Abstract
Background:
Randomized controlled trials (RCTs) that directly compare the efficacy and safety of percutaneous patent foramen ovale (PFO) closure devices have not been conducted. Thus, we performed a network meta-analysis to identify the efficacy and safety of occluder devices.
Methods:
From 1st January, 2000 to 1st May, 2018, we searched Embase, PubMed, and Cochrane Library for RCTs about percutaneous closure devices (such as STARFlex, GORE, and Amplatzer) and medical therapy for cryptogenic cerebral ischemic patients with PFO. The occurrence rate of recurrent stroke, atrial fibrillation (AF), major vascular complication (MVC), headache, transient ischemic attack, and bleeding were compared with the frequentist and Bayesian methods using R statistics.
Results:
We included 3747 patients from six RCTs. The GORE and Amplatzer occluders were found to be significantly associated with a decreased risk of recurrent stroke [relative risk (RR): 0.37 and 0.49; 95% confidence interval (CI): 0.17–0.81, 0.29–0.83, respectively]. Moreover, STARFlex was correlated to an increased risk of postoperative AF and MVCs (RR: 11.66 and 7.63; 95% CI: 4.87–21.91, 2.34–24.88).
Conclusions:
Among the three devices, the GORE and Amplatzer occluders are found to be the most effective in preventing secondary stroke in patients with PFO. Meanwhile, STARFlex is the least recommended device because it cannot decrease the risk of recurrent stroke and is the most likely to cause adverse events.
Keywords
1 Introduction
The prevalence of cryptogenic stroke accounts for about 40% of strokes. Data have shown that the percutaneous closure of patent foramen ovale (PFO) can cause cryptogenic stroke, particularly in young individuals. Furthermore, 50% of patients with cryptogenic stroke are diagnosed with PFO based on transesophageal echocardiography reports. Previous guidelines on the prevention of stroke reoccurrence in patients with stroke and transient ischemic attack did not consider PFO closure as more beneficial than medical treatment [1, 2]. With the development of minimally invasive percutaneous approaches for PFO closure, several randomized controlled trials (RCTs) published within the last few years have pointed out a major historical turning point [3, 4]. That is, they have shown that foramen ovale closure is beneficial in patients with cryptogenic stroke and migraine, and this notion was quickly accepted by the academic community [5, 6]. Different kinds of occlusion devices for the closure of the foramen ovale are available. However, RCTs that directly compare the safety and efficacy of percutaneous PFO closure devices have not been conducted. Thus, this study aimed to perform a network meta-analysis to identify the occluder devices that have the highest and lowest efficacy by evaluating the adverse effects of various devices and drug treatments.
2 Methods
2.1 Search strategies
Two investigators independently performed an electronic search using Embase, PubMed, and Cochrane Library from 1st January, 2000 to 1st May, 2018. The search strategies were based on the following MeSH terms: ((“foramen ovale” OR “patent foramen ovale” OR “patent oval foramen” OR “PFO” OR “right to left shunt” OR “interatrial shunt” OR “heart septal defects” OR “atrial septal aneurysm”) OR (“percutaneous patent foramen ovale closure” OR “percutaneous PFO closure” OR “transcatheter closure”) OR (“occluder device” OR “anticoagulant” OR “antiplatelet” OR “STARFlex” OR “Amplatzer” OR “GORE” OR “Helex”)) AND (“stroke” OR “cryptogenic stroke” OR “transient ischemic attack” OR “death” OR “subarachnoid hemorrhage” OR “hemorrhage” OR “ischemic stroke” OR “atrial fibrillation”) AND “randomized controlled trial”.
The title, abstract, and main text were cautiously assessed. Language or publication status restrictions were not applied. Some supplementary materials listed in the article were also included in the assessment. Discrepancies were settled by mutual deliberations.
2.2 Description of design and selection criteria
The surgical treatment group was defined as the group who underwent foramen ovale closure operation with occluders during the treatment. Generally, anticoagulation and antiplatelet drugs were administered after closure. The drug treatment group was defined as the group who did not undergo any surgical procedures throughout the treatment period. Moreover, anticoagulant or antiplatelet drugs were administered to prevent stroke.
The inclusion criteria included the following: (1) RCTs of PFO closure or medical therapyinduced adverse outcomes and prognosis of PFO in patients with cryptogenic cerebral ischemic stroke, (2) experimental studies that included at least one group who underwent PFO closure, (3) research with a follow-up period longer than 2 years, (4) original studies of humans, and (5) studies written in English. Meanwhile, the exclusion criteria were as follows: (1) studies that did not provide detailed data about the outcomes of different devices during follow-up, (2) animal trials, (3) studies that did not have original data, and (4) duplicate reports. Discrepancies were settled by mutual deliberations.
