Abstract
Background
Epilepsy surgery improves seizure outcomes in Sturge-Weber syndrome (SWS), yet the electrophysiological patterns of postoperative recovery remain poorly characterized. Previous studies suggest that early intervention may yield distinct clinical trajectories, but the associated changes in EEG background activity have not been systematically investigated.
Objective
To investigate whether age of surgery influences the postoperative modulation of EEG background rhythms in children with unilateral SWS, and to evaluate the utility of preoperative EEG asymmetry for lateralizing the epileptogenic hemisphere.
Design
Retrospective cohort study.
Methods
We analyzed children with unilateral SWS who underwent epilepsy surgery, stratified by age at intervention (<2 vs. ≥2 years). Pre- and postoperative scalp EEGs were visually assessed to quantify posterior dominant alpha frequency and slow-wave (delta and theta bands) activity separately in the affected and unaffected hemispheres (AH, UH). Preoperative lateralization accuracy was also evaluated for both frequency bands.
Results
A total of 99 patients were included. Seizure freedom rates were comparable between age groups. However, younger patients exhibited a significantly greater postoperative increase in alpha frequency, particularly in the UH (20 ± 20% vs. 4 ± 10%,
Conclusion
Early epilepsy surgery in SWS is associated with enhanced postoperative modulation of alpha frequency in the UH, possibly reflecting greater neuroplastic capacity during early development. Preoperative alpha asymmetry offers robust lateralizing value. These findings support the clinical utility of background EEG analysis in surgical planning and postoperative monitoring.
Introduction
Sturge-Weber syndrome (SWS) is a neurocutaneous disorder characterized by a high incidence of early-onset, drug-resistant epilepsy. 1 Early seizure onset and high seizure burden are consistently associated with poor cognitive outcomes in SWS. 2 For patients with intractable epilepsy involving large or hemispheric lesions, surgical resection is a crucial therapeutic strategy. 3 At our epilepsy center, previous data indicated that epilepsy surgery achieved seizure freedom in up to 83.33% of SWS cases, 4 which was further validated in our subsequent multicenter cohort. 5
Notably, our prior research revealed significant differences in seizure outcomes and cognitive function depending on whether surgery was performed before or after 2 years of age.4,5 However, the underlying neurophysiological basis for these age-related differences remains unclear, particularly regarding how surgical timing influences fundamental brain activity as reflected in EEG background rhythms—a widely used, noninvasive marker of cortical function and development.
Electroencephalography (EEG) is a fundamental tool for assessing brain electrical activity. It captures a spatiotemporally filtered representation of the local field potential, primarily reflecting the summated postsynaptic potentials from large populations of synchronously active neurons. 6 EEG signals are composed of oscillations across multiple frequency bands, each linked to specific functional states and cognitive processes, that encode distinct information and support specialized computation within neural networks. 7 Clinical EEG interpretation relies on evaluating background activity, including the posterior dominant rhythm, slower background frequencies, and elements of sleep architecture. Changes in these background rhythms reflect widespread alterations in cortical function and integrity. 8
Among the various EEG frequency components, the posterior dominant rhythm (PDR), typically in the alpha range (8–13 Hz in older children and adults, 5–9 Hz in infants), 9 serves as a direct readout of thalamocortical network integrity and synaptic maturation.10,11 Developmentally, the progressive acceleration of alpha frequency is tightly coupled to white matter myelination. 12 In pathological conditions such as epilepsy, chronic seizure activity and structural lesions disrupt thalamocortical circuits, leading to background slowing and disorganized rhythms.13,14 Conversely, surgical removal of epileptogenic tissue can reverse pathological cascades. In pediatric populations, heightened neuroplasticity enables recovery of physiological network dynamics following resection, including normalization of background rhythms and restoration of functional connectivity in regions relieved from chronic epileptic activity. 15
Despite the well-established efficacy of epilepsy surgery in improving clinical outcomes for patients with SWS, it remains unclear to what extent surgical intervention modulates EEG background rhythms, and whether the effects of surgery differ by age at surgery. To address this knowledge gap, we conducted a retrospective study of patients with unilateral SWS to investigate how surgical timing may influence postoperative modulation of EEG background rhythms. Patients were stratified by age at the time of surgery (<2 years vs. ≥2 years), and both preoperative and postoperative EEGs were analyzed. Background activity was evaluated separately for the affected hemisphere (AH) and the unaffected hemisphere (UH), focusing on key components such as the posterior dominant rhythm and slow wave activity. By examining these electrophysiological changes in relation to surgical timing, the study aims to elucidate potential neurophysiological mechanisms underlying the differential epilepsy and cognitive outcomes previously observed between younger and older surgical cohorts. This work may offer novel insights into how early intervention may interact with critical windows of brain plasticity in children with SWS, and whether electrophysiological changes can serve as surrogate markers of network recovery.
