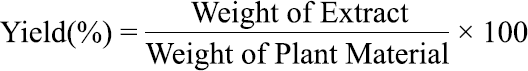Abstract
This study explores the larvicidal potential of a hexane extract from Blepharis ciliaris against Culex pipiens mosquitoes, a known vector of various diseases. The study aimed to find an eco-friendly alternative to chemical insecticides. Promising results were obtained, with the extract showing significant larvicidal activity. The LC50, LC90, and LC95 values against C. pipiens were determined as 125.53, 190.5, and 198.76 µg/mL, respectively. Furthermore, histological observations using a light microscope revealed notable changes in the midgut structure of the treated larvae, including degradation of microvilli, degeneration, and protrusion of epithelial cells and peritrophic membrane, loss of nuclei, and irregular microvilli. These findings suggest that the hexane extract from B. ciliaris has the potential as a natural larvicidal agent against C. pipiens mosquitoes. The MTT assay results indicate that the tested extracts did not exhibit cytotoxic effects on Huh-7 cells, and no morphological changes were observed under light microscopy. The findings of this study suggest that B. ciliaris contains bioactive compounds that could serve as potential bio-larvicides for controlling C. pipiens vectors. Further research should be conducted to assess the efficacy of using this botanical insecticide in small-scale field trials to develop environmentally safe interventions for controlling C. pipiens populations.
Introduction
Plants contain diverse bioactive secondary metabolites to protect themselves against insects and pathogens (Lee et al., 2015). Bioactive secondary metabolites possess a wide range of biological potentials such as toxicity and repellency to insects, antimalarial, antioxidant, antimicrobial, and anticancer properties (Valdivieso-Ugarte et al., 2019).
Various phytochemical compounds have been isolated and identified from different parts of B. ciliaris. The major classes include flavonoids such as genistein-7-O-(6”-O-E-caffeoyl)-áα-D glucopyranoside, naringenin-7-O-(6”-E-p-coumaroyl)-β-D-glucopyranoside, apigenin 7-O-glucoside, and apigenin-7-O-(3”-acetyl-6”-E-p-coumaroyl) glucoside (Rattan. 2023). Additionally, phenolic acid derivatives like verbascoside, cis-verbascoside, protocatechuic acid, vanillic acid, and 4'-O-diglycoside of decarboxy rosmarinic acid have been identified (Dirar et al., 2021). Novel compounds such as apigenin 7-(3”-acetyl-6”-E-p-coumaroyl)glucoside and naringenin 7-(3”-acetyl-6”-E-p-coumaroyl) glucoside have also been isolated (Dirar et al., 2021). Since bioactive secondary metabolites are often a mixture of different compounds, those proven to possess larvicidal and adulticidal properties are believed to have a reduced likelihood of resistance development in mosquito vectors. Consequently, they present a potential substitute for synthetic insecticides (Şengül Demirak & Canpolat, 2022).
Public health is still very concerned about vector-borne infectious illnesses from mosquito bites. Programs to control the C. pipiens vector use a variety of tactics to lower the population and slow the spread of the disease. These tactics include source reduction, which involves eradicating breeding sites; larvicides and adulticidal treatments, which target and kill adult mosquitoes and larvae in their breeding grounds; biological control, which regulates mosquito populations by using natural parasites, competitors, or predators; personal protection; and community education (Beier et al., 2008; Benelli et al., 2016; Shaw & Catteruccia, 2019).
The emergence of resistance in vectors poses a significant challenge to vector control interventions that rely on the use of insecticides. Mosquitoes, including the C. pipiens vector, resist insecticides commonly used in public health, such as pyrethroids, carbamates, and organophosphates (Yahia et al., 2023). In Saudi Arabia, Cx. pipiens populations from Riyadh city showed varying degrees of resistance to several insecticides, such as deltamethrin, lambda-cyhalothrin, beta-cyfluthrin, and bifenthrin, while remaining susceptible to fenitrothion (Al-Sarar, 2010).
