Abstract
Nasutitermes corniger is an arboreal termite that causes economic damage in urban areas, and its control is for chemical insecticides. An alternative for insect control is the use of entomopathogenic fungi and plant extracts, or the synergistic effect of the association of these. The toxicity of aqueous and hydroethanolic extracts of Agave sisalana on the biological aspects of the fungal Cordyceps farinosa, C. fumosorosea and C. javanica and the action of the extracts and their synergistic effect on N. corniger was examined. The effect of the extracts on fungi was evaluated by germination, sporulation and mycelial growth of fungi in Sabouraud medium through the study of biological aspects under laboratory conditions; and control of N. corniger were examined, by ingestion of extracts and of the association extract plus fungus on filter paper, being the percentage of mortality of soldiers and workers of the termite daily. In general, the extracts were inoffensive to the fungi at concentrations 10, 25, 50, 100 and 200 mg.mL−1. The extracts demonstrated termiticidal actions at concentrations tested, causing the mortalities about 100% of the workers after the third and sixth days (LC50: 0.778 to 0.803 mg.mL−1) and 100% of the soldiers between the fourth and seventh days (LC50:0.146 to 0.956 mg.mL−1). The association of the extracts with C. farinosa ESALQ1355 was more efficient in inducing the mortality in N. corniger. These results demonstrated the efficiency in vitro of the sisal extracts in controlling N. corniger termites, alone or in combination with fungi, suggesting their potential joint usefulness in the biological control of insect pests.
Introduction
Insect pests are controlled using chemical insecticides, which can contaminate man and the environment – and their conti-nued use usually results in the appearance of resistant insects (Chen et al. 2004; Pourseyed et al. 2010). Plant extracts and entomopathogenic fungi have been found to be efficient alter-natives for controlling insect pests by maintaining insect po-pulations at low-level equilibriums and limiting their spread (Marques et al. 2004; Sabbour & Abdel-Rahman 2013).
Plant extracts have been tested for insect pest control, due to the insecticidal action of their secondary compounds, besides being easily degradable and not contaminating the environment (Oliveira et al. 2007; Souza et al. 2011). These substances are more beneficial than chemicals, since they are renewable, easily degradable and do not pollute the environ-ment (Oliveira et al. 2007; Souza et al. 2011).
Agave sisalana Perrine ex Engelm (Asparagales: Agava-ceae) or sisal is an herbaceous plant whose fiber of its leaves is used as raw material in the manufacture of ropes, threads, and rugs in Brazilian industry (Costa et al. 2014). Residues and extracts of sisal are constituted by alkaloids, tannins, tri-terpenoids, saponins and flavonoids, which have insecticidal properties (Costa et al. 2014) being efficient controlling ar-thropods such as the mosquito Aedes aegypti (L., 1762) (Dip-tera: Culicidae), the red mite Tetranychus urticae (Koch, 1836) (Acari: Tetranychidae), and the carmine cochineal Dactylo-pius opuntiae Cockerell, 1896 (Hemiptera: Dactylopiidae) (Barreto et al. 2010; Nunes et al. 2015; Lopes et al. 2018).
The fungus Cordyceps fumosorosea (= Isaria fumoso-rosea) (Wize) Kepler, B. Shrestha & Spatafora, C. farinosa (= Isaria farinosa) (Holmsk.) Kepler, B. Shrestha & Spatafora and C. javanica (= Isaria javanica) (Bally) Kepler, B. Shrestha & Spatafora are efficient in controlling in vitro the termites Cop-totermes formosanus Shiraki 1909, Heterotermes tenuis (Ha-gen, 1858) (Isoptera: Rhinotermitidae), Coptotermes gestroi (Wasmann, 1896) (Isoptera: Rhinotermitidae), and Nasuti-termes corniger (Motschulsky, 1855) (Isoptera: Termitidae) (Lopes et al. 2011; Wright & Lax 2013; Lopes et al. 2017).
Nasutitermes corniger is the most common and most important termite pest of the genus, being widely distri-buted in the Americas, from southern Mexico to northern Argentina causing considerable damage to historical buil-dings, collections, and documents, and likewise attacks ur-ban constructions and ornamental tree. In Brazil, it is found in all its territory, especially in northern and northeastern states (Piauí, Bahia, Paraíba, and Pernambuco) (Milano & Fontes 2002; Albuquerque et al. 2012; Mello et al. 2014). N. corniger is highly adaptable to colonization of contrasting habitats in urban, agricultural, and natural environments (Breznak et al. 1982), being found on Moringa oleifera Lam. plantations in Nicaragua (Quiroz-Medina et al. 2021) and it is also encountered in the Bahamas and New Guinea and is responsible for enormous economic damage to wood products (Scheffrahn et al. 2006).
