Abstract
Hydrazinonicotinamide–annexin A5 (HYNIC-Anx), a 99m technetium (99mTc)-labeled agent targeting phosphatidylserine, proved to be sensitive for the detection of apoptosis and thrombosis but is no longer available for clinical use. A mutant of human annexin designed for direct 99mTc labeling (referred to as Anx A5–128) showed improved binding affinity to phosphatidylserine and is expected to be used in humans. We compared both radiotracers with regard to pharmacokinetics and diagnostic ability in animal models. Biodistribution studies were performed in normal rats. Radiolabeled Anx A5–128 and HYNIC-Anx were compared in cardiovascular settings involving phosphatidylserine expression: experimental autoimmune myocarditis and infective endocarditis. Initial blood clearance was faster for Anx A5–128 than for HYNIC-Anx, and tissue biodistribution was similar overall for both tracers. The diagnostic sensitivity of Anx A5–128 was excellent and comparable to that of HYNIC-Anx. Anx A5–128 showed biodistribution and diagnostic ability similar to those of the HYNIC-Anx derivative, supporting its translation to clinical use.
ANNEXIN A5 is an endogenous protein (36 kDa) that specifically binds with nanomolar affinity to phosphatidylserine (PS) exposed by apoptotic cells and activated platelets.1,2 Radiolabeled annexin A5 (Anx) has been successfully evaluated as a molecular imaging agent of apoptotic cells and thrombus, with many potential clinical applications in oncology, cardiovascular diseases, and rheumatology. 1 The most widely used compound designed to 99m technetium (99mTc) labeling has been achieved by conjugation of the hydrazinonicotinamide (HYNIC) chelating group with recombinant human Anx A5 (rh-Anx) 1 in preclinical studies (for a review, see Boersma and colleagues 1 and Rouzet and colleagues 2 ), healthy volunteers, 3 and various oncologic settings in humans.4–8 However, Tait and colleagues showed that random amine-directed modification of Anx, such as HYNIC derivatization, was associated with multiple molecular species and finally reduced its overall membrane-binding affinity. 9 Additionally, preformulated radiolabeling kits (formerly supplied by Theseus Imaging Corp., Worcester, MA) are no longer available for clinical use. 1
Mutants of rh-Anx were developed for direct site-specific 99mTc labeling by the addition of a unique 99mTc-chelating oligopeptide sequence at their N-terminus. 10 The N-terminal region of Anx is located on the concave face of the molecule, opposite the binding site, 11 resulting in unaltered membrane-binding affinity compared to wild-type rh-Anx. 9 One of those derivatives, referred to as Anx A5–128, showed increased in vivo binding on apoptotic cells compared to HYNIC-Anx. 9 Moreover, 99mTc labeling of this new Anx derivative is quite straightforward, with good radiochemical purity, and Good Manufacturing Practices (GMP) grade Anx A5–128 kits designed for 99mTc labeling are undergoing the agreement process for human use in the United States and Europe (Atreus Pharmaceuticals, Ottawa, ON). Additionally, several clinical trials designed to assess Anx A5–128 in various settings are about to begin, and precise preclinical validation is mandatory.
Since preclinical validation has been obtained with HYNIC-Anx, but clinical studies will be carried out with the new Anx A5–128 variant, it is necessary to ascertain whether both forms of radiolabeled Anx are comparable with regard to biokinetics and diagnostic ability in animal models. Diagnostic value has been evaluated in cardiovascular settings in which radiolabeled Anx is likely to meet clinical demand, representative of the two biological pathways involving PS exposure: (1) experimental autoimmune myocarditis because it is associated with cardiomyocyte and lymphocyte apoptosis,12–15 which is likely to be associated with therapy efficacy, 16 and (2) infective endocarditis because the relationship between focal Anx uptake and platelet activation in vegetations has been previously evidenced.17,18
Methods
Experimental Models
The procedures and animal care complied with the principles of animal care formulated by the National Society for Medical Research. This study was conducted under the authorization of the French Directorate of Veterinary Services (No. 75-214) and the approval of the animal ethics committee of our institution.
Experimental Autoimmune Myocarditis
Experimental autoimmune myocarditis was induced in 12 male Lewis rats by immunization with purified rat cardiomyosin. 19 Animals were housed and treated in accordance with institutional guidelines for animals.
