Abstract
Stem cell therapy has been studied intensively as a promising therapeutic strategy toward a cure for diabetes. To study the effect of mesenchymal stem cell (MSC) transplantation for pancreatic regeneration, we monitored the localization and distribution of transplanted MSCs by bioluminescence imaging in a mouse model. Bone marrow MSCs were isolated and transfected with a highly sensitive firefly luciferase reporter gene. To assess the efficiency of MSC transplantation, a partially pancreatectomized (PPx) mouse model was used. Transplanted MSCs were monitored by confocal microscopy and in vivo bioluminescence imaging. Daily blood glucose levels and glucose tolerance were measured. Insulin-secreting beta cells were immunostained, and insulin levels were measured via enzyme-linked immunosorbent assay. Bioluminescence signals were clearly detected from the transplanted MSCs in the pancreatic region regardless of injection route. However, locally injected MSCs exhibited more rapid proliferation than ductally injected MSCs. PPx mice harboring transplanted MSCs gradually recovered from impaired glucose tolerance. Although insulin secretion was not observed in MSCs, transplanted MSCs facilitate the injured pancreas to recover its function. In vivo optical imaging of transplanted MSCs using a highly sensitive luciferase reporter enables the assessment of MSC transplantation efficiency in a PPx mouse model.
DIABETES MELLITUS (DM) is a consequence of inadequate insulin supply or insulin resistance and the inability to respond to high blood glucose levels. It is difficult for DM patients to maintain optimal glucose homeostasis during prolonged periods, despite meeting daily insulin requirements by insulin therapy. Thus, DM patients become susceptible to debilitating complications, such as hypoglycemia, retinopathy, and cardiovascular diseases.1,2 Pancreatic islet transplantation using insulin-producing beta cells that had been differentiated from embryonic stem cells and adult pancreatic stem cells has emerged as a new and promising therapeutic strategy for diabetes.3,4 Recently, a mouse pancreatic stem cell line without genetic manipulation was established to analyze the molecular mechanisms that regulate pancreatic stem/progenitor cell differentiation. 5 However, limited knowledge regarding the in vitro differentiation mechanism has hindered attempts to enhance the quality and quantity of these beta cells. Therefore, it is preferable that the beta cells are replaced in the pancreas autogenously and spontaneously rather than by external islet transplantation.
Mesenchymal stem cell (MSCs), derived from bone marrow, 6 adipose tissue, 7 and most connective tissues, 8 have been recognized as a promising source for cell therapy. Regardless of their tissue source, MSCs have the capacity to differentiate into various cell types, including osteoblasts,9,10 adipocytes,7,11 and neural cells.12,13 Moreover, MSCs support graft protection, tissue revascularization, and immune acceptance. 14 MSCs have been detected in human pancreatic endocrine and exocrine tissue cultures, have resided in the pancreas, 15 and have been derived from chronic diabetes patients expressing C-peptide and insulin. 16 These cells have generated a great deal of interest because of their potential uses in regenerative medicine and tissue engineering.
Molecular imaging techniques using specific imaging probes or reporter genes can provide fundamental information about the survival and localization of implanted cells. Noninvasive in vivo imaging can be used to evaluate transplantation efficiency in individual subjects in the context of cell-based therapy. 17 A variety of molecular imaging modalities, including optical imaging and magnetic-based imaging agents, have been used to track implanted stem cells. Luciferase enzyme-based optical imaging is a highly sensitive method, and the luciferase reporter gene is inherited from mother cell to daughter cells. Because the bioluminescence imaging (BLI) signal is not diluted during cellular proliferation or differentiation, it is possible to represent cell number and localization in terms of the bioluminescence signal intensity. For this reason, luciferase reporter imaging is well suited to monitoring implanted stem cells in a mouse model. In this study, we used a highly sensitive luciferase reporter 18 to monitor the viabilities and distributions of transplanted MSCs injected via different routes. We also visualized the fate of implanted MSCs and evaluated the therapeutic efficacy of MSC transplantation in a partially pancreatectomized (PPx) mouse model during the course of pancreas regeneration.
Materials and Methods
Animals
BALB/c nude mice (8–10 weeks old) were obtained from Orient Bio Inc. (Gyeonggi, Korea). For the transplantation procedure, a female mouse was used as a donor, and a male was used as a recipient. All procedures, including experimental animal treatment and surgical procedures, were reviewed and approved by the Institutional Animal Care and Use Committee of Seoul National University Hospital.
