Abstract
Rhythmicity and oscillations are common features in nature, and can be seen in phenomena such as seasons, breathing, and brain activity. Despite the fact that a single neuron transmits its activity to its neighbor through a transient pulse, rhythmic activity emerges from large population-wide activity in the brain, and such rhythms are strongly coupled with the state and cognitive functions of the brain. However, it is still debated whether the oscillations of brain activity actually carry information. Here, we briefly introduce the biological findings of brain oscillations, and summarize the recent progress in understanding how oscillations mediate brain function. Finally, we examine the possible relationship between brain cognitive function and oscillation, focusing on how oscillation is related to memory, particularly with respect to state-dependent memory formation and memory retrieval under specific brain waves. We propose that oscillatory waves in the neocortex contribute to the synchronization and activation of specific memory trace ensembles in the neocortex by promoting long-range neural communication.
Introduction
Ever since bio-electrical signals could be recorded from the brain, this novel technology opened a new era of neuroscience. One can easily record electroencephalograms (EEG, non-invasive), electrocorticograms (ECog, on the dura), or local field potentials (LFP, invasive) for the summation of activities in a large area of the brain. The recording could also be sufficiently precise to catch the firing of each individual neuron in the brain. It allows scientists to not only observe the summation of neuronal activity/synaptic input at a population level, but also observe spike trains at single cell level in super high temporal resolution with sampling frequency up to (currently) 30 kHz. Recoding human EEG, Hans Berger first identified a typical brain rhythm, alpha oscillation [1], followed by many scientists characterizing delta, theta, beta, and gamma waves in different species from rodents to humans, noting the different characteristics of these waves across species (
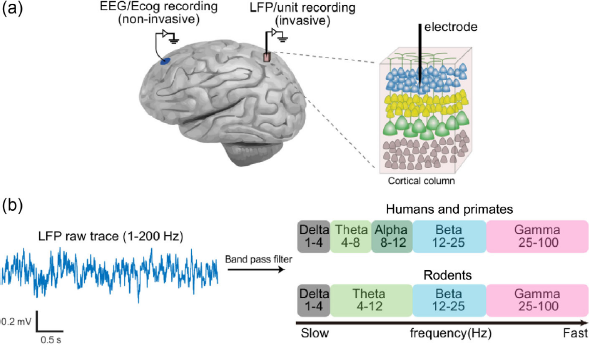
EEG/ECog and LFP recording. (a) Left, EEG/ECog and LFP/unit recording diagram. Right, single electrode recording performed in a specific cortical column. (b) Left, raw LFP sample trace. Right, LFP/EEG signals arbitrarily divided into delta, theta, alpha, beta, and gamma oscillations.
Oscillations can be found in most brain regions including the hippocampus [17], neocortex [9,15,18–32], and thalamus [7,33] and other subcortical regions [34-37]. A typical and well-known oscillation is the hippocampal theta oscillation in rodents (4–12 Hz) [38], which has a macroscopic appearance and can easily be detected in CA1–3 and the dentate gyrus. These oscillations appear predominantly in certain brain states, for example, in humans, delta waves are predominant during sleep while beta waves are predominant during wakefulness [10,39,40]. Therefore, these waves may correlate to unique brain states [41]. Furthermore, studies indicate that distinct brain oscillations correlate to neural system developments [42,43] and high level cognitive functions including social interaction [13,44], language processing [9,45], motor control [9,29,40,46,47], attention [8,9,48-50], sensory information processing [9], working memory [51,52], and long-term memory formation [14,53,54]. Such correlations are present across species from rodents to primates. Although these studies provide insights of how oscillations operate in neural systems, there is still a lack of reliable evidence to establish the causal role of brain oscillations in serving distinct brain functions or to identify the exact information imbedded within these brain oscillations. It is hard to answer the questions, “Are these brain waves functional? If so, what are their functions?” In addition to the correlation between events and brain states, the detailed function and specific mechanisms should be investigated more carefully and meticulously for all brain waves. We will discuss these issues more thoroughly in Section 2.3.
