Abstract
The objective of this study was to successfully synthesize epidermal growth factor receptor monoclonal antibody–conjugated superparamagnetic iron oxide nanoparticles (EGFRmAb-SPIONs) and explore their biocompatibility and potential applications as a targeted magnetic resonance imaging (MRI) contrast agent for the EGFR-specific detection of brain glioma in vivo. After conjugation of EGFRmAb with SPIONs, the magnetic characteristics of EGFRmAb-SPIONs were investigated. Thereafter, the targeting abilities of EGFRmAb-SPIONs with MRI were qualitatively and quantitatively assessed in EGFR-positive C6 glioma cells in vitro and in a Wistar rat model bearing C6 glioma in vivo. Furthermore, the preliminary biocompatibility and toxicity of EGFRmAb-SPIONs were evaluated in normal rats through hematology assays and histopathologic analyses. Statistical analysis was performed using one-way analysis of variance and Student t-test, with a significance level of p < .05. From the results of EGFRmAb-SPION characterizations, the average particle size was 10.21 nm and the hydrodynamic diameter was 161.5 ± 2.12 nm. The saturation magnetization was 55 emu/g·Fe, and T2 relaxivity was 92.73 s−1mM−1 in distilled water. The preferential accumulation of the EGFRmAb-SPIONs within glioma and subsequent MRI contrast enhancement were demonstrated both in vitro in C6 cells and in vivo in rats bearing C6 glioma. After intravenous administration of EGFRmAb-SPIONs, T2-weighted MRI of the rat model with brain glioma exhibited an apparent hypointense region within glioma from 2 to 48 hours. The maximal image contrast was reached at 24 hours, where the signal intensity decreased and the R2 value increased by 30% compared to baseline. However, T2-weighted imaging of the rat model administered with SPIONs showed no visible signal changes within the tumor over the same time period. Moreover, no evident toxicities in vitro and in vivo with EGFRmAb-SPIONs were clearly identified based on the laboratory examinations. EGFRmAb-SPIONs could potentially be employed as a targeted contrast agent in the molecule-specific diagnosis of brain glioma in MRI.
GLIOMAS are the most common aggressive brain tumors, with a very poor outcome. One of the major factors for treatment failure is largely due to challenges in eliminating infiltrating tumor cells. 1 Conventional magnetic resonance imaging (MRI) is still limited in its ability to delineate tumor cells within the normal brain. Cell-specific MRI for detection and treatment monitoring may hold great promise for the targeted therapy of brain gliomas, which is being intensely studied today. 2 The superparamagnetic iron oxide nanoparticle (SPION) delivery system has become a model system that could be used to study the target molecule–specific biodistribution, rapid exertion, and undesired side effects using in vivo small animal MRI.3–6
In addition, a target-specific receptor is the key point in the development of molecular probes in the field of molecule-specific imaging of brain glioma. A detailed description of the brain tumor receptor activity, which is beyond the scope of this article, provides a better understanding of the complicated mechanisms associated with tumor development and progression. As a cellular transmembrane receptor, epidermal growth factor receptor (EGFR) regulates important cellular processes and is linked to a poor prognosis in various human cancers.7–10 An increased expression of both wild-type and mutant EGFR is a common feature in brain glioma, and an aberrant EGFR signaling pathway has been shown to promote the development and progression of malignant tumors in relation to the cellular proliferation, angiogenesis, and metastasis.11–13 Several studies have revealed the diagnostic potential of MRI-coupled fluorescence molecular tomography to determine EGFR status in brain tumors, 14 and SPIONs conjugated with EGFRvIII (mutant EGFR)-specified antibody (EGFRvIIIAb) have also been proved to be feasible for MRI contrast enhancement and therapeutic target of glioblastoma. 15 However, EGFRvIIIAb may fail to target the subgroups of brain tumors with wild-type EGFR and other mutant receptors. This may limit their clinical application in the diagnosis of brain tumors. 16 Shevtsov and colleagues reported that epidermal growth factor (EGF) demonstrated much better targeting ability to all the subgroups of brain tumors with wild-type EGFR. They prepared SPIONs conjugated with EGF for the MRI of brain tumors. 16 As a natural ligand of EGFR, however, it has a potential risk of stimulating tumor growth and angiogenesis.17,18 In this case, the wild-type EGFR monoclonal antibody (EGFRmAb) seems to be more suitable to be used as a target-specific ligand in developing EGFR-targeted nanoprobes of brain glioma.19–21 Moreover, it has not yet been investigated whether EGFRmAb bioconjugated SPIONs (EGFRmAb-SPIONs) can be used for in vivo MRI detection of glioma.
In this study, we synthesized EGFRmAb-SPIONs and explored the magnetic characteristics, biocompatibility, and potential applications as a targeted MRI contrast agent for the EGFR-specific detection of brain glioma in vivo.
Materials and Methods
Materials
SPIONs were provided by Guerbet (Villepinte, France). N-Hydroxysulfosuccinimide (sulfo-NHS) and 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide hydrochloride (EDC·HCl) were purchased from Shanghai Medpep Co. (Shanghai, China). EGFRmAb [EGFR (A-10): sc-373746] was purchased from Santa Cruz Biotechnology (Santa Cruz, CA). All other reagents were analytical grade and used as received. C6 glioma cell lines were maintained in our laboratory and originated from American Type Culture Collection (ATCC).