Myosin purification
Cardiac rat myosin was purified according to the Shiverick technique. 20 Cardiac myosin purity was checked by polyacrylamide gel electrophoresis. The protein concentration was measured using the Bradford technique, and myosin was then emulsified with phosphate-buffered saline (PBS) to obtain a final solution of 4 mg/mL and was stored in aliquots at −20°C.
Rat immunization
To induce autoimmune myocarditis, purified myosin was first emulsified with an equal volume of complete Freund adjuvant supplemented with Mycobacterium tuberculosis H37Ra (Difco) to a final concentration of 2 mg/mL (solution A). Myosin was replaced by an equivalent volume of PBS added to the complete Freund adjuvant/M. tuberculosis solution for control rats (solution B). Six hundred micrograms of rat cardiomyosin in solution A was injected subcutaneously in Lewis rats at day 0 and day 7 (experimental autoimmune myocarditis group), whereas an equivalent volume of solution B was used for the control group (n = 16).
Infective Endocarditis in Rats
Infective endocarditis was induced in 12 male Wistar rats (Janvier, Le Genest-Saint-Isle, France) as described previously. 17 In brief, a polyethylene catheter was inserted into the left ventricle through the right carotid artery in anesthetized rats (ketamine/xylazine). The catheter remained indwelling throughout the experiment to induce an aseptic thrombotic vegetation formation on the aortic valves. Twenty-four hours after catheterization, rats underwent bacterial inoculation (108 colony-forming units of Enterococcus faecalis JH2–2) by the left jugular vein under halothane anesthesia.
Annexin Labeling Procedure
HYNIC-Anx
Freshly eluted sodium pertechnetate (1,110–1,850 MBq) and 50 μg stannous tricine buffer (pH 6) were added to a vial containing 275 μg recombinant human HYNIC-Anx A5 (National Cancer Institute BRB Preclinical Repository, Frederick, MD) and left to incubate for 15 minutes at room temperature. The quality control was performed with instant thin-layer chromatography developed in acid citrate dextrose buffer. The radiochemical purity was 89.5 ± 1.3% (n = 20).
Anx A5–128
Anx A5–128 was provided (Atreus Pharmaceuticals and Advanced Accelerator Applications, Saint-Genis Pouilly, France) as lyophilized radiolabeling kits manufactured according to GMP quality requirements, taken from a batch intended for first-in-human administration. The material was shipped within 6 months after manufacturing. A single-dose vial contains 400 μg of Anx A5–128, 10 μg of stannous chloride as a reducing agent, and other excipients. For radiolabeling, freshly eluted sodium pertechnetate (1,110–1,850 MBq in 0.6–1 mL) was added to the vial and left to incubate for 1.5 hours at room temperature according to the manufacturer's instructions. The quality control was performed with instant thin-layer chromatography developed in acid citrate dextrose buffer. The radiochemical purity was 96.9 ± 0.9% (n = 20). To check for potential high-molecular-weight impurities, the solution was filtered through a 0.22 μm filter (Millex-GV filter, 0.22 μm, 4 mm) in a series of eight radiolabeling procedures of Anx A5–128. The radioactivity loss was 3.7 ± 1.6%, corresponding to the dead volume of the filter (< 10 μL according to the manufacturer's specifications).
Both precursors were stored at −40°C and used within 6 months of receipt.
Biodistribution Studies
Blood Clearance Assessment
Twelve male Wistar rats were anesthetized with urethane (1.22 g/kg). A catheter was inserted into the left carotid for blood sampling. Each animal received a single intravenous injection (penis vein) of 43.6 ± 9.2 MBq (corresponding to 15.7 ± 3.3 μg of protein) 99mTc-Anx-128 (n = 6) or 47.3 ± 4.2 MBq (corresponding to 11.7 ± 1.0 μg of protein) 99mTc-HYNIC-Anx (n = 6) in a 250 μL volume. Blood samples (250 μL) were taken 5, 15, 30, 45, 60, 90, 120, 180, and 240 minutes postinjection. For each sample, one aliquot of 100 μL was taken for well counting (Cobra II, Packard, Meriden, CT).
