Abstract
The present study aimed to evaluate the CO₂ release by infective juveniles (IJs) and the viability of mutualistic bacteria of entomopathogenic nematodes (EPNs) when exposed to synthetic insecticides. Two species of EPNs, Heterorhabditis amazonensis JPM4 and Steinernema carpocapsae All, were tested. The applied insecticides were Vertimec® (abamectin) and Klorpan® (chlorpyrifos). Gas chromatography was employed for CO₂ analysis. The bacteria were isolated and the colony development was evaluated in sterile Petri dishes. Vertimec® triggered greater CO₂ release by the nematodes, associated with changes in their metabolic activity, than Klorpan®. Vertimec® and Klorpan® did not inhibit the bacterial development.
Introduction
Entomopathogenic nematodes (EPNs) (Rhabditida: Heterorhabditidae, Steinernematidae) are key control agents of insect pests because of their association with mutualistic bacteria Xenorhabdus and Photorhabdus, which are released into the hemocoel of insects by infective juveniles (IJs) of the genera Steinernema and Heterorhabditis, respectively, causing septicemia and rapid death of the host within 24 to 48 h (Adams and Nguyen 2002). The use of EPNs as a component of an integrated pest management (IPM) in agricultural systems involves combining them with various other agricultural inputs, including chemical products, fertilizers, and acidity correctors. However, those products may reduce the viability and infectivity of EPNs (Grewal et al. 2001). Thus, the present study aimed to evaluate the CO₂ release by IJs and the activity of mutualistic bacteria of EPNs when exposed to synthetic insecticides to assess factors associated with reductions in EPNs infectivity to insect pests. The selected insecticides in this study have proved no effect on the viability of IJs but reduced their ability to infect G. mellonella larvae (Sabino et al. 2014).
Materials and methods
Rearing of the nematodes
The nematodes used for the bioassay were Steinernema carpocapsae All (isolated from a soil sample in North Carolina, USA) and Heterorhabditis amazonensis JPM4 (isolated from a soil sample in Lavras, Minas Gerais, Brazil), which have potential for the control of Tuta absoluta (Meyrick) (Lepidoptera: Gelechiidae) in tomatoes. They were prepared from stocks maintained as aqueous suspensions (500 IJs/ml) at 16 °C ± 1 °C (Andaló et al. 2011).
The nematodes were reared in the last larval instar of Galleria mellonella L. (Lepidoptera: Pyralidae), according to Kaya and Stock (1997). The G. mellonella larvae were propagated according to the rearing method adapted by Dutky et al. (1964) using the artificial diet modified by Parra (1998), composed of wheat bran, wheat flour, wheat germ, powdered milk, brewer's yeast, glycerin, honey and water.
Bioassay to measure the release of CO₂
The method suggested by Sabino et al. (2015) was used to analyze the CO₂ release by the nematodes to evaluate the effect of insecticides on their metabolic activity. The synthetic insecticides evaluated were Vertimec 18 CE® Syngenta Switzerland active ingredient abamectin, toxicological classification (TC) III, at 1.2 L/400 L water application dose and Klorpan 480 CE® Nufarm Australia active ingredient chlorpyrifos, TC I, at 800 ml/400 L water application dose. These dosages are recommended doses for the control of T. absoluta in a tomato crop.
The method suggested by Negrisoli Jr et al. (2008) was employed to expose the IJs to the insecticides. One liter of each pesticide broth was prepared at double the recommended dose for use in one hectare. One ml aliquot of the mentioned solution was added to a volume of 9 ml water in a Vacuette®-type plastic tube (16 x 100 mm). Following that procedure, 1 ml of suspension containing 1.500 IJs was added to each tube. The following treatments were established for each of the two nematode species: control (distilled water + IJs suspension), IJs suspension + Vertimec®, and IJs suspension + Klorpan®, performing ten replicates per treatment and considering each tube a replicate.
The tubes were maintained in a temperature-controlled chamber at 27 ± 1 °C for 48 h. The gas chromatography analysis was then performed using a GC 2010 gas chromatograph with a thermal conductivity detector (TCD) at 250 °C with a polarity of +50 mV, a 220 °C injector temperature, and He as the loading gas at a linear speed of 50 cm s⁻¹. The initial column (RT-QPLOT - 30 m x 0.32 mm DI x 10 µm) temperature was 50 °C, which was maintained for 3.5 min and then increased at 50 °C min⁻¹ to 150 °C, remaining at 150 °C for 2 min. The injection was conducted in the 1:20 split mode. The total analysis time was 7.50 min. The quantification was performed by external standardization. The bioassay was conducted under aerobic conditions because oxygen entered the tubes when they were opened to add nematodes and insecticides. This has no effect on the results because the amount is negligible for the analyses (Sabino et al. 2015).
Bioassay of mutualistic bacteria
Aqueous suspensions of 1000 IJs/ml of the nematodes S. carpocapsae All and H. amazonensis JPM 4 were prepared to evaluate the effects of the insecticides on the bacteria. One ml aliquot from each suspensions were added to a flat-bottom glass test tube with a diameter of 2 cm and a height of 8 cm. Subsequently, 1 ml of each insecticide, Vertimec® or Klorpan®, prepared at the same concentrations as the previous assay, or distilled water (for the control treatment) were added to each tube, performing ten replicates per treatment. The treatments were as follows: control (distilled water + IJs suspension), Vertimec® + IJs suspension, and Klorpan® + IJs suspension. The tubes were kept in a temperature-controlled chamber at 27 ± 1 °C for 48 h for the subsequent evaluation of colony development.