2.3 Quality assessment
The Cochrane Collaboration Risk Assessment Tool was used to assess the methodological quality of the studies included in the analysis [7]. The methodological design has six aspects, namely, selection bias, performance bias, detection bias, attrition bias, reporting bias, and other biases, that required comprehensive evaluation. Two investigators assessed the quality of the studies, and discrepancies were settled by mutual deliberations.
2.4 Data extraction
The documents were extracted according to a pre-established data extraction table, which comprised data about the age and sex of the patients who were enrolled, treatment methods, drug used and dosage in the different groups, and follow-up time. We extracted the required data from the body of the text and supplementary materials. Two investigators independently performed data collection, and disagreements were resolved by a team discussion.
2.5 Endpoints
The primary endpoint was the occurrence of ischemic stroke, atrial fibrillation (AF), and major vascular complication (MVC). Ischemic stroke was defined as stroke that developed after patients underwent PFO closure or received medical treatments. The major MVCs included myocardial infarction, deep vein thrombosis, peripheral embolism, cardiac perforation, hematoma, and reoperation in general. The secondary outcomes were transient ischemic attacks (TIAs), major bleeding, and headache or migraine, which were common and considered serious complications in the cardiology department.
2.6 Statistical analysis
A network meta-analysis is an extension of the traditional pair-wise meta-analysis, as it combines both direct and indirect information via a common comparator, and obtains estimates of the relative interventional effects on multiple intervention comparisons [8].
Medical therapy was considered as the central reference in the whole network comparison system. Five studies conducted a direct comparison between medical therapy and one PFO closure device. The rest compared the three types of PFO closure device.
Surface under the cumulative ranking curve (SUCRA) was considered one of the most important indicators for evaluating the efficacy of different treatments. SUCRA is advantageous as it simplifies information about the effect of each treatment into a single number, and the whole complex multiple-treatment meta-analysis results were expressed with a few numbers. Moreover, it is most significant when the difference in preference between successive ranks remains the same across the entire ranking scale [9]. The study with a higher SUCRA score will have a higher recommendation level.
The Mantel–Haenszel and DerSimonian–Laird methods for fixed effects were used to estimate the pooled ratio risk (RR), and 95% confidence interval (CI) was accepted as the summary statistics, as implemented based on the frequentist model. I 2, Q test, and the corresponding p value were used to estimate the heterogeneity of the studies. A p value of < 0.05 was considered statistically significant. R software 3.3.3 was used for data analysis. The whole analysis was conducted in accordance with the Cochrane Handbook and Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) Checklist. Codeliberation was a prerequisite for all statistical conclusions.
3 Results
3.1 Demographic characteristics
After a detailed filtration, six RCTs that included four different interventions [medical therapy, Amplatzer® PFO Occluder (Amplatzer), GORE® Helex® Septal Occluder or GORE® CARDIOFORM® Septal Occluder (collectively referred to as GORE), and Cardio-SEAL® STARFlex™ device (STARFlex)] were included in this meta-analysis [4,10 –14]. Moreover, the analysis comprised 3747 patients, and the characteristics of the participants are listed in Table 1. The Amplatzer was the most commonly used closure device (n = 983). All patients received anticoagulation or antiplatelet therapy after surgical closure. The selection flow chart is depicted in Fig. S1. The characteristics of the enrolled population were similar, which indicated assumption of transitivity, and network calculations could then be conducted.
Characteristics of the trials that were included.