Tissue Biodistribution of 99mTc–Anx-128 and 99mTc–HYNIC-Anx
Two groups of male Wistar rats (weight 320–335 g) were injected with an activity of 49.9 ± 8.5 MBq (corresponding to 18.0 ± 3.1 μg of protein) 99mTc–Anx-128 (n = 6) or 54.2 ± 6.6 MBq (corresponding to 13.4 ± 1.6 μg of protein) 99mTc–HYNIC-Anx (n = 6). Animals were euthanized with pentobarbital overdose 60 minutes after intravenous injection of tracer, and relevant organs and tissues were harvested, washed, and weighed. Each tissue and the injected solution were aliquoted for well counting. After correction for residual activity in the injection site and radioactive decay, the results were expressed as the percentage of administered dose per gram of organ (%ID/g) and by the percentage of administered dose per organ (%ID/organ). Blood, muscles, bone, and skin (and hairs) were assumed to represent, respectively, 6%, 40%, 10%, and 18% of the body weight. 17
Imaging Procedures
Myocarditis Model
Imaging was performed 3 weeks after immunization. The natural history of acute autoimmune myocarditis over weeks allows considering the absence of significant disease progression in a time span of a few days. Consequently, each myocarditis rat has been injected (60 MBq) and imaged sequentially with both tracers (99mTc–HYNIC-Anx first in six animals) 2 days apart, assuming that myocardial injury would have remained stable during this period. Additionally, a group of controls was injected with either 99mTc–HYNIC-Anx or 99mTc–Anx A5–128 (n = 8 for each tracer).
Endocarditis Model
Imaging was performed 4 days after bacterial inoculation. Due to the rapid progression of the disease, sequential imaging of the same animal was not feasible. Therefore, endocarditis models were injected (60 MBq) with either one of the tracers (n = 6 for each tracer).
Acquisition and Reconstruction Parameters
Helical single-photon emission computed tomography/computed tomography (SPECT/CT) was performed under intraperitoneal pentobarbital (4 mg/100 g body weight) anesthesia with a four-headed multiplexing multipinhole camera (NanoSPECT/CT plus, Bioscan Inc., Washington, DC). Each head was equipped with a tungsten collimator (rat whole body – high resolution). Flat-panel detector CT was performed first (tube voltage 55 kV, tube current 145 mAs), and then the helical SPECT acquisition was performed with the following parameters: helical scan with 28 projections/rotation plus circular scan at the beginning and at the end of the scan range, matrix size 256 × 256, zoom 1.14 (pixel size 1 mm2). SPECT data were reconstructed using HiSPECT (Bioscan Inc.) iterative reconstruction software on a dedicated workstation and visualized using InVivoScope software with coregistration of SPECT and CT images.
Data Analysis
Myocarditis model
Scintigrams were assessed visually to determine the presence of a focal or diffuse tracer uptake in the myocardium. For in vivo quantification, activity (kBq per voxel) was measured in regions of interest drawn over focal uptake when present or encompassing the left ventricular myocardium (midportion) in the absence of focal uptake and the lung to serve as background activity.
Endocarditis model
Scintigrams were assessed visually to determine the presence of a focal tracer uptake in the heart and measured as previously indicated. The ratio between myocardium to lung activity (uptake ratio) was calculated.
Autoradiography
After completion of SPECT/CT, animals were euthanized with pentobarbital overdose. Tissue samples were frozen and cut into transverse sections of 20 μm thickness, which were exposed in a digital beta imager (BetaIMAGER, Biospace Lab, Paris, France) for 8 hours.
Myocarditis Model
Quantification was performed by calculating the ratio between the activity (mean counts/mm2) of the left ventricular section in the myocarditis models and in controls.
Endocarditis Model
Quantification was performed by calculating the ratio between the activity (mean counts/mm2) of the vegetation and the activity of a region of interest drawn on normal myocardium remote from the vegetation.
Histology and Immunohistochemistry
At sacrifice, transverse sections were frozen for one part (autoradiography) and fixed in buffered paraformaldehyde (4% v/v, 48 hours) for the other to obtain better morphologic definition. Fixed tissues were embedded in paraffin and sectioned at 5 μm thickness. Sections were stained with hematoxylin-eosin for morphologic analysis of cells and nuclei. In myocarditis, sections from both immunized and control rats were used for apoptotic assessment using the Apostain assay (Alexis Corp., Lausen, Switzerland).