The use of entomopathogenic fungi in combination with plant oils and extracts may enhance their efficiency in insect pest control, as well as reducing the damage to the envi-ronment caused by commercial insecticides (Marques et al. 2004; Santos et al. 2015; Silva et al. 2015). In this sense, the objective of this study was to analyze the insecticidal poten-tial of the extracts of A. sisalana and the combination of these with Cordyceps spp. in the control of N. corniger.
Materials and methods
Preparation of the plant extracts
The leaves of A. sisalana were collected from plants growing at the Agronomic Ins-titute of Pernambuco (Brazil) and identified by the Botany Department there. The reference samples were deposited at “Dárdano de Andrade Lima Herbarium” at Agronomic Insti-tute of Pernambuco. After collection, the leaves were washed in distilled water, dried at room temperature, and subsequent-ly triturated. The extracts were obtained according to Lopes et al. (2018). Aqueous extracts of A. sisalana (AELAs) were prepared from 20 g the plant material mixed with 80 mL of a 0.15 M NaCl solution, for a final concentration of 250 mg.mL−1 (w/v). The suspension was agitated for 16 hours at 4 °C, followed by filtration and centrifuging at 10.000 rpm for 15 min (4 °C). Hydroethanolic extracts (HELAs) were prepa-red from 20 g of the plant material infused in 70% ethyl alco-hol (80 mL) for two hours, followed by filtering. The alcohol was then evaporated for 16 hours at 45 °C. The extracts (250 mg.mL−1) were then diluted (in a 0.1% solution of Tween 80) to+ final concentrations of 100, 50, 25, and 10 mg.mL−1.
Species of Cordyceps
For the pathogenicity bioassay, three species of Cordyceps were selected due to the poten-tial in termite in vitro control, using the strains C. farinosa ESALQ1355, C. fumosorosea ESALQ1297 and C. javanica URM4993 in the lethal concentration pre-established, having presented excellent percentages of germination and being effective in the control of N. corniger workers, according to Lopes et al. (2017) (Table 1). C. javanica URM4993 from the URM collection at the Federal University of Pernam-buco/UFPE; C. farinosa ESALQ1355 and C. fumosorosea ESALQ1297 from the ESALQ Collection of Microorganisms at Escola Superior de Agricultura Luiz de Queiroz – ESALQ of the University of São Paulo/USP (Table 1). The fungi were cultivated in Sabouraud-Dextrose-Agar (SDA) for 12 days, after that time, the conidia of each species were transferred to 10 mL of a Tween 80 (0.1%) solution and quantified using a Neübauer chamber, being then diluted to a final concentration of 1 x 107 conidia.mL−1. This conidia solution was used for experiments of compatibility with sisal extracts and to obtain LC50 values using N. corniger workers.
Nasutitermes corniger
Nasutitermes corniger. Parts of termite mounds located at Fe-deral University of Pernambuco (UFPE), State of Pernambu-co (Brazil). They were collected with the aid of a hatchet and conditioned in plastic pots. The samples were identified by Dr. Auristela Correia de Albuquerque, from the Federal Rural University of Pernambuco (UFRPE) / Brazil. The identifica-tion of termites was carried out using the key for neotropical termite genera (Constantino 2002) and by comparing the mor-phological characteristics with the specimens of the species deposited in the Collection of the Order Isoptera of the De-partment of Biology at UFRPE.
The strains of Cordyceps used in the experiments.
Source: Lopes et al. (2017). *95% confidence interval.
Effects of the plant extracts on the fungi strains tested
The effects of the extracts on the Cordyceps spp. were eva-luated by measuring germination, vegetative growth, and the sporulation of the fungal. These experiments were performed on the three strains of Cordyceps used in the toxicological studies, using the two sisal extracts at five different concen-trations, as well as a control (without the sisal extract), tota-ling 33 treatments, with three repetitions. To evaluate conidia germination, a 1 mL suspension (1 x 108conidia.mL−1) of each of the fungal specie was inoculated into 9 mL of each extract concentration with Tween 80 (0.1%) (Tween solution without any extract served as the control) to obtain suspensions with 1 x 107 conidia.mL−1. After one hour, 0.1 mL of those suspen-sions were inoculated into Petri dishes containing SDA, which were subsequently incubated in a BOD chamber (26 ± 1 °C, 80 ± 10% RH). Germination was determined after 16 hours by observing 500 conidia (both germinated and non-germina-ted); the germination percentages were calculated using the formula (G = n x 100/500), following Alves & Pereira (1998) To evaluate vegetative growth and sporulation, the extracts were added to the SAD media in autoclaved Petri dishes while it was still liquid (45 °C) to final concentrations of 10, 25, 50, 100 and 200 mg.mL−1. Filter paper disks (0.3 mm diameter) with 0.01 mL of a conidia suspension (1 x 107 conidia.mL−1) of each strain of Cordyceps tested were placed in the Petri dishes with SDA containing the sisal extracts at test concen-trations and subsequently incubated in a BOD chamber (26±1 °C, 80 ± 10 % RH) for 12 days. Mycelial growth was deter-mined by measuring the diameters of the colonies. To evalua-te sporulation, fragments of the vegetative colonies (cultured as above) were transferred to test tubes containing 10 mL of Tween 80 (0.1%) solution, and the suspensions agitated for approximately 2 min in a vortex mixer; the conidia from each were then quantified by counting using a Neübauer chamber.