Materials and Methods
An overview of the study design is illustrated in Figure 1. Patients with unilateral Sturge-Weber syndrome (SWS) who underwent epilepsy surgery were retrospectively categorized into two cohorts based on age at the time of surgery: a younger cohort (<2 years at time of surgery) and an older cohort (≥2 years). Scalp EEG recordings were collected at two standardized timepoints: during the preoperative assessment (within 3 months prior to surgery) and at the 1-year postoperative follow-up. EEG data were analyzed separately from both the affected hemisphere (AH) and the unaffected hemisphere (UH). EEG background rhythms—alpha (8–13 Hz), theta (4–7 Hz), and delta (1–4 Hz) activity—were identified through expert visual inspection. Overview of study design and EEG background rhythm analysis.
To systematically quantify the changes in EEG background rhythms associated with surgery, a multidimensional analytic framework was applied, incorporating: (i) a spatial dimension (AH vs. UH), (ii) a temporal dimension (preoperative vs. postoperative), and (iii) a modulation dimension, assessing the relative change in each EEG feature, with the following formula described later. This analytic approach enabled systematic evaluation of the impact of surgical timing on neurophysiological plasticity, as reflected by alterations in EEG background rhythms, and provided a robust framework for quantifying interhemispheric differences in recovery dynamics.
Patient Selection
We retrospectively analyzed patients with SWS who underwent epilepsy surgery at Sanbo Brain Hospital from January 2008 to January 2024. Inclusion criteria were as follows: (i) a diagnosis of unilateral SWS confirmed by clinical presentation, neuroimaging, and electroencephalographic findings; (ii) underwent resective epilepsy surgery (focal resection or hemispheric surgery); (iii) availability of preoperative scalp EEG recorded within 3 months prior to surgery and postoperative EEG obtained 12 months after surgery; (iv) clinical follow-up data available at 12 months postoperatively, including seizure outcome assessment; and (v) written informed consent provided by the patient’s legal guardians. Patients were excluded if they were lost to follow-up within 12 months after surgery or if either preoperative or postoperative EEG data were unavailable.
Operative Procedure and Follow-Up
All surgical strategies were determined through multidisciplinary case conferences at the Epilepsy Center, with the goal of achieving complete resection of the epileptogenic zone while preserving functional areas. Surgical planning was based on comprehensive preoperative assessments, including clinical evaluation, scalp EEG, MRI, and additional investigations as necessary. For standardized analysis, both interictal and ictal EEG findings were classified as regional (≤2 involved lobes) or multiregional (>2 involved lobes).
Two primary surgical approaches were employed: focal resection (FR) and hemispheric surgery (HS). All patients were followed for a minimum of 1 year postoperatively. Seizure outcomes at 1-year follow-up were assessed using the Engel Epilepsy Surgery Outcome Scale. 16
Age Stratification
Patients were divided into two cohorts based on their age at the time of surgery: the younger cohort (<2 years) who underwent surgery before 2 years of age, and the older cohort (≥2 years) who received surgery at or after 2 years.
Identification of EEG Visual Features
Scalp EEG recordings were acquired using a 64-channel Nicolet system with electrode placement based on the International 10–20 system. Data were sampled at either 512 Hz or 1024 Hz, with electrode impedance maintained below 5 kΩ. During acquisition, signals were referenced online to a scalp electrode near Cz.
