Abstract
Technetium 99m (99mTc)–annexin A5, a marker of ongoing apoptosis, is supposed to be useful in the detection of metabolically active atheroma. The aim of this study was to determine the potential of 99mTc–annexin A5 for evaluating the therapeutic effects of an angiotensin II receptor type 1 blocker (ARB) (telmisartan) on atherosclerosis. Male apolipoprotein E−/− mice were divided into telmisartan-treated (3 mg/kg/d, n = 10) and control (n = 10) groups. After 16 to 21 weeks of treatment, 99mTc–annexin A5 was injected and cryostat sections of aortic tissues (n = 10–12/aorta) were prepared. The 99mTc–annexin A5 accumulation level in the plaques was evaluated by autoradiography. Serial sections of the plaques were histologically examined to identify the lesion phenotypes (normal vessels, early lesions, atheromatous lesions, and fibrotic lesions), plaque size, macrophage infiltration levels, and lipid deposition levels. Telmisartan treatment significantly decreased the plaque size (0.05 ± 0.05 vs 0.11 ± 0.08, mm2), macrophage infiltration level (0.02 ± 0.02 vs 0.03 ± 0.02, mm2), lipid deposition level (0.01 ± 0.01 vs 0.02 ± 0.02, mm2), and 99mTc–annexin A5 accumulation level (1.30 ± 1.09 vs 2.15 ± 1.91, × 10−6/g). 99mTc–annexin A5 accumulation levels in the plaques positively correlated with macrophage infiltration (r = .69, p < .05) and lipid deposition (r = .66, p < .05) levels. Apoptosis imaging with 99mTc–annexin A5 may be useful for evaluating the therapeutic effects of ARBs on atherosclerosis.
ATHEROSCLEROSIS is a chronic inflammatory disease. The renin–angiotensin-aldosterone system (RAAS) plays an important role in the pathogenesis of atherosclerosis.1,2 Angiotensin II (Ang II), the main mediator of RAAS, promotes endothelial dysfunction, inflammation, and production of reactive oxygen species in atherosclerotic plaques.3,4 The inflammation caused by angiotensin contributes to the development of vulnerable plaques, in part through the activation and induction of apoptosis of macrophages and smooth muscle cells.5,6
Recently, several studies have demonstrated that angiotensin II receptor type 1 blocker (ARB) treatment can attenuate atherosclerotic plaque formation, decrease the cytokine expression level, attenuate inflammation,7,8 and reduce oxidative stress in the vessel wall.9,10 Clinical data also confirmed the efficacy of ARBs (eg, telmisartan) in slowing the progression of atherosclerosis-related-disease and delaying or preventing morbid events in patients with atherosclerosis. 11 Although the beneficial effects of ARBs on plaque morphology can be confirmed by intravascular ultrasonography (IVUS), cardiac computed tomography (CT), magnetic resonance imaging (MRI), or indirect assessment of patient outcomes in clinical settings, 12 the main therapeutic effects, namely, the functional changes of plaques such as attenuated inflammation and apoptosis in the plaques, might be more important and should be targeted for evaluating the therapeutic effects of ARBs.
Molecular imaging of apoptosis using technetium 99m (99mTc)–annexin A5 can detect histologically unstable atheroma in experimental animals. 13 In our previous studies, we found that 99mTc–annexin A5 preferentially accumulates in atherosclerotic plaques and that 99mTc–annexin A5 imaging is potentially useful for evaluating the progression and/or vulnerability of the plaques.14,15
In the present laboratory study, we used 99mTc–annexin A5 to image two groups of apolipoprotein E (apoE) knockout mice, apoE−/− mice, one group treated with telmisartan, an orally active angiotensin II receptor blocker, and control animals that did not receive the angiotensin receptor antagonist, to determine the degree of reduction in atheroma macrophage apoptosis caused by therapy.