Preparation of MSCs
Bone marrow cells were collected by flushing mouse femurs and tibias with Dulbecco's Modified Eagle's Medium (DMEM; Gibco-Invitrogen, Eugene, OR). Collected cells were then cultured in 75 cm2 tissue culture flasks at a concentration of 1 × 10 6 cells/mL using complete DMEM supplemented with 10% fetal bovine serum (FBS; Gibco), 1 mM glutamine, and 1% penicillin/streptomycin (Cellgro-Mediatech, Manassas, VA) in a humidified chamber with 5% CO2 at 37°C. Nonadherent cells were removed by replacing the media after 72 hours, and adherent cells were subcultured after reaching 70 to 80% confluence. A homogeneous cell population was obtained after 3 to 5 weeks. Cells were analyzed by fluorescence-activated cell sorting (FACS) using a BD FACSCanto Flow Cytometer (BD Immunocytometry Systems, San Jose, CA) to confirm MSC characteristics. Murine MSCs were immunephenotyped as CD34, CD45, CD11c, c-kit, and Flk1 negative and as Sca1, CD29, CD44, and CD105 positive using FACS (all staining antibodies from BD Pharmingen, San Diego, CA). MSCs were maintained in culture for no more than 12 in vitro passages.
Luciferase-Expressing MSCs
The 293FT cell line was transfected with retrovirusmediated enhanced firefly luciferase (effLuc). The effLuc construct was kindly provided by Dr. Brian Rabinovich from the MD Anderson Cancer Center. 18 After 48 hours, retroviruses produced in the media were collected and filtered through 0.2 μm pore-size syringe filters. For retroviral transduction, retrovirus was packaged using the Retro-X packaging system (BD Biosciences, Franklin Lakes, NJ) and was introduced into purified MSCs in the presence of polybrene (Sigma-Aldrich, St. Louis, MO) and Lipofectamine LTX with Plus Reagent (Invitrogen). After 2 days, MSCs were stained with anti-mouse Thy-1.1 PE (BD Pharmingen), and MSCs harboring luciferase were sorted out using magnetic activated cell sorting (MACS; Miltenyi Biotec, Bergisch Gladbach, Germany).
Measurement of In Vitro Luciferase Activity
Cultured MSCs were rinsed twice with phosphate-buffered saline (PBS) and were treated with 4 mL trypsin– ethylenediaminetetraacetic acid (EDTA) in T75 flasks for 3 minutes in an incubator at 37°C. To halt the trypsin-EDTA effect, detached MSCs were washed with serum containing DMEM and PBS. Suspended MSCs (5 × 10 5 ) were serially diluted with PBS onto a 24-well plate. D- Luciferin substrate (50 μL, 1.5 mL; Caliper Life Sciences, Hopkinton, MA) was added to the MSCs, and the solution was gently mixed. In vitro luciferase activity was examined using a luminometer (TR717; Applied Biosystems, San Diego, CA).
Transplantation of MSCs
Eight-week-old male mice (Orient Bio Inc.) were used as the recipients. Mice were anesthetized via inhalation of 2% isoflurane in gaseous nitrous oxide and oxygen. Before transplantation, MSCs were loaded with 2 μM of chloromethyl-dialkyl-carbocyanine (CM-DiI) fluorescent dye (Molecular Probes, Eugene, OR) for 5 minutes at 37°C and were further stained for 15 minutes at 4°C. The MSCs were then washed with PBS and resuspended in growth medium for transplantation. For pancreatic ductal injection, MSCs were carefully injected into the duct at the head of the pancreas near the duodenum. For local injection, MSCs were injected into the tail of the pancreas where it attaches to the spleen.
PPx Mouse Model
Approximately 80% of the pancreas, including the attached portion of the spleen, was removed by gentle abrasion with cotton swabs, leaving the major blood vessels supplying other organs intact. MSCs (1 × 10 6 ) were collected with 50 μL PBS or Matrigel (BD Biosciences, San Jose, CA) and were directly transplanted into the remaining (head portion) pancreas of the PPx mouse. Control mice (PPx-Ctr) underwent the same procedure as PPx mice, but pure PBS or Matrigel (50 μL) without MSCs was implanted into each pancreas. PPx-mock indicates PPx-Ctr mice bearing a Matrigel-only implant without MSCs. Animals were fasted for 12 hours before the operation and were allowed free access to standard food and water for 5 hours after the operation. Anesthesia was achieved by inhalation of gaseous nitrous oxygen and isoflurane.
In Vivo BLI
The animals were placed individually in an induction chamber, and anesthesia was induced with 2% isoflurane (Forane, JW Pharm., Seoul, Korea) during in vivo BLI using an IVIS-100 optical imaging device (Caliper Life Sciences). Animals were given D-luciferin substrate (Caliper Life Sciences; 3 mg/100 μL/mouse) via intraperitoneal injection for 15 minutes for the luciferase enzymatic reaction to proceed. BLI was conducted at the acquisition time of 5 minutes using an open filter set. Bioluminescence signal data were collected from a region of interest (ROI) and were expressed as the average photon count per pixel (cm2).