Preparation of SPIONs and EGFRmAb-SPIONs
SPIONs coated with meso-2,3-dimercaptosuccinic acid (DMSA) were synthesized following the reported procedures. 22 The preparation of EGFRmAb-SPIONs was based on the established protocol by using coupling reagents, EDC·HCl and sulfo-NHS. 7 In brief, SPIONs (containing 1 mg iron), EDC·HCl (2 mg), and sulfo-NHS (4 mg) were first mixed in 0.4 mL of sodium acetate solution (1 M, pH 5.0) at 4°C for 30 minutes. Then the solution was separated by magnet, and the supernatant was removed. The pellet was suspended in 0.2 mL phosphate-buffered saline (PBS; 0.1 M, pH 7.4), and then EGFRmAb (1.5 mg/mL, 0.2 mL) in PBS (0.1 M, pH 7.4) was added. After 18 hours at room temperature, the desired product was collected as pellet after magnetic separation and washed with PBS (0.1 M, pH 7.4). The final EGFRmAb-SPIONs were redispersed and stored in PBS at 4°C. Part of the product was then lyophilized to obtain dry solid particles by the Labconco Freeze Dry System (−48°C, 133 × 10−3 mBar; FreeZone Plus 4.5 Liter Cascade Console, Labconco, Kansas City, MO) for further study. Control experiments were carried out similarly without the addition of coupling agent.
Characterization of EGFRmAb-SPIONs
The surface chemical properties of bare SPIONs and EGFRmAb-SPIONs were characterized by Fourier transform infrared spectroscopy (FTIR; VERTEX 70, Bruker, Ettlingen, Germany). For each sample, 2 mg of dried nanoparticles was mixed with 200 mg of potassium bromide and pressed into a pellet for analysis.
Magnetic properties were characterized by a vibrating sample magnetometer (VSM, Model 7404, Lake Shore Cryotronics Inc., Westerville, OH). The hysteresis of the magnetization was measured in a small polypropylene straw using a maximum field of 150 kOe at room temperature.
The size and morphology of nanoparticles were examined on a transmission electron microscope (TEM; Tecnai G2 20, FEI, Eindhoven, the Netherlands) at 200 kV. The particle size was counted through a visual analysis of approximately 200 particles in the TEM micrographs.
The hydrodynamic diameter and the size distribution of the EGFRmAb-SPIONs were measured by a dynamic light scattering instrument (DLS, Zetasizer Nano ZS90, Malvern Instruments Ltd., Worcestershire, UK) running DTS software (Version 7.11, Malvern Instruments Ltd., Worcestershire, UK) and operating at a 4 mW HeNe laser at 633 nm, with an angle of 90° at 25°C. An average hydrodynamic diameter and size distribution were reported from three runs of each sample. Total iron concentrations of EGFRmAb-SPION suspension were determined on a fast sequential atomic absorption spectrometer (Spectr AA 240FS, Varian, Palo Alto, CA). Weighed samples were suspended in nitric acid (2 mL) and perchloric acid (0.5 mL) and then heated at 280°C for 1 hour. The residue was dissolved and diluted into 10 mL of Milli-Q water. Iron concentration was determined at the specific Fe absorption wavelength (248.3 nm).
The T2 relaxivity of the nanoparticles was determined using a 3 T magnetic resonance scanner (Signa HDxt, GE Healthcare, Waukesha, WI) equipped with an eight-channel head coil. The particles were diluted in distilled water, with iron concentrations in the range of 0 to 20 μg/mL.
Samples were transferred to a 96-well plate. T2-weighted (T2WI) images were acquired using a multiecho pulse sequence. The parameters were set as follows: field of view (FOV) = 120 mm, base resolution = 192 × 160, slice thickness = 1.5 mm, interval thickness = 0.5 mm, repetition time (TR) = 2,000 ms, echo time (TE) = 20, 40, 60, 80, 100, and 140 ms. T2 relaxation rates were plotted against the iron concentrations in the particle dilutions. The T2 relaxivity was determined by a linear fit. The reported value was calculated from three samples.
In Vitro Cell Experiments
To evaluate the targeting efficacy of EGFRmAb-SPIONs in glioma cells, the cellular uptake of EGFRmAb-SPIONs was assessed with MRI of C6 rat glioma cell lines. The results were compared to that with SPIONs.
Preparation of EGFRmAb-SPION Solution
C6 glioma cell lines were cultured using a previously described approach.23,24 Briefly, cells were grown in Dulbecco's Modified Eagle's Medium (DMEM) containing 10% fetal calf serum (Gibco, Grand Island, NY) and 1% penicillin/streptomycin in a humidified atmosphere with 5% CO2 at 37°C. Cells were incubated for 1 to 2 days until they reached 80% confluence.
C6 glioma cells were seeded onto six-well plates with 2 mL culture medium at a density of 8 × 106 cells/well and incubated for 24 hours. C6 cells were then incubated with SPIONs and EGFRmAb-SPIONs (dissolved in DMEM) at different iron concentrations (0, 10, 20, 30, 40, and 50 μg/mL) at 37°C for 1 hour. The cells were next washed with PBS three times, added to cell culture medium, and blown gently using a pipette for 15 to 20 times.