Statistical Analysis
Continuous data were expressed as mean and standard deviation and compared by use of unpaired Student t-test in myocarditis (12 myocarditis and 8 controls) and as median and range and compared by use of the nonparametric Mann-Whitney U test in endocarditis (n = 6) (MedCalc version 12.5, MedCalc Software, Ostend, Belgium). The level of significance was set at p < .05.
Results
Blood Clearance and Tissue Biodistribution
After intravenous injection, blood clearance of both tracers followed a two-compartment model (Figure 1). Initial blood clearance (α component) was more rapid for 99mTc–Anx A5–128 with an effective half-life of 10.5 minutes (accounting for 87% of the injected activity) versus 18 minutes (accounting for 90% of the injected activity) for 99mTc–HYNIC-Anx. The slow component was 217 minutes (accounting for 13% of the injected activity) for 99mTc–Anx-128 and 153 minutes (accounting for 10% of the injected activity) for 99mTc–HYNIC-Anx. The blood activity of 99mTc–Anx-128 was consistently lower than that of 99mTc–HYNIC-Anx at each time point (p < .05) but remained below 5% of the injected dose from 30 minutes after injection for both tracers.
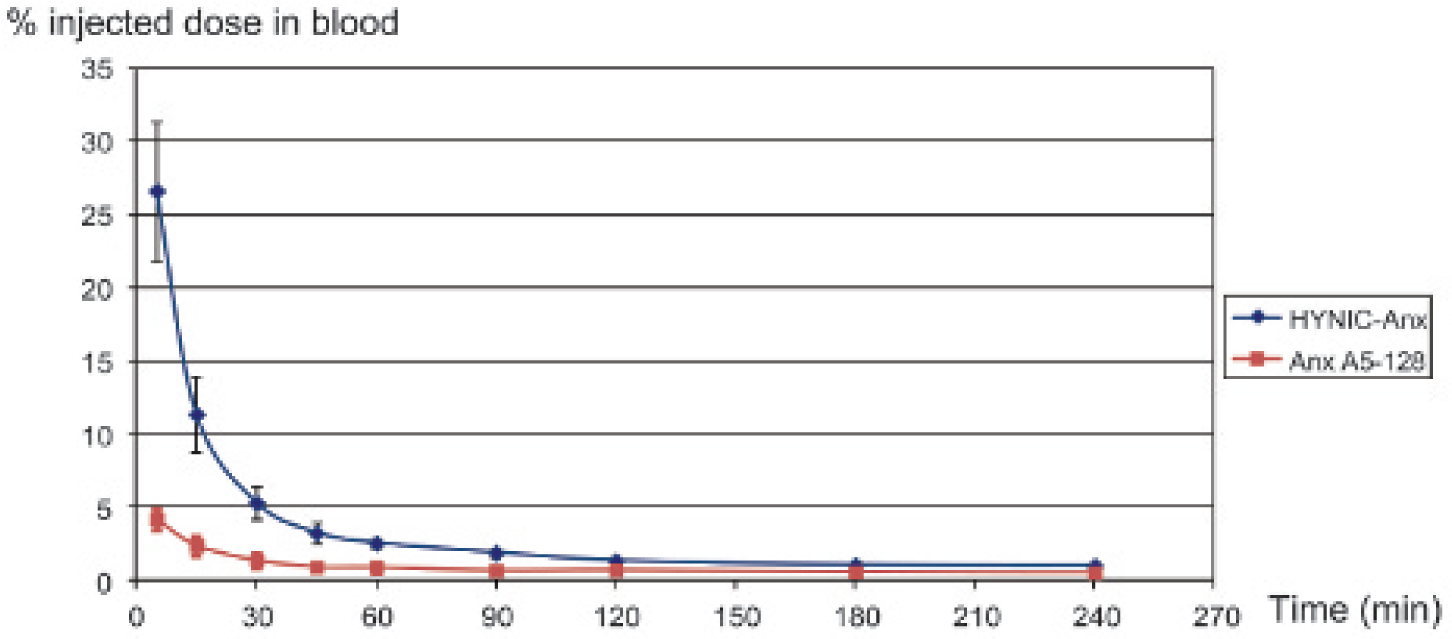
Blood clearance of 99mTc-Anx A5–128 and 99mTc–HYNIC-Anx (n = 6 in each group). After correction for residual activity in the injection site and decay, the results are expressed as a percentage of the administered dose remaining in the blood pool according to time, assuming that blood represents 6% of body weight. Initial blood clearance was faster with 99mTc–Anx A5–128 than with 99mTc–HYNIC-Anx (half-life of rapid component: 10.5 minutes vs 18 minutes, respectively). Blood activity of 99mTc–Anx A5–128 was consistently lower than that of 99mTc–HYNIC-Anx at each time point (p < .05).