Methods
Radiopharmaceuticals
Recombinant human annexin A5 derivatized with hydrazinonicotinamide (HYNIC) was kindly donated by the National Cancer Institute (NCI-Frederick Cancer Research and Development Center, Frederick, MD). HYNIC–annexin A5 was labeled with 99mTc using tricine as the coligand as described previously (specific activity: 4.48 ± 0.15 MBq/μg protein). 16
Animal Studies
Animal care and all experimental procedures were performed with the approval of the Animal Care Committee at Hokkaido University. The male apoE−/− mice (apoE−/−, C57BL/6J; n = 20) used in this study were obtained from Taconic's Gnotobiotic Center (Germantown, NY). After the age of 6 weeks, the mice were weighed and divided into the telmisartan-treated group and the control group. Mice in the telmisartan-treated group (n = 10) were fed a high-fat diet (fat, 21%; cholesterol, 0.15%: diet # TD88137, Harland Teklad, Madison, WI) containing telmisartan (#T1644, LKT Laboratories, Inc., Saint Paul, MN). The dose of telmisartan was set at 3 mg/kg/d, and the food intake and body weight of each animal were monitored weekly to adjust the amount of telmisartan mixed in the diet. Control mice received a telmisartan-free, high-fat diet (n = 10). All animals were kept in a temperature-controlled facility in the Laboratory of Animal Experiments at Hokkaido University on a 12-hour light cycle with free access to food and water.
At 22 to 27 weeks of age, the age-matched animals in the telmisartan group and control group were fasted for 12 hours and then anesthetized with pentobarbital (50 mg/kg body weight, intraperitoneally). 99mTc–annexin A5 (18.5 ± 0.9 MBq/mouse) was intravenously injected into each animal. Two hours after the 99mTc–annexin A5 injection, the animals were sacrificed by exsanguination under deep pentobarbital anesthesia and the aortas were fixed by cardiac perfusion with cold 0.1 M phosphate-buffered saline (pH 7.4) followed by a cold fixative (4% paraformaldehyde, 0.1 M phosphate-buffered saline [pH 7.4]). 14 Ten to 12 samples (average 11 samples/aorta) were dissected from each aorta (Figure 1A), embedded in Tissue-Tek medium (Sakura Finetechnical Co., Ltd.), and frozen in isopentane/dry ice. Serial cross sections of 10 μm (for autoradiographic exposure) or 5 μm (for histochemical staining) thickness were immediately cut and thaw-mounted on glass slides.

Segmentation of the aorta and classification of atherosclerotic lesions harvested from apoE−/− mice. A, Ten to 12 samples were dissected from each aorta: one sample from the innominate artery, two samples from the aortic root, five samples from the thoracic aorta, and four samples from the abdominal aorta. B, Lesions in apoE−/− mice were divided into four categories in accordance with the American Heart Association classification after Movat pentachrome staining as follows: (1) normal vessels, (2) early lesions (types –), (3) atheromatous lesions (types III–IV), and (4) fibroatheromatous/fibrotic lesions (type V).
Plasma Cholesterol, Blood Urea Nitrogen, and Creatinine Levels
Blood samples collected from each animal (approximately 1.5 mL/mouse) were centrifuged after the addition of 100 U of heparin to obtain the plasma samples. Total cholesterol, blood urea nitrogen (BUN), and creatinine analyses were performed on the plasma samples using VET-TEST 8008 (IDEXX, Ellisville, MO). Standards were supplied by the manufacturer.
Histologic Studies
Oil Red O staining and Movat pentachrome staining 17 of serial tissue sections were performed. Immunohistochemical staining with a mouse macrophage-specific antibody (Mac-2, clone m3/38, Cedarlane, Burlington, ON) was also performed in accordance with a standard immunohistochemical procedure, 18 with slight modifications. Briefly, endogenous peroxide activity was blocked for 5 minutes with 0.3% hydrogen peroxide. Thereafter, sections were incubated with a primary antibody (Mac-2, 1:500) for 30 minutes at room temperature. The bound antibody was visualized by the avidin/biotin conjugate immunoperoxidase procedure using Histofine Simple Stain (MAX-PO, Nichirei Bioscience, Japan) and 3,3′-diaminobenzidine tetrahydrochloride (DAB) (Dako, Japan). The sections were counterstained with hematoxylin. The stained specimens were observed under a Biozero BZ-8000 microscope (Keyence, Osaka, Japan), and the obtained images were analyzed with image analysis software (VH-H1A5, version 2.6, Keyence).