This resistance can undermine the effectiveness of vector control programs and jeopardize their success in reducing mosquito populations and preventing the transmission of diseases. The development of resistance in vectors is a complex process influenced by several factors, including the extensive and repeated use of insecticides, genetic mutations, and selection pressure (Nauen, 2007). As a result, alternative strategies and innovative approaches are being explored to overcome the challenge of insecticide resistance (Piedra et al., 2023). These include the development of new insecticides with different modes of action, using insecticide rotation or combination strategies, implementing integrated vector management approaches that combine multiple control methods, and promoting environmental management techniques to reduce mosquito breeding sites (WHO, 2004).
The widespread insecticide resistance phenomenon in addition to the environmental pollution from chemical insecticides and their high operational cost, has led to the need to develop alternative mosquito control approaches (Karunamoorthi & Sabesan, 2013). Developing effective and eco-friendly tools for reducing the burden or eliminating mosquitoes is highly recommended.
It is well known that plant extracts contain insecticidal effects. Numerous plants have been found to have potent insecticidal and repellant effects on both adult mosquitoes and mosquito larvae (Piplani et al., 2019). Exploring the Saudi flora is crucial to identifying plant species that possess secondary metabolites with significant larvicidal activity against mosquito larvae. This research aimed to uncover potential natural sources for effective larvicidal compounds, contributing to the development of eco-friendly strategies for mosquito control.
There are roughly 129 species in the genus Blepharis Juss. of the family Acanthaceae, all found in tropical and subtropical regions (Mahboubi et al., 2013). It is well known that plants in the genus Blepharis have antioxidant, cytotoxic, and antibacterial properties (Bhutkar et al., 2019; Dirar et al., 2021). In traditional medicine, Blepharis ciliaris (L.) B. L. Burtt treats inflammations, coughs, colds, sores, toothaches, skin wounds, and kidney disorders (El-Shanawany et al., 2013).
To our knowledge, there is no report on the insecticidal activity of B. ciliaris. Therefore, this study aims to shed light on the potential insecticidal effects of the B. ciliaris extract harvested in Saudi Arabia, thereby contributing to the ongoing research efforts in utilizing plant-derived products for insect control. By assessing its toxicity and larvicidal activity, valuable insights can be gained, informing the development of environmentally friendly and effective insecticides targeting mosquito populations.
Material and methods
Plant material
Blepharis ciliaris specimens were collected from Al-Qusay'ah region in Al-Kharj Governorate, Saudi Arabia (32°29′00″N, 13°57′00″E) in August 2022. Taxonomic identification of the plants was conducted at the Botany Department of King Saud University in Riyadh, and the voucher specimen (KSU-BRC 0034) is preserved there.
Extraction
The Soxhlet apparatus was used to obtain the B. ciliaris extracts,. The process involved grinding the weighed aerial parts of B. ciliaris (~16 g) and individually extracting them with approximately 500 mL of n-hexane, chloroform, ethyl acetate, and methanol. The extraction occurred in the Soxhlet extractor at around 65 °C for 24 hours. After completion, the residual solvent evaporated, and the resulting extract was quantified and recorded as the percentage yield using the following formula:
The extract was then stored in a glass bottle at 4 °C until further use.
Larvicidal activity of Blepharis ciliaris extract against Culex pipiens
To feed the female mosquitoes, they were offered the blood of albino mice. This blood meal is necessary for the reproductive process of female mosquitoes, as they require the nutrients found in blood to develop and lay eggs.
Larvicidal Activity of B. ciliaris Extracts Against the Third Instar of C. pipiens
Four extracts were tested against the C. pipiens larvae. In each bioassay, 20 third-instar larvae were placed in a 6-well plate containing dechlorinated water. Different concentrations of the extracts were prepared to achieve final concentrations of 62.5, 100, 137.53, and 175 ppm in a 6 mL volume. The mortality of the larvae was recorded at 24 and 48 hours after inoculation with the extracts. Each test was performed in triplicate, and a negative control using 1 % Dimethyl sulfoxide (DMSO) was included. Larvae were considered dead if they did not respond to physical stimuli. The percentage of mortality was calculated. The lethal concentration of the extracts that killed 50% of the larvae (LC50) was calculated using Origin Pro 8.5 Software. The C. pipiens colony was maintained in the insectary under optimal conditions, including a relative humidity of 70% - 80%, a 12:12 light-dark period, and a temperature range of 27 °C -30 °C (Imam et al., 2014).