Although a functional role for these brain waves remains plausible, in this paper we review the recent literature concerning the relationship between brain oscillation and memory. An interesting hypothesis is that oscillation may be involved in state-dependent memory and play a critical role in episodic memory formation. It is believed that the basis of memory formation is the change in synaptic strength between memory traces [55,56], which has been found both in the hippocampus [57] and neocortex [58]. Episodic memory is the collection of past personal experiences that occurred at a particular time and place [59]. For example, imagine that you are eating an apple in your office, you remember many features of the scene, the beautiful painting on the wall (visual cortex), music playing from the radio (auditory cortex), and a sweet apple in your hand (somatosensory cortex). These features would activate neurons in the corresponding cortical areas in a particular brain state, representing distinct features and the whole concept of the context. Given the complexity of the episodic context, the formation of episodic memories involves activation of neurons/memory traces in various brain regions [60]. We propose that oscillation probably plays a key role in both memory trace activation in a specific local cortical area and synaptic linkage enhancement between memory traces stored in distant cortical areas.
What signal are we recording?
When performing local field potential and EEG/ECog recordings, we are recording the extracellular potential difference between the recording electrode and reference electrode. Usually the reference electrode is placed at a site without neural activity, for instance, above the blood vessels. The potential difference you record can then be considered to be the potential change at the recording site (
How does the brain generate oscillations?
A periodic pattern in the ongoing electrical brain signal is referred to as an oscillation. How the oscillation is generated remains a basic question. It is believed that LFPs mainly contribute in two ways: one component is the local ion flux while adjacent neurons fire, the other is the induced neuronal EPSP or IPSP from distant or local input to the local region (
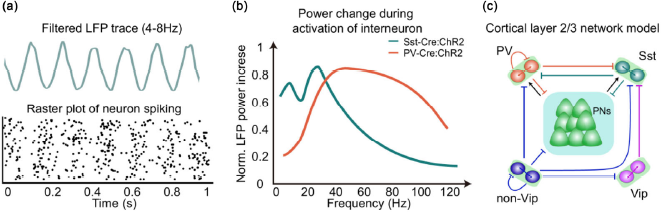
Oscillation generation mechanism. (a) Filtered theta LFP trace and corresponding raster plot of neuronal spiking. (b) Activation of PV+ or Sst+ cortical neuron in Cre-dependent mice induces a corresponding increase in LFP power. However, their frequency sensitivity is different, the Sst+ neuron is more sensitive to the beta band (15–30 Hz) while the PV+ neuron is more sensitive to the gamma band (30–100 Hz). Reconstructing data from Chen et al. [65]. (c) A prevailing circuit model to explain cortical oscillation. PNs, principle neurons.
There does not seem to be a consensus as to whether brain oscillation is functional, which means that the external potential produced by neurons provides functional feedback to nearby neurons. Some scientists have expressed skepticism about the value of brain oscillations, claiming that brain waves and/or EEG/LFP are just epiphenomena of neural normal activity based on serval opinions and pieces of evidence. First, brain oscillations seem to be involved in every type of brain function, even during the development of the neural system. That is to say, it does not have a specific function. Second, the brain wave signal is not as macro as the behavioral level, or micro as the single cell level. Brain waves seem to be a vague phenomenon at the level between them during information transmission. Oscar Herreras highlighted these issues in his recent review: “Indeed, FPs (field potentials) are an epiphenomenon of electrical activity in cell aggregates and as such, spatiotemporal fluctuations may either have a strong relationship or no relationship to the activity in the contributing unit sources.” He