Toxicity efficiency of the sisal extracts on Nasutitermes corniger
The toxicity efficiency analyses of sisal extracts on N. corniger were according to Kang et al. (1990). Filter pa-per disks (4 cm diameter) were impregnated with 0.2 mL of the extract solutions (10, 25, 50, 100, and 200 mg.mL−1) as well as with a 0.1% Tween 80 solution (control) and were subsequently dried at room temperature (24 ± 1 °C). The treated disks were then transferred to Petri dishes (90 x 15 mm) containing a small amount of humid cotton to main-tain humidity levels. A total of 20 termites (4 soldiers and 16 workers) were carefully transferred to the Petri dishes, in the pre-established proportion of 1:4 for N. corniger to maintain the natural interdependence of the castes and guar-antee the maximum survival of those insects outside of their nest, following Vasconcellos & Bandeira (2006). The insects were then carefully transferred to the Petri dishes and main-tained at 26 ± 1 °C, relative humidity ± 80%, in the dark. The experiment was therefore composed of 11 treatments (two types of extracts, five different concentrations of each, and one control) with five repetitions, totaling 100 insects per treatment (20 soldiers and 80 workers). Mortality was evalu-ated daily until the death of the last insect; the survival per-centages and lethal concentrations (LC50) were subsequently calculated. The LC50values obtained were compared by the Chi-square Test.
Evaluations of associations plant extracts with Cordyceps spp. on Nasutitermes corniger
This experiment sought to determine the LC50 of each plant extract with the LC50 of the fungi (Tables 1 and 2). Paper filter disks (4 cm in diameter) were impregnated with 0.2 mL of suspensions containing the plant extracts and the fungi, as well as with extracts and fun-gi separately (which would serve as control treatments). The disks were dried at room temperature and transferred to plas-tic Petri dishes (90 x 15 mm) containing 16 worker termites, which were then maintained at 26 ± 1 °C, relative humidity ± 80%, in the dark. The experiment was composed of 17 treatments (with five repetitions), totaling 80 insects per treat-ment; the percent mortality was evaluated after four days. In order to confirm fungal infection and consequent insect death, disinfection of insects with 70% alcohol, 4% sodium hypo-chlorite and sterile distilled water was carried out to eliminate microorganisms on the surface of the insect and subsequently transferred for a wet BOD chamber (26 ± 1 °C and 80% ± 10% RH) to the exteriorization of the fungal mycelium, ac-cording to Alves (1998). Afterwards, the material was ino-culated in test tubes containing SAD + Chloramphenicol, to obtain the fungal growth in the period of 12 days, without the presence of bacteria. Afterwards, the aspects of the colonies and the characteristics of the microstructures (hyphae, my-celium, conidiophores and conidia) of the re-isolated species were verified by means of mycelial growth techniques in Pe-tri dishes and cover crops, respectively, according to Domsch et al. (2007). The results obtained were compared with the literature (Samson et al. 1988; Lopes et al. 2016) and for the confirmation of re-isolated species and the percentage of mor-tality of the workers of N. corniger.
Statistical analyses
Data concerning the effects of extracts on biological aspects (germination, mycelial growth, and sporulation) of Cordyceps spp. were submitted to analysis of variance (ANOVA) using the SAS ANOVA Proc (SAS Institute, 1999-2001) and the means were compared by the Tukey test at a 5% level of probability. The extracts toxicity on Cordyceps spp. was determined by the Biological Index (IB), obtained by means of the formula IB = 47 [CV] +43 [ESP] +10 [GERM] / 100 (CV = the percentage of vegetative growth; ESP = % of sporulation; GERM = % conidia germi-nation), all in relation to the control. The IB index may vary from: 0 - 41 (toxic), 42 - 66 (moderately toxic) and more than 66 (compatible) (Rossi-Zalaf et al. 2008). Using the termite mortality data from the extracts, the survival rates (%) were determined for each treatment and the data were submitted to the Long-Rank test, using the Kaplan-Meyer method by pairs of isolates, through the SAS Proc Lifetest (SAS - Institute 1999-2001). The mean Lethal Concentrations (LC50) was de-termined after the fourth day of treatment, using Proc Probit Software (SAS - Institute 1999-2001). Data on mortality of termites caused by the combination of fungi + extracts were submitted to analysis of variance (ANOVA) using the SAS ANOVA Proc (SAS - Institute, 1999-2001) and the means were compared by the Tukey test at a 5 % level of probability. The control data of the termites were presented graphically using Software GraphPad Prism (Software Prism 2016).
