Abstract
The humanized antibody (hu1G8) has been shown to localize to prostate stem cell antigen (PSCA) and image PSCA-positive xenografts. We previously constructed hu1G8 anti-PSCA antibody fragments and tested them for tumor targeting and the ability to image prostate cancer at early and late time points postinjection by positron emission tomography (PET). We now then compare the PET imaging and the radioactivity accumulation properties in prostate cancer tumors and nontarget tissues to determine the superior 124I-labeled hu1G8 antibody format. 124I-labeled diabody, minibody, scFv-Fc, scFv-Fc double mutant (DM), and parental IgG were administered into severe combined immunodeficiency (SCID) mice bearing LAPC-9 xenografts and followed by whole-body PET imaging of mice at preselected time points. Regions of interest were manually drawn around tumor and nontarget tissues and evaluated for radioactivity accumulation. The 124I-hu1G8 IgG has its best time point for tumor high-contrast imaging at 168 hours postinjection. The 124I-hu1G8 minibody at 44 hours postinjection results in superior tumor high-contrast imaging compared to the other antibody formats. The 124I-hu1G8 minibody at 44 hours postinjection also has comparable percent tumor radioactivity compared to 124I-hu1G8 IgG at 168 hours postinjection. The 124I-hu1G8 minibody is the best engineered hu1G8 antibody format for imaging prostate cancer.
IMAGING HAS PLAYED an integral role in the successful management of men afflicted with prostate cancer. Anatomic imaging modalities, to an extent, fulfill their role in evaluating the progress of prostate cancer from detection, to staging, to the evaluation of advanced disease. 1 For instance, transrectal ultrasonography (US) is used for biopsy guidance and brachytherapy seed guidance. Biopsies based on US guidance have a sensitivity of 70 to 80% for detection of cancer. 2 Magnetic resonance (MR) has provided information on local tumor extent by having high sensitivity and specificity for detecting extracapsular extension and seminal vesicle invasion. 3 Recently, MR spectroscopy and MR with superparamagnetic nanoparticles have provided information on aggressiveness and detected the presence of prostate cancer cells in lymph nodes, respectively. 3 Computed tomography (CT) has a role in the nodal staging of prostate cancer. However, it is limited and is recommended only for patients who have advanced disease based on high tumor grade and prostate-specific antigen (PSA) levels. 4
Radionuclide imaging has the ability to obtain specific and sensitive tumor information in a short time between tracer injection and subject scanning. However, radionuclide imaging has yet to show a significant role in prostate cancer detection or local staging. For instance, radionuclide imaging such as bone scanning using technetium 99m methylene diphosphonate is useful only for assessing distant spread of prostate cancer to the bone and assessing response to treatment. In addition, bone scans are recommended when the serum PSA has reached high levels (10–50 ng/mL); however, the frequency of a positive bone scan remains very low (≈ 10%). 1 Positron emission tomography (PET) with 2-deoxy-2-[18F]fluoro-D-glucose (18F-FDG) has also reported low sensitivity of 18F-FDG for tumor detection (organ confined or metastatic spread) and cannot differentiate between benign prostatic hyperplasia and prostate carcinoma.5–7
Ideal cancer care is designed to offer patients a maximum of cancer control with a low risk of complications. In prostate cancer, it must first require accurate identification and characterization of the tumor. Despite the information and advantages the above anatomic and radionuclide imaging modalities provide, prostate cancer continues to be the most common malignancy and the second leading cause of cancer-related mortality in American men. 8 Prostate carcinoma has the propensity to spread either by direct extension or metastasizing via lymphatic or blood vessels. Because of this metastatic risk, accurate staging of newly diagnosed patients and identification of sites of recurrence following initial therapy are critical areas to address. It is estimated that 40 to 60% of patients with clinically localized prostate cancer have subdetectable lymphatic or extraprostatic extension of the disease at the time of presentation. 9 Therefore, new targeted imaging agents for prostate cancer detection may provide for more accurate detection, staging, and monitoring of prostate cancer.