The method modified by Voss et al. (2009) for isolating mutualistic bacteria was followed; a 50-µL aliquot was collected from the mixture of nematodes and insecticide and mixed with 50 µL of 2% sodium hypochlorite for 5 minutes in a 9-cm-diameter sterile Petri dish. That process was conducted in a laminar flow hood to promote external asepsis of IJs. The mixture was re-suspended using a micropipette, removing 50 µL and transferring it to the same volume of sterile deionized water. Mixing of the suspension was performed by pipetting up and down and distributing the entire volume (100 µL) onto the nutrient agar (NA) plates (Kaya and Stock 1997) in a sterile glass Petri dish (9 cm in diameter) before spreading the suspension using a Drigalski spatula.
The plates were incubated in a temperature-controlled chamber at 23 ± 1 °C and monitored after 48 and 72 h. The colony development was analyzed by counting the number of colonies per plate. The data recorded for the difference in CO₂ released by both nematode species and the colony numbers per plate were submitted to SISVAR 5.6 software (Ferreira 2011) for analysis of variance (ANOVA) and Tukey's test (P < 0.05). The interaction between time and insecticide product was analyzed separately for each bacterial species.
Results and discussion
The results demonstrated that Vertimec® caused an increase in the concentration of CO₂ released by the IJs of S. carpocapsae All and H. amazonensis JPM 4 when in contact with the insecticide Klorpan®. The release of CO₂ by S. carpocapsae All in contact with Vertimec® was 5.9 x 10⁻⁴ mol/L and with H. amazonensis JPM 4 2.14 x 10⁻⁴ mol/L. In the control treatment the CO₂ release was 3.87 x 10⁻⁴ mol/L for S. carpocapsae All and 1.33 x 10⁻⁴ mol/L for H. amazonensis JPM 4 (Fig. 1).
The insecticide Klorpan® reduced the concentration of CO₂ released by the IJs of S. carpocapsae All (2.88 x 10⁻⁴ mol/L) compared with the control treatment (3.87 x 10⁻⁴ mol/L). There was no statistical difference in the concentration of CO₂ released by H. amazonensis JPM 4 exposed to Klorpan® and in the control treatment (1.33 x 10⁻⁴ mol/L) (Fig 1). The values recorded for the CO₂ concentrations both in the treatment with Klorpan® and in the control were lower than the values obtained in the treatment with Vertimec® for both nematode species tested (Fig. 1).
There were no significant differences between the control, Vertimec®, and Klorpan® treatments (F = 0.39; df = 2; P > 0.05) in the colony development of Xenorhabdus sp. bacteria after 48 and 72 h of exposure to the insecticides (Table 1). However, Photorhabdus sp. development regarding the control treatment differed from the Vertimec® and Klorpan® treatments at both 48 h and 72 h of growth (F = 12.94; df = 2; P < 0.05). Although the products reduced colony development of Photorhabdus sp., they did not cause cell death (Table 1). There was a significant difference between the two sampling times for both Xenorhabdus sp. (F = 37.08; df = 1; P < 0.05) and Photorhabdus sp (F = 14.47; df = 1; P < 0.05), with greater colony development at 72 h than at 48 h, indicating that the products fail to prevent the development of bacteria over time (Table 1).
The results demonstrate that certain insecticides alter the metabolic activity of EPNs. The Vertimec® caused a greater release of CO₂ by both nematode species than Klorpan®. In addition, Vertimec® and Klorpan® delayed the development of Xenorhabdus sp. and Photorhabdus sp., despite failing to cause their death. Studies that examine the physiological and biochemical traits of EPNs must be conducted to support their use in IPM programs. Combined application of nematodes and insecticides should be evaluated under field conditions (Koppenhofer and Fuzy 2008).
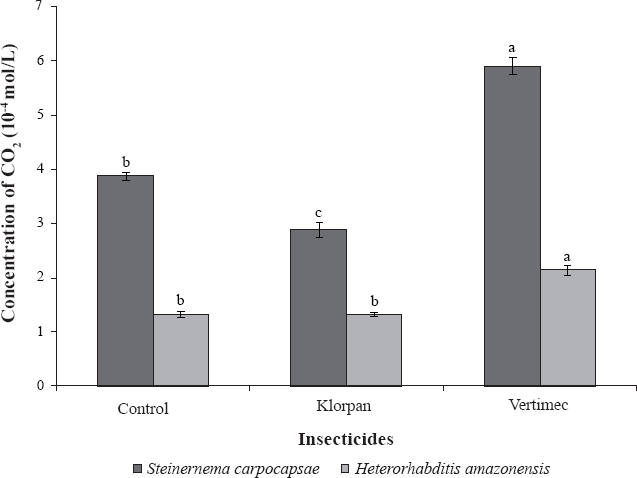
CO₂ release by infective juveniles of Steinernema carpocapsae All and Heterorhabditis amazonensis JPM 4 following exposure to the insecticides Vertimec® and Klorpan®. Means followed by the same letter are not significantly different from each other according to Tukey's test at the 5% significance level.
Number of Xenorhabdus sp. and Photorhabdus sp. colonies development (mean ± SE) after exposure to the insecticides Vertimec® and Klorpan® at two different intervals, 48 and 72 h (27 ± 1 °C, 70 ± 10% relative humidity, RH).
Means followed by the same uppercase letter within a row and lowercase letter within a column are not different from each other according to Tukey's tests at 5% significance.
Footnotes
Acknowledgements
The authors thank the Minas Gerais Research Foundation for financial support.