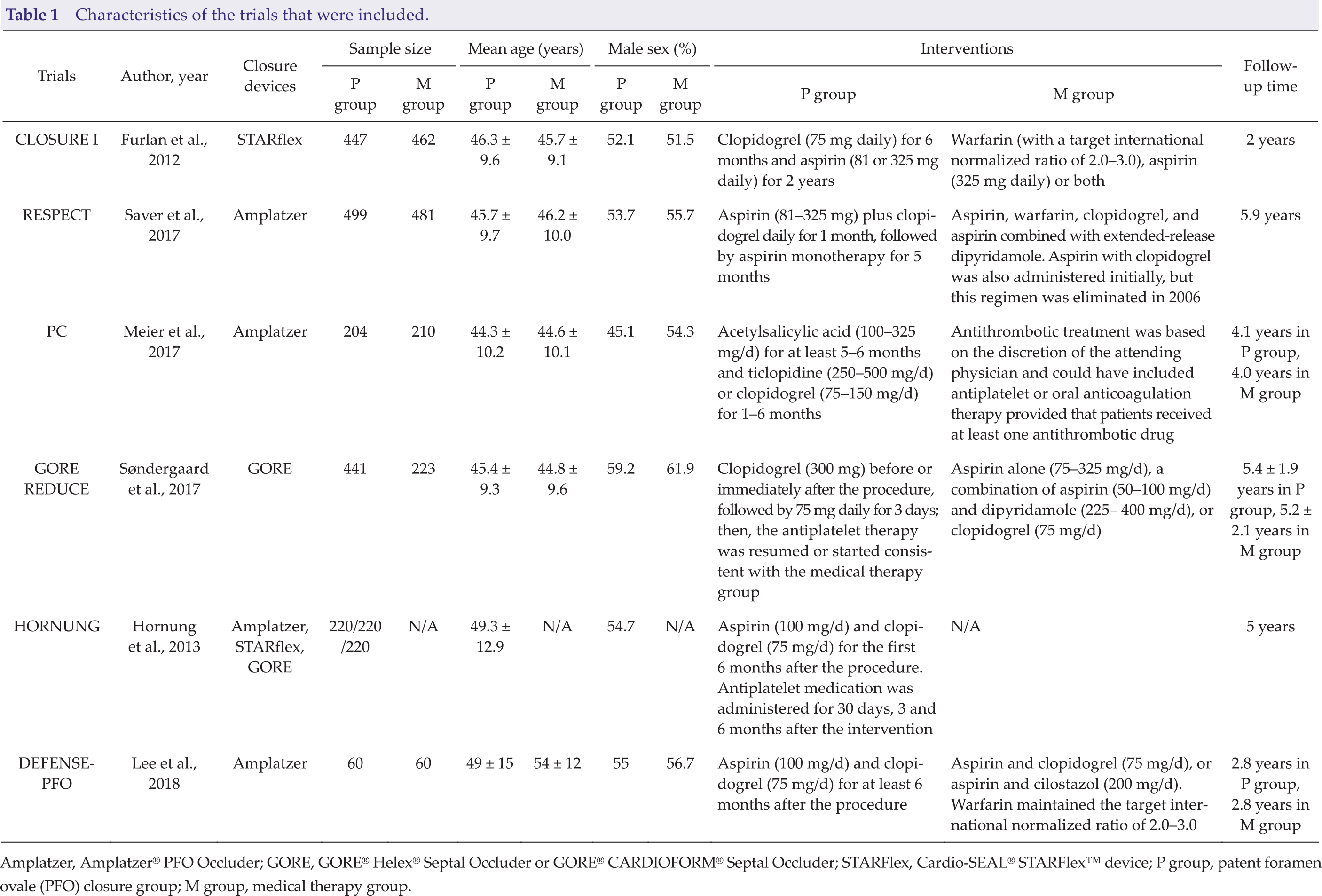
Amplatzer, Amplatzer® PFO Occluder; GORE, GORE® Helex® Septal Occluder or GORE® CARDIOFORM® Septal Occluder; STARFlex, Cardio-SEAL® STARFlex™ device; P group, patent foramen ovale (PFO) closure group; M group, medical therapy group.
3.2 Summary of methodological quality
Most qualified studies were of high quality in terms of methodology. Results for the risk of bias of the included articles using the Cochrane Collaboration Risk Assessment Tool are shown in Figs. S2 and S3. All studies provided complete data of the primary outcome. Some studies included the secondary endpoints. All articles described the principles of random assignment. Five of the six articles that were included have reported about random sequence generation. The blinding methods were not applied to the patients. The number of participants between the groups from the GORE REDUCE study [4] differed, which might have resulted in other potential biases.