Atherosclerotic lesions in apoE−/− mice were divided into four categories in accordance with the American Heart Association (AHA) classification19,20 after Movat pentachrome staining as follows: (1) normal vessels, (2) early lesions (types I–II), (3) atheromatous lesions (types III–IV), and (4) fibroatheromatous/fibrotic lesions (type V) (Figure 1B). Normal regions were defined as healthy vessel walls without visible atherosclerosis. Early lesions were defined as having adaptive thickening of the intima consisting of multilayered or diffuse foam cells with few or no acellular areas (types I–II). Atheromatous lesions contained significant extracellular lipid as lipid pool (type III) or necrotic lipid core superficially covered by a thin fibrous cap (type IV). Fibrotic lesions were defined as lesions in which prominent new fibrous connective tissue has formed, with (type Va) or without (type Vc) lipid core, or partially calcified (type Vb). 15 Total plaque size was measured on Movat pentachrome–stained specimens, macrophage-infiltrated regions were measured on Mac-2-stained specimens, and lipid deposition level was measured on Oil Red O–stained specimens.
99mTc–Annexin A5 Levels in the Myocardium
The myocardium of each mouse was also collected, and the radioactivity in the myocardium was measured using a well-type scintillation counter. The 99mTc–annexin A5 accumulation level was expressed as percent injected dose per gram of myocardium and normalized by animal body weight (%ID × kg/g).
Autoradiographic Studies
The distribution of 99mTc–annexin A5 in atherosclerotic plaques was determined by autoradiography. 15 Briefly, the 10 μm cryostat cross sections of aortic tissues were exposed to phosphor imaging plates (Fuji Imaging Plate BAS-SR 2025, Fuji Photo Film Co., Ltd., Japan), together with a set of calibrated standards. 21 The autoradiographic exposure was performed for 12 hours to detect the radioactivity of 99mTc. The exposed imaging plates were scanned with Fuji Bio-imaging Analyzer BAS-5000 (Fuji Tokyo, Japan; internal resolution of 25 μm), and the obtained images were analyzed with an image analysis software (Multi Gauge, version 3.0, Fuji Photo Film).
Coregistration of Images and 99mTc–Annexin A5 Accumulation Evaluation
Coregistration of autoradiographic and histologic images was performed as described in our previous report. 15 Briefly, the raw data of autoradiography digitally scanned using the BAS-5000 and the corresponding histologic images microscopically captured using the Biozero BZ-8000 were transferred to one computer. Autoradiographic images were magnified and analyzed with Multi Gauge image analysis software (version 3.0), and histologic images were simultaneously magnified and analyzed with VH-H1A5 image analysis software. Regions of interest were manually drawn on the atherosclerotic plaques, and the 99mTc–annexin A5 accumulation level was normalized by the injected dose and animal body weight (%ID × kg or %ID × kg/mm2). 15 The 99mTc–annexin A5 level in plaques was expressed as a ratio with respect to myocardial accumulation levels, with the assumption that the tissue density was 1 g/cm3 (g−1 or no unit). 22
Statistical Analysis
Numerical parameters are expressed as mean ± SD. The unpaired Student t-test was carried out to test the significance of difference in each parameter between the telmisartan-treated group and the control group. One-way analysis of variance and the Bonferroni post hoc test were performed to compare the 99mTc–annexin A5 accumulation levels among different lesion categories. Linear regression analysis was performed to determine the correlation of 99mTc–annexin A5 accumulation level with histologic findings (areas positively stained with Mac-2 or Oil Red O). A value of p < .05 was considered statistically significant.