All features were visually assessed using a bipolar double-banana montage. A 0.5-100 Hz bandpass filter was applied. To ensure objective evaluation, all EEG recordings were independently rated by two board-certified epileptologists (W.J. and W.M.Y.) who were blinded to age group, surgical type, surgical side, and all postoperative outcomes. Discrepancies were resolved by consensus, and consensus ratings were used for all subsequent analyses. To quantify inter-rater reliability, we randomly selected 30 patients (60 hemispheres, representing 30.3% of the full cohort) and calculated Cohen’s kappa coefficients and percentage agreement for alpha, theta, and delta background activity in both the UH and AH. The results are summarized in Supplemental Table 1.
During resting-state wakefulness, posterior dominant rhythm (PDR) was evaluated using age-appropriate frequency criteria: for children < 2 years, a broader low-frequency band (5-9 Hz) was used to account for developmental immaturity; for children ≥ 2 years, the conventional alpha range (8-13 Hz) was applied. To differentiate physiological PDR from overlapping background theta activity (particularly in children < 2 years), we applied strict criteria based on topography, reactivity, and morphology. PDR was defined as sinusoidal, reactive rhythms maximally prominent over posterior regions (O1/O2, P3/P4), distinct from diffuse or irregular pathological theta activity. Slow wave activity, including delta (0.5-4 Hz) and theta (4-8 Hz) bands, was assessed based on amplitude and spatial distribution. The center frequencies of alpha and slow wave activity obtained from visual inspection were used in subsequent quantitative comparisons between age groups and hemispheres.
Modulation of EEG Background Rhythms
To analyze the impact of surgery on EEG background rhythms, each patient’s brain was divided into the AH and the UH based on comprehensive clinical and neuroimaging evaluations.
Quantitative analysis focused on changes in the center frequency of background rhythms from the preoperative to the 1-year postoperative EEG. The degree of modulation was expressed as the percentage change in center frequency, calculated using the following formula:
Here, Frequencypre denotes the center frequency of the rhythm in either the AH or UH before surgery, and Frequencypost corresponds to the frequency at the 1-year postoperative follow-up. This index was used purely as a relative effect size to compare the magnitude and direction of pre-to postoperative change between age groups, without implying a linear physiological relationship between alpha frequencies. These modulation values were then used to compare postoperative changes in EEG background rhythms between the younger (<2 years) and older (≥2 years) surgical cohorts.
Statistical Analysis
Patients were stratified into two groups based on age at the time of surgery: a younger cohort (<2 years) and an older cohort (≥2 years). Continuous variables were summarized as mean ± standard deviation (SD) for normally distributed data, or as median with interquartile range (IQR) for non-normally distributed data. Categorical variables were summarized as counts and percentages (n [%]). The Normality was assessed using the Shapiro–Wilk test.
To assess age-related differences in postoperative modulation of EEG background rhythms, percentage changes in the center frequency of rhythms were compared between the younger and older cohorts, separately for the unaffected hemisphere (UH) and affected hemisphere (AH). Between-group comparisons were conducted using unpaired two-tailed t-tests. Analyses were performed in the overall cohort and further stratified by surgical type (focal resection [FR] vs. hemispheric surgery [HS]).
The concordance between EEG-based lateralization and the actual surgical hemisphere was evaluated using the chi-square (χ2) tests and Cohen’s kappa coefficient (
The false discovery rate (FDR) correction was applied to account for multiple comparisons, and P < 0.05 (FDR-adjusted) was considered statistically significant. All statistical analyses were conducted using Python, version 3.0.