Results
The A. sisalana extracts reduced the survival indices of both the workers and soldiers of N. corniger at all of the concen-trations tested, differing from the control treatment (in which the insects survived until the 11th day) (p < 0.05). Exposure to the aqueous (AELAs) and hydroethanolic (HELAs) ex-tracts at concentrations of 200 mg.mL and 100 mg.mL re-sulted in the deaths of 100% of the workers after the third and fourth days of exposure; the other concentrations result-ed in the deaths of 100% of the workers between the fifth and sixth day (Figs. 1A and 1C). The soldiers treated with the EAAs solutions survived until the fourth day when exposed to extract concentrations of 50 mg.mL−1, 100 mg.mL−1, and 200 mg.mL−1 (Fig. 1B), whereas the HELAs solutions result-ed in deaths of 100 % of the soldiers after the fifth day at the same concentrations (Fig. 1D). At lower concentrations the soldiers survived until between the sixth and seventh day. These data differed significantly from the control treat-ment, in which all insects remained alive until the 11th day (p < 0.05).
There were no significant differences between the LC50 indices of the workers and soldiers of N. corniger exposed to the AELAs and HELAs, with LC50 of 8.0 mg.mL−1and 7.0 mg.mL−1, and 9.7 mg.mL−1and 9.5 mg.mL−1, respectively (Table 2).
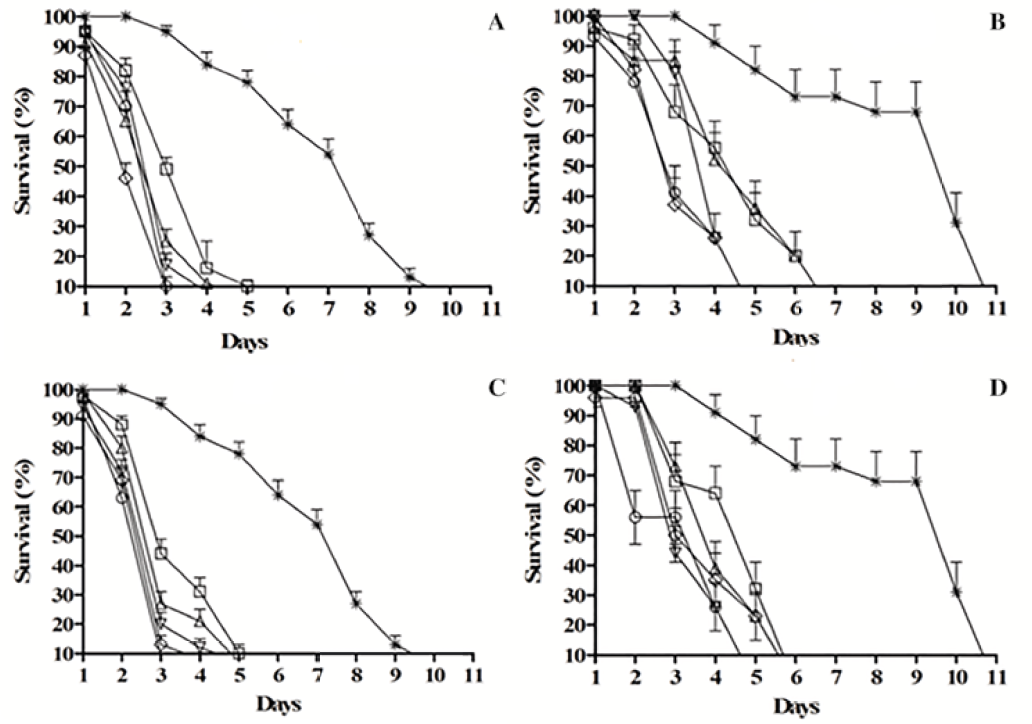
Daily survival (%) of workers and soldiers of Nasutitermes corniger treated with aqueous and hydroethanolic extracts of Agave sisalana evaluated until death of the last individual by the Long-Rank test: AELAs applied to workers (A) and soldiers (B). HELAs applied to workers (C) and soldiers (D), at concentrations of 10(□), 25(△), 50 (+), 100 (x), 200 mg/L (♦), and the control (*). Each point represents the mean ± SE of five repetitions.
