s to 1980s by Professor Jean-Michel Gracies, seems to be more accurate and widely accepted to use. It pays more attention to the muscle reaction to specific velocities (low, normal, and fast). Measurement is recorded by X/Y (X indicates 0 to 5 rating scores, while Y implies the degree of angle at which the muscle reaction occurs) at three different velocities, respectively (Table 4) [26].
The Tardieu Scale [26].

4.3 Assessment of range of motion
When the articulations are deformed, the range of motion decreases. In this situation, spasticitydecreasing treatments are not enough. Orthopedic surgery, such as lengthening of the contracted muscles and transferring tendons, must be considered [6].
4.4 Gross motor function
The Gross Motor Function Classification System (GMFCS) describes a five-level classification based on a child’s self-initiated movement [27, 28]. It is primarily used for children with cerebral palsy; however, if used properly, it can be used more widely especially on the elders (Table 5). It is important to determine the level based on the patient’s usual performance in daily life, rather than on their best capability [29, 30].
The Gross Motor Function Measure (GMFM) assesses gross motor function in five dimensions: lying and rolling; sitting; crawling and kneeling; standing; walking, running and jumping. GMFM-88 is used for calculating raw and percent scores for each of the five dimensions to give a total percentage score. GMFM-66 converts the scores to an interval level total score using the Gross Motor Ability Estimator software. Either of the GMFM versions takes a long time in applying, approximately 30–60 minutes [31, 32].
5 Technical principles for SPN
Conservative treatment is typically efficient in patients with mild and even moderate spasticity. Surgeries should be given to the patients whose muscle tone significantly increases (Ashworth Score of 3 and higher).
SPN represents neurotomy of motor nerve fibers which are innervating spastic muscle(s). Therefore, the neurotomy should not be performed at the level of the nerve trunk which consists of sensory and motor nerve fibers. On the other hand, the motor nerves should also be identified proximally to the branches from the trunk.
In order to maintain the rebalance of muscle tone between the agonist and antagonist muscles, appropriate resection of the nerves is performed on the motor branches. Excessive rate of resection could cause muscle weakness; in contrast, insufficient resection could result in spastic syndrome recurrence [33]. Until now, although SPN is accepted all over the world, there is no consensus on the technical details [35]. Most neurosurgeons suggest sectioning of 50%–65% of all branches in a targeted muscle [8, 33 –36]. The objective of neurotomy is to be effective and to avoid of recurrence of spasticity. Under the microscope, variable proportions of fascicles are resected to ensure the accuracy. Then the neurosurgeons resect 5 mm of the fiber and then coagulate resected ends by bipolar forceps to prevent their regrowth. The resection rate can be evaluated by comparing muscle responses to intraoperative electrical stimulation. If the response after proximal stimulation is still intense, further resection should be undergone. Intraoperative electromyography can help determine the degree of nerve resection with more objective evidence.
Anesthesia is unique for SPN surgeries because it is performed under general anesthesia without long-lasting muscle relaxants. Using long-lasting muscle relaxants for SPN prevents the evaluation of the effectiveness of SPN intraoperatively (the muscle cannot contract under electrical stimulation).
Consensus is also needed on the nomenclature for surgical management of spasticity [34]. “Selective neurotomy” is a common term for this surgery now [37 –39], but “partial selective denervation” was used by Brunelli in 1983, and Fouad [40] and Gras et al. [41] used “selective peripheral neurectomy” and “hyperse-lective neurotomy”, respectively.
5.1 Neurotomy of musculocutaneous nerve
Neurotomy of the musculocutaneous nerve is indicated in spastic elbow flexion, in which the affected muscles are the biceps brachii, brachialis, and brachioradialis. Along its way, the musculocutaneous nerve divides into motor branches for biceps brachii, brachialis, and brachioradialis and ends as a major sensory nerve and lateral cutaneous nerve of the forearm.
With experiences of the surgery, we will describe the procedures as following. A 3-cm longitudinal incision is made on the proximal medial 1/3 of the upper arm, between the biceps brachii and triceps brachii (Fig. 1, A). The nerve always displays the most proximal motor branch to the biceps brachii and a distal motor branch to the brachialis [42]. When stimulating the nerves of biceps brachii or brachialis, elbow flexion occurs. Attention should be paid to the fact that the median nerve is going medially to the musculocutaneous nerve. But when the median nerve is stimulated, wrist flexion, finger flexion, and forearm pronation will occur. The nerves contributing to the biceps brachii and brachialis are selectively denervated, while the terminal cutaneous branch is completely preserved. With the aid of an intraoperative microscope, the epineurium is incised to allow dissection of the nerve fascicles. Motor fascicles are distinguished from sensitive fascicles with the help of a nerve stimulator [37].