Results
A total of 99 patients with SWS were enrolled after the application of strict inclusion and exclusion criteria (Figure 2). Based on age at the time of surgery, patients were stratified into a younger cohort (<2 years, n = 53) and an older cohort (≥2 years, n = 46). Flowchart of patient selection and surgical classification.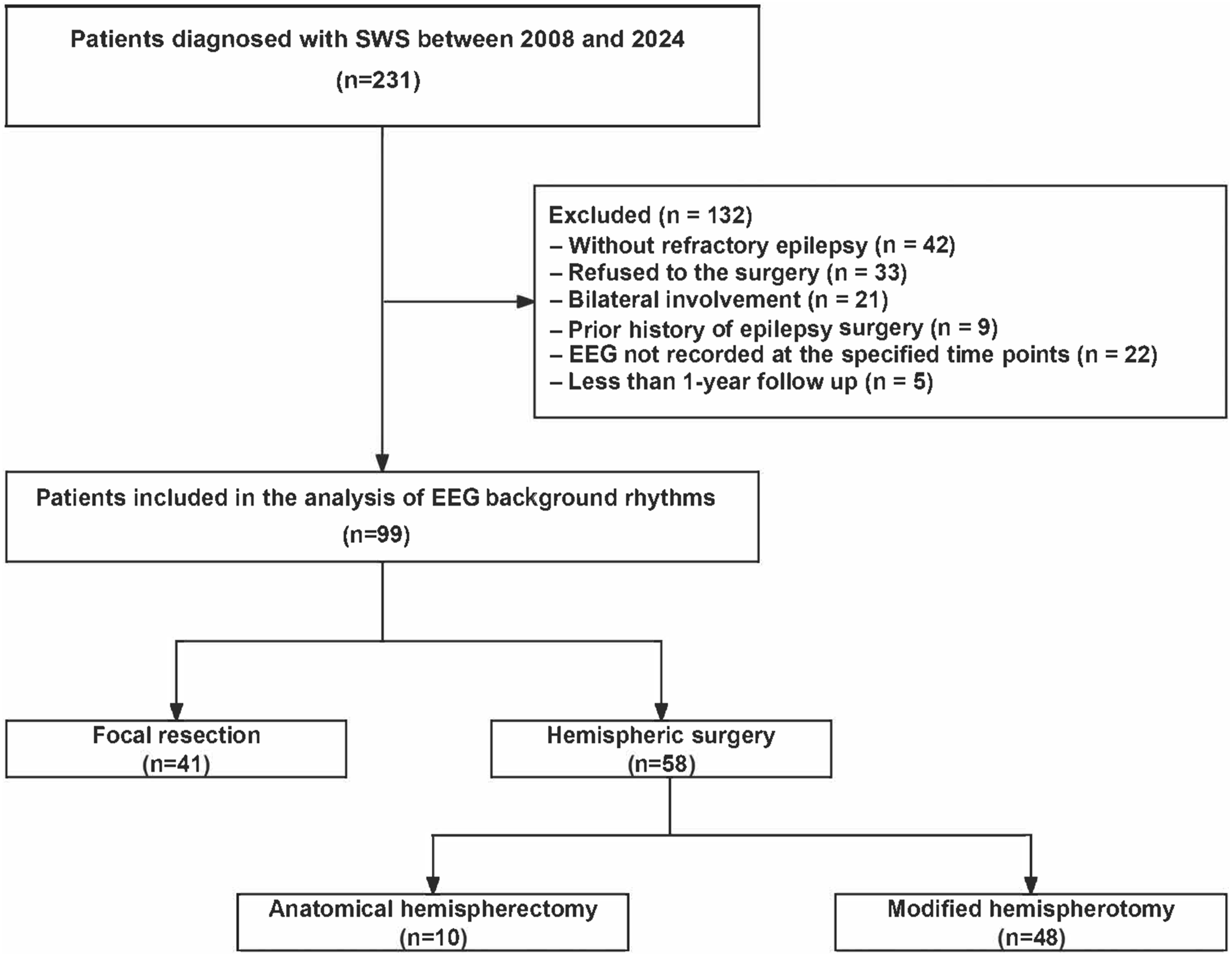
Clinical Characteristics
Clinical Characteristics of Patients With SWS

No significant differences were found between groups with respect to seizure type (focal vs. generalized,
Surgical laterality (right vs. left hemisphere) did not differ significantly between groups (
Preoperative Lateralization of EEG Background Rhythms
Preoperative Lateralization of EEG Background Rhythms

PPV: Positive Predictive Value, NPV: Negative Predictive Value.
For alpha activity, the lateralization demonstrated a high level of concordance with the surgical side (χ2 < 0.001). This yielded an overall accuracy of 86.9%, with a sensitivity (for left-sided surgery) of 85.7%, specificity (for right-sided surgery) of 88.0%, positive predictive value (PPV) of 87.5%, negative predictive value (NPV) of 86.3%, and a Cohen’s κ coefficient of 0.74, indicating substantial agreement.
