Abstract
Increases in depressive behaviors have been reported in patients experiencing chronic pain. In these patients, the symptoms of pain and depression commonly coexist, impairing their lives and challenging effective treatment. The hippocampus may play a role in both chronic pain and depression. A reduction in the volume of the hippocampus is related to reduced neurogenesis and neuroplasticity in cases of chronic pain and depression. Moreover, an increase of proinflammatory factors and a reduction of neurotrophic factors have been reported to modulate the hippocampal neurogenesis and neuroplasticity in chronic pain and depression. This review discusses the mechanisms underlying the depressive-like behavior accompanying chronic pain, emphasizing the structural and functional changes in the hippocampus. We also discuss the hypothesis that pro-inflammatory factors and neurotrophic factors expressed in the hippocampus may serve as a therapeutic target for comorbid chronic pain and depression.
1 Introduction
Pain, including both sensory and emotional— affective components, may protect organisms from either potential or real tissue damage. However, disruptive pain that exceeds its biologically useful function may develop from chronic pain that lasts from three to six months, or even longer [1]. Chronic pain, which is a significant cause of disability worldwide, causes both physical and psychological discomfort and raises huge medical costs [2, 3]. Due to differences in populations and inclusion criteria, the prevalence of chronic pain in different countries has been reported as ranging from 8% to 60%, and it seriously affects the working lives and the social and daily activities of people affected by it [4]. Chronic pain commonly coexists with major depressive disorder (MDD) [1]. Up to 80% of patients with chronic pain have comorbid depressive symptoms [5]. Moreover, patients with chronic pain may also experience increased anxiety and deficits of both learning and memory [6]. Chronic pain may change a patient’s emotional state and induce depressive-like behaviors [7].
A literature suggests that, in a patient experiencing comorbid chronic pain and depression, the severity of pain is predictive of the time required for the remission of the patient’s depressive conditions following treatment [8]. Such findings indicate the existence of a mutual relationship between chronic pain and depression: depressive conditions may reduce the pain threshold, and painful conditions may contribute to depressed states [8]. In fact, pain is a major obstacle to achieve full remission during treatment for depression, and pain and depression may share the same neurobiological mechanisms [9].
Previous brain imaging studies have revealed the key brain regions involved in pain processing; these regions include the sensory areas (e.g., the primary somatosensory cortex, primary motor and supplementary motor cortices, secondary somatosensory cortex, insular cortex, anterior cingulate cortex, and thalamus), cognitive areas (e.g., the prefrontal and parietal cortices), and both memory and emotional areas (e.g., the amygdala and the hippocampus, and the subcortical structures of basal ganglia) [10 –12].
In particular, the hippocampus, as one of the central components in the limbic system, has been found to exhibit abnormalities in cases of both chronic pain and depression [13, 14]. Reduced volume of the hippocampus has been found in patients experiencing chronic pain [15, 16] and depression [17]. The findings of a cross-species study involving both humans and animals indicate that abnormalities in the hippocampus, including reduced volume, neurogenesis, and synaptic plasticity, reflect certain functional abnormalities, such as negative affective states and emotions, associated with chronic pain [6]. Similarly, there have been reports of an association between the reduced volume of the hippocampus and the severity of depression [18, 19]. In addition, the reduced hippocampal volume, affected similarly by chronic pain and depression, has been found to be related to reduced neurogenesis and neuroplasticity in both human patients and rodent models [20]. This may represent a neuropathological mechanism underlying comorbid chronic pain and depression [21]. Moreover, inflammation is known to be an important modulator of the hippocampal neurogenesis. Both pain and depression are thought to be associated with inflammatory responses in the region of the hippocampus and can alter hippocampal neurogenesis [22–24]. The related pathophysiological mechanisms are summarized in Fig. 1.