3.3 Primary endpoint: stroke
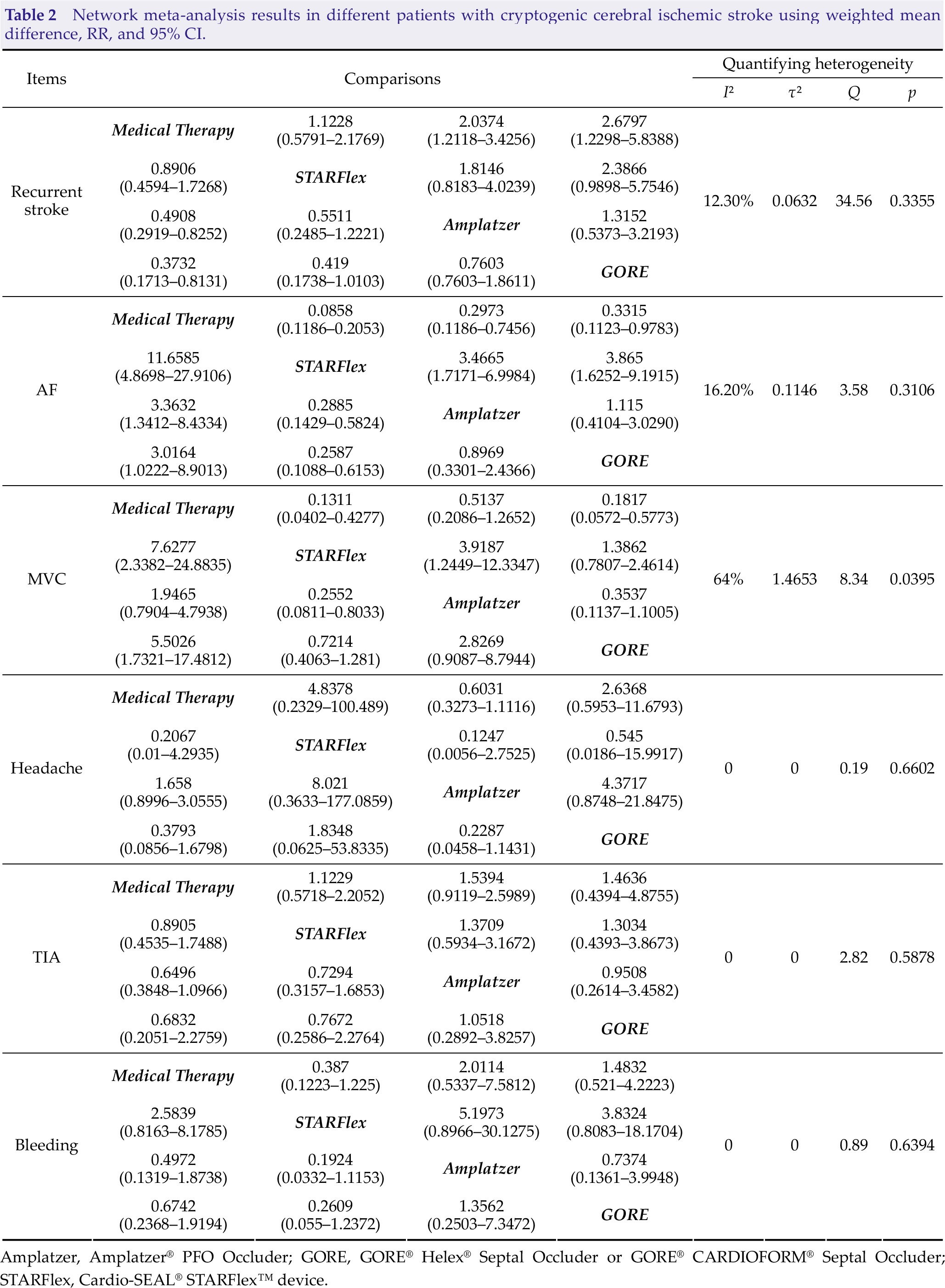
Attention was paid to the outcome of stroke in six randomized trials. To validate the efficacy of the PFO closure devices that were included, we conducted a pair-wise estimation, and the network diagram of these studies is presented in Fig. S4(A). The GORE and Amplatzer have lower reoccurrence rates of stroke than drug therapy based on the pair-wise comparison (RR: 0.37 and 0.49; 95% CI: 0.17–0.81 and 0.29–0.83, respectively). However, a difference was not observed between STARFlex and drug-only therapy [RR: 0.89; 95% CI: 0.46–1.73; Fig. 1(A)]. The result was consistent in the network analysis, and patients who underwent PFO closure with the GORE had the best prognosis (Table 2). According to the network comparison result, the group who underwent PFO closure with the GORE (89.76%) had the highest SUCRA score, followed by the group in which the Amplatzer (73.31%) was used. Meanwhile, the medical treatment group (12.53%) had the lowest SUCRA score, which indicated that the GORE might be most effective in preventing recurrent stroke (Table 3). The heterogeneity and inconsistency tests had a low level of heterogeneity (I 2 = 12.3%, τ² = 0.0632, Q = 4.56, and p = 0.3355). Eliminating any study did not significantly affect the overall outcome using the fixed effect model.