In Vitro MRI
After incubation with various nanoparticles, the cells were washed and then dispersed and suspended by 300 μL 0.5% agarose gel. Samples were then quickly transferred to a 96-well plate. MRI was performed on the same 3.0 T magnetic resonance scanner (Signa HDxt) using the same parameters mentioned above.
In Vitro Cytotoxicity
Human normal liver cells (HL-7702) and human umbilical vein endothelial cell (ECV304) were used for cell viability studies according to a previous report. 7 The EGFRmAb-SPION solution was added into a dilution series (cell medium containing 0, 25, 50, 75, and 100 μg/mL Fe, respectively). The control group was prepared with the culture medium of SPIONs at the corresponding Fe concentration. After 24, 48, and 72 hours of incubation, 20 μL of 3-(4, 5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) (5 mg/mL) was added to each well. After incubation for 4 hours, formazan crystals were solubilized by 100 μL of isobutanol in the incubator overnight. The absorbance of each well was read on a microplate reader (1420 multilabel counter, PerkinElmer, Waltham, MA) at 560 nm. The cell viability (%) relative to the control wells containing cell culture medium without nanoparticles was calculated by the equation
where [A]test is the absorbance value of the tested cells and [A]control is the absorbance value of the control group. The average result was calculated from six samples.
In Vivo Animal Experiments
This study was approved by the Institutional Animal Care and Use Committee (IACUC), and all procedures conformed to the National Guides for the Care and Use of Laboratory Animals.
Animal Model Creation and Housing
Experiments were performed on adult male Wistar rats (250–300 g) provided by the Center of Experimental Animals in our institution. Animals were maintained in a temperature- and humidity-controlled environment under a standard 12-hour light and 12-hour dark cycle with ad libitum access to food and water. 25 The animal condition was closely monitored throughout the experimental period.
Wistar rats (n = 12) were anesthetized with 10% chloral hydrate (intraperitoneal, 0.4 mL/100 g body weight [BW]) and placed in the prone position in a stereotaxic frame (Stoelting, Wood Dale, IL). After the calvarium was exposed by a midline scalp incision from the nasion to the superior nuchal line, a hole was drilled into the brain basal ganglia (1.0 mm posterior and 3.0 mm lateral to the bregma). A 20 μL microinjector was then used to inject 10 μL of the 2 × 106 C6 cells, which were suspended in DMEM. The injection was at a rate of 0.5 μL/min and lasted 10 minutes. The needle was then withdrawn after another 10 minutes to prevent the possible leakage of solution. The skin was closed with nonmagnetic sutures, and the rats were allowed to recover. All surgical procedures were performed using aseptic techniques.
Confirmation of Brain Tumor Model
After operation, the individual rat was fed in a single cage. In vivo T2WI MRI was performed to confirm the success of brain tumor model after tumor growing for 11 days. All MRIs were performed on a 3.0 T magnetic resonance system (Signa HDxt), with a research coil insert designed specifically for rat imaging. The parameters of axial T2WI are as follows: TR = 3,000 ms, echo time (TE) = 50 ms, FOV = 60 mm, excitations (NEX) = 6, base resolution = 192 × 160, slice thickness = 1.5 mm, interval thickness = 0.5 mm. After MRI examinations, histologic specimens of brain tumor tissues were obtained and stained with hematoxylin-eosin (H&E) followed by standard clinical laboratory protocols.
Nanoparticle Injection and MRI
After rats were anesthetized with 10% chloral hydrate (intraperitoneally; 0.4 mL/100 g BW), precontrast MRIs were obtained prior to the injection of the nanoparticles and served as the baseline. A research coil insert designed specifically for rat MRI was used when the rat was placed in the imaging chamber. Then SPIONs and EGFRmAb-SPIONs (12 mg iron/kg BW) were administered via intracarotid delivery for the contrast-enhanced MRI. The time course of nanoparticle distribution in the rat brain was monitored using a 3.0 T magnetic resonance scanner (Signa HDxt). The parameters of axial T2WI included TR = 3,000 ms, TE = 50 ms, FOV = 60 mm, excitations (NEX) = 6, base resolution = 192 × 160, slice thickness = 1.5 mm, interval thickness = 0.5 mm; the parameters of axial T2 mapping were listed as follows: TR = 1,500 ms; multiple TE = 10, 20, 30, 40, 50, 60, 70, and 80 ms; FOV = 60 mm; base resolution = 192 × 160; slice thickness = 1.5 mm. Relaxivity was measured within region of interest (ROI). All ROI with the same diameter were manually placed at the center of the tumor area on the same slice of T2 mapping images before or after administration of the contrast agent. Change in R2 was quantified by the following formula:
where R2 [pre] and R2 [post] were R2 values of the tumor acquired before and at 0.5, 2, 24, and 48 hours after administration of the contrast agent, respectively.
Histologic Analysis
In addition to the physical and neurologic evaluations, histologic analysis of tissues was performed in all the experimental rats. After 48 hours of nanoparticle injection and MRI examinations, the rats were deeply anesthetized and perfused with 0.9% sodium chloride (200 mL) and 4% paraformaldehyde (250 mL). Then brain tissues were removed and soaked in 4% paraformaldehyde for at least 24 hours; the paraformaldehyde-fixed sections were then embedded in paraffin and sectioned. Sections were subsequently stained with H&E and Prussian blue according to the standard clinical laboratory protocols. 26 Tissue sections were reviewed by a pathologist with expertise in veterinary pathology. EGFR immunohistochemistry was performed (day 13 after tumor implantation) after deparaffinization of tissue sections. Rabbit polyclonal to EGFR primary antibody and biotinylated goat antirabbit secondary antibody were processed for immunohistochemical visualization.