For both tracers, the highest concentrations were found in kidneys (and urine) and then liver, spleen, and muscles (Table 1). Bone marrow presented a mild tracer uptake, which was also detectable on SPECT. It should be noted that the physiologic uptake was very low on myocardium and brain. Tissue biodistribution assessed 60 minutes after injection showed a lower uptake of 99mTc–Anx A5–128 compared to HYNIC-Anx in liver and thyroid, whereas the uptake was greater with 99mTc–Anx A5–128 in heart and lungs.
Tissue Biodistribution of 99mTc-Anx A5–128 and 99mTc-HYNIC-Anx, 1 Hour after Intravenous Injection

ND = not determined.
p < .02
p = .04
p = .03.
Myocarditis
Visual analysis of SPECT/CT was positive with both tracers in all animals with myocarditis and negative in controls (Figure 2 and Figure 3). The general pattern was a moderate and heterogeneous myocardial tracer uptake in myocarditis rats, with additional intense focal uptake in 8 of 12 rats. In case of additional focal uptake, their location and intensity were similar with both tracers (see Figure 3, A and B). Quantitative analysis of myocardial focal uptake/lung ratio in myocarditis and control groups was 1.7 ± 0.6 versus 1.0 ± 0.3, respectively (p = .006), with 99mTc–HYNIC-Anx and 1.7 ± 0.5 versus 1.2 ± 0.2, respectively (p = .003), with 99mTc–Anx A5–128. Uptake ratios were not significantly different between tracers in the myocarditis and control groups (Figure 4).
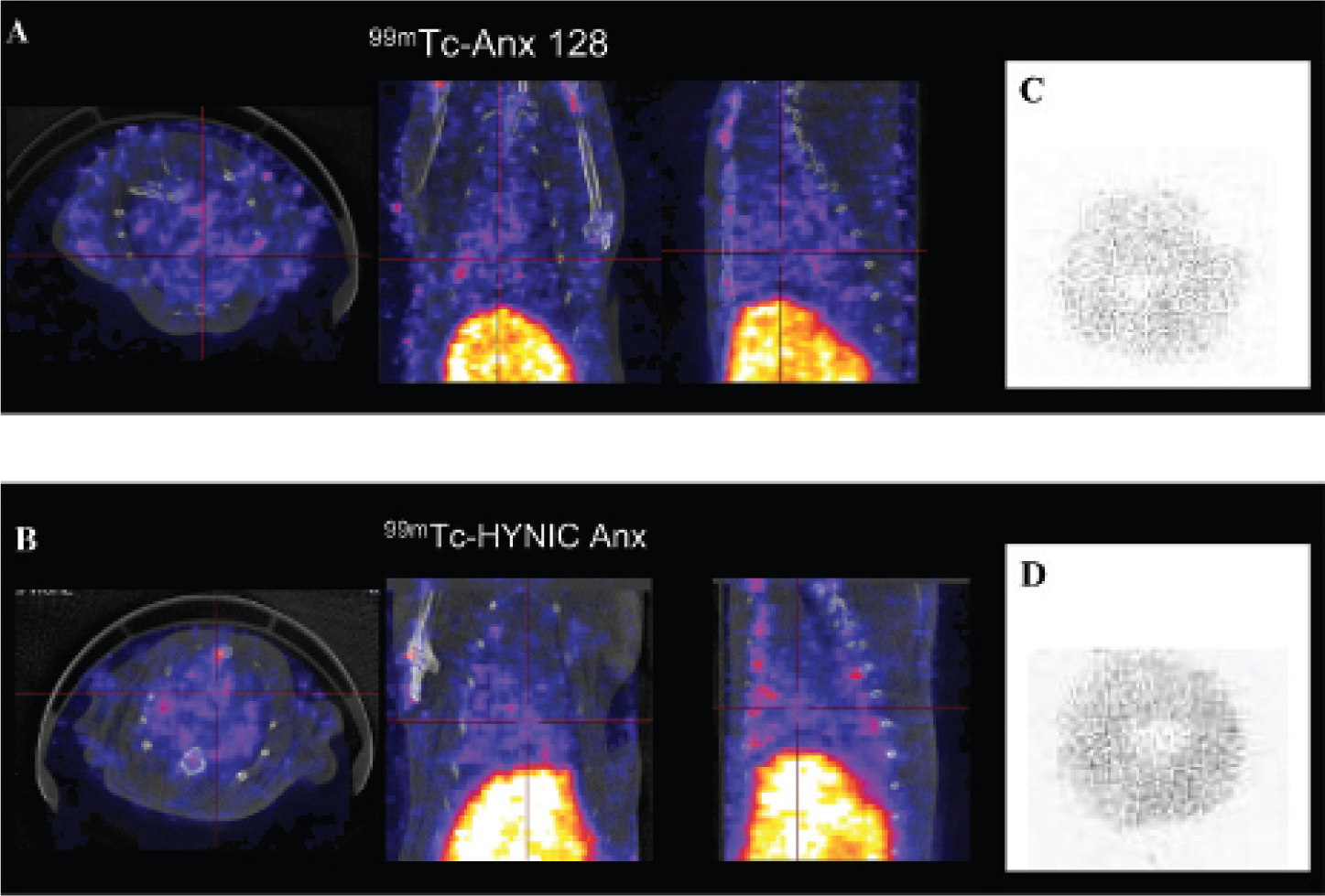
Representative images in control rats. No abnormal uptake was detectable on SPECT/CT in the heart area either with 99mTc–Anx A5–128 (A) or with 99mTc–HYNIC-Anx (B). On autoradiography of a myocardial slice, the tracer uptake was faint and homogeneous with both tracers (C, D).

Experimental autoimmune myocarditis in rat (SPECT/CT) 3 weeks after immunization. Images are normalized for maximal activity (kBq)/pixel because the greatest value is outside the cardiac area. Acquisitions were performed 1 hour after intravenous injection of 99mTc–Anx A5–128 (A) or 99mTc–HYNIC-Anx (B) 2 days apart in the same animal (the natural history of acute autoimmune myocarditis over weeks allows considering the absence of significant disease progression in a time span of a few days). Comparable focal uptake is detectable on the anterior wall with both tracers (arrow). Comparative analysis of autoradiography (C) and histology (D, hematoxylin-eosin stain, ×10 original