Results
Body Weight, Plasma Cholesterol and BUN/Creatinine Levels, and Blood 99mTc–Annexin A5 Level
No significant difference in body weight or total cholesterol level was observed between the telmisartan-treated and control groups. Mice in the telmisartan-treated group showed significantly higher plasma BUN and creatinine levels than those in the control group (Table 1). The blood 99mTc–annexin A5 level was significantly higher in the telmisartan-treated group than in the control group. 99mTc–annexin A5 levels in the myocardium and other organs were also higher in the telmisartan-treated group than in the control group (Table 2).
Comparison of Body Weight, Plasma Cholesterol Level, Plasma BUN, and Creatinine Levels between Telmisartan-Treated and Control Groups

BUN = blood urea nitrogen; NS = not significant.
Data represent mean ± SD (n = 10/group).
Comparison of 99mTc–Annexin A5 Accumulation (%ID X kg/g) in Different Organs between Telmisartan-Treated and Control Groups 2 hours after Injection

Data represent mean ± SD (n = 10/group).
Effects of Telmisartan on Plaque Morphology and 99mTc–Annexin A5 Levels
Table 3 shows the number of lesions classified into four categories in the telmisartan-treated and control groups. The statistical analysis demonstrated that telmisartan treatment significantly decreased the number of atherosclerotic plaques (telmisartan-treated mice versus control mice: 5.6 ± 1.7 vs 8.0 ± 1.9, lesions/aorta). In particular, the number of atheromatous lesions markedly decreased (1.1 ± 0.6 vs 4.0 ± 2.5, lesions/aorta). Normal and early lesions accounted for about 90% of lesions in the telmisartan-treated group, whereas a high proportion of atheromatous lesions was observed in the control group.
Comparison of Lesion Numbers in Each Category between Telmisartan-Treated and Control Groups

NS = not significant.
Data represent mean ± SD (n = 10/group).
Representative images and quantitative results of microscopy of the stained specimens and autoradiography are shown in Figure 2. Telmisartan treatment significantly decreased the plaque size (telmisartan-treated mice versus control mice: 0.048 ± 0.052 mm2 vs 0.105 ± 0.083 mm2, p < .05) (see Figure 2, A and B), macrophage infiltration level (0.020 ± 0.017 mm2 vs 0.029 ± 0.022 mm2, p < .05) (see Figure 2, A and C), and lipid deposition level (0.011 ± 0.014 mm2 vs 0.020 ± 0.017 mm2, p < .05) (see Figure 2, A and D). The 99mTc–annexin A5 accumulation level in the plaques of telmisartan-treated mice was also significantly lower than that of control mice (1.30 ± 1.09 vs 2.15 ± 1.91, × 10−6/g) (see Figure 2, A and E).