In Vivo Toxicity in Normal Rats
The normal rats were randomly divided into four groups (n = 6 for each group): a control group and three groups injected with saline, EGFRmAb-SPIONs, or SPIONs (12 mg Fe/kg BW) via intracarotid delivery. At the twenty-first day after injection, rats were euthanized. Four milliliters of blood was collected from the femoral artery and sent to the clinical laboratory for analysis of liver toxicity, nephrotoxicity, anemia, and bone marrow toxicity by biochemical markers, such as aspartate aminotransferase (AST), alanine aminotransaminase (ALT), total protein (TP), albumin (ALB), blood urea nitrogen (BUN), and creatinine (CREA), and for blood cell analysis, including white blood cells (WBCs), red blood cells (RBCs), platelets (PLTs), and hemoglobin (Hgb). Thereafter, rats were perfused with sodium chloride (250 mL) and then euthanized. Series of tissue specimens from the heart, liver, spleen, kidney, and brain of the rats were collected for histologic examination. All tissues were stained with H&E according to the standard clinical laboratory protocols and reviewed by a pathologist with expertise in veterinary pathology.
Statistical Analysis
All the measurements for statistical analysis were repeated three times. The results were expressed as mean ± standard deviation. Statistical analysis was performed with a commercial statistical software package (SPSS 21.0, Chicago, IL). Statistical significance between the mean values was calculated by using one-way analysis of variance and Student t-test. A difference of p < .05 was considered statistically significant.
Results
Characterizations of EGFRmAb-SPIONs
The FTIR spectra of SPIONs and EGFRmAb-SPIONs are shown in Figure 1. The results show that peaks at 640 and 590 cm−1 from SPIONs and EGFRmAb-SPIONs, respectively, were typical Fe-O absorption bands. After conjugation with EGFRmAb, the amide band peak around 1,655 cm−1 and the carbonyl band peak around 1,549 cm−1 were observed, suggesting that the conjugation was successful. The saturation magnetization of EGFRmAb-SPIONs and SPIONs was found to be 55 emu/g·Fe and 70 emu/g·Fe, respectively (Figure 2).

Characterizations of nanoparticles: Fourier transform infrared spectroscopy spectra of superparamagnetic iron oxide (Fe3O4) nanoparticles (SPIONs) and EGFRmAb-SPIONs.
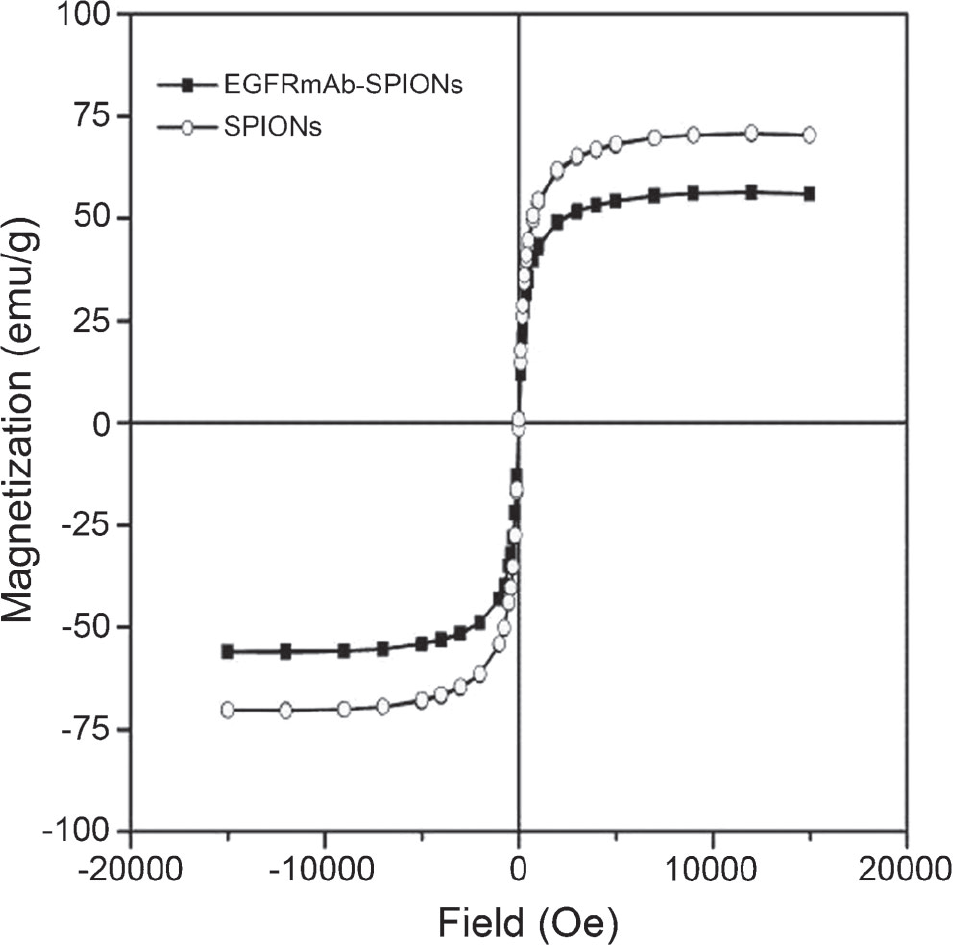
Magnetization curve of nanoparticles at room temperature.