proposed that the EEG/LFP signal is a mixture of several confounding factors, containing a mass of information concerning the neuronal firing, remote source regions, cellular basis, and geometry of neuronal structures, among others [83]. It is quite difficult to identify a particular and consistent relationship between the wave and behavior or stimulus input. In fact, Kohn et al. stated that they were unable to obtain a consistent relationship between gamma power and its peak frequency in the monkey visual cortex after performing sophisticated recordings with various visual stimulation [84,85]. This evidence may support that brain waves
On the other hand, evidence has shown that LFPs causally influence neuron spiking activity and cognitive brain function. Regarding spiking, Buzsaki et al. applied transcranial electrical stimulation (TES) to the rat cortex and examined neural activity. They observed that neuronal spike probability is modulated by the induced electric field; neurons are more likely to spike in the trough of the induced electrical field [87]. Recently, Frohlich and McCormick showed a similar result by recording multiunit activity (MUA) and LFPs by applying an electric field to brain sections [88]. Concerning cognitive function, Marshall et al. claimed that brain oscillation is not a mere by-product of neuronal activity but does have an effect on cognitive function. Their research demonstrated that TES-induced slow oscillation enhances declarative memory performance in humans, although its amplitude was quite weak (~100 μV) [89]. Observational results in physiological conditions also support the aforementioned results. Neuronal spike activity was found to be entrained to the theta cycle trough. Neurons were more likely to fire during the down state of field potentials with a slight delay, implying LFPs causally mediate spike probability [90,91]. These reports show that power-weak but effective external potentials can fulfill a functional role. However, we should bear in mind that these results are based on artificial electrical field application and may not represent the normal physiological situation.
Regardless of whether brain waves are epiphenomena or play a functional role in the brain, it is a splendid and simple tool for understanding thought and behavior. Unquestionably, it reflects neuronal firing at a population level, and informs us generally of characteristics such as brain state or pathological diagnoses, such as epilepsy, or even predicts cognitive function, such as memory performance [92]–[94].
Oscillations, brain states, and state-dependent memory
Oscillation and brain state
Humans possess various brain states. The brain state is affected by factors including: emotion, level of consciousness, and centrally acting drugs. Some brain states are always accompanied by specific brain oscillations (
Cortical oscillation is associated with brain state in both humans and rodents.
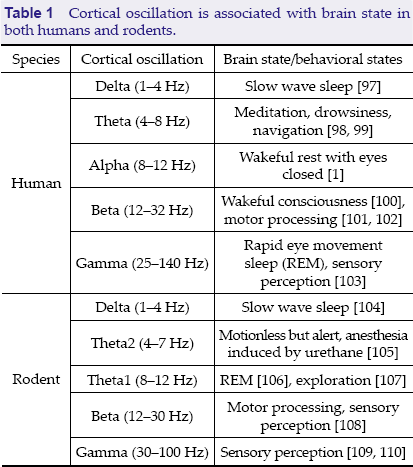
Cortical oscillation is associated with brain state in both humans and rodents.

The diversity of brain state classification for different brain oscillations indicates the varying functions of brain oscillations. Although many states correspond to a dominant oscillation, the actual situation is that one dominant oscillation coexists with other oscillations, e.g., theta oscillation often coexists with gamma oscillation. Accordingly, a brain state is a mixture of various oscillations with one dominant activity band.
Mammals can spontaneously switch between brain states. However, drugs targeting the central nervous system can specifically change the brain state. For example, alcohol abuse can cause a power decrease in the theta and delta bands [95], nicotine causes a power rise in the gamma band [30], and chronic use of ketamine N-methyl-D-aspartate (NMDA) receptor antagonist can lead to an attenuation of power and a power peak shift in the gamma band [96].