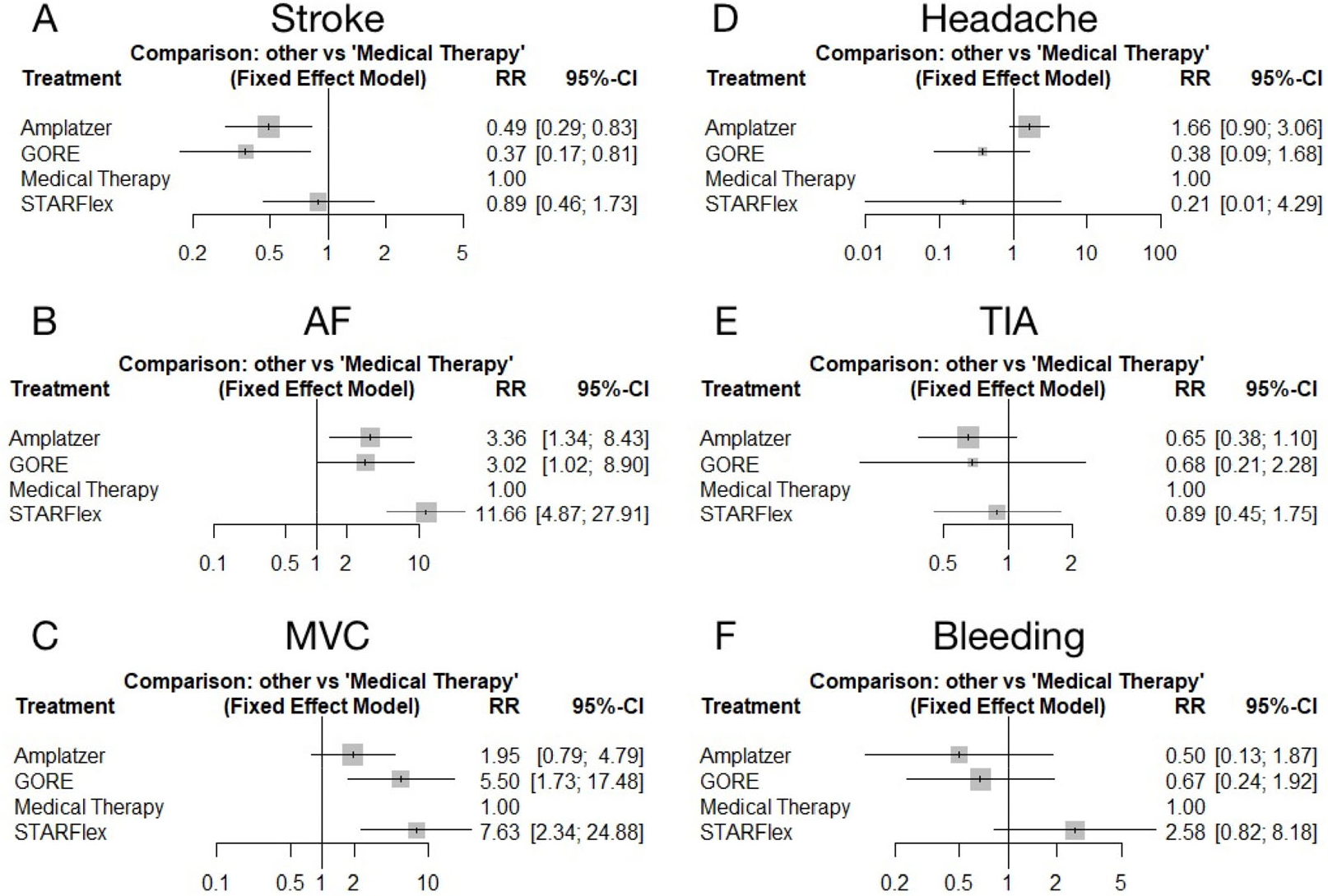
Direct meta-analysis of the calculation results. (A) All closure strategies compared with medical therapy for stroke. (B) All closure strategies compared with medical therapy for atrial fibrillation (AF). (C) All closure strategies compared with medical therapy for major vascular complications (MVC). (D) All closure strategies compared with medical therapy for headache. (E) All closure strategies compared with medical therapy for transient ischemic attack (TIA). (F) All closure strategies compared with medical therapy for bleeding. Amplatzer, Amplatzer® PFO Occluder; GORE, GORE® Helex® Septal Occluder or GORE® CARDIOFORM® Septal Occluder; STARFlex, Cardio-SEAL® STARFlex™ device.