Histological observations
The treated and control fourth-instar larvae of C. pipiens were fixed in paraformaldehyde for 24 hours. Following the standard method (Al-Mekhlafi et al., 2021), the fixed samples were prepared and sectioned using a microtome (Leica RM2126, Germany). These sections were stained with hematoxylin-eosin and examined under a light microscope (Olympus, Japan). Photographs were captured of the midgut cells of C. pipiens in both the treated larvae and the control group, allowing for comparing and observing any alterations or variations between them.
Cytotoxicity and cell morphology
The cytotoxicity of the solvent extract obtained from B. ciliaris was assessed using the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium Bromide (MTT) test on the Huh-7 cell line, following the methodology outlined by Abutaha (2015).
For this experiment, Huh-7 cells were cultured in DMEM medium (Gibco) supplemented with 10% phosphate buffer saline PBS (Gibco) and maintained at 37 °C in a humidified atmosphere with 5% CO2. The cells were regularly passaged using trypsin/EDTA (Gibco) and PBS solution. After 24 hours of incubation, the cells were exposed to different concentrations (500, 250, 125, 50, and 25 µg/mL) of the hexane extract suspension obtained from B. ciliaris Control cells treated with 0.1% methanol were also included in the experiment.
Following treatment, the cells were incubated with 100 µL of MTT solution for 2 hours without light at 37 °C. The supernatant was then removed, and the resulting MTT crystals were dissolved in 1000 µL of DMSO with shaking at 37 °C for 5 minutes.
To assess cell viability, the absorbance at 570 nm was measured using an ELISA reader (ChromMate, England). The concentration of the plant extract that led to 50% cell death (IC50) was determined from the respective dose-response curves using Origin Pro 8 software.
Additionally, changes in cellular morphology before and after treatment with the hexane extract were examined using a phase-contrast inverted microscope (Leica, Germany).
Statistical Analysis
Statistical analysis of the experiments was conducted with triplicate measurements, and the means, along with standard deviations, were computed. Larval mortality data were evaluated using one-way ANOVA, and Tukey's test was subsequently employed for post hoc comparisons. The statistical analyses were carried out using SPSS software (version 15, USA). A p-value below 0.05 was considered indicative of statistical significance. The IC50 value was calculated using OriginPro 8 software. The data, consisting of extract concentrations and corresponding cell viability percentages, were plotted to generate a dose-response curve. A sigmoidal function was applied through nonlinear curve fitting to model the relationship between concentration and viability. The software then determined the IC50 value based on the fitted curve, ensuring an accurate assessment of the inhibitory potential of the extract.
Results
The yield of the extracts
The Soxhlet apparatus extraction of B. ciliaris (Fig. 1) resulted in the following extract yields: hexane extract yielded 1.8 % wt/wt, chloroform extract yielded 3.49 % wt/wt, ethyl acetate extract yielded 0.7 % wt/wt, and methanol extract yielded 14.6 % wt/wt.

Blepharis ciliaris harvested from the Kingdom of Saudi Arabia, Riyadh.
Larvicidal activity of the extract
The initial experiment was conducted at a concentration of 500 µg/ml. Extracts of ethyl acetate, chloroform, and methanol showed no mortality rate. However, the hexane extract demonstrated promising efficacy, with mortality rates reaching 85%, 91.67%, and 95% at the highest concentration of 175 µg/mL after 24, 48, and 72 hours, respectively. Conversely, at the lowest concentration of 62.5 µg/mL, the mortality percentages were 18.33 %, 21.16 %, and 27.67%. The calculated LC50 values were 125.53, 115.46, and 99.90 at 24, 48, and 72 hours, respectively. The statistical analysis results showed significant differences between concentrations and different times at a significant level of 0.5 (Table 1).