Immunocytochemistry
Pancreatic tissue sampling was performed on days 6 and 28 after MSC transplantation. Samples were fixed with 4% paraformaldehyde solution in PBS (USB, Cleveland, OH) for 24 hours at room temperature and then were embedded in paraffin. Pancreatic sections (4 μm thick) were dewaxed in xylene, and endogenous peroxidase activity was blocked using 0.5% H2O2 in methanol for 30 minutes. Sections were then washed in PBS and blocked with normal goat serum or normal horse serum for 30 minutes at room temperature. Tissue sections were incubated with a 1:800 dilution of goat antiluciferase (Millipore, Billerica, MA), a 1:500 dilution of rabbit antiinsulin, a 1:100 dilution of rabbit antiamylase (Sigma-Aldrich), and a 1:250 dilution of rabbit anti-CK20 (Santa Cruz Biotechnology, Santa Cruz, CA). Slides were incubated with primary antibodies overnight at 4°C before being washed in PBS and incubated with the fluorescent dye-conjugated secondary antibodies (antirabbit–Alexa Fluor 488; antigoat–Alexa Fluor 488; antirabbit–Alexa Fluor 594, Invitrogen) for 1 hour at room temperature.
Determination of Blood Glucose Levels and Intraperitoneal Glucose Tolerance Test
Fasting blood glucose concentrations were measured using a Contour TS Blood Glucose Meter (Bayer HealthCare, Mishawaka, IN) in blood obtained from the mouse tail vein after a 5-hour fast. After transplantation, blood samples were taken from the tail vein of PPx mice for 1 month. To examine normal variations in blood glucose, an intraperitoneal glucose tolerance test (IPGTT) was performed in mice that underwent transplantation with MSCs or pure Matrigel at 3, 6, and 12 days (n = 6). Mice were intraperitoneally injected with 1 mg glucose/g body weight after fasting overnight (1416 hours). Blood samples were obtained from the tail vein at 0, 5, 10, 15, 20, 30, 60, and 120 minutes and were analyzed for glucose level using a Contour TS Blood Glucose Meter (Bayer HealthCare).
Insulin Enzyme-Linked Immunosorbent Assay
We used a mouse insulin enzyme-linked immunosorbent assay (ELISA) kit (Millipore, MA) for the quantification of insulin from PPx mouse sera or coculture media. PPx mouse sera were isolated from blood samples obtained at 3, 6, 9, and 12 days after PPx, and coculture media were collected 3 days after MSC culture with pancreatic duct cells. For sandwich ELISA, we followed the manufacturer's recommendation. Briefly, insulin molecules from samples were incubated in microtiter plates coated with monoclonal mouse antirat insulin antibodies for binding to occur. Unbound materials were then washed from samples. Biotinylated secondary antibodies were added, and samples were incubated for 2 hours. Free enzyme conjugate was removed by washing. Immobilized antibody-enzyme conjugates were quantified by horseradish peroxidase activity at 450 nm.
Coculture of Primary Pancreatic Cells with MSCs
Pancreatic tissues were collected from normal 12-week-old male nude mice and were digested with 5 mg collagenase type 4 (Worthington Biochemical, Lakewood, NJ) in serum-free DMEM (Welgene, Daegu, Korea) in a 37°C shaking water bath for 20 minutes. The digested pancreatic cells were washed five times with PBS, and undigested cells (eg, blood vessels and lymph nodes) were removed manually using a pipette. The isolated pancreatic cells were plated with MSCs (1:1) on cover slips in 12-well plates. The cell mixture was cultured with DMEM supplemented with 10% heat-inactivated FBS (Hyclone, Waltham, MA) and 1% penicillin/streptomycin (Gibco) for 3 days until adhesion. No growth factors or glucose was added to the culture medium to exclude their effects on cell proliferation and differentiation during culture.
Statistical Analysis
Data were expressed as the mean ± standard deviation (SD) of the mean. Mean and SD values were calculated from at least four experiments. Significant differences between two groups were detected using Student unpaired t-tests. Differences between more than two groups were identified using one-way analysis of variance (ANOVA) followed by the Dunnett test with SigmaPlot statistical software (Systat Software, San Jose, CA). Statistical significance was accepted at p values of < 0.05 (*) and < 0.001 (**).