Lethal concentrations (LC50) of Agave sisalana extracts on Nasutitermes corniger.
95% confidence interval.
Significant analyses by the Chi-square Test.
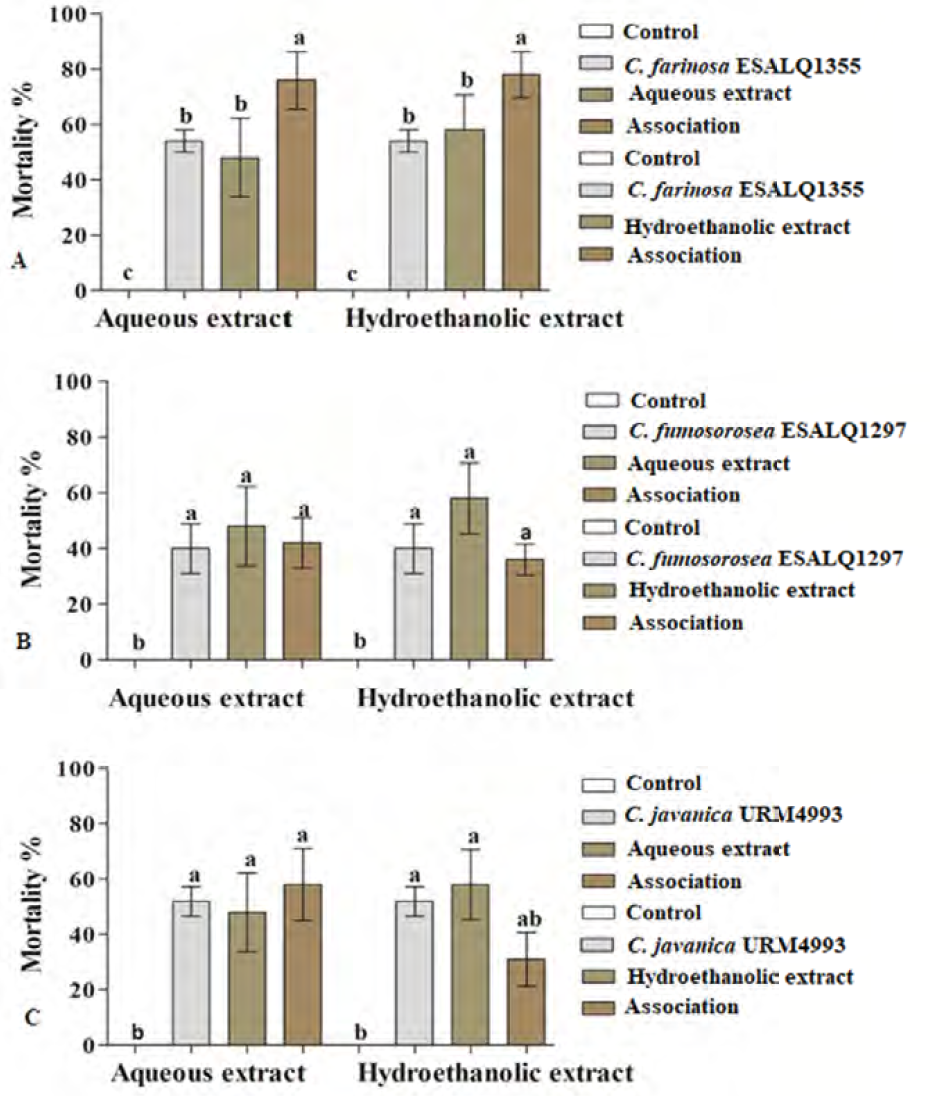
Effects of the association of fungal strains and aqueous and hydroethanolic ex-tracts of Agave sisalana on the workers of Nasutitermes corniger: Cordyceps farinosa ESALQ1355 (A) (df: 5, F= 8.7812, p < 0.01), Cordyceps fumosorosea ESALQ1297 (B) (df = 5, F = 5.8253 p < 0.01), and Cordyceps javanica URM4993 (C) (df = 5, F = 5.7618, p<0.01). Different letters on the bars indicate statistical differences between them by the Tukey test (p = 0.05).
The effects of the A. sisalana extracts on the biological aspects of the fungal strains can be observed in Table 3. No decreases in the germination percentages of the conidia of any of the strains were noted at the different extract concentra-tions, except with C. fumosorosea ESALQ1297 (which de-monstrated a significant decrease in germination (33%) when exposed to the highest concentration of the aqueous sisal extract (p < 0.05). The different AELAS did not negatively affect the fungal strains at lower concentrations (10 mg.mL−1 and 25 mg.mL−1) in terms of micellar growth and sporulation, which did not differ from the control treatment (p < 0.05). The HELAs, however, did promote diminished micellar growth of C. farinosa ESALQ1355 and C. fumosorosea ES-ALQ1297, although these extracts were well-tolerated by C. javanica URM4993 at low concentration (10 mg.mL−1 and 25 mg.mL−1). These hydroethanolic extracts also negatively im-pacted sporulation, except in C. farinosa ESALQ1355, which demonstrated compatibility with extract treatments at concen-trations of 10, 25, and 50 mg.mL−1, which did not differ from the control (p < 0.05).