Network meta-analysis results in different patients with cryptogenic cerebral ischemic stroke using weighted mean difference, RR, and 95% CI.
Results of the SUCRA rankings for different treatments.
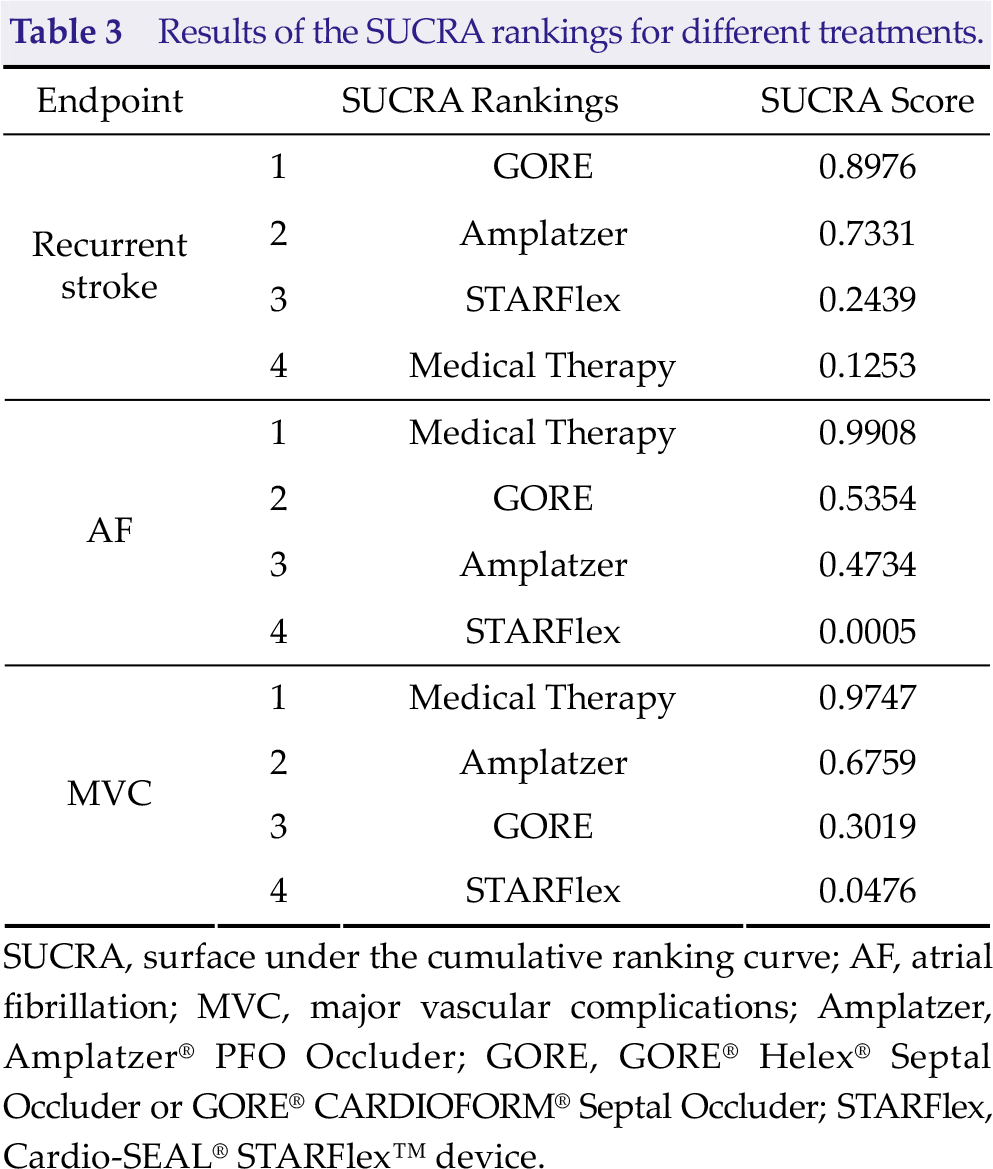
SUCRA, surface under the cumulative ranking curve; AF, atrial fibrillation; MVC, major vascular complications; Amplatzer, Amplatzer® PFO Occluder; GORE, GORE® Helex® Septal Occluder or GORE® CARDIOFORM® Septal Occluder; STARFlex, Cardio-SEAL® STARFlex™ device.
Amplatzer, Amplatzer® PFO Occluder; GORE, GORE® Helex® Septal Occluder or GORE® CARDIOFORM® Septal Occluder; STARFlex, Cardio-SEAL® STARFlex™ device.