For slow wave activity, lateralization remained statistically significant (
Collectively, these findings indicate that alpha rhythm asymmetry is a more reliable and consistent lateralizing marker compared to slow wave asymmetry in determining the hemisphere requiring surgical intervention.
Postoperative Modulation of EEG Background Rhythms
To explore whether age at surgical intervention influences postoperative neurophysiological adaptation, we analyzed changes in EEG background rhythms stratified by age groups (<2 years vs. ≥2 years). Given the heightened neuroplastic potential in early developmental stages, we specifically examined modulation in alpha activity and slow wave features to evaluate age-dependent effects on neural recovery.
While we report data for both hemispheres, our primary metric for neurodevelopmental recovery is the unaffected hemisphere (UH). This focus is essential because in patients undergoing extensive hemispheric surgery (HS), the affected hemisphere (AH) is functionally disconnected or resected, rendering its postoperative rhythm less reflective of physiological maturation and more susceptible to volume conduction or residual isolation effects.
Modulation of Alpha Center Frequency
To quantify age-related differences in alpha rhythm modulation following surgery, we calculated the percentage change in alpha center frequency from the preoperative baseline to the 1-year postoperative follow-up. Group comparisons were made between younger (<2 years) and older (≥2 years) cohorts (Figure 3; Supplemental Table 1). Modulation of alpha center frequency in surgical patients.
In the overall cohort (n = 99), both the UH and AH exhibited significantly greater alpha modulation in the younger cohort. Specifically, the UH alpha frequency increased by 20 ± 20% in younger patients compared to 4 ± 10% in older patients (
When stratified by surgical type: in the focal resection (FR) group (n = 41), the UH showed significantly greater modulation in younger patients (17 ± 12%) than in older patients (1 ± 8%;
These findings indicate that younger children, particularly those undergoing surgery before age two, demonstrate robust modulation of alpha activity postoperatively—most notably in the UH. This enhanced alpha modulation likely reflects greater neuroplastic potential during early brain development, underscoring the importance of early surgical intervention in optimizing functional recovery.
Modulation of Slow Activity
Postoperative changes in slow wave activity were quantitatively assessed and compared between the younger and older cohorts (Figure 4; Supplemental Table 2). Modulation of slow wave center frequency in surgical patients.
In the overall cohort (n = 99), modulation of slow wave activity in the UH did not differ significantly between the younger (5 ± 15%) and older (4 ± 18%) cohorts (
When stratified by surgical type: in the FR group, there were no significant age-related differences in slow wave modulation in either the UH (younger: 4 ± 14% vs. older: 2 ± 22%,
These findings indicate that, unlike alpha rhythms, slow wave activity shows only limited age-related modulation following surgery. The lack of consistent modulation across surgical subtypes suggests that slow wave reorganization may be less sensitive to developmental timing than alpha oscillations.
Discussion
Our findings demonstrate that age at the time of surgery emerged as a critical determinant of postoperative electrophysiological recovery in Sturge–Weber syndrome (SWS). Children undergoing surgery at a younger age (<2 years) showed a marked postoperative increase in background alpha rhythms, most prominently in the unaffected hemisphere (UH). This age-dependent acceleration was substantially greater than that observed in older children (≥2 years), consistent with a time-limited opportunity to reverse epilepsy-related network dysfunction and support more typical developmental trajectories. By contrast, slow-wave activity exhibited relatively little postoperative change, suggesting it may reflect more enduring structural pathology.