magnification; E, inlet ×40 original magnification) on the same slice showing colocalization of annexin uptake (99mTc–HYNIC-Anx) and areas of myocyte damage, edema, and cell infiltration. The background of Apostain staining was low in the control heart (negative controls, F; ×40 original magnification), whereas it was intense in tissue areas characterized by myocyte disappearance and immune cell infiltrates (G; ×40 original magnification). Both injured cardiomyocytes and immune cell infiltrates were positive for Apostain.

Comparison of uptake ratios in rat models of experimental autoimmune myocarditis (n = 12) injected sequentially with 99mTc–HYNIC-Anx and 99mTc–Anx A5–128 (2 days apart, 99mTc–HYNIC-Anx first in six animals) and in controls (n = 8 for each tracer). Data are presented as mean and 95% confidence interval.
Autoradiography showed diffuse Anx myocardial uptake with more intense and focal hot spots in the myocardium of rats with myocarditis, whereas the uptake was faint and uniform in controls. Histologic analysis showed injured myocytes that disappeared and mononuclear cell infiltrates diffusely distributed throughout the myocardium, with clusters frequently located in the subepicardium. Comparative analysis of autoradiography and histology performed on the same slices showed colocalization of Anx uptake (whatever the form) and myocardial damage (see Figure 3, C and D), characterized by myocyte disappearance and immune cell infiltrates (see Figure 3, E–G). Both injured cardiomyocytes and immune cell infiltrates were positive for Apostain.
Infective Endocarditis
Macroscopic examination at sacrifice showed that left-sided vegetations were localized mainly on the aortic valves but could also be observed on the aortic wall along the catheter path and in the left ventricle (apex), as previously described. 17 Visual analysis of SPECT/CT was positive with both tracers in all endocarditis rats. This focal uptake was intense in the aortic valve area and could be associated with foci localized in the ascending aorta or left ventricular apex (Figure 5, A and B). Quantitative analysis of vegetation uptake on SPECT was (median and range) 2.5 (1.4–9.6) with 99mTc–HYNIC-Anx versus 4.7 (2.1–8.7) with 99mTc–Anx A5–128 (p = .3) (Figure 6A). Vegetation to remote myocardium ratio on autoradiography was (median and range) 8.1 (2.8–31) with 99mTc–HYNIC-Anx and 11 (2.2–24) with 99mTc–Anx A5–128 (p = .4) (Figure 6B).