3.4 Primary endpoint: AF
Direct and network comparisons: five RCTs were included in the analysis of AF after PFO closure or medical therapy. The network diagram of these studies is depicted in Fig. S4(B). A direct comparison showed that patients who underwent PFO closure generally had an increased incidence of AF compared with patients who received treatment with drugs [Fig. 1(B)]. The STARFlex had an increased risk of AF by more than 10 folds compared with treatment with drugs alone (RR: 11.66; 95% CI: 4.87–21.91; Table 2). As for network calculation, while medical therapy (99.08%) is advantageous in the entire hierarchy, the STARFlex (0.05%) had the lowest rank in the recommended order, which indicated that the use of the device for foramen ovale closure may have an increased risk of AF (Table 3). Quantifying heterogeneity was acceptable (I 2 = 16.2%, τ 2 = 0.1146, Q = 3.58, and p = 0.3106).
3.5 Primary endpoint: MVC
A pair-wise comparison of six studies showed that the STARFlex had an increased risk of vascular accidents in patients with PFO compared with conservative treatment with drugs [RR: 7.63; 95% CI: 2.34–24.88; Fig. 1(C)]. The difference in vascular complications between the Amplatzer device and drug therapy was not statistically significant (RR: 1.95; 95% CI: 0.79–4.79; Table 2). The network diagram of the selected studies is depicted in Fig. S4(C). By contrast, the use of medications (97.47%) had the highest recommendation ranking in the SUCRA score in the network analysis, indicating that it might be safer than surgery for PFO patients in terms of vascular adverse effects. The STARFlex (4.76%) might cause the highest number of vascular complications. Meanwhile, the Amplatzer (67.59%) was considered the safest device (Table 3). Notably, the heterogeneity was relatively high (I 2 = 16.2%, τ² = 0.1146, Q = 3.58, and p = 0.3106). The heterogeneity of MVC was attributed to the GORE REDUCE trial, and after excluding the studies by Søndergaard et al., the network comparison heterogeneity decreased (I 2 = 0%, τ² = 0, Q = 0.13, and p = 0.9348). We speculated that the source of heterogeneity might be correlated to the varying number of individuals included in each study group. Correspondingly, the results of the pairwise and network comparisons and the SUCRA rank findings remained relatively stable (Tables S1 and S2, Fig. S5).
3.6 Secondary endpoint
Headache, TIAs, and major bleeding were the outcomes of the comparison after a long-term follow-up. Some studies included the incidence of the three endpoint events. The network diagrams of the studies about these endpoints are depicted in Figs. S4(D)–(F). A pair-wise comparison of the enrolled studies did not show any difference in the three evaluation index [Figs. 1(D)–(F)]. The indirect comparison results were consistent with those of the direct comparison, which did not indicate any significant difference between all treatments (Table 2). The quantifying heterogeneity was low. The stability and reliability of the combined results were acceptable.
4 Discussion
Study results about whether closure is required for patients with PFO and cryptogenic stroke switched from negative to affirmative. Studies with longer follow-up time and larger sample size make conclusions more reliable. Patients with PFO can benefit from closure. Thus, choosing an appropriate closure device became a problem worthy of attention. At present, only one study has conducted a direct comparison of RCTs of occlusion devices [13], which is insufficient for clinical guidance. Thus, the present study aimed to further compare the advantages and disadvantages of using closure devices, not to reassess the notion whether foramen ovale closure is more effective than drug treatment in preventing the recurrence of cryptogenic stroke. Compared with previous meta-analyses [15, 16], this systemic review and network meta-analysis did not only include a higher number of studies and evaluation indicators to obtain comprehensive conclusions from indirect comparisons but also chose the most suitable and clinically effective closure device, and this is considered the strength of the study. The ideal device for transcatheter PFO closure should facilitate complete and rapid defect closure, and it must be safely, easily implanted and adaptable [17]. Although the device might be associated with coagulation disorders, preventing the formation and shedding of thromboembolism in patients who underwent PFO closure cannot be ignored. PFO closure with the GORE is superior to other devices in terms of preventing recurrent stroke; the STARflex has less protective effects than treatment with medications. This result is consistent with that of previous studies. According to previous reports, both the Amplatzer and the GORE® HELEX® devices can be considered the least thrombogenic [14, 18–20]. However, the STARflex device resulted in frequent thrombus formation [18].
