Abstract
Recent years have witnessed the fascinating development of imaging approaches to studying neural activities; this progress has been based on an influx of ideas and methods from molecular biology and optical engineering. Here we review the design and application of genetically encoded indicators for calcium ions, membrane potential and neurotransmitters. We also summarize common strategies for the design and optimization of genetically encoded neural activity indicators.
Introduction
Imaging the activity of a specific group of neurons in real time has long been a goal of neuroscientists. Many chemical indicators have been developed to monitor neuronal activities by converting intrinsic parameters of neuronal activity into optical signals such as fluorescence changes.
A major goal of neuroscience is to determine the precise functions of different neuronal cell types [1,2]. However, except for a few semi-genetically encoded indicators, chemical indicators are unable to target specific cell types in the living mammalian brain [3]. Furthermore, the loading procedure has limited the usage of chemical indicators. Genetically encoded neural activity indicators (GENAIs) are a class of biological molecules that can indicate neural activity, usually through changes in fluorescence. Compared with synthetic chemical indicators, GENAIs can be expressed in cells, thus facilitating long-term observation of individual cells [4] while enabling the targeting of specific cell types through genetic techniques.
GENAIs can be classified into several groups according to their usage (
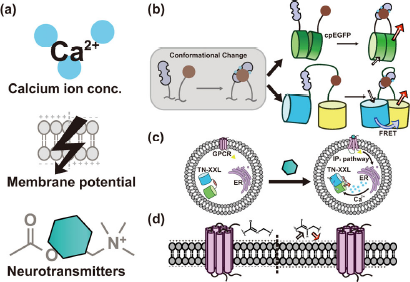
Classification and common strategies of neural activity indicators. (a) GENAIs can be classified into calcium indicators (GECIs), voltage indicators (GEVIs) and synaptic transmission indicators (GESTIs) by their target physical parameter. These signals could be transformed into fluorescence signal by (b) amplifying the signal induced structural variation by conformation-sensitive fluorescence protein such as circularly permuted FP (cpFP, upper) or fluorescence resonance energy transfer (FRET, lower). (c) Hybrid neurotransmitter indicator CNiFER senses its ligand by GPCR and indirectly reports such signal by genetically encoded calcium indicator TNXXL. (d) Archaerhodopsin based GEVI could directly transform the voltage variance signal to optical signal without other additional structure.
Genetically encoded calcium indicators (GECIs), which sense the concentration of Ca2+ ions, a second messenger that faithfully indicates neural activity;
Genetically encoded voltage indicators (GEVIs), which can be used to visualize changes in neuron membrane potential.
Genetically encoded synaptic transmission indicators (GESTIs), which sense the presence of common neurotransmitters and provide a fluorescence readout.
GENAIs usually indicate neural activity through fluorescence changes. Because most signals generated by GENAIs are recorded by optical systems, either traditional cameras or sophisticated microscopes, optical performance is the most critical feature of GENAIs. Some important factors are summarized below (
Signal intensity and signal-to-noise ratio
The contrast of an image is proportional to the intensity difference ΔI between two image areas, divided by the average image brightness I, as shown in the following equation:
In fact, contrast is often the only output that can be measured from a GENAI, because normalizing the captured intensity to an absolute level of neural activity is difficult. Neural activities are usually recorded as ΔF/F, the ratio of the fluorescence intensity to the average recorded intensity. Without a high-intensity fluorescence signal source, the measured neural activity is easily overwhelmed by the noisy background: the signal I may be too weak to be distinguished from background noise. In addition, GENAIs themselves must maintain a high signal-to-noise ratio (SNR) to distinguish their on and off states. For a neural indicator designed for in vivo recording, labeling specificity also substantially contributes to the final contrast: poor labeling specificity or densely labeled neurons lead to high background and neuropil contamination. GENAIs combined with selective expression methods and sparse labeling techniques can circumvent this problem and therefore are especially useful in recording axonal or dendritic activities.
Temporal resolution
Real-time imaging of certain biological processes requires rapidly responding indicators. In many attempts to improve GENAIs, the major focus has been enhancing temporal resolution. If the temporal resolution of the indicators is insufficient, information loss occurs and may preclude certain experimental designs.