EGFRmAb-SPIONs and SPIONs had similar morphologic characteristics (Figure 3). The average particle size was 6.95 ± 0.93 nm for SPIONs and 10.21 ± 1.27 nm for EGFRmAb-SPIONs. The hydrodynamic diameter was 71.45 ± 1.08 nm for SPIONs and 161.5 ± 2.12 nm for EGFRmAb-SPIONs (Figure 4). The Bradford method was adopted to determine the quantity of EGFRmAb in an equal amount of the conjugates. In this way, the amount of EGFRmAb was 0.14 mg per 1 mg SPIONs.

Transmission electron microscopy images of SPIONs and EGFRmAb-SPIONs. The original magnification is x100,000 without staining.

Hydrodynamic diameter of SPIONs and EGFRmAb-SPIONs.
Both SPIONs and EGFRmAb-SPIONs markedly reduced the T2 relaxation time. With the increasing EGFRmAb-SPION concentration, the signal intensity decreased. The T2 relaxation rate (R2 = 1/T2, s−1) of EGFRmAb-SPIONs was the linear fit with the iron concentration. The T2 relaxivity at 3 T was 165.72 mM−1s−1 for SPIONs and 92.73 mM−1s−1 for EGFRmAb-SPIONs (Figure 5).
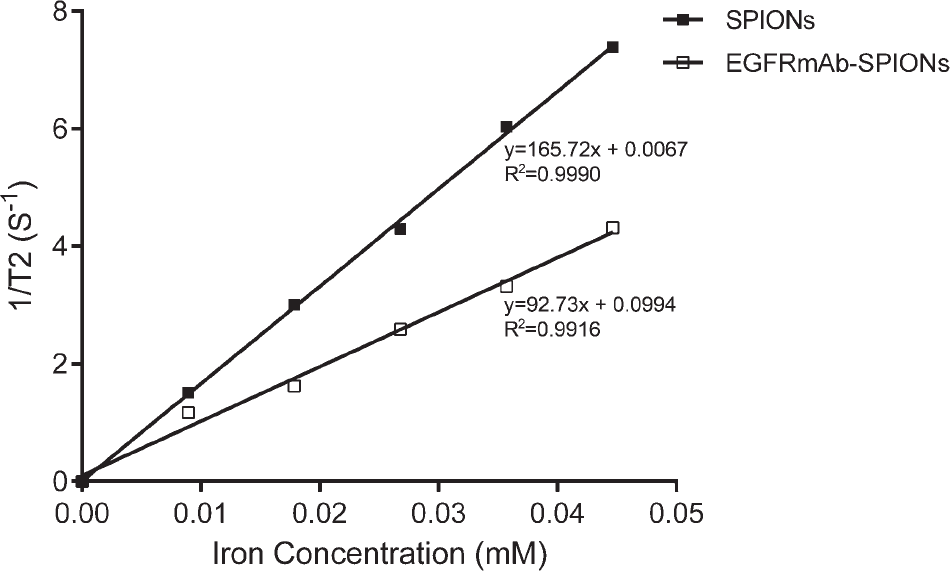
Magnetic properties of nanoparticles: T2 relaxivity of SPIONs and EGFRmAb-SPIONs. The T2 relaxation rate (1/T2, s−1) was the linear fit with the particle concentration. The T2 relaxivity was 165.72 mM−1s−1 for SPIONs and 92.73 mM−1s−1 for EGFRmAb-SPIONs.
In Vitro Cell Experiments
The MRI of the cells treated with EGFRmAb-SPIONs showed a significant negative contrast enhancement (significant decrease in signal intensity; p < .05) at each point of iron concentration compared to the tumor cells incubated with SPIONs (Figure 6). Cellular uptake of EGFRmAb-SPIONs showed a marked dose-dependent manner, whereas only negligible contrast was observed from the cells treated with SPIONs.

In vitro T2-weighted MRI of C6 glioma cells treated with both SPIONs (upper row) and EGFRmAb-SPIONs (lower row) at different iron concentrations.
The results showed that both EGFRmAb-SPIONs and SPIONs did not affect the viability of HL-7702 cells and ECV 304 cells in the range of 0 to 100 μg/mL iron concentration (Figure 7). It was found that all tested cells grew normally even when exposed to the high concentration, and no evident cytotoxic effects were observed.