Variations in mortality of Culex pipiens larvae treated with Blepharis ciliaris as a function of concentration and time.
Vertical capital letters indicate significant differences between concentrations, and small horizontal letters indicate differences between time intervals. Significant differences were assessed using one-way ANOVA followed by Tukey's test, with p < 0.05.
Histological observations
The observations under the light microscope showed that hexane treatment at LC50 125.53 µg/ml affected the midgut cells of the third-instar larvae. 24 h after treatment, the midgut showed severe morphological alterations. The midgut showed degradation of microvilli (DMV); degeneration and protruding of epithelial cells (DE), and peritrophic membrane (DPM); loss of nuclei, irregular and damage of microvilli (Fig. 2 A and B). In the control group, the brush border in the epithelial cells of the midgut were all completely tight and intact (Fig. 2 C and D).
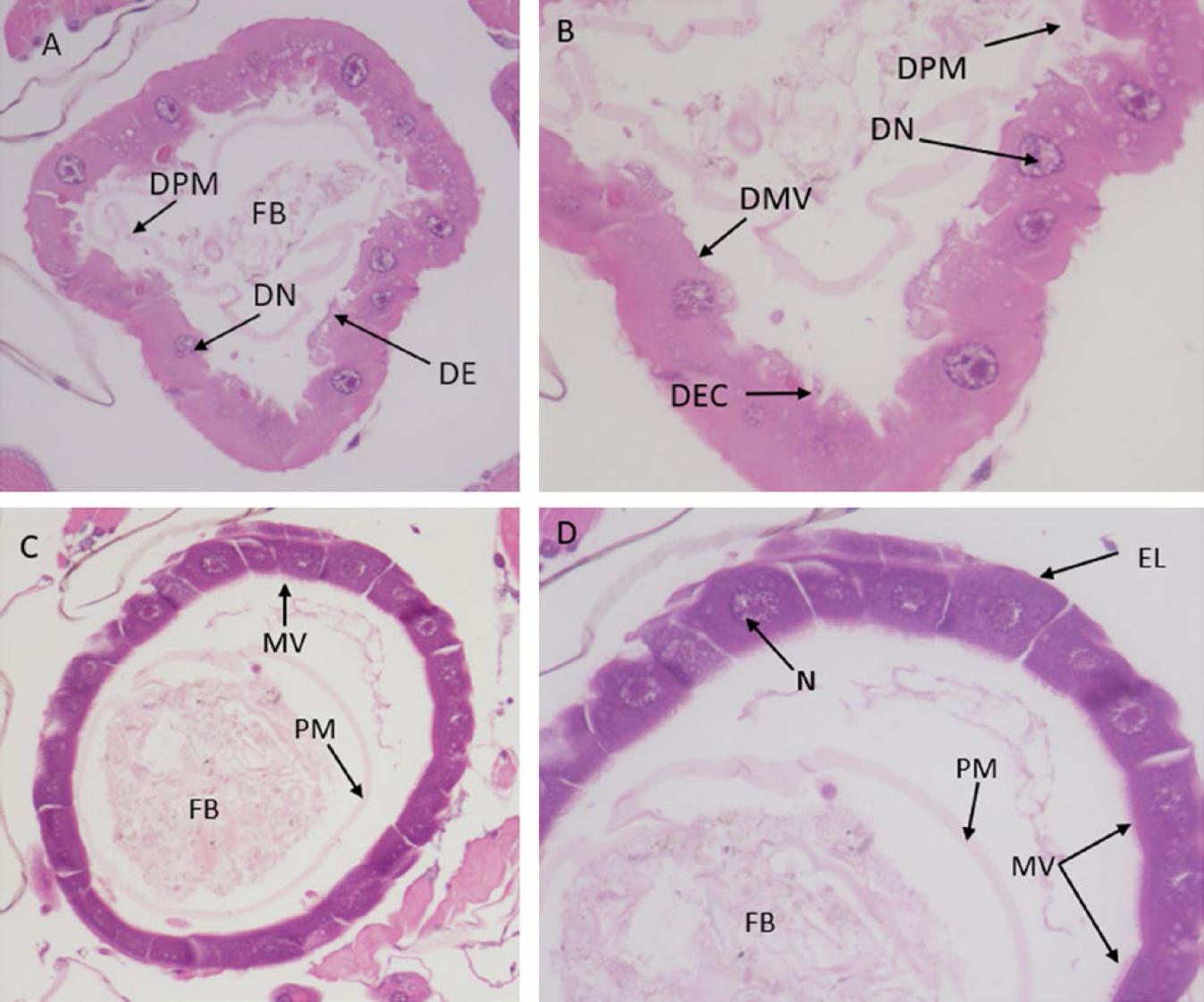