Dynamic range
Dynamic range (DR) refers to the range of target physical values that can be measured by a GENAI. In most cases, DR is limited at one end of the range by saturation and at the other end by random noise or uncertainty in signal levels, which are often referred to as the sensitivity of an indicator. If the DR of the indicator cannot cover the entire range of a changing biological parameter, the indicator is unable to perfectly record the entire biological process.
Photostability
For most GENAIs using fluorophores as an output, the readout relies on fluorescence excitation by a light source. Although techniques such as non-linear multiphoton excitation can partially decrease phototoxicity and the chance of photobleaching, photostability remains a key factor for fluorophores used in long-term observation. Beside the techniques involving decreasing the photon energy of the input light or the intensity or time-span of light exposure, indicator design using more robust fluorophores that are less prone to bleaching is another practical method to obtain high photostability.
Although all GENAIs have the same goal in transforming neural activity into measurable optical changes, different strategies have been used to achieve this goal (
FRET, another way to amplify the conformational changes of protein domains (
Some hybrid genetically encoded neural activity indicators function indirectly through transcoding the target physical parameter to be measured into an intermediate form, which in turn is transformed into an optical signal. These indicators often consist of a pair of separated sensing and reporting units working cooperatively.
Quantification of free [Ca2+] changes can be used as a reliable proxy for neural activity, because dramatic changes result from action potential firing and synaptic transmission. Chemical calcium indicators can sense the concentration changes of Ca2+ ions, thus indirectly indicating neural activity. In early practice, such quantification was widely used in functional in vivo imaging; however, the complicated loading process and lack of cell-type specificity have strongly limited its use. As an alternative, GECIs can be easily expressed in a specific group of neurons through genetic techniques and subsequently enable long-term observation, as do other GENAIs (
Summary of Genetically encoded calcium indicators.
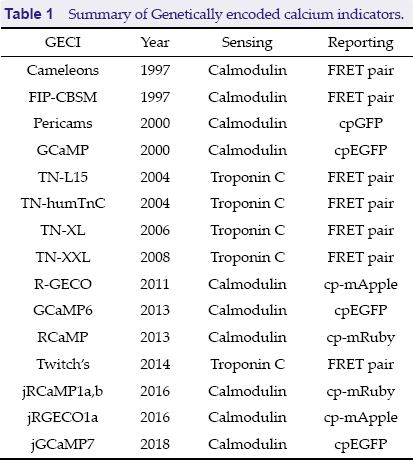
Summary of Genetically encoded calcium indicators.

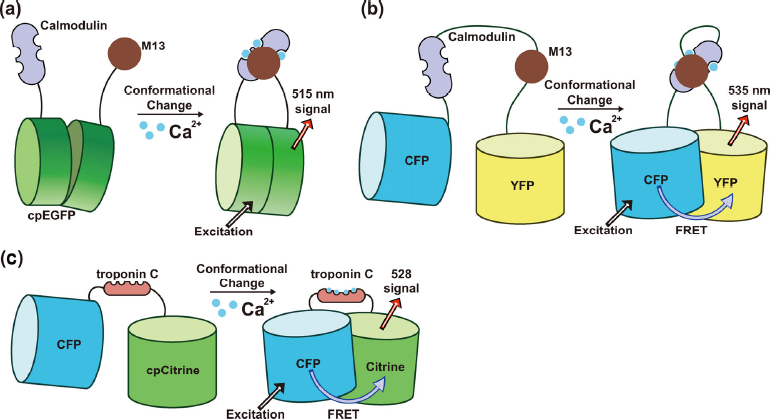
Genetically encoded calcium indicators. (a) A cpFP-calmodulin based GECI, the calcium binding cause structural variance of CaM and M13 domain and amplified through a circularly permuted GFP. (b) FRET-calmodulin based GECI, the calcium-induced structural variance is reported by a FRET pair. (c) Skeletal muscle troponin C (TnC) based GECI, the calcium-induced structural variance is reported by FRET between two fluorescent protein domains.
CaM has a relaxed linear structure in the absence of Ca2+ binding (
Beyond the cpFP based CaM-M13 indicators, another important class of widely used calcium signal indicator tools is a series of fluorescent indicators called Cameleons (
Troponin C based calcium indicators
Skeletal muscle TnC based calcium indicators are another large family of GECIs (
Specialized GECIs
Some application-specific GECIs have also been developed in recent years. By combining the photo-regulated features of photoactivatable (pa) or photoconvertible (pc) FPs with allosterically modulated cpFPs, Hoi et al. developed a “highlightable” GECI that can convert output spectra by illumination [26]. Through library screening and structure-guided mutagenesis, Fosque et al. further developed a calcium modulated photoactivatable ratiometric integrator (CaMPARI), which can provide a snapshot of activated neurons by permanently marking activated indicator molecules under violet light illumination. With short timescale violet light illumination, CaMPARI can be used to record neuron activity, thus providing a powerful tool for optical functional mapping [27].