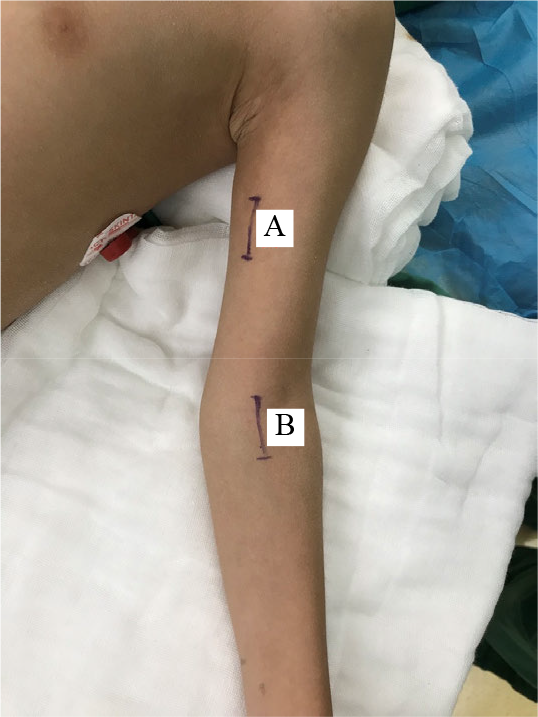
The incision of musculocutaneous nerve and median nerve surgeries. A, the incision of musculocutaneous nerve is on the proximal medial 1/3 of the upper arm, between biceps brachii and triceps brachii. B, the incision of median nerve is above the flexion line of the elbow, medially to the pronator teres.
5.2 Neurotomy of median nerve
Spastic wrist flexion, finger flexion, thumb adduction, and forearm pronation are indications for neurotomy of median nerve. Median nerve converges from the medial and lateral nerve fascicles of brachial plexus, goes along with the brachial artery, and passes through the elbow. The median nerve divides into pronator teres, flexor carpi radialis, flexor digitorum superficialis, and palmaris longus branches in the cubital fossa. At the level of the anterior forearm, it gives branches for the flexor digitorum profundus, flexor pollicis longus, and pronator quadratus muscles. It forms the fibrous arch in the palm, controlling flexor pollicis brevis, abductor pollicis brevis, opponens pollicis, etc., and communicates with ulnar nerve [37].
When doing the surgery, a 3-cm longitudinal skin incision is performed above the flexion line of the elbow, medially to the pronator teres, passing through the elbow (Fig. 1, B). Thereafter, the median nerve is taken medially to the pronator teres, deep under the bicipital aponeurosis (lacertus fibrosus), which is then cut. A sharp dissection is performed to separate the branches of the median nerve. The branch of pronator teres is on the ulnar side, which can be retracted to view its muscular branches. Then if we move or pull the pronator teres laterally and proximally, as well as the flexor carpi radialis muscle medially and distally at the same time, the muscular branches of the flexor carpi radialis and flexor digitorum superficialis muscles become exposed. Sometimes, it may be easier to dissect other branches of the median nerve after dividing the fibrous arch of the flexor digitorum superficialis [37]. However, those branches are so fine that they may be undifferentiated from the branches of the ulnar and radial nerves. The authors found that median neurotomy isn’t effective in spastic thumb deformation.
5.3 Neurotomy of obturator nerve
Neurotomy is often recommended for patients with spastic adductor muscle groups when their walking is hampered by crossing of the lower limbs. The main muscles are the adductor longus, adductor brevis, adductor magnus, and gracilis, which are innervated by the anterior branch of the obturator nerve. The spasticity of hip adductor muscle groups can induce reversion of upper femur and finally cause maldevelopment and dislocation of hip joint. Therefore, it is important to relieve the spasticity of the hip adductor muscles. The anterior branch of the obturator nerve is located between the adductor longus and the adductor brevis.
When doing the surgery, the patient is asked to lie on the operation bed, with hip joint flexed and abducted. A transverse incision is made below the groin, centered on the prominence of the adductor longus tendon (Fig. 2). When the adductor longus is pulled up and medially, with the gracilis pulled laterally, the adductor brevis is exposed. Separating the inter-wall between the adductor longus and the adductor brevis, the anterior branch of the obturator nerve becomes exposed and can be cut selectively according to the degree of spasticity and affected muscles. This neurotomy is always companied with tendon lengthening or releasing.