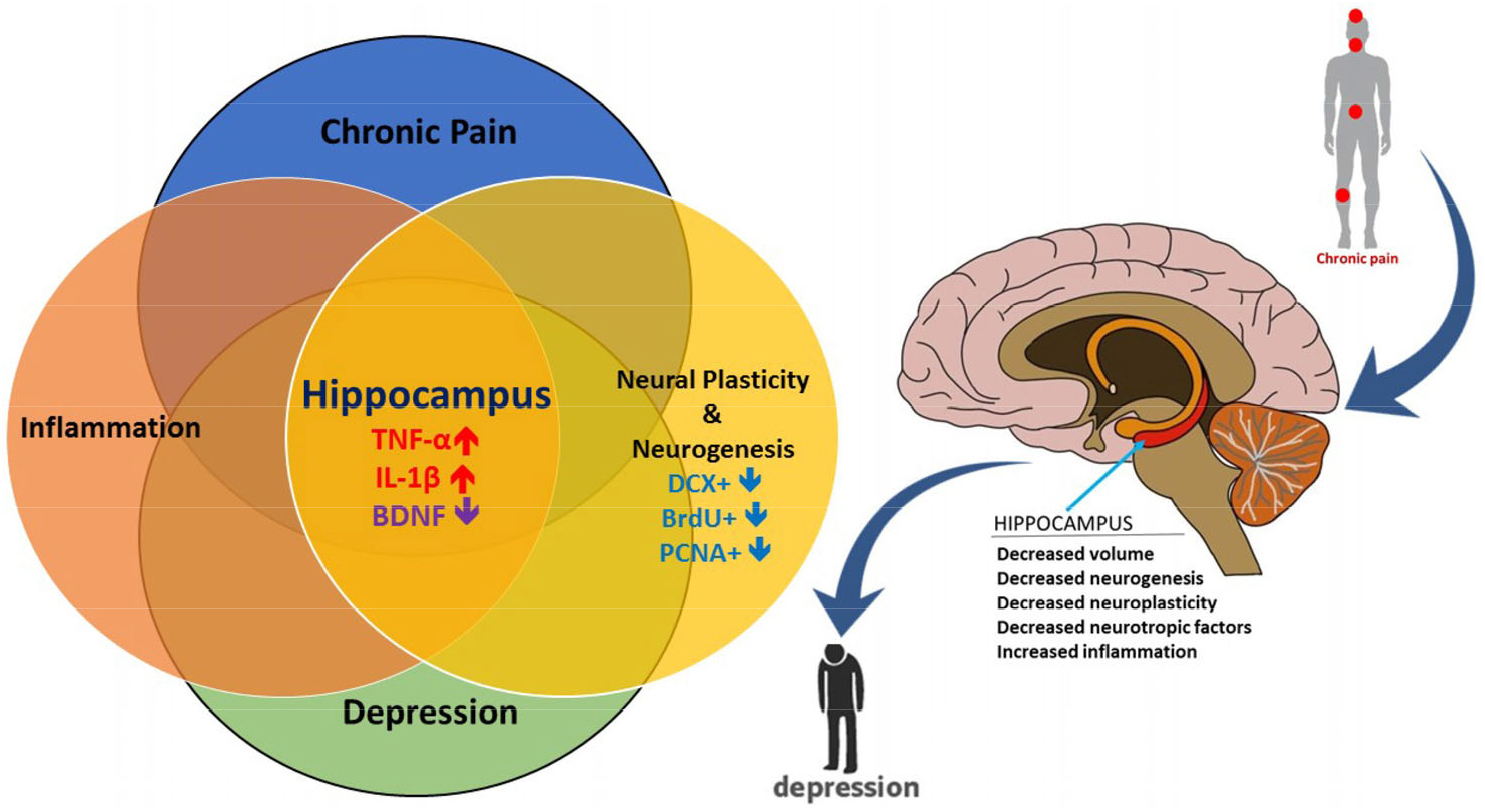
The mechanism underlying chronic pain-induced depression. Structural and functional changes in the hippocampus subserved a link between chronic pain and depression. Pain-induced depression was associated with increased levels of inflammatory factors, such as TNF-α and IL-1β, and reduced levels of BDNF in the hippocampus. TNF, tumor necrosis factor; IL-1β, interleukin-1β; BDNF, brain-derived neurotrophic factor; DCX+, doublecortin positive cells; BrdU+, bromodeoxyuridine positive cells; PCNA+, proliferating cell nuclear antigen positive cells.
Here, we review the literature to discuss the interrelationship of chronic pain and depression, with a particular focus on the hippocampus as a potential common denominator for chronic pain and depressive-like behaviors. Knowledge of the associations between these two conditions in the hippocampus, linked through a common neuroanatomical pathway and structure and a common inflammatory neuromodulator, would help in the treatment and management of comorbid chronic pain and depression.