Photomicrographs of the midgut of three instar larvae of C. pipiens. (A) and (B) Cross-section in the midgut treated with the LC50 dosage of Blepharis ciliaris extract, showing the effect after 24 h of exposure. Degraded microvilli (DMV), degenerating epithelial cells (DEC), degenerating peritrophic membrane (PM), and degenerating nuclei (DN). (A = 200× and D = 400×). (C) and (D), cross-section of the midgut of untreated larvae showing normal epithelial cells (Ep), peritrophic membrane (PM), food bolus (FB), microvilli (MV), nuclei (N), and normal epithelial layer (EL) (C = 200× and D = 400×).
Cytotoxicity assay and morphological study
The results showed that none of the tested extracts exhibited cytotoxicity against Huh-7 cells (Fig. 3) and did not induce any morphological modifications, such as cell shrinkage, detachment of cells, or loss of the typical spindle shape, in Huh-7 cells when compared to the untreated cells.
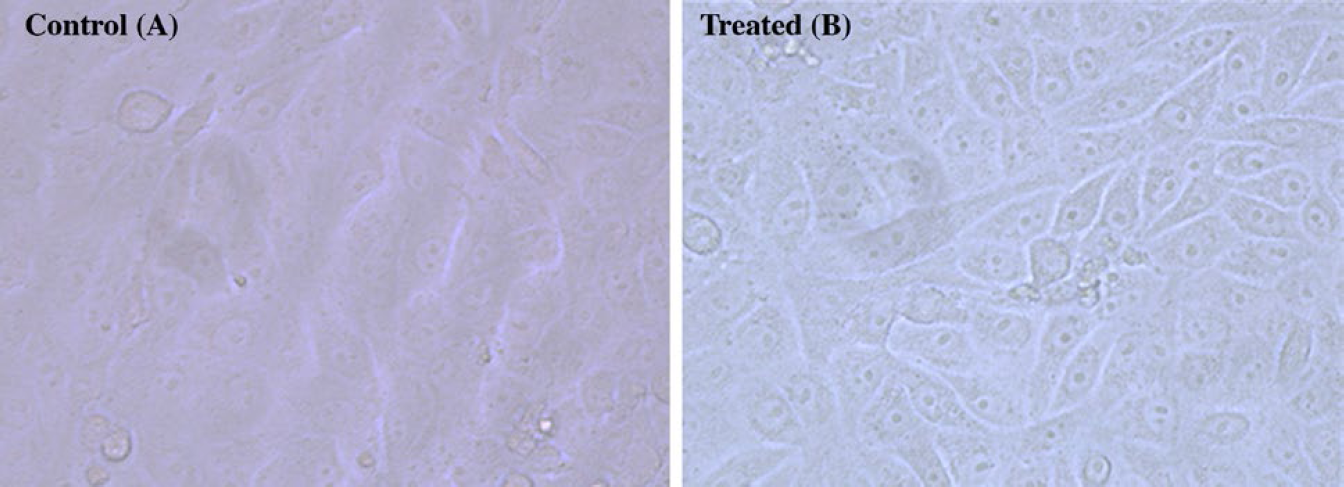
Morphology changes in Huh-7 cells after hexane extract treatment at 24 hours were recorded using SEM. (A) untreated Huh-7 at 24 hours, (B) hexane-treated Huh-7 cells at 24 hours.