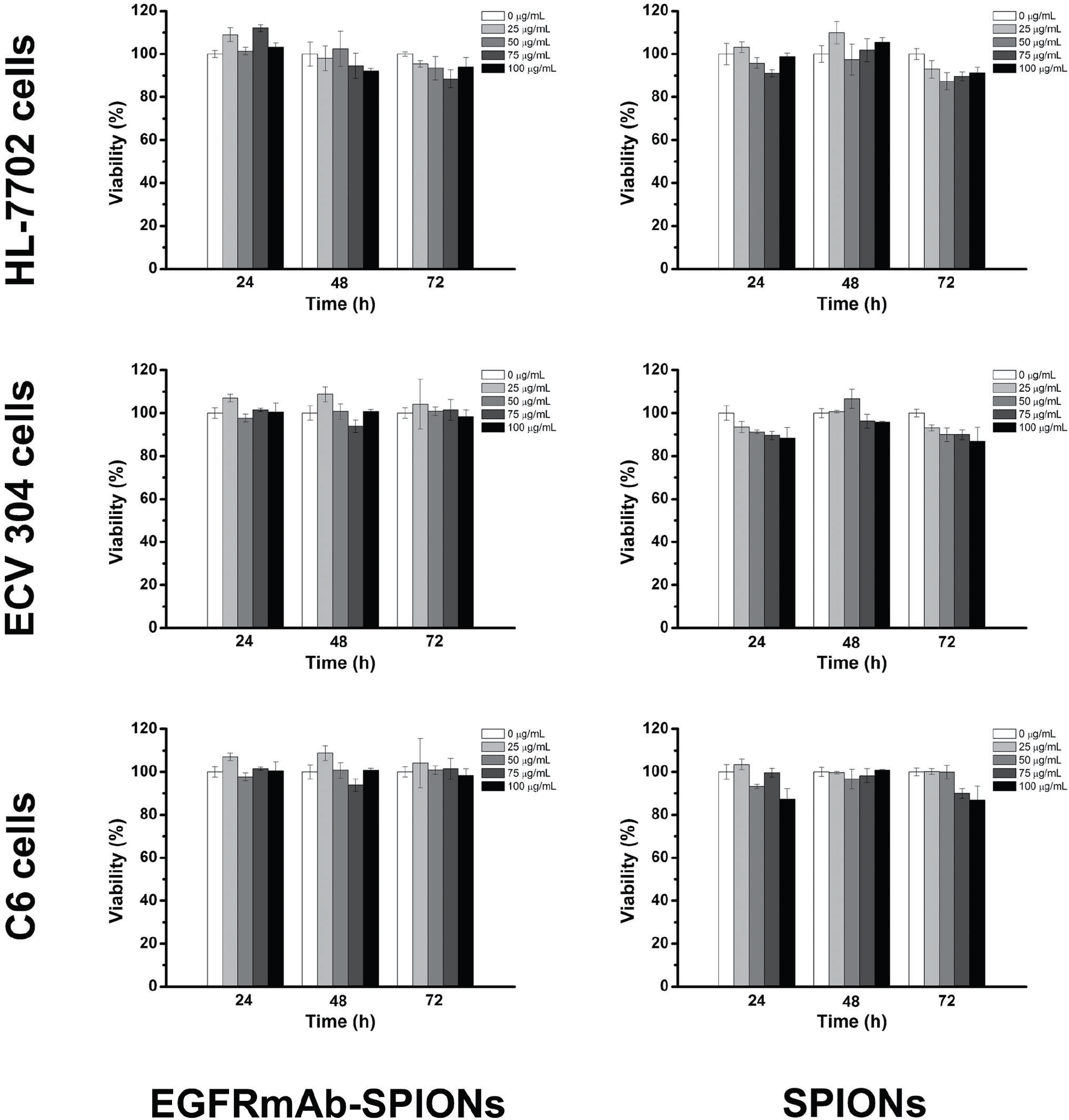
The cytotoxicities of EGFRmAb-SPIONs (left column) and SPIONs (right column) were assessed by MTT assay in vitro based on the viabilities of HL-7702 cells (top row), ECV 304 cells (middle row), and C6 cells (lower row) in the range of 0 to 100 μg/mL (iron concentration).
In Vivo Animal Experiments
Figure 8 shows a series of T2WI images of rat brain tumor after injection with EGFRmAb-SPIONs and SPIONs, respectively, and Figure 9 demonstrates R2 changes in tumor over time in two groups. T2WI MRIs of brain glioma administered with EGFRmAb-SPIONs exhibited a region of apparent hypointensity within glioma from 2 and 48 hours, with the maximal contrast at the time point of the 24 hours. The maximal R2 value was increased by 30% compared to baseline. Meanwhile, T2WI MRIs of brain glioma administered with SPIONs showed no visible signal changes within the tumor over the same period, and maximal R2 increased less than 10% at the 24-hour time point. The difference in R2 changes was statistically significant between two groups (p < .01).