2 Comorbidity of pain and depression in chronic pain patients
Pain is an unpleasant sensory experience that is accompanied by affective components, including feelings of annoyance, anxiety, sadness, and depression. Pain is known to be a main risk factor for the development of depression in patients with chronic pain, and there has been considerable research into the close relationship between pain and depression [25].
A longitudinal 12-year cohort study reported a significant correlation between the onset of depression and the baseline, chronicity, and severity of pain [26]. Depression and anxiety in chronic pain patients are acknowledged as being pain-related disabilities [27], given that studies have shown that 30%–100% of patients with chronic pain are affected by depression; this prevalence is several times higher than that in the general population [28, 29]. Moreover, the severity of chronic pain may hinder treatment for depression. From 51.8% to 59.1% of depression patients have reported experiencing chronic pain [30, 31]. Before antidepressant treatment commenced, the presence and severity of baseline pain were able to predict negative outcomes after treatment [32].
Combined administration of available antidepressants and analgesics is recommended to treat these comorbid disorders, but the efficacy of treatment was not satisfactory [33]. In addition, a higher rate of functional interference has been identified in the comorbid occurrence of pain and depression. In a cross-sectional study, patients with MDD and chronic pain experienced more interference in daily living activities (2.1–4.6 times) than depressed patients who were free of pain did [34]. Recently, the administration of analgesic drugs, such as opioids and benzodiazepines, in antidepressant therapy has provided solid evidence confirming the link between depression and chronic pain [21]. Thus, analgesics are also used to ameliorate the depression-like behaviors caused by neuropathic pain [35, 36].
3 Altered hippocampal structure in patients with chronic pain and depression
Many studies have demonstrated hippocampal abnormalities in patients experiencing chronic pain. Reduced volume of the hippocampus has been reported in patients with different types of chronic pain, such as knee osteoarthritis [37], chronic back pain, and complex regional pain syndrome [6], and also in elderly nondemented patients with chronic pain [16]. The hippocampus is an important brain structure that plays a role in specific types of learning paradigms and is active primarily in the storage and retrieval of long-term explicit memories [38]. As a subjective sensation, pain also involves the cognitive integration of awareness with memory and emotional responses. The hippocampus can facilitate and process direct and indirect nociceptive inputs of pain [24]. The septohippocampal neurons receive direct nociceptive inputs from the spinal cord [39, 40], and the indirect inputs innervate the hippocampus via the spinothalamic and parabrachial ascending pathways [41]. In chronic pain, 70% of patients experience attention and memory deficits [42, 43], which are associated with disruption of attention affected by painrelated sensory inputs and the formation of working memory in the hippocampus [44, 45]. Furthermore, the hippocampal-related abnormalities, including the deficits in recognition [46] and short-term memory [47], have been reported in animal models of chronic pain [48]. Thus, changes in the hippocampus caused by chronic pain may also be associated with the chronic pain comorbidities of learning and memory deficits.
Cognitive impairments, such as anxiety and depression, have been reported often in patients experiencing chronic pain [49]. In human studies, patients with chronic pain have been shown to have higher scores in tests of anxiety and depressive symptoms, confirming the well-documented relationship between chronic pain and affective disorders [50, 51]. As with chronic pain, reduced volume of the hippocampus has been demonstrated in patients suffering from anxiety and depressive disorders [19], and it may regulate the depressive phenotype [52].
Therefore, the hippocampal abnormality may be a link between chronic pain and depression, indicating that depression and cognitive deficits in chronic pain disorders may be associated with changes in the hippocampus. Recent research findings show a significant correlation between changes in the hippocampus induced by chronic pain and anxiety-like behaviors that are normally related to the depressive condition [6].
4 Neurogenesis and neuroplasticity in the hippocampus following chronic pain and depression
Despite the comorbidity of chronic pain and depression having been shown to be related to reduced volume of the hippocampus, the underlying molecular mechanism remains unclear. Hippocampal abnormalities, including impaired neuroplasticity and enriched-environment neurogenesis, have been associated with a different mechanism in chronic pain conditions [53]. At least two specific anatomical regions, the subventricular zone of the lateral ventricles and the subgranular zone of the dentate gyrus in the hippocampus, have been reported to be involved in the neurogenesis of the adult mammalian brain [54, 55].