Discussion
The rise of insecticide resistance and growing concerns over the environmental impact of synthetic insecticides have spurred the exploration of alternative solutions such as plants. Among these, products utilizing plant secondary metabolites have gained attention as potential biological agents for mosquito larvae control (Senthil-Nathan, 2020). Plants possess a diverse array of compounds for developing environmentally friendly alternatives to traditional insecticides, providing a sustainable approach to mosquito control (Souto et al., 2021).
In this study, various solvents with different polarities were used to extract the plant and its effect on the larval development of C. pipiens was evaluated for the first time. The objective was to identify a novel bio-larvicidal agent targeting C. pipiens. The results revealed that the extract exhibited larvicidal activity, with inhibition of larval development observed in a time- and concentration-dependent manner. However, the results of this study demonstrate the presence of bioactive compounds in the extract; however, their activity may appear weak. This could be due to the active compounds being present in very small amounts, which may reduce activity when influenced by other co-occurring compounds in the extract. Previous studies have documented this phenomenon, where compound production fluctuates seasonally, with the highest levels detected in the autumn and winter (Hejna et al., 2024). Other studies have shown that bioactive compounds in low concentrations may be masked or their effects modulated by other compounds within the extract, leading to either synergistic or antagonistic interactions (Caesar & Cech, 2019). The significance of these findings suggests that further isolation and analysis of the individual components are crucial. Advanced analytical methods such as Nuclear Magnetic Resonance (NMR) and Liquid Chromatography-Mass Spectrometry (LC-MS) should be employed to accurately identify and characterize these compounds (Bucar et al., 2013; Sasidharan et al., 2011).
By isolating and studying the individual components, a clearer understanding of their bioactivity and potential value can be obtained.
Similar larvicidal activities were reported from different botanical extracts against C. pipiens larvae as Magnolia foveolata leaf extract against the larvae of four mosquitoes Culex fuscocephala Theobald, Culex quinquefasciatus Say, Aedes albopictus Skuse, and Aedes aegypti (Linnaeus, in Hasselquist) (Diptera: Culicidae), with 24-h LC50 less than 40 µg/mL (Trinh et al., 2023). Another study reported the larvicidal effects of leaf extracts from Peltophorum ptero carpum (DC.) Backer ex K. Heyne (Fabaceae) were investigated against A. aegypti and C. quinquefasciatus mosquitoes. The results showed that the methanol extract exhibited the highest larvicidal activity against both mosquito species, with LC50 values of 111.77 ppm (A. aegypti) and 158.93 ppm (C. quinquefasciatus). The chloroform and n-hexane extracts also demonstrated larvicidal activity, with LC50 values of 170.74 and 169.18 ppm (chloroform extract) and 219.14 and 203.48 ppm (n-hexane extract) for Ae. aegypti and C. quinquefasciatus, respectively (Yagoo et al., 2023).
The results of the present study were extended to evaluate the histopathological changes of hexane extract on C. pipiens larvae. This investigation aimed to determine whether the inhibitory effects on larval development were attributed to alterations in the midgut structure induced by the presence of hexane extract. The mosquito midgut involves critical physiological processes, including enzyme secretion, nutrient absorption, ion transport, osmoregulation, defense against pathogens, and metamorphosis. It plays a vital role in mosquitoes’ overall survival and development (Christophers, 1960; Procopio et al., 2015; Sina & Shukri, 2016). Many researchers have shown that botanical extracts can impact the mosquito midgut's structure and function. These extracts have been reported to induce alterations in the midgut, affecting its morphology, cellular integrity, peritrophic matrix, enlargement of intercellular spaces, and cytoplasmic vacuolisation and overall survivability of the insects (Abutaha & Al-Mekhlafi, 2014; Al-Mehmadi & Al-Khalaf, 2010; Gusmão et al., 2002). By targeting the midgut, these extracts can potentially interfere with vital functions, rendering mosquitoes more susceptible to control measures and reducing their ability to transmit diseases.