Results
Establishment of Luciferase-Expressing MSCs
The MSCs had small cell bodies (Figure 1A, left) and expressed several specific markers, such as Sca1 as assessed by FACS staining (data not shown). To establish highly sensitive luciferase-expressing MSCs, the cells were infected with a retroviral vector containing the effLuc reporter construct, 18 and the MSC population harboring effLuc was selected via MACS using a Thy-1.1-specific antibody. FACS analysis indicated 29.8% transduction efficiency (Figure 1B). Expression of effLuc was specifically detected in the cytoplasm of the sorted cells by immunofluorescence staining (Figure 1A, right), and the luciferase activity of the MSCs gradually increased as the cell number increased (Figure 1, C and D). To test the optical resolution of luciferase-expressing MSCs in vivo, we injected MSCs into the pancreatic tail of a mouse and visualized them (Figure 1, E and F). The values of total photon flux in the region of interest (ROI) from an MSC-bearing mouse were dependent on cell number. We also verified that the MSCs used in this study retained their original characteristics after reporter transfection by measuring the expression of several MSC-specific markers (data not shown). We also confirmed the differentiation of MSCs into osteoblasts using osteoblast-specific alizarin red staining (data not shown). These findings indicate that retroviral transfection of the reporter did not change the characteristics of the MSCs.

Establishment of luciferase-expressing bone marrow–derived mesenchymal stem cells (MSCs). A, The general morphology of MSCs was assessed by hematoxylin-eosin (H & E) staining, and luciferase expression was confirmed by immunofluorescence staining. Scale bar: 100 μm. B, FACS analysis indicated a 29.8% efficiency of luciferase reporter gene transduction. C, Luciferase-expressing MSC cells were visualized using an IVIS-100 imaging system, and the cell number is indicated by a color range. D, The luciferase activities of MSCs in vitro were represented with a value of total photon flux in the region of interest (ROI) and were dependent on cell number (N = 3). E, In vivo luciferase activities from MSCs that were injected to the pancreatic tail of a mouse were visualized using an IVIS-100 imaging system. F, The values of total photon flux in the ROI from an MSC-bearing mouse were dependent on cell number (N = 4).
Noninvasive Imaging of MSCs after Transplantation into the Pancreas by a Different Injection Route
Similar to the human pancreas, the mouse pancreas can be approximately divided into a pancreatic head, body, and tail. Each part can be readily identified based on its relationship to adjacent organs; the area near the duodenum is considered the head, and the area near the spleen is considered the tail. As shown in Figure 2A, the mouse pancreas is located under the stomach and is attached to the duodenum and spleen. We aimed to identify the distribution of the MSCs according to injection routes after transplantation into the pancreas. We injected MSCs via two different routes: (1) ductal injection through the pancreatic duct, which is located at the head part of the pancreas, and (2) local injection into the tail part of the pancreas. We also loaded CM-DiI fluorescent dye into the MSCs just before transplantation so that we could visualize MSC localization in pancreatic tissue sections (Figure 2B). Locally injected MSCs exhibited a strong fluorescence signal. At 7 days after transplantation, MSCs injected through the pancreatic duct were detected throughout the whole pancreas (Figure 2C). In contrast, locally injected MSCs were clearly localized in the area of injection (Figure 2D). Noninvasive imaging was also performed to monitor the localization of transplanted MSCs in vivo (Figure 2, E and F). Regardless of injection route, transplanted MSCs were increased during the first week. Transplanted MSCs were clearly observed in both injection routes; however, locally injected MSCs showed stronger fluorescence and bioluminescence signals.

Imaging of mesenchymal stem cells (MSCs) transplanted into the pancreas by different injection routes. A, Anatomy of mouse pancreas and transplantation routes of MSCs. B, Tissue section of pancreas after MSC transplantation. CM-DiI fluorescent dye (red) was loaded into MSCs before transplantation. Scale bar: 50 μm. C, Grafted MSCs at 7 days after injection through the pancreatic duct. Luciferase signals were detected mainly over the whole area of the pancreas and the duodenal papilla. D, Grafted MSCs at 7 days after local injection. Luciferase signals were detected only in the transplanted region. E, Noninvasive in vivo imaging of transplanted MSCs was conducted to monitor the localization of MSCs by injection through the pancreatic duct. F, Noninvasive in vivo imaging of transplanted MSCs by local injection. Five animals received the MSC injection models through the pancreatic duct, and five animals received local injections. Du = duodenum; Li = liver; Sp = Spleen.
Noninvasive Imaging of Transplanted MSCs in the PPx Mouse
A model of pancreatic injury was established via PPx.19,20 Because 80% of the pancreas was surgically removed and only the head portion of the pancreas was retained in the nude mouse, MSCs were locally injected into the pancreas to prevent leakage of MSCs from the injured pancreas. After PPx, a mixture of MSCs and Matrigel (or PBS) was directly injected into the remaining portion of the pancreas. Bioluminescence signals from the transplanted MSCs were then detected at the implantation site for 28 days after transplantation. In vivo luciferase signals of MSCs injected without Matrigel in PPx mice (MSCs in PBS, Figure 3A, top) peaked on day 16 and resulted in the spread of MSCs into the duodenum and the intestine near the pancreas. However, luciferase signals from MSCs with Matrigel peaked on day 8 and decreased continuously until day 28, and MSCs implanted with Matrigel showed limited spread within the area of injection (Figure 3A, bottom). Localization of implanted MSCs in the pancreas was also confirmed by BLI after laparotomy on day 28 (Figure 3B). Thus, our BLI system clearly visualized the distribution and survival of transplanted MSCs.