A promising cell surface protein to target for molecular imaging of prostate cancer is prostate stem cell antigen (PSCA), a cysteine-rich 123–amino acid glycosylphos-phatidylinositol-anchored glycoprotein overexpressed in prostate carcinoma. 10 PSCA was discovered through genes upregulated in primary prostate cancer cells passaged through xenografts in severe combined immunodeficiency (SCID) mice. 10 PSCA is expressed in > 80% of local and metastatic disease 11 and has been correlated with increased tumor stage, grade, and progression to androgen independence. 12 In addition, PSCA has low levels of expression in a very limited number of normal tissues.11,13
Antibody-based imaging approaches for noninvasive targeting of specific molecular phenotypes in prostate cancers have been attempted, and it has been demonstrated that they have the potential to play a role in the development and implementation of targeted molecular imaging in prostate cancer patient management. Indium 111 (111In)-labeled capromab pendetide (ProstaScint, Cytogen Corporation, Princeton, NJ) is a clinically available imaging monoclonal antibody (mAb) to detect lymph node metastases in prostate cancer. 1 Recently, a humanized antibody (hu1G8) was generated specific to human PSCA. 14 Preclinical studies with 111In-labeled intact hu1G8 (111In-hu1G8) showed good tumor to carcass and tumor to blood ratios in biodistribution studies at 72 hours postinjection with high contrast images of prostate cancer xenografts obtained at 196 hours postinjection. 14 As with ProstaScint, molecular imaging of tumors using radiolabeled hu1G8 (molecular weight 150 kDa) appears to require long imaging wait times due to a long mAb circulation half-life (1–3 weeks) typical of IgGs, resulting in low tumor to background ratios at early time points. 15
Newer developments in radioimmunoimaging by PET with engineered antibody fragments have resulted in a highly sensitive technique for imaging tumors in vivo due to high antigen specificity and optimum pharmacokinetics, resulting in high tumor to background ratios. 16 Due to their rapid pharmacokinetics, these engineered antibody fragments can clear quickly from circulation and nontarget healthy tissues. Coupled with rapid and adequate fragment uptake into tumors, engineered antibody fragments result in high tumor to background ratios at earlier times after injection.17–20 These impressive tumor-targeting and pharmacokinetic qualities have now allowed engineered antibody fragments to be used as attractive alternatives for radionuclide imaging of tumors in humans at early time points after injection (18-48 hours).21,22
In our laboratory, engineered antibody fragments have been extensively evaluated in preclinical models for tumor detection of colorectal carcinoma at early and late time points by microPET.16,23,24 For example, engineered antibody fragments specific for the carcinoembryonic antigen (CEA) have been generated. T84.66 bivalent engineered antibody fragments were assembled, (VL-linker-VH-hinge-CH3)2, to produce a minibody and (VL-linker-VH)2 to produce a diabody. The VL is the light-chain variable region, VH is the heavy-chain variable region, and CH3 is the human IgG1 third constant domain. Both fragments demonstrated in nude mice bearing LS174T human colon carcinoma tumors to have fast clearance kinetics, rapid tumor targeting, and lower normal organ uptake compared to the parent antibody.24,25 PET image evaluations of both the minibody and the diabody resulted in similar tumor region of interest (ROI) to background ROI and tumor ROI to soft tissue ROI at 18 hours postinjection. 23 Imaging PSCA-expressing prostate cancer tumors with engineered antibody fragments has also previously been evaluated in male SCID mice with subcutaneously implanted androgen-dependent LAPC-9 xenografts. Scout imaging at early and later time points postinjection with iodine 124-(124I; t1/2 = 100.8 hours) labeled hu1G8 diabody and 124I-hu1G8 minibody was employed to evaluate high-contrast tumor imaging with respect to nontarget tissues.18,19 PET imaging revealed that the 124I-hu1G8 minibody produced high-contrast tumor images at 21 hours postinjection. 18 However, the 124I-hu1G8 minibody also demonstrated a longer blood half-life (t1/2β = 11.2 hours) compared to other similarly studied minibodies targeting colorectal and breast cancer tumors.18,24,26 The 124I-hu1G8 diabody was evaluated at 4, 12, and 20 hours postinjection and found to have its best imaging capability at 12 hours postinjection, which is faster compared to the T84.66 diabody.19,23 Thus, the hu1G8 fragments have displayed unique imaging properties distinct from our experience with other cancer systems; therefore, the ideal imaging time and engineered antibody format for imaging PSCA-expressing tumors remain to be seen in the LAPC-9 tumor–bearing SCID model of prostate cancer.