According to the value of the Biological Index (BI), the extracts were considered compatible with the fungi, at the concentrations tested, except aqueous, classified as toxic for C. fumosorosea ESALQ1297, at 200 mg.mL−1 concentra-tion, presenting a biological index of 36.34 (Table 4).
The termiticidal activities associated with the sisal ex-tracts combined with the Cordyceps strains against N. corni-ger workers can be seen in Figure 2. The associations of the AELAs and HELAs extracts with C. farinosa ESALQ1355 were efficient in causing the death of approximately 78% of the workers, with significant increases in the mean mor-tality as compared to applications of only the aqueous (47%) hydroethanolic (61%) extracts, or just the C. farino-sa ESALQ1355 strain (53%) (p < 0.05) (Fig. 2A). The as-sociations of the extracts with the fungal strains C. javanica URM4993 and C. fumosorosea ESALQ1297 did not result in synergetic actions in controlling N. corniger, as their combined the uses did not result in increases in the mean mortality rates caused by the fungi acting alone (p < 0.05) (Fig. 2B, C).
Effects of aqueous and hydroethanolic extracts of Agave sisalana on the germination (%), growth (cm), and sporulation (1 x 107 conidia.mL−1) of Cordyceps.
Means followed by the same letter in the same column do not differ significantly at a 5% level of probability by the Tukey test. *Germination, **Growth, ***Sporulation
Compatibility of Cordyceps with extracts of leaves of Agave sisalana.
*Biological Index: C-compatible (above 66), MT-moderately toxic (42-66), T-toxic (0-41).
Discussion
Our data showed the efficiency of A. sisalana extracts in dimi-nishing the survival rates of workers and soldiers of the termite pest N. corniger, indicating that the extracts contain primary and/or secondary metabolites with insecticidal properties. Plants are known to be rich sources of bioactive compounds that demonstrate toxicity to termites, repel them, or inhibit their consumption of potential food sources (Scheffrahn 1991; Chen et al. 2004) but that are, at the same time, inoffensive to humans and the environment (Omena et al. 2007).
The AELAs and HELAs extracts showed insecticidal ac-tivity in relation to N. corniger, causing the deaths of all the workers and soldiers at a concentration of 10 mg.mL−1 (after five and seven days, respectively); at higher concentrations, insect survival varied between three and five days, with LC50 values varying between 7.0 mg.mL−1 and 9.7 mg.mL−1. These data corroborate those of Santana et al. (2010), who repor-ted the susceptibility of N. corniger to extracts of Bowdichia virgilioides Kunth (Fabaceae), with all the termites dying af-ter the fourth day when treated with extract concentrations of 100 mg.mL−1 (LC50 7.2 mg.mL−1); likewise ethyl acetate extracts of Anadenanthera colubrina (Vell.) Brenan (Faba-ceae), provoked the death of 100% of the insect individuals after seven days when tested at concentrations of 25 mg.mL−1, 50 mg.mL−1, and 100 mg.mL−1 (LC50 17.3 mg.mL−1). Other stu-dies proved that a lectin (CrataBL) extracted from the bark of Crataeva tapia L. (Capparaceae) was an efficient insectici-de against N. corniger workers, causing the deaths of all of the individuals after the sixth day of treatment (LC50 0.475 mg.mL−1) (Araújo et al. 2012).
The termiticidal actions of sisal extracts are apparently related to the toxicity of primary and secondary metabolites produced by that plant, indicating that these compounds con-tain toxic properties against N. corniger workers and soldiers (or render their natural foods impalpable). Studies of the re-sidual liquids and extracts of A. sisalana have been shown to be efficient against A. aegypti mosquito and the red mite T. urticae (Barrêto et al. 2010; Nunes et al. 2015). Bioactive compounds produced by plants, such as alkaloids, tannins, terpenoids, glycosides, phenolic compounds, flavonoids, and phenylpropanoids are known to have attractant, deterrent, and insecticidal properties (Chen et al. 2007; Melo-Santos et al. 2009); flavonoids are widely distributed among plant species and show insecticidal actions against termites (Oh-mura et al. 2000). Primary metabolic compounds such as lec-tins have been shown to control N. corniger under laboratory conditions, and the lectins extracted from the bark (MuBL) and heartwood (MuHL) of Myracrodruon urundeuva Fr. All. (Anacardiaceae) were also found to be efficient in controlling that termite, causing the deaths of 100 % of the workers (LC50 0.374 mg.mL−1and 0.974 mg.mL−1) and soldiers (LC50 0.432 mg.mLand 0.78 mg.mL−1) (Napoleão et al. 2011). Simi-larly, a lectin (BmoRoL) extracted from the roots of Bauhinia monandra Kurz (Fabaceae) demonstrated termiticidal activi-ties against workers and soldiers of an arboreal termite after 12 days of exposure (LC50 0.09 mg.mL−1and 0.395 mg.mL−1), demonstrating its potential bio-technological use against tho-se pests (Souza et al. 2011).