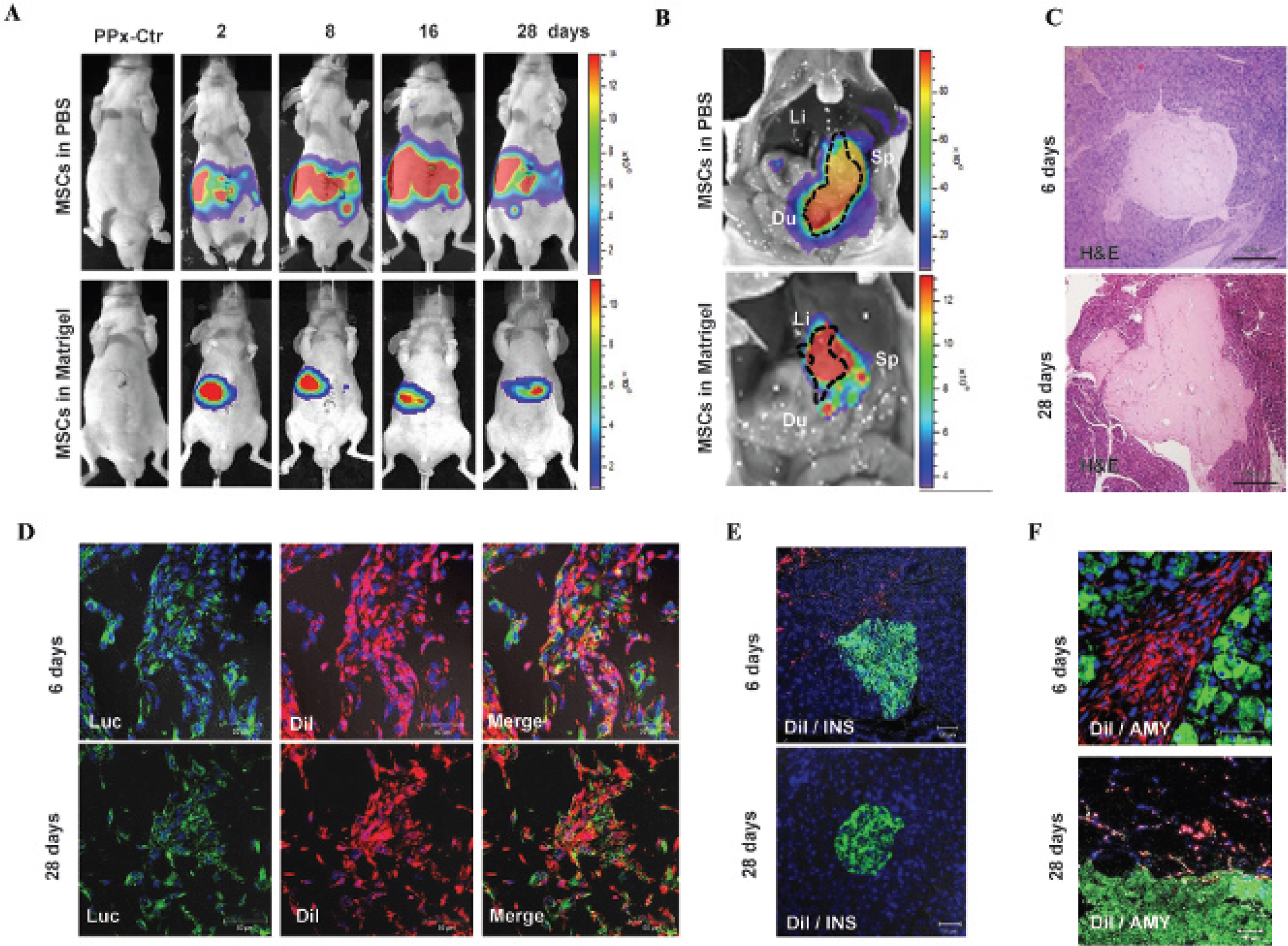
Imaging of mesenchymal stem cells (MSCs) after transplantation in the partially pancreatectomized (PPx) mouse model. A, In vivo bioluminescence imaging of MSCs was monitored on the ventral side for 28 days. B, Bioluminescence imaging of MSCs after sacrificing the animal on day 28. MSCs locally injected with phosphate-buffered saline (PBS) were widely distributed inside the abdominal area, whereas MSCs injected with Matrigel were localized in the pancreas near the injection site. C, MSCs transplanted within Matrigel were detected at days 6 and 28 in pancreatic tissue by hematoxylin-eosin staining. Scale bar: 200 μm. D, Transplanted MSCs were visualized by immunofluorescence using antiluciferase (green). Prior to transplantation, MSCs were loaded with CM-DIL (red) and detected by fluorescence imaging. E, MSCs with CM-DiI signals (red) were located near insulin-positive cells (green) on day 6 or were not placed near insulin-expressing cells on day 28. F, Transplanted MSCs (red) were observed close to the amylase-positive exocrine area (green) on days 6 and 28. CM-DiI signals (red) from MSCs were not colocalized with insulin- or amylase-positive cells on day 6 or 28. Scale bar: 50 μm in D, E, and F. Three animals received the MSC injection models with PBS and three animals with Matrigel, and five animals received the MSC local injections with Matrigel. Two animals were sacrificed on day 6 after transplantation, and three animals were sacrificed on day 28. Ctr = control.