Infective endocarditis in rat (SPECT/CT). Both acquisitions were performed 1 hour after intravenous injection of 99mTc–Anx A5–128 (A) or 99mTc–HYNIC-Anx (B), in different animals. A focal uptake is detectable in the aortic valve area with both tracers (arrow). Comparative analysis of autoradiography (C) and histology (D, hematoxylin-eosin stain, ×10 original magnification; E, Alcian blue stain, ×40 original magnification) on adjacent slices showing localization of annexin uptake (99mTc–Anx A5–128) matching with vegetations (arrow) on the aortic valve and aorta root. LV = left ventricular.

Comparison of uptake ratios between 99mTc–Anx A5–128 and 99mTc–HYNIC-Anx in infective endocarditis models (n = 6 in each group). The results are presented as box plots for SPECT (A) and autoradiography (B) and show no significant difference between tracers.
Comparative analysis of autoradiography and histologic slices demonstrated localization of Anx uptake (whatever the tracer) at the peripheral layer of vegetations, corresponding to the site of vegetation growth through platelet recruitment from the blood pool (Figure 5, C–E), without significant positive cell for apoptosis, as previously described.17,18
Discussion
This study shows that Anx A5–128 is comparable to HYNIC-Anx as a molecular imaging agent of exposed PS with regard to biodistribution and diagnostic ability in rat models of cardiovascular diseases. Blood clearance of Anx A5–128 is more rapid than that of HYNIC-Anx, but without a significant impact on contrast of images. The diagnostic sensitivity was excellent for both tracers, with an abnormal uptake detectable in all animals with autoimmune myocarditis or infective endocarditis. The first model is characterized by an acute autoimmune injury of the myocytes containing myosin associated with immune cell infiltrate within the myocardium (see Watanabe and colleagues 21 for a review). In our experiments, both injured myocytes and immune cells could be positive for Apostain, providing evidence that both cell types could expose PS on their membrane and bind Anx, due to antibody-mediated myocyte injury leading to myocyte death for one part and immune cell maturation involving both immune cell proliferation and selection through apoptosis of nonrelevant cells for another part. In humans, evidence of cardiomyocytes' membrane leakage in acute myocarditis was obtained in vivo by use of indium 111–labeled antimyosin antibodies. 22 In such patients, antimyosin antibody uptake pattern was either diffuse or focal and not systematized, comparable to that obtained with Anx in the present study.
The model of infective endocarditis is characterized by the development of active vegetations, involving platelet aggregates, which highly expose PS as a link between platelet activation and fibrin formation,17,18 generating intense uptake of Anx A5 in the absence of significant apoptosis. 2
Blood Clearance
The most striking difference between Anx A5–128 and HYNIC-Anx was blood clearance, mainly the rapid component during the first hour after injection. Less than 2% of the injected activity of Anx A5–128 remained in the blood pool 30 minutes after intravenous injection, which confers an advantage for the tracer in early imaging, particularly in cardiac and vascular applications. In our previous work aiming to assess the diagnostic ability of HYNIC-Anx in endocarditis, we found that the contrast was optimal between 1 and 2 hours after injection. 17 In previous studies evaluating Anx A5–128 uptake in animal models of apoptosis, data acquisition (in vitro and in vivo) was also performed 60 minutes after injection.9,23 However, this may not apply to tissues such as tumors, where penetration of imaging agents is necessary.
Extensive biodistribution of HYNIC-Anx in rats has been reported by Ohtsuki and colleagues, with a blood clearance pattern similar to ours. 24 Conversely, we found some differences in tissue biodistribution such as kidney and spleen uptake, whereas liver, lung, and blood-pool activity were comparable. To date, there are no published data of tissue biodistribution of Anx A5–128 in rats, but we found a quite similar uptake to that reported by Tait and colleagues in mice for kidney and spleen. 23 However, they reported eightfold greater renal uptake for HYNIC-Anx compared to Anx A5–128. We did not find such a difference, and in our study, renal uptake was comparable for both forms, in the range of values reported by Ohtsuki and colleagues, 24 thus suggesting a species-related difference. The slightly greater thyroid uptake with 99mTc–HYNIC-Anx compared to 99mTc–Anx A5–128 is likely to be related to lower radiochemical purity.