Surgical Timing in SWS
SWS is a clinically heterogeneous neurocutaneous disorder that often requires individualized and time-sensitive management. 17 Epilepsy is its most common neurological manifestation and typically begins within the first year of life. 18 Ongoing seizures are a major risk factor for subsequent cognitive and behavioral impairments, thereby underscoring the need for early and effective intervention. 3 EEG is central to diagnostic assessment and longitudinal management in patients with early-onset epilepsy. 19 For individuals who remain drug-resistant after adequate trials of at least two appropriately selected and tolerated anti-seizure medications, referral for comprehensive surgical evaluation is recommended. 20 In SWS, epilepsy surgery is an established option for drug-resistant cases, with favorable seizure outcomes and an acceptable safety profile, and has been associated with improved longer-term developmental trajectories in selected patients.5,21
In our prior work, surgery before 2 years of age was associated with better seizure control and cognitive outcomes than later intervention.4,5 Building on that observation, we hypothesized that earlier surgery may create a more permissive neurophysiological milieu for network reorganization. Our current findings are consistent with this hypothesis. Younger children exhibited a larger postoperative increase in alpha frequency, most notably in the UH. Given the rapid maturation of alpha rhythms in early childhood, part of this increase likely reflects normal development. 22 However, the disproportionate UH acceleration suggests release from epilepsy-related suppression and a resumption of a more typical maturational trajectory. Because we did not collect contemporaneous longitudinal neuropsychological measures in this cohort, any direct inference from EEG modulation to cognitive recovery remains indirect. Nonetheless, these data support the view that timely intervention may maximize the opportunity for functional network recalibration by reducing epileptic burden during an age-sensitive period of brain development.
Alpha Frequency Modulation and Neuroplasticity
The posterior dominant alpha rhythm is a core feature of the awake human EEG and carries clinically meaningful information in its amplitude, phase, and—particularly—its frequency. 23 Although historically viewed as an “idling” rhythm, alpha activity is now understood to support cognition, including attention, memory, and sensory integration, 24 by shaping the timing of neural excitability and coordinating communication across distributed networks. 25 Peak alpha frequency is not a fixed property: it shows substantial heritability and is influenced by genetic factors affecting thalamocortical circuitry, and it increases with age along a nonlinear developmental course, with the steepest gains in early childhood and a plateau in adolescence.9,26,27
Against this background, we interpreted postoperative alpha frequency modulation through both maturational and functional lenses. Consistent with normative development, children operated on before age two showed larger increases in alpha frequency, most prominently in the UH. While part of this effect likely reflects expected age-related acceleration, the disproportionate UH change suggests that reducing epileptic burden may release the structurally preserved hemisphere from remote suppression, enabling a “catch-up” trajectory of thalamocortical rhythmogenesis.
Alpha oscillations have also been proposed to provide a temporal framework for perception and cognition, 28 such that faster alpha frequencies are associated with finer temporal precision in sensory perception. 29 In that context, the postoperative increase in UH alpha frequency—especially in infants—may indicate restoration of network dynamics that are permissive for later cognitive development. However, because contemporaneous longitudinal neuropsychological assessments were not available in this cohort, any direct inference from alpha modulation to cognitive improvement should be considered provisional.
Although alpha frequency also increased in the AH, its interpretation is inherently constrained after extensive surgery. Postoperative AH signals may be influenced by reduced generator volume, disconnection, residual isolated cortex, or volume conduction from the contralateral side. For these reasons, UH alpha frequency modulation is likely the more robust electrophysiological marker of postoperative functional recovery in SWS.
Postoperative Slow Wave Modulation
Slow background activity—most notably in the delta (0.5-4 Hz) and theta (4-8 Hz) bands—is a routine component of waking EEG interpretation and is highly sensitive to cortical dysfunction. Across a range of neurological conditions, excess focal or hemispheric slow activity is commonly interpreted as a marker of impaired cortical excitability and disrupted large-scale network organization.30,31 In SWS, this physiology is plausibly linked to the underlying angioma burden and its downstream effects, including chronic hypoperfusion and progressive parenchymal injury. Consistent with this framework, our preoperative analyses showed that slow-wave asymmetry provided clinically useful lateralizing information, with a moderate classification accuracy (66.7%), supporting its role as an adjunct marker of hemispheric compromise in surgical evaluation.
In contrast to the more consistent postoperative changes observed in alpha frequency, slow-wave modulation after surgery was modest and showed limited age dependence. In the UH, postoperative changes in slow-wave power did not differ meaningfully between younger and older children. This relative stability likely reflects the mixed biological meaning of slow activity in pediatrics: delta and theta rhythms are expected to be more prominent early in life, yet they can also index persistent structural and metabolic disturbance. These overlapping interpretations reduce the sensitivity of slow-wave measures for detecting postoperative functional recalibration, particularly over a one-year interval.