Recent research has investigated how memory is regulated by the brain state. Jun and her colleges demonstrated that if a fear memory is encoded in a specific brain state, then this memory can be recalled more efficiently in the same brain state as encoding [111]. It was first demonstrated that low dose MK801 (a non-competitive NMDA receptor antagonist) can induce delta oscillation in the cortex through downregulating activity in the mammillary body (MB). Moreover, directly inhibiting MB activity via chemogenetic inhibition induces cortical delta oscillation in awake mice. These results reveal that activity in the MB is related to the brain state. By controlling the brain state through activity in the MB, they found mice that acquired fear memory in the delta state can only recall in the delta state, and if the fear memory was encoded in the normal state (i.e., theta), this fear memory can then only be recalled in the normal state (
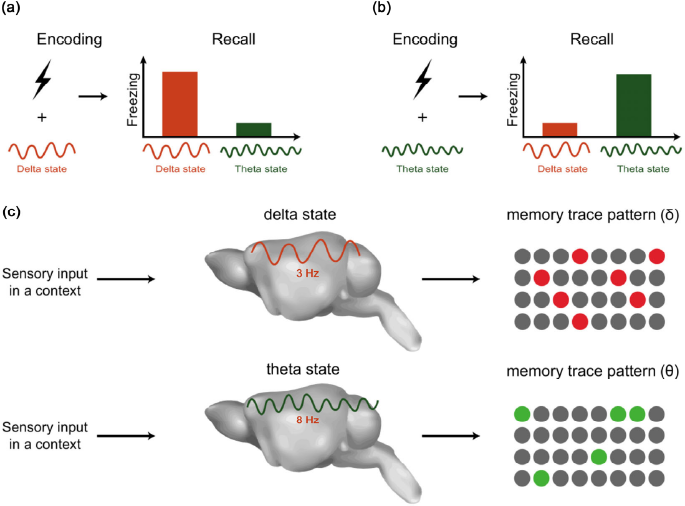
State-dependent memory and memory trace pattern. (a, b) State-dependent fear memory. (a) Fear memory encoding in the delta state and sufficient memory recall in the delta state. (b) Fear memory encoding in the theta state and recall in the theta state. (c) Separate memory trace generated while the internal state interferes with the external stimuli input.
Studies have employed EGFP as a reporter of neuronal activity under the control of the immediate early gene, EGR1 (EGR1-EGFP mice) to investigate cortical neuronal activity in a very large population. We found that distinct memory traces are distributed in many cortical regions and are mainly located in cortical layer 2/3 [60,113]. Different contexts would be reflected in distinct memory trace ensembles. Similarly, several groups have reported cortical memory trace detection. Furthermore, labelling memory traces with ChR2 and reactivating memory traces both in the hippocampal dentate gyrus (DG) or cortical retrosplenial cortex (RSC) region lead to memory recall [56-58]. Therefore, successful memory encoding and recall require a memory trace ensemble pattern to be generated and then reactivation.
As mentioned in Section 3.2, state-dependent memory can only be recalled in the specific state. This implies that local regions generate disparate memory trace patterns during different brain oscillations (
Oscillations, synchronization and memory
What is local synchronization?
When discussing state-dependent memory, we are actually talking about the LFP power of a specific frequency band, which is the amplitude of the oscillation. The amount of co-firing neurons across frequencies determines the power spectrum of the local region. Therefore, the LFP power reflects the degree of synchronization of neuronal firing within a local brain region, referred to as local synchronization (
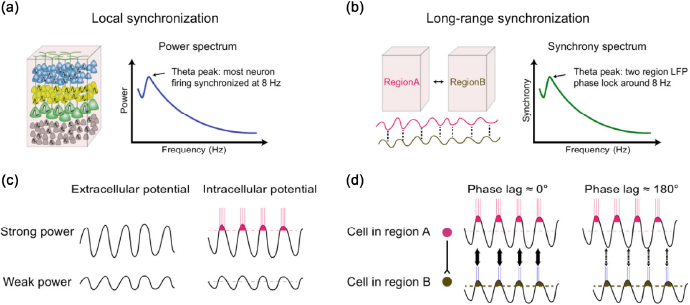
Local synchronization and long-range phase synchronization. (a) Local synchronization. Left, the majority of local neurons fire together at the same frequency. Right, one of the sample power spectra shows a dominant theta power during animal exploration. (b) Long-range phase synchronization. Left, the demonstration of phase relations between two LFPs from two cortical regions. Right, representative synchrony spectrum during animal exploration. (c) Influence of extracellular potential on neuronal firing probability and frequency. Neurons under greater extracellular oscillation power conditions have a higher firing probability at that frequency. (d) Influence of extracellular potential phase on synaptic strength between two cells from two cortical regions. Synchronized LFP with low phase lag promotes synaptic strength while high phase lag does not.