Diagnostic Ability
The site-specific labeling of the new variant Anx A5–128 at the N-terminus of the protein, distant from the binding site, was associated with an increase in PS binding affinity and greater uptake in vivo on the apoptotic cells of mice liver after treatment with cyclophosphamide, compared to HYNIC derivatization. 9 The faster blood clearance of Anx A5–128 (see Tait and colleagues 23 and the present study) is likely to result in the lowest background activity and potentially better image contrast compared to HYNIC-Anx. The present study shows uptake ratios greater with Anx A5–128 than with HYNIC-Anx consistently in apoptosis and thrombus, but without a significant difference. Although larger samples may have allowed us to reach significance, the difference remains small between both tracers, for three main reasons: (1) even if the binding affinity of HYNIC-Anx is slightly lower than that of Anx A5–128, it is still in the nanomolar range, allowing stable binding of the imaging agent to the target; (2) the remaining activity of HYNIC-Anx in the blood pool 1 hour after injection is only around 3% of the injected activity, with a very limited impact on image contrast; and (3) on normal myocardium, nonspecific binding measured as tissue activity in control animals was slightly greater with Anx A5–128 compared to HYNIC-Anx. Taken together, these results show that the new Anx variant provides images of quality at least equivalent to HYNIC form, with excellent diagnostic value.
Clinical Applications
Our group 14 and others 12 reported on the increased HYNIC-Anx uptake in acute and subacute animal models of myocarditis and on the correlation of HYNIC-Anx cardiac uptake and number of apoptotic cells in this setting. There is a clinical demand for better diagnosis and characterization of inflammatory cardiac diseases that is not met by current diagnostic tools. 25 Cardiac magnetic resonance does not allow for precise identification of immune injury or early therapy monitoring, 26 and fluorodeoxyglucose positron emission tomography (FDG–PET) is hampered by physiologic cardiac uptake of the tracer. As well, although diagnosis of endocarditis relies on clinical data, biology, and echocardiography, recent studies using whole body scanning with FDG-PET showed that half of septic embolisms were not detected by morphologic imaging alone.27,28 Even if FDG-PET is now readily available in routine practice, its diagnostic value is likely to be hampered by the physiologic uptake of the tracer by heart and brain. In this regard, by targeting specifically PS exposed on the membrane surface, radiolabeled Anx offers the advantage of being devoid of significant cardiac and cerebral uptake, thus allowing us to perform a comprehensive evaluation of the disease in a single scan. Clinical translation of Anx imaging is therefore strongly desirable, and the present study supports its assessment in diseases such as myocarditis or endocarditis. However, these settings are relatively rare, and further validation in conditions affecting larger populations is required. Hence, a large body of preclinical data supports the potential clinical usefulness of radiolabeled Anx to target apoptosis in monitoring the response to therapy in cancer 1 and rheumatoid and other autoimmune diseases.29,30 In the setting of atherothrombosis, Anx uptake may be regarded as a marker of plaque vulnerability by targeting both apoptotic cells (such as macrophages and smooth muscle cells) and activated platelets as key components of intraluminal thrombus. 31
Conclusions
The present study showed that Anx A5–128 was comparable to HYNIC-Anx with regard to biodistribution and diagnostic ability in a model of apoptosis (myocarditis) and in a model of platelet activation (endocarditis) in rats. Blood clearance of Anx A5–128 is more rapid than that of HYNIC-Anx, but without a significant impact on contrast of images. This study suggests that the new variant Anx A5–128 may represent an attractive alternative to HYNIC-Anx and supports the clinical translation of radiolabeled Anx A5–128 imaging.
Footnotes
Acknowledgments
We would like to thank Atreus Pharmaceuticals and Advanced Accelerator Applications for supplying the Anx A5–128 labeling kits and the National Cancer Institute BRB Preclinical Repository for supplying the HYNIC-Anx labeling kits.
Financial disclosure of authors: Donato Barbato is an employee of Advanced Accelerator Applications.
Financial disclosure of reviewers: None reported.
