Abstract
The circadian clock controls daily rhythms in animal physiology, metabolism, and behavior, such as the sleep‐wake cycle. Disruption of circadian rhythms has been revealed in many diseases including neurodegenerative disorders. Interestingly, patients with many neurodegenerative diseases often show problems with circadian clocks even years before other symptoms develop. Here we review the recent studies identifying the association between circadian rhythms and several major neurodegenerative disorders. Early intervention of circadian rhythms may benefit the treatment of neurodegeneration.
1 Introduction
From hormone secretion to immune response, from locomotor activity to learning and memory, most of our bodily functions are under control of the circadian clock [1
–4]. The molecular clocks enable animals to anticipate daily environmental changes and adjust their physiology and behavior. Studies in several model organisms, especially fruit flies and mice, reveal the fundamental mechanism of the circadian clock, which is a conserved negative transcription‐translation feedback loop [2
–6]. Circadian transcription activators bind to the promoter region of clock‐controlled genes and activate their rhythmic transcription in different tissues. Among these clock‐controlled genes, some are critical circadian repressors. These repressor proteins gradually accumulate in the cytoplasm and provide negative feedback to this loop through inhibition of the activators on their own transcription [2
–6]. In flies, CLOCK (CLK) and CYCLE (CYC) are the main transcription activators, while PERIOD (PER) and TIMELESS (TIM) are the key repressors (Fig. 1). TIM also functions as crucial protein for light entrainment in flies, as light triggers the degradation of TIM and resets the molecular clock. In addition to the core feedback loop, there is also a second loop to fine control the expression of
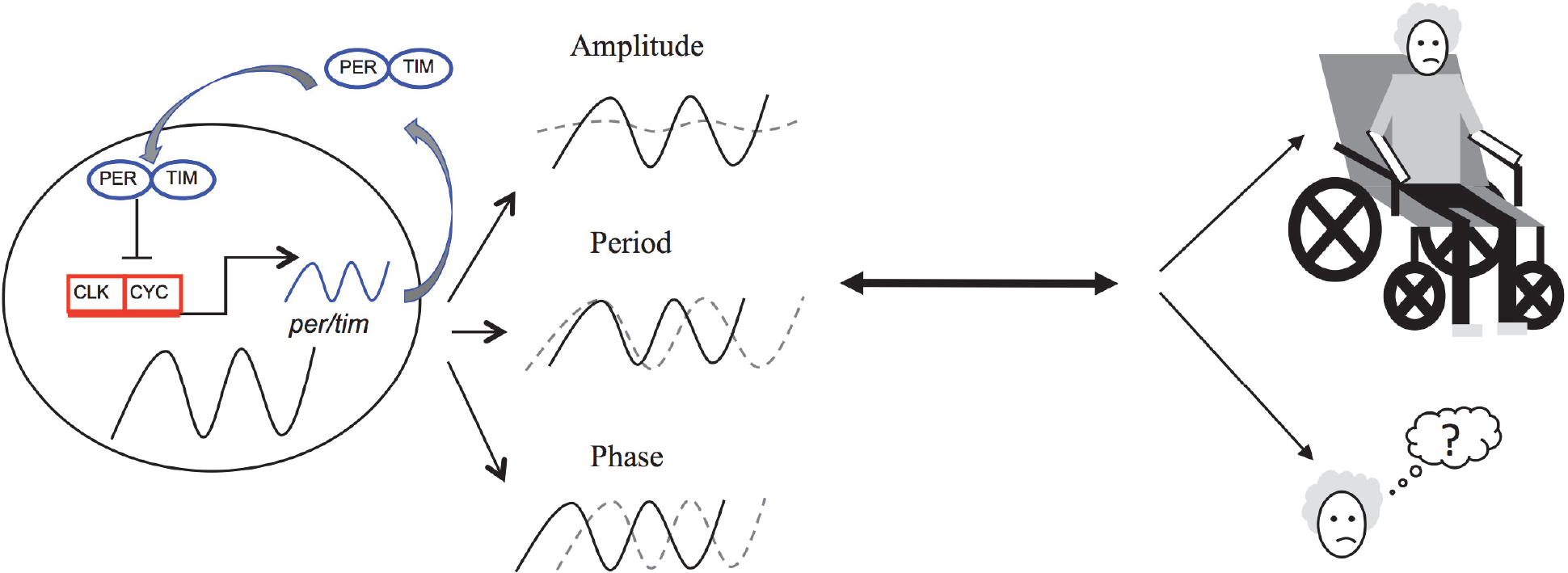
Interaction of circadian clocks and neurodegeneration. Circadian rhythms are generated by a conserved negative transcriptional translational feedback loop. Transcription activators (CLK/CYC, red) bind and activate rhythmic transcription of clock‐controlled genes. PER and TIM (blue ovals) are critical circadian repressors that accumulate in the cytoplasm and inhibit their own transcription. Circadian rhythms disruption is often shown as amplitude dampening of circadian oscillation, changing of circadian period, and shifting of circadian phase (dashed lines). A tight association is found between disruption of circadian rhythms and neurodegeneration. The bidirectionality between circadian rhythms and two major symptoms of neurodegenerative diseases (movement disorder and dementia) is focused in this review.