Compared to medical treatment, percutaneous closure, particularly with the STARflex device, was more likely to lead to atrial arrhythmias and MVCs, and this notion has been proven in a previous research [18]. Based on the characteristics of the surgery, we assessed the cause of multiple postoperative AF. Most atrial arrhythmias and MVCs occurred during the surgical and perioperative periods. Residual shunt after closure, left atrial enlargement, and old age might be the risk factors for the increased risk of AF [19]. In addition, compared with the medication group, the perioperative electrocardiogram monitoring of the PFO closure groups had greater accuracy, which might lead to an increase in the detection rate of AF [21]. Therefore, in patients with PFO and cryptogenic stroke, proper monitoring and prevention of AF are important after closure during the perioperative period [22]. PFO occlusion-related AF is usually self-limiting. Perioperative AF events are not likely associated with new strokes because an increase in stroke events was not observed in the interventional occlusion group. The occurrence of MVCs, including bleeding and vascular hematoma formation, might differ between surgical procedures and operators’ manipulations. Moreover, considering that the MVCs vary in different studies and the incidence of adverse events is relatively low, heterogeneity and volatility of statistical results might occur.
No difference was observed in the occurrence rate of headache, TIA, and major bleeding between different therapies. TIA is a transient neurological deficit attributed to focal brain, retinal, or spinal cord ischemia. The clinical symptoms of TIA do not exceed 1 h, and there is generally no evidence of cerebral infarction. Some neurological disorders, such as epileptic seizure, Meniere’s syndrome, and migraine, are also known to recur [23 –25]. TIA lacks a specific diagnosis; thus, accurate assessments are required to prevent misdiagnosis and missed diagnosis. The statistics of the studies about TIA may not be completely reliable. Major bleeding events include spontaneous intracerebral hemorrhage and hemorrhagic transformation of acute ischemic stroke during the anticoagulant or antiplatelet period. Most major bleeding events occur during the medication period [26], which indicate that the process of a simple PFO closure might not increase the risk of bleeding.
As mentioned previously, the STARFlex is commonly associated with the development of thrombus and atrial arrhythmias. Based on previous studies, the main technical difficulty of the device was its nonretrievable nature; thus, complete removal is required to correct its position [18]. During this process, the myocardium and blood vessels could be damaged. Currently, no direct evidence has shown which treatment option is better. Studies with larger sample size and longer follow-up time must be conducted to validate the reliability of this conclusion. The extended follow-up trial of RESPECT in 2017 has shown a diametrically opposite result compared with the study in 2013 [11, 27]. Thus, we only included studies with follow-up periods of more than 2 years. Long-term follow-up and observations are more robust than the evaluation of short-term outcomes.
Oral anticoagulants, single antiplatelet therapy, and dual antiplatelet therapy were included and compared in different medical therapies [16]. These regimens have differential effects on the recurrence of cryptogenic stroke. Since the closure group used anticoagulant and antiplatelet therapy after operation and the therapies included in the study were different, we could not further subdivide the medication group to explore which drug treatment is more effective in this meta-analysis, and this is one of the limitations of this study. However, this current research aimed to identify the most appropriate closure devices via comparison, and a detailed drug treatment can be used as a reference. From a statistical point of view, different kinds of drug treatments do not affect the safety and efficacy of closure devices.
Moreover, in terms of limitation, although more studies were included in the current study than in previous studies, our sample size was still relatively small. Thus, larger multi-arm studies that directly compare different treatments must be conducted to obtain evidence-based medical conclusions. The outcomes included in each study were not completely consistent, and the difference might be attributed to small-sample effects.
5 Conclusion
PFO closure has advantages as it prevents secondary stroke. Among the devices, the GORE and Amplatzer had the best performance with cumulative ranking probability. However, the use of occluders increases the risk of adverse events, such as AF and MVCs, due to occlusion of the foramen ovale. Meanwhile, the STARFlex is the least recommended device as it is most likely to cause adverse events. However, no difference was observed in terms of the occurrence of headache and TIAs between the use of closure devices and treatment with medications.
Supplemental Material
Supplemental Material, BSA20190014_Supplementary_material - Efficacy and safety of percutaneous patent foramen ovale closure devices for recurrent stroke: A systemic review and network metaanalysis
Supplemental Material, BSA20190014_Supplementary_material for Efficacy and safety of percutaneous patent foramen ovale closure devices for recurrent stroke: A systemic review and network metaanalysis by Hao Nie, Yang Hu and Zhouping Tang in Brain Science Advances
Footnotes
Conflict of interests
The authors declare that they have no conflict of interests.
Financial support
This study was supported by grants from the National Natural Science Foundation of China (No. 81471201, No. 81873750).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