We introduced two novel fragments and incorporated the intact mAb into this study. We evaluated the capabilities of five matched engineered antibody formats (IgG, scFv-Fc, scFv-Fc double mutant [DM], minibody, and diabody; Figure 1) at different time points to determine statistically the best engineered format and accompanying time point for imaging LAPC-9 xenograft prostate cancer. The goal is to evaluate the effect that engineered format variations on antibodies have on clearance and tumor targeting to elucidate which is the faster clearing format that can still penetrate into prostate cancer tumors and provide high-contrast images by PET.

Schematic of evaluated anti-PSCA hu1G8 humanized IgG and generated engineered antibody fragments. Line (gray) connecting VL and VH genes for scFv-Fc fragments and minibody is an 18–amino acid GlySer rich linker. The diabody is engineered with a shorter GGGSGGGS linker. For minibody, indirect fusion to the human IgG1 CH3 domain is accomplished via the human IgG1 upper and middle hinge and continued by a GlySer linker peptide 10 residues in length (dotted line). Mutations H310A and H435Q on the Fc of the scFv-Fc DM fragment are indicated by an asterisk.
Materials and Methods
Prostate Cancer Cells
The human prostate cancer cell line LAPC-9 used in this study was derived from a human metastatic bone lesion and has been reported in previous studies.14,27–29 Briefly, LAPC-9 tumors were excised from male SCID mice and minced. The explants were then allowed to incubate in PrEGM (Clonetics, San Diego, CA) after digestion in 1% Pronase (Sigma-Aldrich, St. Louis, MO) for 20 minutes at room temperature. Tumor mixture was passed through a 40 μm cell strainer, stained with 0.4% trypan blue (Sigma-Aldrich, St. Louis, MO), and counted. Single LAPC-9 cell suspensions were obtained, and 1 × 106 cells were immediately subcutaneously inoculated in SCID mice. Tumors were allowed to grow for 2 weeks and imaged at a size of approximately 500 mg.
Animals
Eight- to 10-week-old male SCID mice were maintained under pathogen-free conditions under a protocol approved by the University of California, Los Angeles (UCLA) Chancellor's Animal Research Committee. Animals were housed in accordance with the UCLA Department of Laboratory Animal Medicine guidelines.
Protein Expression, Screening, Propagation, Purification, and Size Characterization
Hu1G8 IgG, minibody, and diabody were produced and purified as previously described.14,18,19 Hu1G8 scFv-Fc was engineered by excising the human IgG1 CH3 gene from the plasmid encoding the hu1G8 minibody and subsequently subcloning the human IgG1 Fc gene behind the hinge of the minibody gene. The scFv-Fc DM was produced by site-specific mutagenesis polymerase chain reaction. Histidine 310 (Kabat numbering) was mutagenized for an alanine and histidine 435 for a glutamine producing the H310A/H435Q double mutation. Both constructs were subcloned into the mammalian expression vector pc3.1 (Invitrogen, Carlsbad, CA) and then transfected into NS0 mouse myeloma cells as described 26 and were placed under neomycin (Sigma-Aldrich) selection (0.9 mg/mL). Hu1G8 scFv-Fc protein selection from protein-producing clones was based on protein expression level evaluated by enzyme-linked immunosorbent assay (ELISA) and Western blot as described. 26 High-expressing clones were expanded to terminal cultures, and the level of scFv-Fc expression was assayed by ELISA using serial dilutions of harvested supernatants with reference minibody of known concentration as a standard. ScFv-Fc fragments were purified by cation exchange chromatography. 18 Size and PSCA binding were analyzed by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE), high-performance liquid chromatography (HPLC), and flow cytometry as previously described. 18