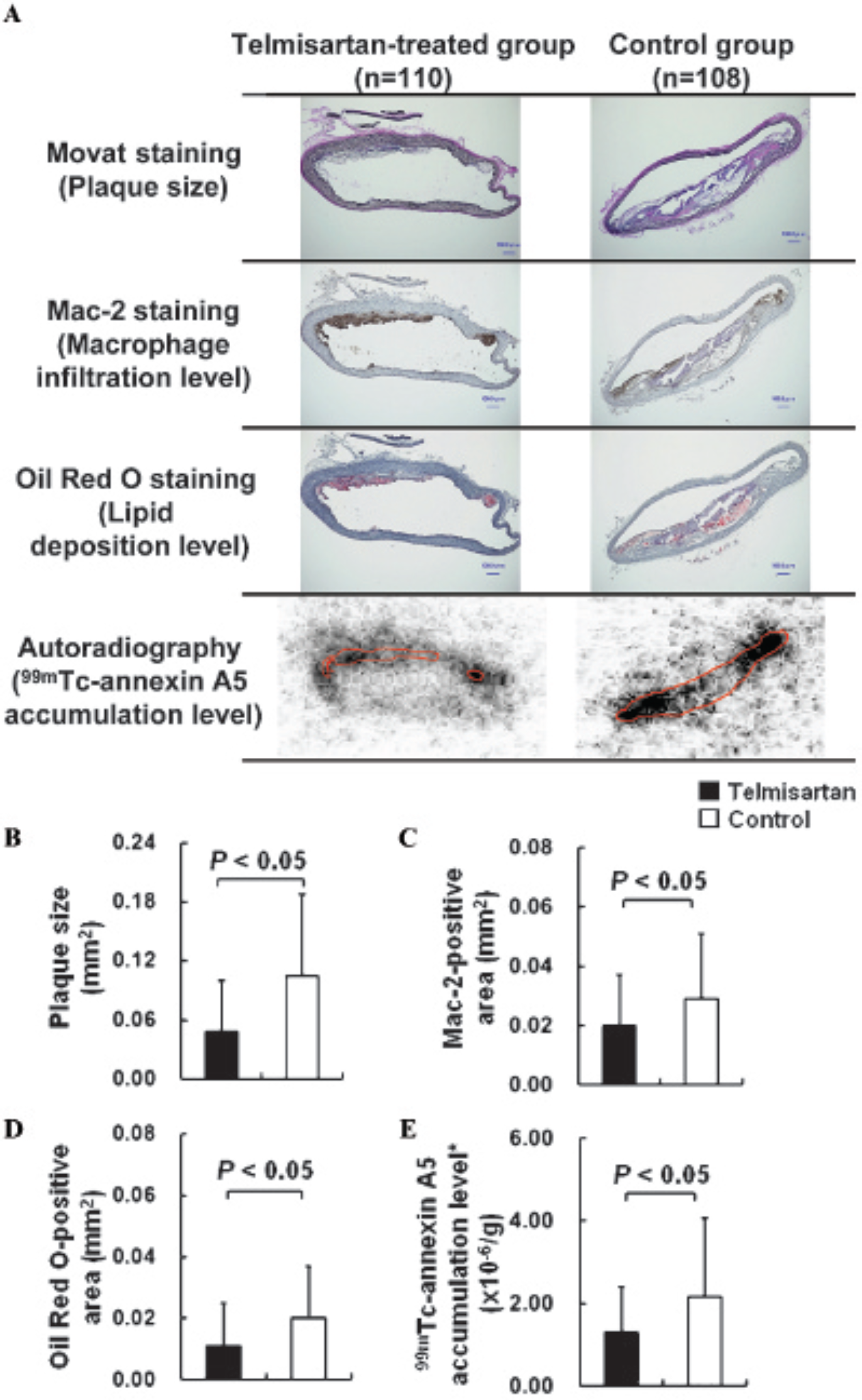
Comparison of plaque size, macrophage infiltration, lipid deposition, and 99mTc–annexin A5 accumulation levels in plaques between the telmisartan-treated and control groups. A, Serial cryostat sections (5 μm) subjected to Movat pentachrome staining, Mac-2 staining, and Oil red O staining are shown. Radioactivity exposures were performed on serial cross sections (10 μm) to obtain corresponding autoradiograms. B to E, Quantitative evaluation of plaque size (whole intimal areas) (B), Mac-2-positive areas (macrophage infiltration) (C), Oil Red O–positive areas (lipid deposition) (D), and 99mTc–annexin A5 accumulation (E) in the telmisartan (▪) and control (□) groups. *Ratio of accumulation level in plaques with respect to that in myocardium.
Relationships between 99mTc–Annexin A5 Level and Histologic Findings
The relationships between the 99mTc–annexin A5 level and morphologic parameters are shown in Figure 3. The 99mTc–annexin A5 level positively correlated with the levels of macrophage infiltration (r = .69, p < .05) and lipid deposition (r = .66, p < .05) (see Figure 3).