Because CaM based GECIs such as GCaMPs may interfere with gating and signaling of L-type calcium channels and therefore disrupt Ca2+ dynamics and gene expression, Yang et al. developed an improved indicator, GCaMP-X, by incorporating an additional apoCaM-binding motif. GCaMP-X exhibits far fewer side-effects than regular GCaMP indicators while retaining good calcium-sensing properties [28]. Because many GECIs use CaM as their sensing domain and may have the same drawbacks, this work may inspire the creation of a series of GECIs with few side-effects.
Genetically encoded voltage indicators
GEVIs indicate neural activities by allowing for visualization of neuronal membrane potential (
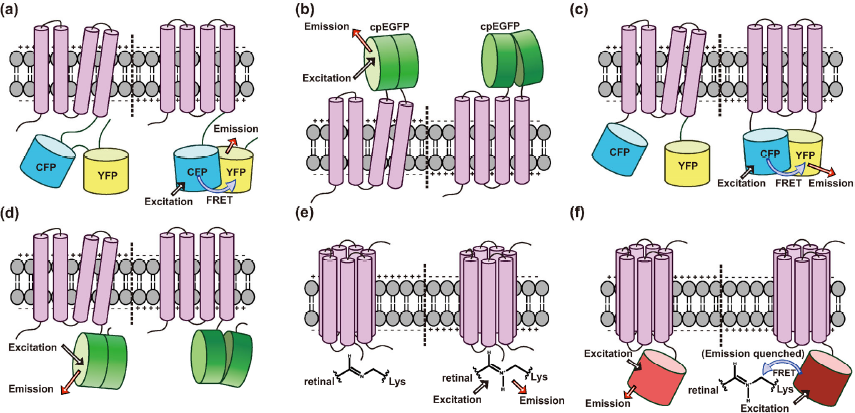
Overview of GEVI structural designs. (a) A VSFP based GEVI uses FRET as its reporting module. The structural variance of VSFP driven by depolarization of membrane potential is reported by fret between two fluorophore domains. (b) An extracellular cpFP-VSFP based voltage indicator. (c) A FRET-VSFP butterflies chimeric protein design. (d) Fast-dynamics and sensitive single-cpFP based ASAP. (e) Archaerhodopsin based GEVI, can directly transform the action potential signal to optical signal. (f) Archaerhodopsin-FRET based GEVI. The emission of FP is quenched by FRET with protonated retinal Schiff base at depolarizing voltage. This method provides higher fluorescent intensity and higher-method compatibility spectral properties.
A major class of GEVIs, the voltage-sensitive fluorescent proteins (VSFPs), including VSFP2 and its variants, are based on the paddle domain of voltage-gated phosphatase (VSP), mainly Ci-VSP (
Summary of genetically encoded voltage indicators.