Radiolabeling and Small-Animal PET Imaging
Two hundred micrograms of hu1G8 IgG and antibody fragments were radioiodinated with 500 to 600 μCi of 124I (sodium iodide in 0.02 M NaOH; radionuclide purity > 99%) provided by Advanced Nuclide Technologies (Indianapolis, IN) using the Iodogen method, and labeling efficiency was determined as described. 30 Mice were injected via the tail vein with 124I-hu1G8 minibody (50 mg, specific activity: 66.6 ± 7.4 kBq/μg, n = 4), 124I-hu1G8 diabody (50 mg, specific activity: 92.5 ± 7.4 kBq/μg, n = 5), 124I-hu1G8 scFv-Fc DM (50 μg, specific activity: 77.7 ± 11.1 kBq/μg, n = 3), 124I-hu1G8 scFv-Fc (50 μg, specific activity: 70.3 ± 7.4 kBq/μg, n = 4), or 124I-hu1G8 IgG (50 μg, specific activity: 7.40 ± 11.1 kBq/μg, n = 3) in saline/1% human serum albumin (Sigma-Aldrich). Mice were anesthetized with isoflurane (2%) in oxygen at a flow rate of 2 mL/min during PET acquisition. At selected time points, whole-body scans were performed with a 10-minute acquisition time (one bed position) using a Focus microPET scanner (Concorde Microsystems Inc., Knoxville, TN). Images were reconstructed using a filtered back-projection reconstruction algorithm, corrected for 124I attenuation, 31 and displayed using the AMIDE software package (UCLA, Los Angeles, CA). 32 Mice were also imaged by non–contrast-enhanced microCT using a CT II imaging scanner (ImTek Inc, Knoxville, TN) with a 12-minute acquisition time. CT images were created using Feldkamp reconstruction at 200 μm resolution and coregistered with the PET scans for anatomic reference and accuracy. 33
Image-Based Cylindrical ROI Analysis
Reconstructed images were loaded into AMIDE and three-dimensional cylindrical ROI were drawn within the LAPC-9 tumor and major anatomic organs (ie, heart, lung, liver, and soft tissue arm muscle [adjacent side of xenografts]) throughout different imaging time points in the same mouse. ROI cylinders(3-8) were manually drawn within LAPC-9 xenografts to fill the entire tumor volume space. The appropriate positioning of ROI was verified by looking at each of the coronal, sagittal, and transverse orientation PET/CT images. For ROI analysis in normal organs, three cylindrical ROI per organ were drawn either in the transverse (liver) or coronal (heart, lungs, and muscle) projections. Cylindrical ROI volumes per organ were of similar size, and placement within the organ was consistent. ROI PET mean activity (MAROI) units were expressed as MAROI per cubic millimeter. The MAROI/mm3 in the tumors and nontarget tissue were then decay corrected beginning with the initial imaging time point analyzed. The decay-corrected MAROI/mm3 values were then multiplied by the administered dose (MBq) injected with the particular 124I-labeled engineered antibody. Coronal projections are used to show the radioactivity accumulation in the tumor and major organs evaluated at each time point. For ROI analysis of 124I-hu1G8 IgG, the radioactivity intensity of images was scaled off the first imaging time point (72 hours). For ROI analysis of 124I-hu1G8 engineered fragments, the radioactivity intensity of images was scaled off the 21-hour time point of mice injected with the 124I-hu1G8 minibody.