The utilization of fungi as bio-insecticides requires the previous selection of fungal species and strains to determine their virulence, their reproductive aspects, and their mass pro-duction under artificial culture conditions (Ambethgar 2009; Lopes et al. 2011). These pathogens can be used in associa-tion with plant extracts to increase their effectiveness, althou-gh it will be necessary to examine the extract concentrations most compatible with them.
In general, the A. sisalana extracts were compatible with the fungal strains tested here at the lowest concentrations, not causing any detectable inhibitory effects to their sporulation, germination, or growth. Studies show that the compatibili-ty of entomopathogenic fungi with insecticides or chemical substances derived from plants is important when considering their combination for the control of insect pests (Amjad et al. 2012, Silva et al. 2015; Santos et al. 2015). Studies examining the compatibility of chemical products natural our metabo-lic compounds with entomopathogenic species of Cordyceps have revealed diverse effects on their growth, sporulation, and germination (Marques et al. 2004; Demirci et al. 2011; Amjad et al. 2012). The effects of the oil derived from Azadirachta indica A. Juss. (Meliaceae) on the vegetative growth, sporu-lation, and germination of the fungus Metarhiziuim aniso-pliae (Metchnikoff) Sorokin, Beauveria bassiana (Balsamo) Vuiillemin, and C. farinosa were examined by Marques et al. (2004), who found that the plant oil reduced fungal grow-th and sporulation but did not affect conidia viability. Xu et al. (2011) likewise examined the larvicide synergism of C. fumosorosea in combination with the secondary metabolic compound 20-hydroxyecdysone and concluded that the latter did not negatively influence germination, vegetative growth, or fungal conidia production. Matsuura & Matsunaga (2015), however, reported that the pheromones ethyl n-butyl-n-bu-tyrate and 2-methyl-1-butanol extracted from the termite queen of Reticulitermes speratus (Kolbe, 1885) (Isoptera: Rhinotermitidae) had antifungal properties that significant-ly reduced growth, sporulation and conidia germination in C. farinosa and M. anisopliae. The effects of certain chemi-cal products on micellar growth and conidia germination in C. farinosa were reported by Demirci et al. (2011), althou-gh tebuconazole, penconazol, and nuarimol were not found to affect conidia germination or micellar growth. Likewise, Gurulingappa et al. (2011) evaluated the actions of nine in-secticides on the fungus Lecanicillium lecanii (Zimmermann) Gams & Zare and reported that thiamethoxam and acetami-prid did not affect conidia germination or micellar growth. Micellar growth and conidia germination in C. fumosorosea and Lecanicillium muscarium (Zimmerman) Viegas varied according to the concentrations and types of pesticides they were exposed to, with azocyclotin being the most toxic to co-nidia germination; acetamiprid was found to be the insectici-de most compatible with C. fumosorosea (Amjad et al. 2012).
The extracts of A. sisalana did not cause severe damage to the conidia germination, the mycelial growth of Cordyceps strains, this fact was confirmed by the values of the biological indexes, which, in general, did not cause toxicity to the tested strains. The selection of entomopathogenic fungi compatible with plant extracts is essential to determine the most efficient strains to be tested in association with these products in the control of insect pests.
Previous studies have reported the effectiveness of ento-mopathogenic fungi in the laboratory to control Nasutitermes species, with mortality percentages greater than 40%, highli-ghting the Isaria (Cordyceps) and Metarhizium species (Lo-pes et al. 2017, Quiroz-Medina et al. 2021). However, the effectiveness of entomopathogenic fungi is not always repro-duced in field experiments, as reported by Quiroz-Medina et al. (2021), who found that M. anisopliae and B. bassiana caused the death of 24% and 7%, respectively, on N. corniger castes. The failure of fungal infections can be attributed to the actions of insects against the pathogens, as they isolate and remove infected individuals from termite mounds, pre-venting the spread of fungal spores; in addition, they produce inhibitory and defensive metabolites, such as pheromones and terpenoids with antimicrobial action (Rath, 2000; Rosengaus, 2000; Yanagawa et al. 2012; Matsuura and Matsunaga, 2015). The use of plant extracts in association with fungal species may have stressed those termites and facilitated the fungal infection. As such, associations of entomopathogenic fungi with chemical insecticides or plant extracts can amplify their effects against insect pests while diminishing environmental damage (Amjad et al. 2012).