Examination of Transplanted MSCs with Matirigel in Pancreatic Tissue
Hematoxylin-eosin staining of the pancreas indicated that the area transplanted with the MSC-Matrigel complex was readily distinguishable in pancreatic tissue at days 6 and 28 (Figure 3C). Although the luciferase activity of the MSCs was slightly reduced at day 28 compared to day 6, these cells survived for the entire 28 days. The distribution of the MSCs within the Matrigel was clearly detected using a luciferase-specific antibody (Figure 3D). To track all of the implanted MSCs at the cellular or tissue level, MSCs were stained with CM-DiI, a fluorescent cell tracker, prior to transplantation. Merged fluorescence results indicated that CM-DiI persisted in the MSC cytoplasm on days 6 and 28. To verify whether the implanted MSCs differentiated into insulin-secreting cells, an immunohistochemistry assay using an insulin-specific antibody was conducted in tissue slices containing margins of the MSC-Matrigel complex (Figure 3E). On day 6 after PPx, we identified several MSCs within the Matrigel that were close to the insulin-expressing pancreatic cells, but the MSCs were not stained with insulin-specific antibody. On day 28, MSCs were not detected in insulin-positive normal pancreatic tissue. These morphologic features of MSCs were also observed in adjacent amylase-positive cells in the exocrine portion of the pancreas (Figure 3F). These indicated that transplanted MSCs were not directly differentiated into insulin-secreting beta cells.
Monitoring of Blood Glucose Levels
Following PPx, dysfunctional glucose regulation generally results in extreme hyperglycemia because the number of insulin-secreting beta cells decreases. To demonstrate functional recovery of the implanted MSCs in the PPx mice, we monitored fasting blood glucose levels every 2 days and tested glucose tolerance via an IPGTT at 3, 6, and 12 days after transplantation. Since Matrigel contains growth factors, MSCs transplanted with Matrigel were compared to transplantation of Matrigel alone (PPx-mock) to exclude the effects of Matrigel and to elucidate MSC effects on pancreas regeneration. The fasting blood glucose levels of the PPx-mock group were elevated for 0 to 9 days after PPx, and these blood glucose levels decreased gradually until day 28 because regeneration from the remaining pancreas occurred after PPx (Figure 4A). The blood glucose level of MSC-transplanted mice (PPx-MSC) was lower than that of the pure Matrigel group (PPx-mock) during the 28-day monitoring period. We then evaluated serum insulin levels at 3, 6, 9, and 12 days after PPx. Serum insulin levels did not differ between the MSC-transplanted group (PPx-MSC) and the pure Matrigel-transplanted group (PPx-mock) at 3 days after PPx. However, serum insulin levels gradually increased at 3, 6, 9, and 12 days in the MSC-transplanted group compared to the mock group (Figure 4B). These data suggest that transplanted MSCs may help remaining beta cells to recover functional insulin secretion after pancreatic injury. Consequently, we performed glucose tolerance tests during the 3- to 12-day period to assess functional recovery (Figure 4C). At 3 days after transplantation, the time-dependent blood glucose levels in the pure Matrigel group and the MSC-transplanted group were not significantly different. However, MSC-transplanted mice recovered glucose tolerance activity at day 6 compared to the pure Matrigel-transplanted mice. Sixty minutes after glucose injection at day 12, the glucose concentration in the MSC-transplanted group was significantly decreased compared to those in the pure Matrigel-transplanted group. The MSC-transplanted group subsequently recovered glucose tolerance similar to the normal mouse group. These data suggest that MSCs facilitate the recovery of pancreatic function after PPx.