Statistical Analysis
Multiple group comparison evaluations were performed. For analysis of tumor targeting at the inspected imaging time points, defined by radioactivity in the tumor versus nontarget tissues, repeated measures one-way analysis of variance was performed followed by a Dunnett post test for evaluating the significance of the muscle, liver, lung, and heart versus the reference tumor (Prism version 2.0, GraphPad, San Diego, CA). An additional posttest for linear trend over the imaging time points of mice injected with 124I-hu1G8 IgG was applied to determine significance in clearance of radioactivity in the tumor and nontarget tissues. F ratios (dfbetween, dfwithin) and p values are reported. Time activity curves of radioactivity in the tumors versus nontarget tissues were fitted to a nonlinear regression curve with a weighted (1/Y 2 ) laurentzian distribution. Weighted sum of squares is reported. For comparative analysis of percent radioactivity in the tumors between hu1G8 antibodies at the selected imaging times, a nonparametric Kruskal-Wallis test followed by a Dunn post test was performed. Statistical significance was considered at p < .05. All data are plotted as mean and standard deviation.
Results
Imaging of LAPC-9 Tumor-Bearing SCID Mice Injected with 124I-hu1G8 IgG
On inspection from the coronal small-animal PET projections, radioactive accumulation in the mouse was highest at 72 hours. Therefore, mouse images at 120, 144, and 168 hours were scaled to the same threshold as the 72- hour time point and evaluated for radioactivity accumulation in the tumors and nontarget tissue (Figure 2A).
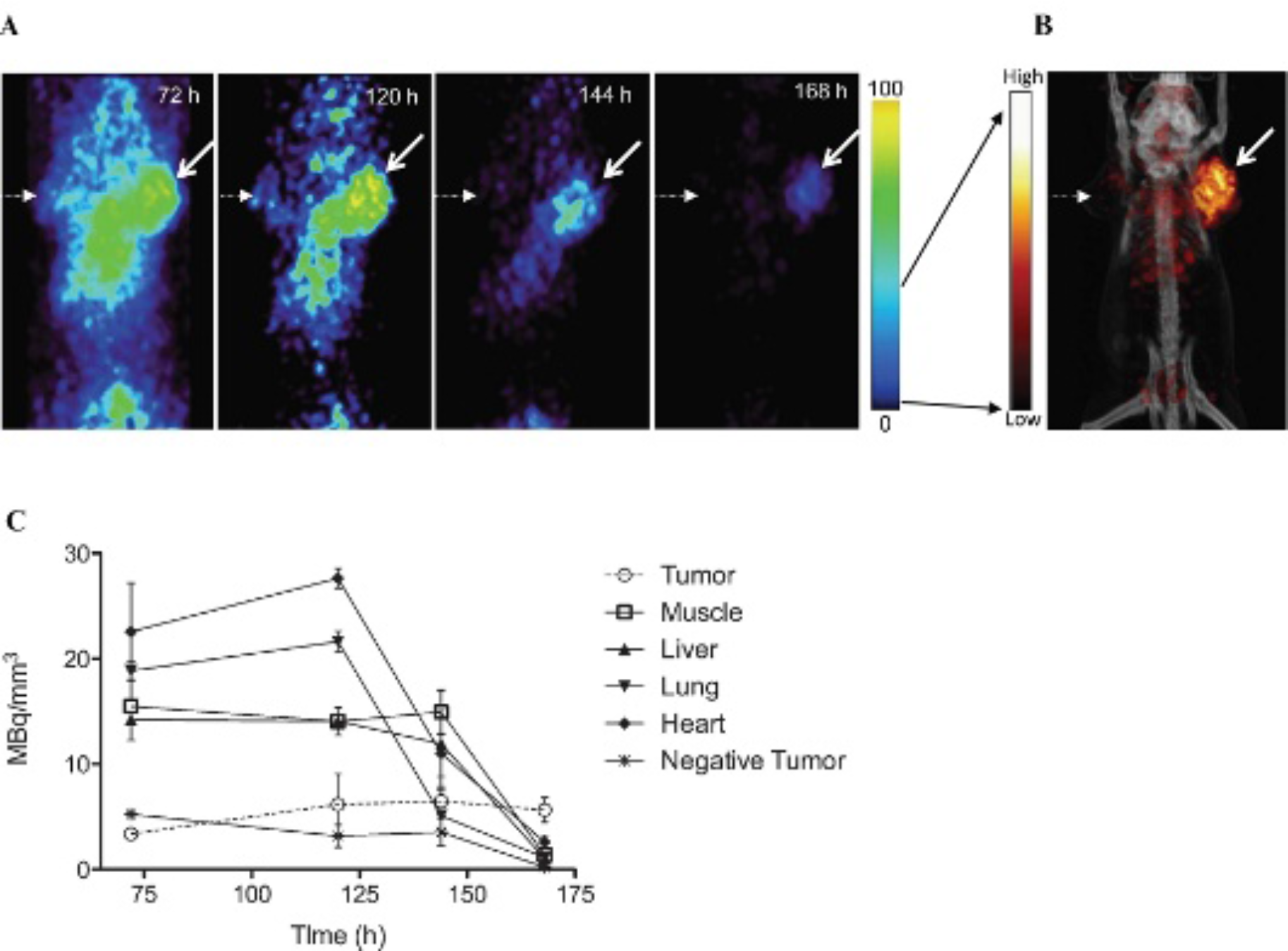
Serial small-animal PET imaging and ROI tumor and organ activity evaluation of mice injected with 124I-hu1G8 IgG. A, Comparison of PET images of SCID mice at 72, 120, 144, and 168 hours postinjection. Mice bearing PSCA-positive (LAPC-9; arrow) and PSCA-negative (PC-3; dotted arrow) xenografts in SCID mice are shown. Mice at different time points are on a common scale. B, The 168-hour time point was rescaled (black arrows) to illustrate excellent contrast achieved with the 124I-hu1G8 IgG at 168 hours. C, Tumor and organ activity (MBq/mm3) over time as measured by image ROI analysis of microPET data sets. Mean ± SD is shown and has been corrected for 124I decay.