Regression analyses of tracer accumulation and intraplaque macrophage infiltration and lipid deposition levels. Relationships between the 99mTc–annexin A5 accumulation level and Mac-2-positive areas (macrophage infiltration) (A) and between the 99mTc–annexin A5 accumulation level and Oil Red O–positive areas (lipid deposition) (B) were examined by regression analyses. *Ratio of accumulation level in plaques with respect to that in myocardium.
To determine the ability of 99mTc–annexin A5 localization to distinguish unstable plaques from normal vessels and stable plaques, the accumulation of 99mTc–annexin A5 was compared in each lesion category of the treated and control groups. In both the telmisartan-treated and control groups, the normal vessels showed low 99mTc–annexin A5 accumulation levels, similar to that in the myocardium (1.09 ± 0.49–fold for the telmisartan-treated group, 1.25 ± 0.71–fold for the control group). The 99mTc–annexin A5 accumulation level was significantly higher in early lesions (1.91 ± 1.12– and 1.97 ± 1.09–fold, respectively, for the telmisartan and the control groups). In atheromatous lesions, 99mTc–annexin A5 uptake increased further (2.49 ± 0.96–fold for the telmisartan-treated group, 2.59 ± 1.12–fold for the control group). In fibrotic lesions, 99mTc–annexin A5 uptake was relatively low (1.44 ± 0.63–fold for the control group). No fibrotic lesion was observed in the telmisartan-treated group (Figure 4).

Comparison of 99mTc–annexin A5 accumulation levels among different lesion categories. A = atheromatous lesions; E = early lesions; F = fibrotic lesions; N = normal vessels. *Ratio of accumulation level in plaques with respect to that in myocardium. †No fibrotic lesion was observed in the telmisartan-treated group.
Discussion
In our apoE−/− mouse model, telmisartan effectively reduced the progression of atherosclerosis. The 99mTc–annexin A5 accumulation level in plaques was significantly decreased by telmisartan treatment, in concordance with the reduction of plaque size, macrophage infiltration, and lipid deposition (see Figure 2). The 99mTc–annexin A5 accumulation level in plaques positively correlated with the macrophage infiltration and lipid deposition levels (see Figure 3).
The correlation of the 99mTc–annexin A5 accumulation level with the intraplaque macrophage infiltration and lipid deposition levels is the basis of monitoring the therapeutic effect of telmisartan on atherosclerosis in this study. The main cellular population that undergoes apoptosis in atherosclerotic plaques is the foamy mononuclear infiltrates. 23 Telmisartan treatment effectively reduced the monocyte/macrophage infiltration in plaques, which consequently reduced the source of apoptotic cells, which was quantitatively reflected by the decrease in the 99mTc–annexin A5 accumulation level in plaques. The data from this study suggest that the attenuation of apoptosis by telmisartan treatment can be monitored with 99mTc–annexin A5 accumulation.
Over the last two decades, the indications for treatment with angiotensin receptor–blocking drugs have markedly increased.24,25 In addition to the efficacy of ARBs in controlling blood pressure, the applicability of these agents to reduce cardiovascular risk in patients with different cardiovascular conditions has also been noted. 25 Studies on the potential use of molecular imaging for evaluating therapeutic effects on atherosclerosis, however, have extensively focused on statins, the lipid-lowering agents used for treating hyperlipidemia, using 99mTc–annexin A5 or 18F-fluorodeoxyglucose (18F-FDG).26–28
It is important to discuss the 99mTc–annexin A5 accumulation level in each lesion category. Despite the decrease in 99mTc–annexin A5 level in whole-lesion areas by telmisartan treatment (see Figure 2), the accumulation level in each category was not altered between the control and treated groups (see Figure 4). These findings support the accuracy of 99mTc–annexin A5 for evaluating plaque characteristics and the feasibility of this technique for evaluating the therapeutic effects of ARBs on atherosclerosis.