Monitoring blood glucose levels after mesenchymal stem cell (MSC) transplantation in a partially pancreatectomized (PPx) mouse model. A, The fasting glucose levels of PPx mice were elevated for 0 to 9 days after PPx, and these blood glucose levels decreased gradually until day because regeneration from the remaining pancreas occurred after PPx. The PPx-MSC group transplanted with Matrigel showed lower blood glucose levels than the PPx-mock group (Matrigel without MSCs) during the course of pancreatic regeneration in a PPx model. B, The serum insulin levels of mice with transplanted MSCs (PPx-MSC) were gradually increased. In contrast, serum insulin levels in the PPx-mock group were not changed. C, Blood glucose tolerance was monitored after glucose injection at 3, 6, and 12 days posttransplantation. Normoglycemia was not achieved at 120 minutes in either the MSC-transplanted (PPx-MSC) or the PPx-mock group on day 3 after PPx. After 6 days, the PPx-MSC mice displayed a greater decrease in glucose levels from 30 to 120 minutes than did the mice transplanted with pure Matrigel (PPx-mock). The glucose levels in the PPx-MSC mice were similar to those of normal control mice at day 12. *p < .05 and **p < .001 compared to the PPx-mock group. Data are shown as mean ± SD. Three animals received the MSC injection models with PBS and three animals with Matrigel, and five animals received the MSC local injections with Matrigel. Two animals were sacrificed on day 6 after transplantation, and three animals were sacrificed on day 28.
MSCs Contribute to the Proliferation of Insulin-Positive Beta Cells In Vitro
MSCs were cocultured with primary pancreatic cells to assess whether MSCs help generate insulin-producing beta cells from primary pancreatic cells or directly differentiate into pancreatic cells. Primary pancreatic cells in coculture with MSCs adhered to the plate faster than primary pancreatic cells in culture without MSCs (Figure 5A). MSCs were then tracked in coculture by staining with CM-DiI cell tracking dye (Figure 5B). Primary pancreatic cells were cytokeratin 20 positive, and MSCs were located at the periphery of primary cells. Insulin-expressing pancreatic beta cells were detected near the luciferase-positive MSCs (Figure 5C). Interestingly, we observed that the number of insulin-positive cells increased with the addition of MSCs in coculture (Figure 5D). Moreover, secreted insulin levels in cocultured media of primary pancreatic cells and MSCs were increased compared to those of pure pancreatic cells in culture (Figure 5E). These results suggest that MSCs assist in the generation of beta cells and the elevation of insulin secretion.

In vitro coculture of primary pancreatic cells (PanCs) and mesenchymal stem cells (MSCs). A, Isolated PanCs adhered to the culture dish 3 days after seeding, and newly formed cells were grown from the original primary PanCs. In coculture, primary PanCs with the MSCs adhered early and grew faster. B, Cocultured cells were tracked by DiI staining (red) of MSCs and by immunofluorescence staining with cytokeratin 20 (green) as a marker of pancreatic duct cell. C, Luciferase-expressing MSCs (green) and insulin-positive pancreatic cells (red) were not merged in a same cell cluster. D, The number of insulin-positive cells in the 3-day coculture of MSCs and primary pancreatic cells increased more than that of the pure pancreatic cell culture group. E, Secreted insulin levels in coculture media were increased approximately 1.5-fold relative to pure pancreatic cells in culture at day 3. Scale bar: 20 μm in A, 50 μm in B and C. N = 3.
Discussion
DM is primarily caused by injury to or impaired function of pancreatic cells. The adult pancreas is an organ of slow renewal, and its growth occurs mainly by duplication of existing cells rather than by neogenic growth.21,22 For this reason, tissue regeneration was not regarded as a viable option for the treatment of DM. However, recent reports have suggested that regeneration can be induced in the adult pancreas under specific experimental conditions,19,23,24 and tissues with slow renewal rates can be stimulated to produce new cells at higher rates. For example, subtotal pancreatectomy induces neogenic regeneration of the pancreatic tissue.19,20,25,26
MSCs are an attractive option for clinical regenerative therapy. MSCs can be isolated from patients easily and are capable of providing trophic support without an innate immune response. When cells with stem cell–like capabilities were transplanted with islet cells, survival and insulin function were highly increased,27,28 and the stem cell–like cells protected grafted islets from apoptosis. 29 Moreover, Madec and colleagues reported that adult MSCs can protect beta cells from autoimmune responses. 30
Many studies have examined beta cell regenerative treatment of DM by implanting stem cells into the injured region of the pancreas. To treat DM, islets from several origins have been transplanted into kidney capsules in murine models.31,32 For transplantation, human primary islets have been introduced using encapsulation 33 and methoxypolyethylene glycol 34 to reduce or eliminate the need for immunosuppression. Because the successful delivery of a sufficient quantity of islet cells into the recipient pancreas has been challenging, favorable injection routes and matrices must be investigated. Toward this end, the in vivo visualization of grafted cells is significant and necessary because the proper localization and prolonged survival of transplanted cells are important to successful transplantation.