Radioactivity in nontarget tissue and in the PSCA-negative tumor decreased markedly where only residual radioactivity remained at 168 hours postinjection. Radioactivity in the LAPC-9 xenograft appeared to accumulate higher from 72 to 120 hours, plateau between 120 and 144 hours, and decrease by 168 hours postinjection, respectively. Mice injected with 124I-hu1G8 IgG at 168 hours achieved the highest tumor to background contrast, especially regarding the central cavity in the abdominal region, and resulted in a clear image of the PSCA-positive LAPC-9 tumor (Figure 2B). Decay-corrected cylindrical ROI analyses of the constructed images were compared for radioactivity in the tumor versus nontarget tissue and PSCA-negative tumor at each time point. At 72 and at 120 hours postinjection, radioactivity in the mouse heart, lung, liver, and muscle was significantly higher than in the LAPC-9 tumor (F72 h (5,2) = 9.16, p = .0017; F120 h (5,2) = 38.35, p < .0001; Figure 2C). By the 144-hour time point, radioactivity remained stable in the tumor, whereas radioactivity in the heart and lung markedly decreased. At 168 hours postinjection, radioactivity accumulation in the PSCA-positive tumor was significantly higher over the nontarget tissue and PSCA-negative tumor (F168 h (5,2) = 10.79, p = .0009). We also compared the clearance in radioactivity accumulation in the PSCA-positive and -negative tumors and nontarget tissue over the four time points evaluated by PET ROI analysis. The muscle, liver, lung, heart, and PSCA-negative tumors all displayed a significant decrease in radioactivity (pmuscle = .0148, pliver .0030, Plung < .0001, Pheart .0059, and Pnegative tumor .0104). PET image inspection and ROI analysis demonstrate that the LAPC-9 radioactivity in the tumor remains over time, whereas radioactivity in nontarget tissue significantly decreases due to washout from nonspecific binding, resulting in the radioactivity in the tumor versus nontarget tissue at 168 hours being the best scan time, and thus now serves as a benchmark for comparison with the 124I-hu1G8 fragments.