With the rapidly increasing aging population, neurodegenerative disorders become remarkable health issues. Neurodegeneration can affect many brain regions, including the basal ganglia, the cerebellum, or the spinal cord, and the diseases that cause neurodegeneration can be grouped into cognitive neurodegeneration (also known as dementia) or motor neurodegeneration (also known as movement disorder, or in some cases ataxia) [17]. The most common dementia‐causing disease is Alzheimer’s disease (AD). In 2017, 7.5 million people in the United States alone suffered from AD, and that number is predicted to rise to 8.4 million by 2030. Parkinson’s disease (PD) is also a common neurodegenerative disorder that affected at least 630,000 people in 2017, and that number is expected to double by 2050. Neurological diseases (including ataxias, dementias, back pain, migraines, epilepsy, and others) are estimated to cost the US economy approximately $780 billion annually [18]. Even though different neurodegenerative diseases may have different manifestations of symptoms, it is worth noting that most neurodegeneration will eventually cause cognitive decline [19].
Circadian rhythm disruptions can be caused by defects of the main components of circadian rhythms, including core clock, the input pathway for environmental entrainment, and the output pathway. Decline in circadian rhythms has been well documented in aging populations [20, 21]. Even though there is no definitive conclusion about casual effects of circadian rhythm disruption and neurode‐generation, it is clear that a bidirectional association between disruption of circadian clocks and neurodegenerative disorders exists. In the following part, we will review the recent studies and discuss this bidirectionality.
2 Neurodegenerative disorders leading to circadian rhythm complications
One of the most prominent outputs of circadian clocks is sleep‐wake cycles. Sleep disruptions in patients with neurodegenerative disorders (NDs) may not be a priority for patients, and often go undiagnosed [22]. These sleep problems are now understood to have greater implications in neurodegenerative diseases than previously believed, and may become a more common topic of conversation between doctors and patients. However, while sleep is the most obvious and commonly known output of circadian rhythms, there are other parts of circadian rhythms that have major implications for our daily health and functioning [23], and more research is indicating that maintaining robust circadian rhythms is an important factor in long term health [24]. For example, endocrine function can be inhibited by irregular circadian rhythms, which is unsurprising according to the role melatonin plays in the endocrine system [25]. In mammals, it has been found that regulated circadian rhythms allow the immune system to anticipate the highest risk of interaction with a pathogen during the day, suggesting a deeply evolved trait [26, 27].
2.1 Dementias
Dementias are neurodegenerative diseases that affect the central nervous system causing cognitive decline, and are often associated with aging [28]. One of the major symptoms for AD is severe dementia. AD is a neurodegenerative disease resulting from an accumulation of amyloid‐beta (Aβ) peptide in the brain. This accumulation is correlated with increased wakefulness and disruptions in sleep patterns [29, 30]. Patients with AD experience more severe sleep disruptions than healthy age‐matched controls [28] and experience a phase delay in their sleep [31]. The suprachiasmatic nucleus (SCN), is the central pacemaker for mammalian circadian systems and has been implicated in many neurodegenerative diseases [32]. Recent evidence in mice models has shown that the circadian clock influences plaque formation and Aβ activity. Researchers knocked out the core clock gene
Melatonin participates in a temporally strict clock gene regulation pathway via the mammalian pars tuberalis [38, 39], and melatonin levels oscillate according to the time of day [40]. This oscillation can become disturbed and irregular in AD patients as the disease progresses. Melatonin is necessary for regulation of sleep and circadian rhythms in humans and has been shown to promote brain resilience against neurodegeneration in transgenic mice. When the transgenic AD mice were given 10 mg/kg of melatonin per day for 6 months, researchers found this treatment significantly reduced the cognitive impairment seen in the AD mice [41]. Melatonin has also been shown to have neuroprotective effects in mice treated with scopolamine, which is known to induce AD‐like dementia [42]. Serum melatonin concentrations are also strongly correlated with body temperature, mood, and performance [43]. Growing evidence indicates that disruption of melatonin secretion rhythms, including amplitude, peak phase and total abundance is related with dementia, AD and mild cognitive impairment patients [44]. Based on a pioneer study comparing post‐mortem human pineal, the circadian difference of melatonin abundance (day
Frontotemporal dementia (FTD) is a group of dementias, which is characterized by selective degeneration of the frontal lobe of the brain. FTD can be difficult to distinguish from Alzheimer
2.2 Ataxias and polyglutamine disorders
Movement problems are another common indication of neurodegenerative disorders, which are triggered by defects in the peripheral nervous system. Ataxias are a class of neurodegenerative diseases that are characterized by impaired mobility. They are divided into subgroups including hereditary (autosomal dominant or autosomal recessive) and non‐hereditary (acquired or sporadic degenerative) ataxias [54].