Taken together, slow-wave activity is most informative for characterizing preoperative hemispheric dysfunction and lesion burden, whereas alpha frequency modulation—especially in the UH—better captures dynamic, age-sensitive aspects of postoperative network recovery. Accordingly, slow-wave metrics may complement, rather than substitute for, alpha-based markers when tracking neurophysiological restoration after epilepsy surgery in SWS.
Limitations
Several limitations should be acknowledged. First, the retrospective design limits causal inference, and unmeasured confounding—such as heterogeneity in vascular malformations, cumulative seizure burden, or comorbid neurodevelopmental conditions—may have influenced both surgical timing and EEG trajectories. Second, scalp EEG has limited spatial resolution and may underestimate the contribution from deeper cortical or subcortical structures. Third, although the sample is relatively sizable for a rare pediatric disorder, subgroup analyses—particularly those stratified by surgical type or hemisphere—may have been underpowered to detect more subtle interactions. Finally, this cohort was drawn from a single center with a specialized pediatric epilepsy surgery program. As such, referral and selection biases are likely: patients evaluated and operated on at our center may differ systematically from the broader SWS population in disease severity, lesion laterality, access to care, and family preference, and our surgical expertise and perioperative pathways may not be fully generalizable to other centers.
Despite these limitations, our findings provide clinically relevant evidence that EEG background rhythms—especially alpha frequency in the UH—may serve as sensitive electrophysiological markers of early postoperative network recovery. Prospective, multicenter longitudinal studies incorporating standardized referral criteria, multimodal imaging, and repeated neurocognitive assessments are needed to validate generalizability and to clarify the relationship between EEG modulation and functional outcomes.
Conclusion
In summary, postoperative modulation of EEG background rhythms in children with SWS varies by age at intervention, with the most prominent age-related effects observed in alpha frequency. Surgery performed before two years of age was associated with a greater postoperative increase in alpha frequency in the UH, consistent with greater capacity for developmental network recalibration early in life. These results support the use of background EEG rhythms as practical, noninvasive markers to complement pre- and postoperative assessment and to help inform surgical timing. Future prospective studies integrating EEG, neurodevelopmental assessments, and long-term outcomes are needed to clarify how EEG changes relate to functional outcomes to refine individualized treatment strategies for pediatric SWS.
Supplemental Material
Supplemental Material - Modulation of EEG Background Rhythms in Sturge-Weber Syndrome: Impact of Surgical Timing on the Unaffected Hemisphere
Supplemental Material for Modulation of EEG Background Rhythms in Sturge-Weber Syndrome: Impact of Surgical Timing on the Unaffected Hemisphere by Jing Wang, Kun Lv, Zeming Wang, Xiongfei Wang, Yuguang Guan, Jian Zhou, Tianfu Li, Mengyang Wang, Yujiao Yang, Guoming Luan in Journal of Central Nervous System Disease.
Supplemental Material
Supplemental Material - Modulation of EEG Background Rhythms in Sturge-Weber Syndrome: Impact of Surgical Timing on the Unaffected Hemisphere
Supplemental Material for Modulation of EEG Background Rhythms in Sturge-Weber Syndrome: Impact of Surgical Timing on the Unaffected Hemisphere by Jing Wang, Kun Lv, Zeming Wang, Xiongfei Wang, Yuguang Guan, Jian Zhou, Tianfu Li, Mengyang Wang, Yujiao Yang, Guoming Luan in Journal of Central Nervous System Disease.
Footnotes
Acknowledgements
We extend our sincere gratitude to all the patients whose data were included in this study. We would also like to thank the dedicated clinical and EEG laboratory teams, as well as the staff and technicians of the Epilepsy Center at Sanbo Brain Hospital, for their invaluable assistance in patient identification and data collection.
Ethical Considerations
This study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Ethics Committee of Sanbo Brain Hospital, Capital Medical University (Approval No. SBNK-YJYS-2025-052-01).
Consent to Participate
Written informed consent was obtained from all patients or their legal guardians.
Author Contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (Grant No. 82471476 and 2022ZD0205000), the China Association Against Epilepsy Research Fund–UCB Pharma Fund (CU-2023-052), the Laboratory for Clinical Medicine, Capital Medical University (2025LCMCMU02).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets generated and/or analyzed during the current study are not publicly available due to patient privacy but are available from the corresponding author on reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