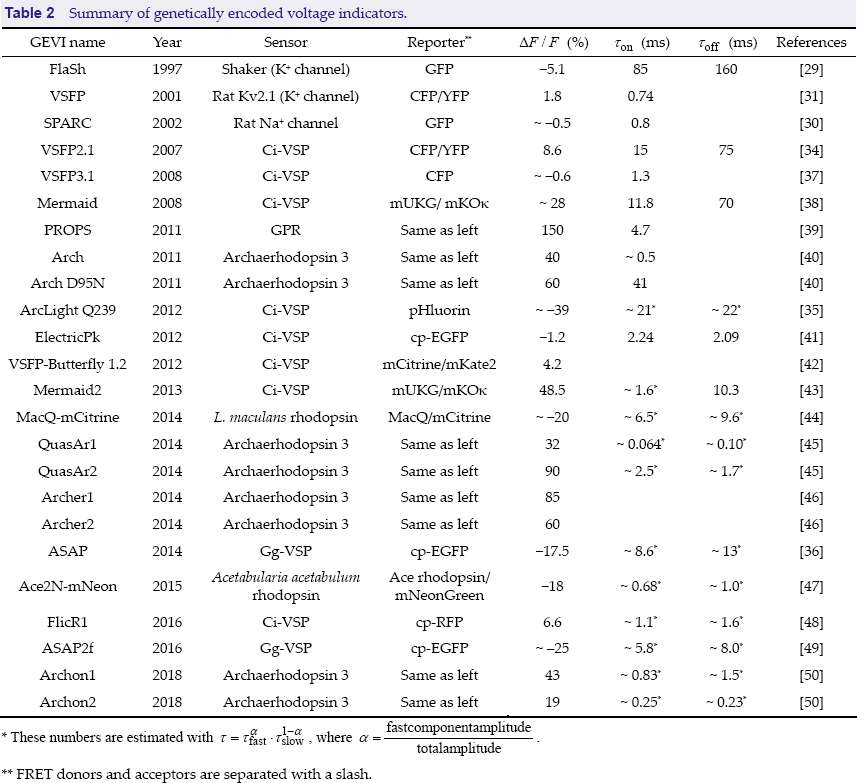
Summary of genetically encoded voltage indicators.

These numbers are estimated with τ = τfastα · τslow1-α, where
FRET donors and acceptors are separated with a slash.
Another class of GEVIs using an opsin-based mechanism was introduced after VSFPs (
Recent progress in action potential imaging
In recent work, in 2018, Piatkevich et al. developed a robotic cell picker to screen hundreds of thousands of proteins in a few hours and produced the high-performance opsin-based GEVI Archon1 [50]. Archon1 is improved in multiple aspects and exhibits excellent localization, SNR, sensitivity, response speed and photostability, as well as full compatibility with optogenetic control [50]. Because action potential has a millisecond timescale, currently, only state-of-the-art sCMOS cameras can provide a sample rate (~ 1000 FPS) permitting in vivo action potential imaging. However, these devices are generally large and are unable to be integrated into head-mountable devices for imaging in freely moving animals, and they usually have a limited field of view. To remedy these defects, some ongoing projects aim to build miniature microscopes capable of high-speed action potential imaging (https://web.archive.org/web/20180912222322/ http://grantome.com/grant/NIH/R21-EY028381-01).
Genetically encoded synaptic transmission indicators
GESTIs are a class of neural activity indicators that provide a signal response to specific neural transmitters (
Summary of Genetically encoded synaptic transmission indicators.
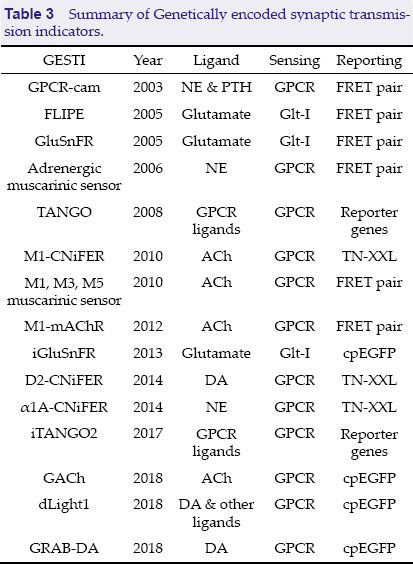
Summary of Genetically encoded synaptic transmission indicators.

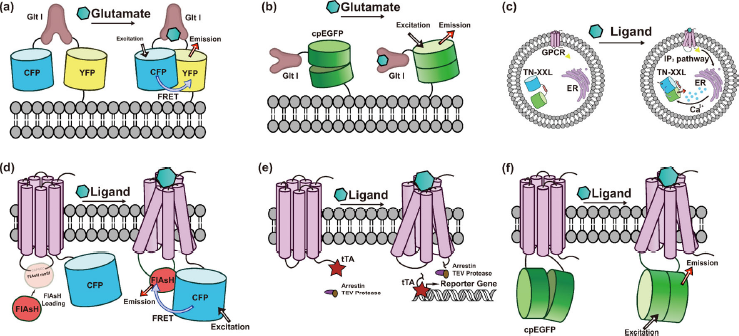
Genetically encoded synaptic transmission indicators. (a) GluSnFR, a GltI-FRETbased glutamate indicator. (b) iGluSnFR, a GltI-cpFP based glutamate indicator. (c) Cell-based neurotransmitter fluorescent-engineered reporters (CNiFERs). (d) Hybrid GPCR-FRET indicators using biarsenical chemical dye FlAsH as one of its fluorophores. (e) TANGO assay, sensitively reports subtype specific GPCR activation through cleaving and releasing the transcriptional activator, tTA fused to ligand-activated GPCRs by a TEV protease-human-arrestin chimeric protein. (f) cp-FP based GPCR neurotransmitter probes which provide superior spatiotemporal precision.