The abnormal CAG repeat is a form of inherited trinucleotide repeat expansion known as polyglutamine, and has been associated with six types of ataxias (including Spinocerebellar ataxia type 2, SCA2), spinal bulbar muscular atrophy (SBMA), dentatorubral‐pallidoluysian atrophy (DLPRA), and Huntington
There are over 40 types of SCAs, and many are understood to be caused by either polyglutamine expansions, untranslated repeat expansions in non‐coding regions, or point mutations [54, 57]. SCA2 has been studied extensively. SCA2 is a neurodegenerative disorder that contains an abnormal expansion of the CAG repeat in the
Similarly, Huntington
Parkinson
Parkinson
3 Circadian disruptions leading to neurodegeneration
While circadian disruptions are often considered to be symptoms of neurodegenerative diseases, recent evidence has shown that circadian disruptions (CDs) can often precede onset of other neurodegenerative symptoms in several types of neurodegenerative disorders (ND) patients by years. This has caused researchers to look at circadian rhythm disruptions in a different light: rather than being just a symptom of NDs, perhaps CDs play a more active role in the origination of the diseases. As mentioned above, circadian rhythms are not solely presented as sleep, but also hormone release and core body temperature oscillations [27]. While circadian rhythms are responsible for the timing of the sleep‐wake cycle, disruptions in circadian rhythms are distinct from disruptions in sleep. The two can be distinguished by circadian rhythm biomarkers such as melatonin. There are also sleep disorders that are not a result of any differences in circadian rhythms.
Researchers found that
While there are other important outputs of the circadian rhythm, sleep is the most significant. REM behavior disorder (RBD) is a sleep disorder characterized by loss of muscle atonia during REM sleep. This results in patients acting out their dreams and occasionally hurting themselves or others. An estimated 80%–90% of patients with RBD are at risk for developing an ND, most commonly a synucleinopathy [73, 74]. A majority of idiopathic RBD cases that are comorbid with development of a ND are synucleinopathies. Why RBD and synucleinopathies are so strongly correlated is unknown [75]. One follow‐up study on patients diagnosed with RBD found that nearly 81% of those patients developed either a Parkinsonism or a dementia (
Environmental factors, particularly shift work, have been linked to an increased likelihood of developing neurodegenerative diseases [27]. While light is the main input, or zeitgeber, for the circadian rhythm, alternative inputs include social interactions, eating and exercise [23]. Circadian rhythm disruption, which is commonplace for those who work night shifts, may increase likelihood for developing AD, and the increased prevalence of night shifts in today’s society could have significant implications for Alzheimer’s disease in the future [77].
An interesting area of research is studying the chronotypes and rhythmicity of polar populations. Though more work needs to be done to understand how polar populations develop neurodegenerative disorders, there have not been studies indicating a significant reduction in sleep time, but studies that indicate a phase delay in winter months, when it is dark for two months [78]. There is evidence for increased depressive symptoms during winter months at more northern populations (
Light pollution and the increased availability and prevalence of artificial light have caused concern for researchers looking at sleep and health. One study found that in
4 Summary
Even though some discrepancies of changes for specific markers such as melatonin rhythms have been observed, there is no doubt that clear association between circadian rhythm disruption with neurodegenerative disorders exist. In the future, more research needs to be conducted to better understand the role that circadian rhythms have in disease progression or prediction. The literature has pointed toward a, "which came first, the chicken or the egg?" problem that will require some cleverly designed experiments to determine the answer to. For example, sleep disturbances may be caused by neurodegeneration of the sleep modulating structures [81]. The next major step in the field is to determine whether the neurodegenerative disease or the circadian disruptions came first in the lives of the millions of patients living with NDs. Together, these conclusions indicate bidirectionality in the disease onset and progression and the regulation of circadian rhythms.
Footnotes
Conflict of interests
The authors declare no conflict of interests in this work.