The associations of the EAAs and EHEAS extracts with C. farinosa ESALQ1355 increased its pathogenicity to N. cor-niger workers, with percentage mortalities much greater than either the fungus or the extracts alone – demonstrating that the extracts increase the potency of C. farinosa ESALQ1355 in infecting the termites, making them most potent at concentrations of 6.66 x 104 conidia.mL−1 in conjunction with LC50 doses of the extracts. Similar results were reported by Xu et al. (2011) in their analyses of the action of C. fumosorosea against Plutella xylostella L., 1758 (Lepidoptera: Plutellidae) when associated with different concentrations of 20-hydrox-yecdysone; the percentage mortalities of the insects were re-lated to the concentrations of each component in the solution, with their cumulative effects being more evident with greater exposure times. Similarly, Santos et al. (2015) examined the insecticidal actions of extracts (water and hydro ether) of Ri-cinus communis L. (Euphorbiaceae) and Poincianella pyra-midalis (Tul.) L.P. Queiroz (Fabaceae) and their effectiveness in association with the F. incarnatum-equisetispecies com-plex (FIESC) against the cochineal insect D. opuntiae, finding a synergistic effect of the fungus with an aqueous extract of R. communis that caused 100% mortality of the insects. The combined utilization of plant extracts with entomopathogenic fungi can increase the efficiency of the biological control of insect pests and reduce the economic costs and environmen-tal impacts of chemical insecticides (Marques et al. 2004; Ambethgar 2009). The association of extracts of A. sisalana with C. farinosa ESALQ1355 may facilitate the penetration of the teguments of the worker termites by their conidia due to stress provoked by the extracts. It has been shown that after the deaths of termite workers exposed to entomopathogenic fungi, the conidia are easily dispersed to other members of the colony during their social interactions, increasing the ki-lling potential of the fungal inoculum in the nest environment (Chouvenc et al. 2011; Lopes et al. 2011). Termites' frequent diverse environments, which can make them susceptible to infections and promote the rapid transmission of pathogens (Hamilton et al. 2011).
Conclusions
The termite N. corniger causes severe economic damage in urban areas in northeastern Brazil as well as other countries throughout the world, creating the necessity for modern te-chniques that can control those pests without impacting the environment with chemical pollutants. Our results demons-trated the extracts of A. sisalana is compatibility with all the Cordyceps fungal strains tested, end the insecticidal activi-ties of A. sisalana extracts on the soldiers and workers of N. corniger, as well as the efficiency of the association of those extracts with C. farinosa ESALQ1355 on workers, indicating their potential joint usefulness in the biological control of N. corniger.
Origin and funding
The present work is a result from a essay on biological and alternative control of insect pests carryed out at the Federal University of Pernambuco - UFPE and the Agronomic Ins-titute of Pernambuco - IPA, Brazil. The financial for the re-search activities, was provided by the Fundação de Amparo à Ciência e Tecnologia do Estado de Pernambuco - FACE-PE, grants APQ-1133-2.07/12, and IBPG-0040-2.08/09; the National Scientific Development Council – CNPq, grant no. 314082/2009-3; the High Education Human Development Coordination – CAPES, grant PROCAD/NF_519/2010, and the Brazilian Northeastern Bank - BNB.
Author contribution
Rosineide da Silva Lopes and Vera Lúcia de Menezes Lima conceived and designed research. Elza Áurea de Luna Alves Lima, Luciana Gonçalves de Oliveira and Rosineide da Silva Lopes analyzed data. Rosineide da Silva Lopes, Mônica Cristina Barroso Martins and Geiziquele de Lima conducted experiments. Auristela Correia de Albuquerque identified the termite species. All authors wrote, critically revised and approved the manuscript.
Conflict of Interest
The authors declare that they have no conflict of interest.
Footnotes
Acknowledgements
Appreciate the Brazilian agencies: “Fundação de Amparo à Ciência e Tecnologia do Estado de Pernambuco” (FACEPE) grant (APQ-1133-2.07/12, and IBPG-0040-2.08/09), “Con-selho Nacional de Desenvolvimento Científico e Tecnoló-gico” (CNPq; grant no. 314082/2009-3), “Coordenação de Aperfeiçoamento de Pessoal de Nível Superior” (CAPES; grant PROCAD/NF 519/2010), and “Banco do Nordeste do Brasil” (BNB) for their financial support.