Adult hippocampal neurogenesis is associated with learning and memory [56], and findings suggest that reduced neurogenesis in the hippocampus region is involved in various neuropsychiatric disorders, including anxiety [57], depression [58], stress [59], and impaired contextual fear conditioning [60]. Importantly, research has shown that reduced neurogenesis in the adult hippocampus correlated with cognitive decline and mood alterations is associated with chronic pain [6, 53]. The neurogenic hypothesis of depression suggests that depression is caused by impaired adult hippocampal neurogenesis and that newly generated neurons in the adult brain play a key role in regulating mood and are associated with antidepressant efficacy [61]. Similarly, several preclinical studies [20, 62] have reported a correlation between persistent pain and either reduced hippocampal neurogenesis or a blunted response of hippocampal neural progenitor cells to proneurogenic stimuli [20, 62].
Based on findings related to the rate of neurogenesis, as quantified by counting the number of bromodeoxyuridine (BrdU+)-positive cells (as a marker of neurogenesis) in the hippocampal dentate gyrus (DG), the number of BrdU-labeled cells remains unchanged following exposure to acute nociception (induced by formalin) [41]. However, a reduction in the number of BrdU+ cells in DG was recorded in animals exposed to prolonged nociception (via application of Complete Freund’s Adjuvant for 21 days to achieve hyperalgesia) [63]. In addition, impaired neurogenesis has been demonstrated in the depression-like behavior associated with peripheral neuropathy that persists following resolution of prolonged tactile hypersensitivity (at least three weeks) [64]. As these data demonstrated impaired neurogenesis in both chronic pain and depression, decreased neurogenesis may also contribute to the depression following chronic pain.
The phenomenon of neural plasticity, which refers to changes in neuronal synapses and pathways to adapt to new experiences in either healthy or damaged conditions [65], is known to be a fundamental characteristic observed in the development of chronic pain [66]. Studies using animal models have demonstrated that persistent peripheral nerve injury impairs long-term potentiation at CA3–CA1 synapses in the hippocampus, indicating reduced synaptic plasticity in the hippocampus following chronic pain [47]. According to the neuroplasticity hypothesis of MDD, the dysfunction of neural plasticity is a basic pathomechanism of the disorder [61]. Moreover, loss of volume of the hippocampus has been shown to be correlated with reduced neurogenesis and neuroplasticity following chronic pain-induced depression in both human patients and rodent models [21, 67]. To summarize, these findings have shown that reduced neural plasticity can occur in both depression and chronic pain. Depression-like behavior induced by chronic pain may be associated with reduced neural plasticity in the hippocampus.
5 Proinflammatory factors in the hippocampus following chronic pain and depression
There is mounting evidence that the immune system plays an important role in the etiology of chronic pain-induced depression [68]. Changes in the function and structure of the hippocampus have been suggested to be associated with increased proinflammatory cytokines in hippocampal tissue following nerve injury [47]. In particular, neuroinflammation of the hippocampus is key to the development of depressive and anxious disorders following chronic pain [69, 70]. Correlation between abnormal expression of cytokines in the hippocampus and chronic pain-like behavior has been demonstrated in animal models [23, 71]. Reduced proliferation and neurogenesis in the hippocampus, along with an increase in proinflammatory cytokines (CD86+) and a reduction in neuroprotective (CD163+) microglia/ macrophages, indicates increased activity of microglia/macrophages, which may lead to the suppression of neurogenesis reported in chronic neuropathic pain [72]. Furthermore, the reduction in neurogenesis may be related to the increases in other hippocampal proinflammatory cytokines, such as TNF-α [47] and IL-1β [23, 71], following nerve injury.