In this study, we calculated the 99mTc–annexin A5 accumulation level in plaques as a ratio with respect to that in the myocardium. We used this calculation not only because the tracer distribution in plaques can be visualized as a contrast of lesion to background ratio in SPECT imaging but also because we wanted to address the concern regarding the renal effects of ARBs. Clinical trials also indicated that ARB administration potentially induces adverse effects on renal function, worsens azotemia, and decreases the glomerular filtration rate (GFR). 29 Because 99mTc–annexin A5 undergoes significant renal excretion, the decreased GFR by ARB treatment may consequently delay tracer elimination. Based on this consideration, we examined plasma BUN and creatinine levels, which are commonly used to estimate GFR. As shown in Table 1, telmisartan treatment increased plasma BUN and creatinine levels, which indicate the reduced GFR, and may consequently have delayed the clearance of 99mTc–annexin A5 from the blood in apoE−/− mice. Along with the delayed blood clearance, 99mTc–annexin A5 showed significantly decreased distribution in the kidneys but increased distribution in other organs, including the liver, muscle, and heart, in the telmisartan-treated mice (see Table 2). It is also known that chronically high levels of BUN and creatinine adversely impact red blood cells and cause an increase in phosphatidylserine exposure with time, 30 which may result in the increased blood-pool activity with telmisartan treatment. However, when the 99mTc–annexin A5 accumulation level was calculated as a lesion to background ratio, this parameter could accurately reflect the plaque characteristics and the therapeutic effects of telmisartan on atherosclerosis (see Figure 2 and Figure 3). It should be noted that the lesion to blood or lesion to muscle ratio is a more general index than the lesion to myocardium ratio. However, the lesion to myocardium ratio is preferable for evaluating tracers that target atherosclerotic plaques in coronary arteries. 31 ARBs can effectively reduce the incidence of cardiovascular events, and the effects of ARB on atherosclerotic plaques in the coronary artery seem to be more critical. Thus, the lesion to myocardium ratio was used in this study to indicate the potential contrast to neighboring tissues for in vivo imaging.
As a limitation, we did not perform in vivo SPECT imaging in this study. The plaques (0.005–0.32 mm2 in cross sections) were far smaller than the spatial resolution of our small-animal SPECT system 32 and most others (ranging from 0.3 to 2 mm in full width at half-maximum). 33 Although Isobe and colleagues, using a dual-head micro-SPECT (X-SPECT; Gamma Medica, Inc., Northridge, CA) with micro-CT, showed that 99mTc–annexin A5 accumulation in atherosclerotic plaques is distinguishable in apoE−/− mice, 34 higher spatial resolutions and quantitative capabilities are needed for evaluating therapeutic effects than for distinguishing plaques from surrounding regions. Therefore, we used an autoradiographic method to quantitatively compare 99mTc–annexin A5 accumulation in plaques between the treated and control groups. It is necessary, however, to validate the results of this autoradiographic study by in vivo SPECT imaging. Further study using a larger animal model with spontaneous atherosclerosis (eg, Watanabe heritable hyperlipidemic rabbits) may solve this problem.
Because we focused on the suppression of apoptosis in plaques, we only used 99mTc–annexin A5 in the present study. However, 18F-FDG is the most preferable and widely used tracer for atherosclerosis. It is also very interesting to evaluate antiinflammatory effects by 18F-FDG PET because suppression of inflammation is one of the antiatherogenetic therapeutic proprieties of telmisartan. 8 Comparative studies using 99mTc–annexin A5 and 18F-FDG would clarify the potential of both tracers, including issues on the lower accumulation levels of annexin A5 than 18F-FDG in atherosclerotic lesions,14,15 and may provide further information on the antiatherogenetic effects of telmisartan from different biologic viewpoints.
We used a dose of 3 mg/kg/d and a duration of 20 weeks, which were found to show certain therapeutic effects on atherosclerosis. 35 The therapeutic effects were confirmed by the histopathologic studies in the present study. However, the antiapoptotic effect of telmisartan on atherosclerotic plaques might differ depending on the treatment dose and duration. More variations in animal groups treated with different doses and durations will bring more variations in outcomes, which will be useful for further clarifying the quantitative potential of 99mTc–annexin A5 for evaluating the therapeutic effects of ARBs on atherosclerosis.