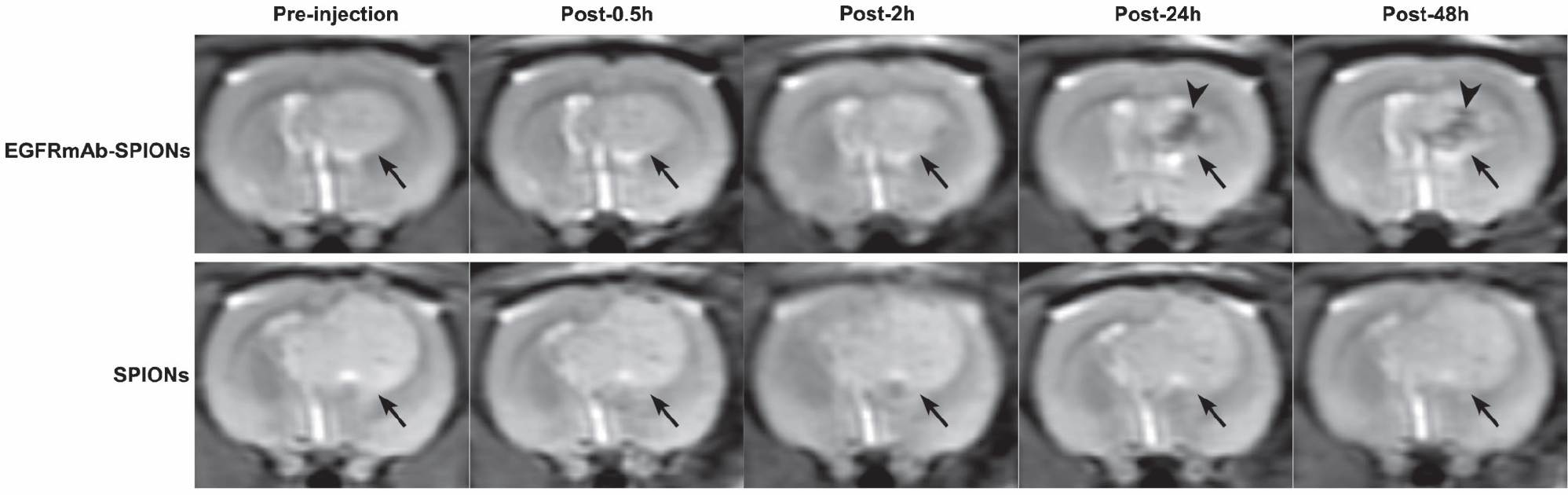
In vivo T2-weighted images of rat brain bearing C6 glioma (arrow) after administration of EGFRmAb-SPIONs (upper row) and SPIONs (lower row). After the injection of contrast agents, the negative enhancement within the tumor (arrowhead) was seen only on the images with EGFRmAb-SPIONs, with timing up to 48 hours compared to SPIONs.

R2 changes before and 0.5, 2, 24, and 48 hours after the injection of SPIONs and EGFRmAb-SPIONs. After injection of EGFRmAb-SPIONs, R2 significantly increased from 2 to 48 hours, with a maximal change of 30% at the 24-hour time point. Meanwhile, the maximal R2 change with SPIONs was less than 10% at 24 hours.
Prussian blue staining of the tumor tissue 48 hours after injection showed positive blue-staining iron particles only in the EGFRmAb-SPION group (Figure 10, A and B) but negative in the SPION group (Figure 10, C and D). EGFR overexpression was found to be positive and was demonstrated as yellow spots in the cytoplasm of cells in both the EGFRmAb-SPION (Figure 11A) and the SPION (Figure 11B) group. This finding indicated that the negative contrast enhancement of brain glioma on T2WI was due to the accumulation of EGFRmAb-SPION nanoparticles (see Figure 8).

Histochemical analysis of the tumor tissues: A and B, stained with Prussian blue in the EGFRmAb-SPION group (original magnification x100); C and D, barely stained with Prussian blue in the SPION groups (original magnification x400).

Immunohistochemical analysis of the tumor tissues and the corresponding EGFR expressed in the EGFRmAb-SPION (A) and the SPION (B) groups. The original magnification is x400.
In Vivo Biocompatibility Study
Compared to the control group, the levels of the biochemical markers in the three treated groups on the seventh day after injection showed no significant changes (all p > .05), and all values were within the normal ranges (Table 1). From the results of histologic analysis, no apparent in vivo toxicities were observed in the animals receiving the nanoparticles compared to the rats in the control group (Figure 12).

Hematoxylin-eosin–stained tissue sections from mice receiving no injection (control) and injected with saline, EGFRmAb-SPIONs, and SPIONs 7 days after the injection. Tissues were harvested from heart, liver, spleen, lung, kidney, and brain. The original magnification is x200.
Biochemical Analysis and Blood Analysis of Normal Rats on the Seventh Day after Treatment (n = 6)

ALB = albumin; ALT = alanine aminotransferase; AST = aspartate aminotransferase; BUN = blood urea nitrogen; CREA = creatinine; EGFRmAb-SPION = epidermal growth factor receptor monoclonal antibody–conjugated superparamagnetic iron oxide nanoparticle; Hgb = hemoglobin; PLT = platelet; RBC = white blood cell; SPION = superparamagnetic iron oxide nanoparticle; TP = total protein; WBC = white blood cell.
Discussion
The following main findings were obtained in this study: (1) EGFRmAb-SPIONs were successfully synthesized; (2) EGFRmAb-SPIONs could be uptaken specifically and efficiently by C6 glioma cells in vitro and targeted to a rat model bearing C6 glioma in vivo; thus, using EGFRmAb-SPIONs could produce a better tissue contrast in brain glioma and improve the detection of tumor cells on MRI; and (3) the EGFRmAb-SPIONs exhibited good biocompatibility and safety for potential clinical applications. In this context, EGFRmAb-SPIONs possessed the characteristics of long retention in glioma and higher R2 and could be efficacious as a targeting contrast agent for brain glioma MRI in vivo.