TNF-α is an integral determinant/component of nociception involved at all neuroaxis levels contributing to the pathogenesis of chronic pain [73 –75]. TNF-α has been shown to be essential to the cognitive experience of pain and chronic pain associated with mood changes [24]. Hippocampal noradrenergic neurotransmission, which plays an important role in the neurogenesis [76], could be dampened following elevated hippocampus TNF-α levels due to the peripheral nerve injury in the sciatic nerve chronic constriction injury (CCI) model [77, 78]. In the absence of any peripheral injury, intracerebroventricular (ICV) microinfusion of TNF-α into the hippocampus induced chronic pain-like symptoms, indicating that the hippocampus was a site of the nociceptive action of TNF within the central nervous system [24]. Studies have revealed that increased levels of TNF-α in the brain activate the α2-adrenergic receptor (α2AR), thus inhibiting the release of norepinephrine in the hippocampus of animal models following chronic pain [77, 79]. This finding suggests that the administration of antidepressants, such as adrenergic drugs, in rodents reduced the animals’ sensitivity to pain by decreasing the production of TNF, thereby inducing the increased release of norepinephrine [80].
IL-1β is another cytokine that is potentially overexpressed in supraspinal brain regions, particularly in the hippocampus, and that underlies the mechanism of behavioral and neurogenesis changes in neuropathic pain [22, 23]. Increased levels of neuronal IL-1β in the contralateral ventral hippocampus have been suggested to be linked with changes in astrocyte and microglial reactivity in the CCI model [81]. Moreover, IL-1β has been reported to reduce neurogenesis in the elderly [82], and it may play a similar role in neuropathic pain [22]. IL-1β knock-down in the hippocampus has also ameliorated lipopolysaccharide-induced depressive- and anxiety-like behaviors [83]. In the mouse model of spared nerve injury, the administration of the IL-1 receptor antagonist alleviates the effects of neuropathic pain on depression-like behaviors [69].
In addition, activation of the Nod-like receptor protein (NLRP) inflammasomes, which results in the direct maturation of caspase-1 and then induces production of the proinflammatory cytokines (IL-1β and IL-18), has been implicated in neuroinflammation-related diseases [84]. Recent findings have shown that hippocampal NLRP1 inflammasome activation contributes to the depression-like behaviors induced by neuropathic pain in rats [70]. Taken these findings together, we believe that the neuroinflammation in the hippocampal region may play a key role in paininduced depressive behaviors.
6 Neurotrophic factors in the hippocampus following chronic pain and depression
Brain-derived neurotrophic factor (BDNF) expressed in neurons and in immune cells has been shown to be involved in neurogenesis, the formation of synaptic plasticity and memory [85, 86]. The receptor of BDNF is tropomyosinrelated tyrosine kinase B (TrkB). TrkB has a critical role in signaling pathways associated with the function and development of the mammalian nervous system [87]. Abnormal expression of BDNF during either nociceptive or inflammatory processes has been reported [88]. Moreover, reduced expression of BDNF has been found in the hippocampus of animals exposed to painful stimuli [89]. BDNF has been reported to regulate both spinal and peripheral sensitivity in various animal models of chronic pain [90]. Moreover, BDNF has been shown to be a crucial signaling molecule between microglia and neurons, which represents an essential link in the transmission of neuropathic pain, and blocking this pathway may be a strategy for treating neuropathic pain [91]. In addition, reduced levels of serum BDNF have been found in patients with major depressive disorder, whereas successful antidepressant treatment increases the BDNF levels in these patients [92, 93]. By modulating synaptic plasticity throughout the peripheral and central pain circuits, BDNF may change pain sensitivity and, more importantly, the level of pain-induced depression. Therefore, BDNF is a potential new therapeutic target for treating chronic pain-induced depression in the near future.
7 Conclusion
The data presented in this review show that chronic pain-induced depression may suppress adult neurogenesis in and synaptic plasticity of the hippocampus. Increases in the number of inflammatory factors and reductions in the number of neurotrophic factors in the hippocampal region may underlie the pathology of this condition. The development of new therapeutic strategies to both increase the neurotrophic factors, such as brain-derived neurotrophic factor, and inhibit the inflammation may attenuate depressive behaviors in patients with chronic pain and can be considered in future clinical studies.
Footnotes
Conflict of interests
The authors have declared that no competing interests exist.
Acknowledgments
This work was supported by the National Natural Science Foundation of China (No. 31671141, No. 31822025), the Informatization Special Project of Chinese Academy of Sciences (No. XXH13506-306), and the Scientific Foundation project of Institute of Psychology, Chinese Academy of Sciences (No. Y6CX021008).