PBPs compose a large superfamily of members that bind various chemicals including neuro-transmitters (
Hybrid synaptic transmission sensors
Attempts to address the limitations of single GEStIs have yielded several hybrid sensors (
Another problem in GPCR-based GESTI design is that FRET pairs used to amplify conformational variance signal are large and thus cause protein trafficking problems. Because chemical dyes are much smaller than fluorescent proteins, some hybrid GPCR-based GESTIs use the biarsenical chemical dye FlAsH (fluorescein arsenical hairpin binder) to act as one of the fluorophores in the FRET pair [13,58], thereby decreasing the indicator size.
GPCR-activation-based sensors
Among the GESTIs, GPCR-activation based sensors (GRABs) are a promising series of GESTIs that consist of a ligand specific GPCR and a reporter to amplify the structural variation of the 7-TM rhodopsin domain of these GPCRs (
Beyond the GESTIs used to amplify the ligand-binding signal via the intrinsic GPCR signaling pathway described in Section 4.2, the TANGO assay developed by Barnea et al. provides another practical method to monitor subtype specific GPCR activity by cleaving and releasing the transcriptional activator tTA fused to ligand-activated GPCRs via a TEV protease-human β-arrestin chimeric protein. The tTA dependent reporter gene can thus be expressed in response to subtype-specific GPCR activation [59]. The improved iTANGO2 assay achieves a higher temporal resolution (on a scale of minutes) than the original TANGO assay (on the scale of several hours) by adding a light inducible protease system to the ligand-gated gene expression system [60]. However, limited by the gene expression, the temporal resolution of reporter gene-based methods is still incompatible with the fastest single molecule indicators. FRET is a common method to construct high temporal resolution indicators. Efforts to design dual-fluorescent protein GPCR indicators have resulted in several FRET-based GEStIs. FRET-based GRABs have superior temporal resolution [18,61], but because of the relatively small conformational changes of GPCRs, most of these indicators do not have ideal temporal-resolution and sensitivity. As mentioned in Section 4.2, the size of these dual-FP FRET indicators is still a problem for in vivo study. In recent studies creating a high-temporal solution GPCR-based GEStI, a conformationally sensitive fluorescent protein has been inserted in the third intracellular loop of GPCRs, because the intracellular loop between the TM and TM undergoes a relatively large conformational change upon ligand binding. In addition, these GPCR-based sensors must be carefully designed to avoid perturbing the intrinsic physiology in the cell of interest [9]. Jing et al. have constructed the ACh indicator GACh, which has high sensitivity (EC50 ≍ 1 μM), high SNR (≍14) and fast kinetics (τ ≍ 200-800ms) while maintaining an endogenous receptor level on/off specificity and photostability [62]. A similar approach has been applied to the construction of the DA indicator [57,63] and several other indicators including norepinephrine, serotonin, melatonin and opioid [57]. These single-FP based GRABs enable spatiotemporally precise measurements to indicate their corresponding neurotransmitters.
Discussion
The development of GENAIs in recent decades has provided powerful tools for cell-type-specific neuroscience studies. These indicators have been conceived, constructed and improved to meet the growing requirements of functional imaging. Efforts to improve GENAIs have mainly focused on their optical properties including photo-stability, fluorescence intensity, signal sensitivity, dynamic properties and compatibility with optical neuronal activity manipulating methods. However, for many real-life applications, all these requirements cannot be satisfied simultaneously, and certain applications may require distinct properties that could be achieved by a single indicator. The next generation GENAIs could be optimized for specific uses, such as the jGCaMP7 series variants, as recently reviewed by Luo et al. [2]. These variants with different sensitivity, temporal resolution and baseline intensity can satisfy the diverse requirements of applications such as neuropil observation and wide-field imaging.
Finally, given how the field has developed in recent years, further developments of GENAIs that are ultrafast, ultrasensitive and compatible with manipulation methods are likely to boost further study of brain function.