Comparative Evaluation of LAPC-9 Tumor versus Nontarget Tissue in SCID Mice Injected with Different Engineered 124I-hu1G8 Fragments
Having established that the best prostate cancer imaging time point within the time frame evaluated with the 124I-hu1G8 IgG was at 168 hours postinjection, we then evaluated the different hu1G8 formats. A multiple comparison test evaluated if faster-clearing 124I-hu1G8 engineered antibody fragments could produce comparable prostate cancer tumor images at earlier times between tracer injection and subject scan. The scFv-Fc DM was designed to determine if mutations of Fc residues important for FcRn binding resulted in faster clearance and a high proportion of radioactivity in the tumor versus nontarget tissue. The scFv-Fc (no mutations) through pilot studies was previously determined to have high-contrast tumor imaging of prostate cancer tumors at 120 hours postinjection and served as an additional comparative reference. As both scFv-Fc formats are novel, we report here their biochemical characterization. The engineered hu1G8 scFv-Fc and scFv-Fc DM fragments were successfully expressed and yielded levels of 2 to 5 mg/L in terminal culture. Analysis of purified scFv-Fcs by SDS-PAGE (Figure S1A, online version only) showed that the relative size migration in comparison with hu1G8 IgG and hu1G8 minibody was consistent with a covalent dimer of ≈ 105 kDa. Under reducing conditions, both scFv-Fcs migrated as a monomer consistent with its predicted molecular weight of 52.5 kDa. Size exclusion chromatography verified that the scFv-Fcs eluted as a uniform peak at a time (28 minutes), corresponding to a correctly folded dimer of expected molecular weight of 105 kDa (Figure S1B, online version only). Flow cytometry showed that the anti-PSCA scFv-Fc and scFv-Fc DM fragments could discriminate for LAPC-9 cells (Figure S1C, online version only). The 124I-hu1G8 scFv-Fc DM demonstrated two different profiles of tissue radioactivity at 4 hours versus 20 hours postinjection. At 4 hours, there was significantly higher radioactivity in nontarget tissue with only modest radioactivity levels in the LAPC-9 tumor (F(4,2) = 13.06, p = .0014; Figure 3A, left panel). Sixteen hours later, at the 20-hour imaging time point, radioactivity levels markedly decreased with only minimal radioactivity decrease in the tumor. The 20-hour time point ROI analysis resulted in only the heart as the nontarget tissue with significantly higher radioactivity compared to the LAPC-9 tumor (F(4,2) = 4.89, p = .0272). The reference scFv-Fc format demonstrated at 12° hours postinjection that the heart was also the only nontarget tissue with significantly higher radioactivity compared to the LAPC-9 tumor (F(4,3) = 7.22, p = .0050; Figure 3A, right panel). Thus, the scFv-Fc DM successfully produced comparable radioactivity levels in the LAPC-9 tumor versus nontarget tissues relative to the scFv-Fc at 20 hours versus 120 hours, respectively. Inspection of microPET images of mice injected with 124I-hu1G8 scFv-Fc DM revealed high radioactivity in the central cavity containing major organs at 4 hours (Figure 3C) versus a decrease in radioactivity in nontarget tissue, with the exception of the heart, at 20 hours postinjection (Figure 3D). The 20-hour imaging time point with 124I-hu1G8 scFv-Fc DM was comparable to the 120-hour imaging time point of mice injected with 124I-hu1G8 scFv-Fc (Figure 3E). These images are in agreement with ROI analysis where tumor radioactivity accumulations in the tumor and nontarget tissue between the scFv-Fc DM at 20 hours and the scFv-Fc at 120 hours are comparable (see Figure 3A).