As another limitation of this study, we did not compare the changes in blood pressure in the telmisartan-treated mice and control mice. However, the same treatment protocol in previous reports by other groups may be suitable as a reference for reviewing the changes in this parameter. Significant hypertension can be observed in aged apoE−/− mice, 36 and the elevated blood pressure can be significantly reduced by telmisartan treatment at a dose of 3 mg/kg/d. 35 Our data demonstrate the antiatherogenic effects of telmisartan; however, the possibility remains that these effects may be secondary to the antihypertensive actions of telmisartan. A recent animal study, however, identified significant reductions in atherosclerotic lesion area and intraplaque macrophage infiltration in the absence of any change in blood pressure in apoE−/− mice that received low-dose telmisartan treatment (0.3 mg/kg/d). 35 Moreover, clinical studies also demonstrated that ARBs, but not other antihypertension drugs, such as diuretics or calcium channel blockers, can effectively suppress atherosclerosis and stabilize plaques.37,38
The lack of histologic examination that reflects the apoptosis rate in atherosclerotic plaques is another drawback of this study; however, the feasibility of using 99mTc–annexin A5 for imaging apoptosis in atherosclerotic plaques was already proved by our previous data and previous reports by other groups.13–16
In addition to antiapoptosis, telmisartan has various antiatherosclerotic effects. The most frequently reported benefit of telmisartan on the cardiovascular system is the suppression of inflammation and oxidative stress, with peroxisome proliferator–activated receptor gamma (PPARγ) activation in addition to angiotensin type 1 receptor blockade. 39 Moreover, recent investigations have also indicated the capability of telmisartan to attenuate vascular endothelial growth factor (VEGF)-driven angiogenesis, 40 proinflammatory integrin expression, 41 intraplaque matrix metalloproteinase expression, and intraplaque hemorrhage. 42 Because these substances or pathologic processes are critical to the formation of vulnerable plaques, the above-mentioned therapeutic effects of telmisartan may also be worth evaluating and may have more clinical relevance. In addition to 99mTc–annexin A5 and 18F-FDG, novel radiolabeled agents such as 111In-RP748, 99mTc-MPI, and 18F-RGD have also been recently proposed as potential tracers for assessing particular biologic processes.43,44
The data in this study may also explain the mechanism underlying the cardiovascular benefits of ARBs: the significant decrease in plaque number suggests that ARB treatment can effectively suppress plaque progression; the significantly decreased number of atheromatous lesions indicates that ARB treatment can prevent the formation of unstable plaques and, consequently, may reduce the risk of plaque rupture and cardiovascular events; and the significantly decreased macrophage infiltration and lipid deposition levels in the plaques also suggest that ARB treatment can effectively attenuate plaque vulnerability.
Conclusions
Telmisartan treatment significantly reduced atherosclerotic plaque formation and decreased intraplaque macrophage infiltration and lipid deposition levels in apoE−/− mice. The 99mTc–annexin A5 accumulation level in the plaques was simultaneously reduced, in parallel with reduced macrophage infiltration and lipid deposition. Our data indicate the potential and accuracy of 99mTc–annexin A5 imaging for evaluating the therapeutic effects of ARBs on atherosclerosis.
Footnotes
Acknowledgments
We would like to thank the staff of the Department of Nuclear Medicine and Central Institute of Isotope Science, Hokkaido University, and the Facility of Radiology, Hokkaido University Medical Hospital, for supporting this work. We also thank the NCI-Frederick Cancer Research and Development Center for providing annexin A5.
Financial disclosure of authors: This study was partially supported by the Project for Developing Innovation Systems from the Ministry of Education, Culture, Sports, Science and Technology, Government of Japan. This research was also partially supported by a Grant-in-Aid for General Scientific Research from the Japan Society for the Promotion of Science.
Financial disclosure of reviewers: None reported.