It is very important that an excellent MRI contrast agent have appropriate magnetic properties. In this study, SPIONs were composed of Fe3O4 nanoparticles, which provided five times magnetization in comparison with Fe2O3 nanoparticles. The saturation magnetization of EGFRmAb-SPIONs was found to be 55 emu/g·Fe. The R2 relaxivity value of EGFRmAb-SPIONs was 92.732 s−1mM−1, which was higher than that of a specific MRI contrast agent used in the detection of lymph node metastasis and resulted in a better contrast effect.27–29 In addition, magnetic resonance contrast enhancement of EGFRmAb-SPIONs was evaluated at 3.0 T by acquiring images of agarose samples with various iron concentrations, and a good linear correlation between R2 and iron concentration was established. When the T2 relaxation rate of the superparamagnetic nanoparticles increases, its ability to shorten the proton relaxation time becomes stronger; thus, the magnetic resonance contrast between tissues becomes more obvious. 30 Meanwhile, administration of EGFRmAb-SPIONs did not induce evident cytotoxic effects, indicating that it was well tolerated and safe at the doses evaluated in this study. These results indicate that EGFRmAb-SPIONs have a relatively small size, fine dispersion ability, and high R2; thus, they are suitable and safe for use as a negative MRI contrast agent.
From previous studies, EGFR was overexpressed in 40 to 50% of all glioblastoma,31,32 and EGFR overexpression has been shown to be correlated with glioma grade and a poor prognosis.31,33–36 Although iron oxide nanoparticles conjugated to a purified antibody specific to the EGFRvIII can be used for MRI contrast enhancement and therapeutic targeting of brain glioma,15,37 its clinical application is still limited, as mentioned above. In addition, both EGFRmAb-SPIONs and SPION-EGF have similar physiochemical properties and MRI characteristics, 16 and EGFRmAb is an appropriate target ligand of glioma. Based on these, it suggested that MRI used with EGFR-targeted contrast agent might be widely applied in monitoring and diagnosis of glioma in vivo.
Intravascular delivery of MRI contrast agents to brain tumor lesions is one of the most challenging goals of diagnostic MRI. Chertok and colleagues found that magnetically mediated retention of iron oxide nanoparticles in brain tumors after intravascular administration was feasible.38,39 High local concentration of MRI contrast agents is required to establish the diagnosis of brain glioma. MRI visually confirmed the accumulation of gum arabic–coated magnetic nanoparticles at the 9L glioma tumor site following intracarotid administration. 40 Previous studies also revealed that intracarotid administration in conjunction with magnetic targeting resulted in a 30-fold increase in tumor entrapment of polyethyleneimine-modified magnetic nanoparticles compared to that seen with intravenous administration, 39 suggesting that an advantage with the carotid arterial administration is due to higher exposure of the brain tumor vasculature to the MRI contrast agent on its first pass through circulation. The results explored in this study demonstrate that R2 changes at the tumor site increased by 30% in comparison with the precontrast images of the tumor, and the area of signal change in the tumor increased gradually until 48 hours after intracarotid injection of EGFRmAb-SPIONs, suggesting that EGFRmAb-SPIONs could be captured in brain glioma lesions following intracarotid administration. In addition, choosing appropriate pulse sequences, such as susceptibility weighted imaging, can improve the tumor delineation when performing molecular imaging.41,42 In this study, all MRI protocols were based on a standard spin echo sequence, mainly aiming at the consistently quantitative measurements of magnetic properties of the nanoparticles.
A major concern in the development of a new nanomaterial for biomedical application is the potential toxicity. In this study, the biocompatibilities of the EGFRmAb-SPIONs were verified both in vitro and in vivo using a cell-based assay and an animal model. It was found from the MTT assay that the tested cells (HL-7702 cells and ECV 304 cells) grew normally even when exposed to a high concentration of EGFRmAb-SPIONs, and no evident cytotoxic effects were observed. In addition, the toxicity was also performed in normal rats in vivo. There were no apparent toxicities observed in the tissue specimens from the animals receiving the EGFRmAb-SPIONs in comparison with the control group (see Figure 8). When compared to the control group, the levels of the biochemical markers in the three treated groups had no significant changes at 7 days after injection, and all the levels were within the normal range (see Table 1). Therefore, the biocompatibility of the EGFRmAb-SPIONs should be acceptable.
Conclusions
It has been demonstrated that EGFRmAb-SPIONs have superior biochemical properties, good specificity, and acceptable safety to target glioma both in vitro and in vivo and serve as an EGFR-targeted negative contrast enhancement agent in glioma MRI in vivo. Such glioma-targeted EGFRmAb-SPIONs can be used to identify the specific molecular subtypes of the brain glioma, which would provide new promising strategies for the target therapy and treatment monitoring of the glioma in the clinical setting.
Footnotes
Acknowledgments
We would like to thank the Analytical and Testing Center of Huazhong University of Science and Technology for the work-related analysis.
Financial disclosure of authors: This work was supported by grants from the National Natural Sciences Foundation of China (No. 30870702, 81171308), National Program of the Ministry of Science and Technology of China during the “12th Five-Year Plan” (ID: 2011BAI08B10), and National Basic Research Program of China (973 Program, 2012CB932500, 2015CB931800, and 2015CB931802).
Financial disclosure of reviewers: None reported.