The observed damage in the midgut region of the larvae treated with the hexane extract may have impaired digestive absorption processes and the regenerative cells responsible for midgut maintenance and repair. This damage likely interfered with larval mosquito development and compromised their overall survival. The disruption of these essential functions in the midgut can have significant consequences for the larvae, affecting their ability to obtain nutrients, undergo proper growth, and successfully complete their life cycle.
Toxicity studies often involve many animals, which can be time-consuming and expensive, and limit the testing of multiple compounds simultaneously. Huh-7 cells, a widely used model of human liver cells, offer a simpler and controlled in vitro system that can provide reliable data over time. In vitro metabolomics systems enable the identification of organ toxicity, assessment of the toxicological profile of various substances, prediction of compound toxicity, and investigation of molecular mechanisms underlying toxicity. These systems offer highly controlled environments suitable for regulatory purposes and, importantly, eliminate the need for animal testing (Quintás Soriano et al., 2023; Ramirez et al., 2018).
Statistical analysis of metabolite profiles in 1114 cell supernatants and 3556 intracellular samples from 7 experiments conducted over a span of 3 years using liver cancer cell lines demonstrated the robustness and reproducibility of the metabolomics in vitro method in capturing experimental variability over time (Ramirez et al., 2018). Our results suggest that all the extracts tested were not toxic to the Huh-7 cells, indicating the potential safe use of the plant. However, it is important to note that further studies are necessary to evaluate its toxicity using other cell lines, including normal cells.
A key limitation of this study is the lack of precise chemical characterization of the compounds responsible for the observed activity. Due to the complexity of the extract and the possible presence of bioactive compounds in trace amounts, further research is needed to isolate and accurately quantify these components. Additionally, assessing the extract's toxicity across various cell lines, including normal cells, is essential for a comprehensive understanding of its potential effects. Since different cell types may exhibit varying sensitivities or respond differently to specific compounds, conducting further experiments using a broader range of cell lines will help provide a more complete toxicity profile for the plant extract.
Conclusions
The extraction of B. ciliaris using a Soxhlet apparatus yielded varying percentages of extracts, with the highest yields obtained from methanol (14.6%) and chloroform (3.49%) extracts. Among these, the hexane extract demonstrated significant larvicidal activity against Cx. pipiens larvae, with mortality rates reaching up to 95% at the highest concentration tested (175 µg/mL) after 72 hours. Histological examination revealed severe morphological alterations in the midgut cells of third-instar larvae treated with the hexane extract, including microvilli degradation and peritrophic membrane damage. However, cytotoxicity assays on Huh-7 cells showed no adverse effects of the hexane extract, indicating its safety on mammalian cells. These findings suggest the potential of the hexane extract of B. ciliaris as a natural larvicidal agent for mosquito control, warranting further investigation into its bioactive compounds and mode of action.
Origin and Funding
This project was funded by Researchers Supporting Project number (RSP2025R112), King Saud University, Riyadh, Saudi Arabia.
Author Contribution
Fahd A. Al-Mekhlafi, Nael Abutaha, and Abdulrahman Zaid M. Almohssin conceived research. Abdulrahman Zaid M. Almohssin, and Nael Abutaha conducted experiments. Fahd A. Al-Mekhlafi, Nael Abutaha, and Abdulmani H. Al-Qahtni analysed data, and conducted statistical analyses. The manuscript was written by Nael Abutaha, Fahd A. Al-Mekhlafi, Muhammad A. Wadaan, and Mohammed S. Al-Khalifa.
Conflict of Interest
The authors declare no conflict interests.
Footnotes
Acknowledgements
The authors express their sincere appreciation to the Researchers Supporting Project number (RSP2025R112), King Saud University, Riyadh, Saudi Arabia.