We monitored transplanted MSCs by molecular imaging using a bioluminescence reporter gene to investigate the best possible injection route and the most favorable matrix. Our aim was to estimate the potential capability of MSCs in the regenerating pancreas. Although fluorescent proteins such as green fluorescent protein are widely used, chemiluminescence proteins such as luciferase are powerful tools because they do not require an external source of light. In this study, we observed the location and survival of grafted MSCs using in vivo luciferase activity monitoring without sacrificing the animals. Our optical reporter system was useful for tracking MSCs introduced via pancreatic ductal injection or local injection. Although each injection route has advantages and disadvantages according to the patient's situation or the type of model, our reporter was sensitive enough to evaluate the efficiency of MSC transplantation (see Figure 2). MSCs introduced by pancreatic ductal injection were observed throughout the whole pancreas (see Figure 2, C and E). Locally injected MSCs were more localized to the site of injection but also proliferated more at the injection site (see Figure 2, D and F). In both cases, the proliferation of MSCs was observed after transplantation.
In the PPx model, local injection seemed to be more applicable than ductal injection because the pancreas was partially injured and grafted MSCs could leak from the PPx site of the pancreas. To prevent the leakage of MSCs from the injured pancreas, we used Matrigel, which is frequently used to generate tumor xenografts. A temporal increase in luciferase activity in the MSC-transplanted area was observed until 8 to 16 days after transplantation in the PPx model. A gradual decrease in the in vivo luciferase signal was observed 8 to 28 days after transplantation (see Figure 3A). Similar to in vivo imaging, immunostaining results indicated that the luciferase signal was decreased at 28 days in the pancreatic tissue (see Figure 3D). Thus, the survival of implanted MSCs lasted for 1 to 2 weeks and was continuously reduced in the pancreas of the nude mouse.
Interestingly, some of the transplanted MSCs were detected near insulin-secreting cells in the tissue on day 6 (see Figure 3E, top) and in a primary cell coculture experiment (see Figure 5C). However, implanted MSCs and insulin-positive cells were observed in completely different sites on day 28 (see Figure 3E, bottom). Therefore, we conclude that implanted MSCs did not directly differentiate into any kind of pancreatic cell, including islet or acinar cells, either in vivo or in vitro. Nevertheless, mice implanted with MSCs mostly recovered from glucose tolerance dysfunction (see Figure 4A). In support of our findings, Sordi and Piemonti proposed that stem cells also act as “feeder” cells for islets and as supporters of graft protection, tissue revascularization, and immune acceptance. 14 However, another study reported that human MSCs exhibit the cellular characteristics of pancreatic progenitors by expressing transcription factors that are known to be necessary for the normal development of the endocrine pancreas. 35 MSCs derived from chronic diabetes patients express pancreatic hormones such as C-peptide and insulin. 16 Although these reports share the opinion that implanted MSCs may be capable of differentiating into pancreatic cell types in an in vivo context, our observations did not suggest that implanted MSCs could differentiate into a pancreatic cell lineage, such as insulin- or amylase-secreting cells (see Figure 3, E and F).
We tried to determine how MSCs participate in the regenerating pancreas without differentiating into insulin-secreting beta cells. We could not find any evidence that MSCs differentiate directly into insulin-positive cells or cytokeratin 20–positive duct-like cells (see Figure 5, B and C). However, we found that an increase in insulin-positive cell numbers was dependent on MSC addition (see Figure 5D) and that insulin secretion from beta cells was increased (see Figure 5E) in a coculture of primary pancreatic cells and MSCs. These results suggest that transplanted MSCs promote the injured pancreas to recover its function by promoting insulin production in residual beta cells.
To summarize, we successfully established a population of reporter-transfected MSCs that enabled us to track transplanted MSCs for investigating the MSC-mediated functional recovery of the injured pancreas in a PPx mouse model. Although transplanted MSCs did not differentiate into insulin-secreting cells, they facilitated the functional recovery of glucose regulation in the partially removed pancreas region. In vivo imaging using the highly sensitive luciferase reporter is a useful tool to evaluate the efficiency of MSC transplantation in a mouse model by visualizing the localization and distribution of transplanted cells.
Footnotes
Acknowledgments
Financial disclosure of authors: This work was supported by the Future-based Technology Development Program (No. 2010-0028755), the Global Core Research Center (No. 20110030680) of the National Research Foundation (NRF) from the Ministry of Science and ICT & Future Planning, and grants (A110805, A101446, HI13C1299020013) from the Korea Healthcare Technology R&D Project, Ministry of Health & Welfare, Republic of Korea.
Financial disclosure of reviewers: None reported.