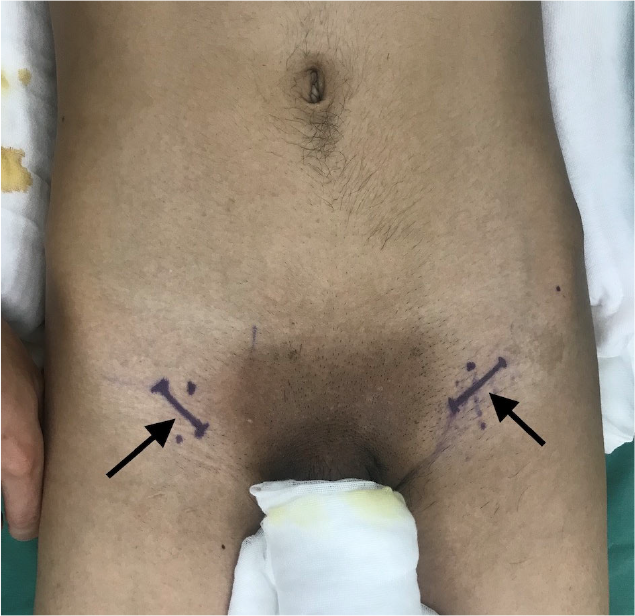
The incision of obturator nerve (indicated by the arrows) is below the groin, centered on the prominence of the adductor longus tendon.
5.4 Neurotomy of femoral nerve
The indication for neurotomy of femoral nerve is the stiffness of knee joint and genu recurvatum caused by quadriceps femoris spasticity. The quadriceps muscle can interfere with gait by limiting knee flexion during the swing phase. In addition, the patients should be older than 6 years, with muscle tone being more than grade 2 and muscle strength more than grade 4, knee tendon hyperreflexia, patellar clonus, high patella, obvious stiffness of the knee joint, and genu recurvatum. In the quadriceps femoris, the rectus femoris plays the crucial part, with the vastus lateralis being involved in 67% of cases, in maintaining the posture and movement. The vastus medialis muscle is the key muscle for the knee joint, and the vastus intermedius muscle terminates in the joint capsule. So we insists on choosing the branches of the rectus femoris and the vastus lateralis muscles as the targeted nerve fibers.
The incision extends from 0.5 cm below the groin, longitudinally going along with femoral artery for about 3 cm. The dissection passes medially to the sartorius muscle and exposes the trunk of the femoral nerve, which divides into 2–3 branches of the rectus femoris and the vastus lateralis muscles. Generally, about 50% of the rectus femoris branch is cut, while the vastus lateralis muscle branch is cut by 1/4–1/3. After that, 5 mm of the nerve fascicles should be resected to avoid recurrence. Electrical stimulation is essential, given the degree of resection and the large number of sensory fascicles, in this nerve, that must be spared.
5.5 Neurotomy of sciatic nerve
Neurotomy of sciatic nerve is for spastic knee flexion deformity, whose responsible muscle group consists of hamstring muscles, including semitendinosus, semimembranosus, and biceps femoris muscles. Sometimes, knee flexion can be caused by medial and lateral gastrocnemius muscles, which should be differentiated.
The longitudinal incision starts on the posterior thigh in the axis of midpoint between the ischial tuberosity and tip of the femoral greater trochanter and ends at about 3–4 cm distally. The sciatic nerve is surrounded with adipose tissue, companied with vessels. The biceps femoris is separated lengthwise uncovering a colossal sciatic nerve trunk. On the bottom of the incision, the branch for biceps femoris muscle and then the branches for the semitendinosus and semimembranosus muscles can be dissected and selectively resected.
5.6 Neurotomy of tibial nerve
Neurotomy of tibial nerve is indicated for talipes equinus caused by spastic triceps surae, a varus caused by spastic tibialis posterior muscle, with or without claw toes which are caused by spastic flexor hallucis longus and flexor digitorum longus.
Over the recent years, a transverse incision in the popliteal fossa has been adopted (Fig. 3), which gives a much better aesthetic result. Following division of the popliteal fascia and the tibial nerve trunk and its regional branches, including the nerves of the medial and lateral heads of the gastrocnemius, branch of the soleus is encountered. The neurosurgeons will selectively resect the nerve fibers after assessing the responsible muscles. The soleus has been demonstrated to be almost fully responsible for the pathogenesis of spastic plantar flexion allowing us to spare the gastrocnemius [43]. But other researchers [44, 45] also showed that it takes up 75% of cases for the soleus muscle to be responsible for ankle flexion spasticity, while only 12.5% of cases for the gastrocnemius to be the main target. Equinus or equinovarus is the most common symptoms in lower limb spastic patients. There are many targeting nerve branches in tibial nerve neurotomy; therefore, identifying those branches is of utmost importance.