Except for studies of LFP, a number of studies of human cognition are based on EEG recording because of its non-invasive property and the convenience of recording large brain areas. By measuring the event-related potential (ERP) that occurs with various sophisticated tasks, researchers can examine the relationship between the ERP and subject's cognitive function. Researchers continuously record EEG signal in one or more target scalp regions during a specific task, and then align this signal with the onset of each trial to obtain an average EEG trace or average power spectrum related to a specific task. These ERP traces essentially reveal the transient synchronization of synaptic inputs or neuronal firing in large areas under the whole recording site during the task, e.g., during visual stimulation. It reveals the transient response of a brain region. The ERP during cognitive processes has been widely studied, including in attention [114–117], perception [118], language processing [119,120], empathy [121], and memory [122–126]. As an example of a study of attention, Fang et al. showed that the saliency map is created in a V1 region by monitoring the C1 component of the ERP in V1 [115].
There was no direct evidence demonstrating how memory trace activity is mediated by brain oscillations before research published Tanaka and his colleagues this year [127]. Tanaka et al. not only described the relationship between non-memory traces and oscillation in the hippocampus, but also described how memory traces are modulated by theta oscillation in CA1. They labeled activated CA1 neurons (c-Fos+) during the animals’ context exploration and explored the spiking property and oscillation. They found that memory traces are more modulated by theta oscillations. Memory traces tended to burst fire and phase lock to theta oscillation more than non-memory trace cells. Moreover, these c-Fos+ cells carry less spatial information than c-Fos-cells, suggesting these cells may encode information concerning the whole context but not a specific place. Although memory traces in the hippocampus have been characterized, these data only display an observational correlation between theta waves and memory traces. The mechanism of how the network between memory traces, non-memory traces, and interneurons is constructed to underlie memory processing is still unclear. Furthermore, cortical memory traces in layer 2/3 that interact with brain oscillations remain elusive.
However, a computational model has been proposed by Guan et al. in 2016 [113]. In this model, activation of the memory trace causes the initiation of a local oscillator that leads to a specific frequency power enhancement. This synchronization of the local regional neuronal firing can temporally affect the information coding in downstream targets. Inversely, the oscillatory input from layer 4 and layer 1 to layer 2/3 initiates the distinct oscillation of memory traces. This conversion of temporal information to spatial neuronal patterns may be one of the potential mechanisms of how oscillations contribute to memory trace activation specifically. Another idea has been provided by Capogna et al. [128]. In their review, despite the focus on memory trace activation in the amygdala, they proposed that theta and gamma oscillation synchronization in the basolateral amygdala with interconnected areas during fear and reward memory retrieval is critical to generating specific memory trace patterns. This reliable synchrony enhances the spike probability of memory traces that are active in the amygdala and receive input from interconnected regions simultaneously. Even so, experimental data are required to prove these ideas.