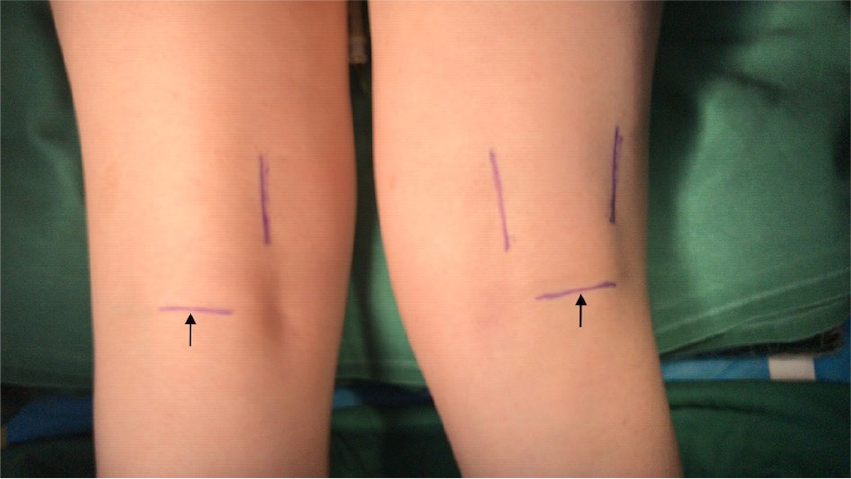
The incision of tibial nerve (indicated by the arrows) is in the popliteal fossa.
The medial and lateral gastrocnemius branches always exit from both sides of the tibial nerve trunk or posteriorly from a common origin. One or two nerves of the soleus muscle arises from the tibial trunk at the level of the tendinous arch of the soleus [45].
5.7 Neurotomy of superficial and deep peroneal nerve
Neurotomy of superficial peroneal nerve is indicated for treatment of talipes varus caused by spastic peroneus longus and shortened peroneal muscle, after careful analysis of the etiology. Neurotomy of deep peroneal nerve is for varus of the anterior part of the foot caused by spastic tibialis anterior muscle.
5.8 Others
Neurotomy of ulnar nerve and radial nerve are seldom performed due to their unsatisfactory results and added complications. Neurotomies of anterior thoracic nerves (nn. thoracales anteriores) and thoracodorsal nerve are also seldom performed because of their difficulty.
In our experience, the effectiveness of this procedure on spastic syndrome of the upper limbs may be lower than that of the lower limbs. However, some researchers say otherwise, for example, Dekopov et al. stated that “SPN for the upper limbs ensures a steady reduction of spasticity” [33].
6 Complications and recurrence
The outcome depends on the preoperative assessments, surgical procedure, and rehabilitation. If there are complications after SPN, rehabilitation will be delayed, which may reduce the efficacy of postsurgical reha-bilitation.
6.1 Sensory disorders
About 2 months post-SPN, a number of patients report sensory disorders, as several sensory fascicles might have been involved in the operation.
6.2 Incision infection
Every surgical incision carries a risk of infection, especially in the case of obturator and femoral nerves SPN because of their proximity to the groin area.
6.3 Muscle strength reduction
As the resection of the nerve fibers is targeted on motor fibers, after surgery, it is thought that muscle strength declines. But in fact, most of the patients can recover in a short time. It is assumed that other motor nerves can compensate and motor end-plate can be reformed.
6.4 Recurrence
Recurrence is believed to be related to nerve regrowth. After neurotomy, the nerve endings of α motoneurons sprout in order to compensate for the partial denervation. New motor end plates appear as well, and the size of motor units increases to compensate for the nerve loss [6]. If the fibers are resected for about 5–10 mm, followed by coagulation of resected ends by bipolar forceps, it is impossible for fiber regrowth to occur. Additionally, formation of other nerve fibers or other circuits in the spinal cord may be the causes for recurrence.
7 Conclusion
SPN is useful for focal or multifocal spasticity which is the main clinical manifestation in brain injury patients and SCI patients. In our opinion, SPN is a good choice, especially for elder patients after stroke, due to its relatively low invasiveness and good safety profile.
In addition, the goal of surgical therapy is not necessarily for the patient to walk independently [3]. We expect all the patients to be able to walk normally, but for some patients, especially those who have severe motor dysfunction, it would be ideal enough to stand or walk with a flat foot using a walking stick. A surgical goal in a spastic quadriplegic child, who is bound to a wheelchair, may be focused on improved and easier care for him/her.
Footnotes
Conflict of interests
The authors declare no conflict of interests in this work.