Long-range phase synchronization
The benefits of superior multichannel recording technology [129-131] and the establishment of mathematical calculations for the phase of discrete biological signals [132,133] have enabled scientists to inspect the phase relationship between brain regions. The phase relationship is revealed by calculating the synchrony level, which is the phase difference between two LFP/EEG signals (
Long-range phase synchronization and synaptic plasticity
Neurons are large, tree-like structures, whose axon and dendrites are capable of spanning over 1,000 mm. Such a complex structure endows neurons in distinct regions the ability to exchange information over long distances. It has been proposed that long-range cell ensemble phase synchronization contributes to synaptic strength. Synchronized waves in two regions with low phase lag cause the pre-synaptic cell and post-synaptic cell to fire simultaneously or with a small delay. According to the Hebbian rule, “cells that fire together, wire together” and hence, synaptic strength between two cells is strengthened (
Long-range synchronization in episodic memory formation
As mentioned previously, numerous studies suggest that episodic memories are stored and distributed throughout the brain. Memory traces can be found in various sub-regions of the neocortex, dentate gyrus, and amygdala. Based on this background, a central question is how does the brain orchestrate the symphony of different brain regions with distinct functions that are distributed across the brain to effortlessly form a memory trace network? One promising clue to the problem is the long-range neural oscillation synchronization which is correlated with perceptual binding and may play a role in transient, long-range coordination of distinct brain regions [71,141-144]. As early as 2001, R. T. Knight pointed out that theta synchronization across the cortex in humans may serve a coordinating function by widely recording 64-channel ECog over the temporal and frontal cortex [103]. Buzsaki's research previously demonstrated similar theta synchronization in rodents [145]. Moreover, theta phase synchronization enables the interaction of gamma oscillation entrainment between regions [14]. This evidence suggests that large scale phase synchronization in the theta band coordinates neurons interconnecting over the cortical network. Furthermore, this process may coordinate the connection between memory traces in episodic memory processing. Observational evidence shows that EEG/ECog theta synchronization at the neocortical level both during memory encoding and retrieval is positively correlated to the performance of the memory task in humans [146-148]. Recently, Hanslmayr et al. demonstrated the causal effect of cortical synchronization on association memory [14]. They “manipulated” the synchronization between the auditory and visual cortex by changing the amplitude of auditory and luminance of visual stimuli, and found that during the epoch of reliable synchrony, the subject obtained better association memory, while exhibiting poor memory with poor synchrony. In the current case, they manipulated the synchrony by changing the stimulus properties and not directly manipulating the electrical potential in related brain regions. Thus, more experimental data are still required for dissecting the causality between oscillation and memory.
Animal experimental data unveiled the importance of the theta wave; a lack of theta oscillation leads to an episodic memory formation deficit. Lesions of the medial septum lead to large attenuation of the theta wave in the hippocampus, causing a serve deficit of spatial memory formation [149,150]. However, directly artificially inducing theta waves in the hippocampus using an optogenetic method is unable to rescue the memory formation deficit caused by medial septum lesions [150]; meanwhile, theta coupling between the neocortex, entorhinal cortex, and hippocampus is required [5,26,151]. Taken together, successful memory encoding requires theta and gamma entrainment in the interaction between the medial temporal lobe and neocortex.
Conclusion
This paper reviewed recent studies that investigated the potential role of oscillations in memory processes. First, we introduced the methodology and basic concepts of neural electrophysiology and how it can be utilized in brain cognitive function research, including that of episodic memory. Then, we focused on the properties and generation mechanisms of oscillations, followed by the discussion of brain states and state-dependent memory. Lastly, we discussed investigating its role in neuronal spike probability and timing, coordination of larger scale brain region interactions, and memory trace interconnection, covering local synchronization and long-term phase synchronization. In conclusion, the generation of oscillations in neural networks by the interplay of inhibitory and excitatory neurons is meaningful for cognitive function. It determines the brain state framework and interferes with external stimuli or internal signals to generate separate memory trace ensembles in each local region, while the phase synchronization of this oscillation provides the opportunity for interconnection of memory traces. However, memory processes remain to be further investigated. Future directions may include investigating the oscillations more specifically in the neocortex and medial temporal lobe for the laminar structure of oscillation properties and their interconnection dynamics, and the mechanisms by which these oscillations contribute to memory selection in layers 2 and 3.
Footnotes
All contributing authors have no conflict of interests.
This paper is jointly supported by grants from NSFC (Nos. 31371059 and 31671104). The work was partially funded by the National Natural Science Foundation of China and the German Research Foundation (DFG) in project Crossmodal Learning, NSFC (61621136008)/DGF TRR-169 to J.-S. G. J.-S. G. is supported by Beijing Nova program (2015B057).