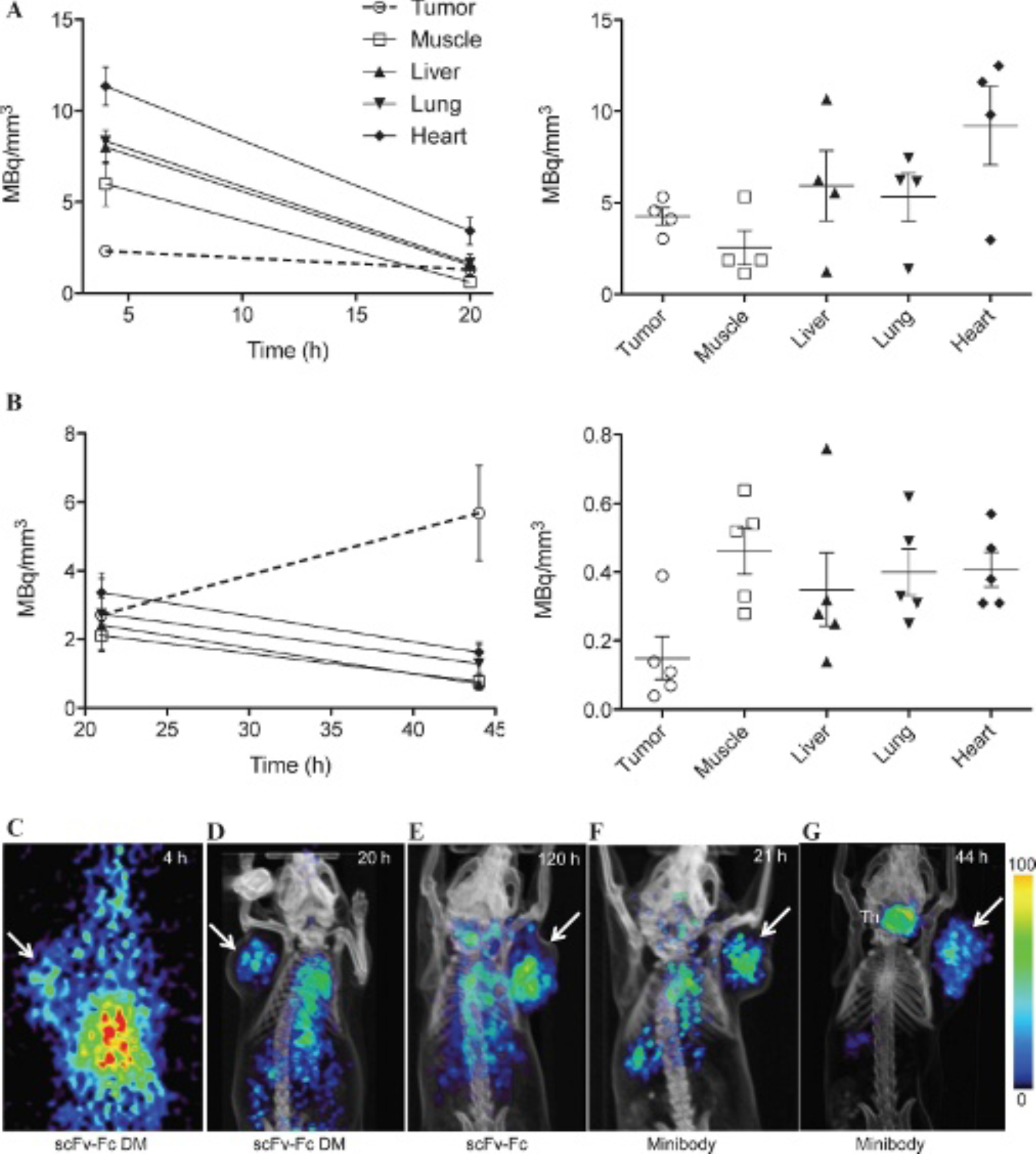
ROI analysis of tumor versus nontarget tissue radioactivity accumulation of different 124I-hu1G8 engineered formats and small-animal PET imaging comparison. A, Tumor and nontarget tissue radioactivity in mice injected with 124I-hu1G8 scFv-Fc DM (left panel) at 4 and 20 hours postinjection and 124I-hu1G8 scFv-Fc (right panel) at 120 hours postinjection. B, Tumor and nontarget tissue radioactivity in mice injected with 124I-hu1G8 minibody (left panel) at 21 and 44 hours postinjection and 124I-hu1G8 diabody (right panel) at 12 hours postinjection. Comparison of PET images of SCID mice bearing LAPC-9 tumors (arrow) injected with 124I-hu1G8 scFv-Fc DM at (C) 4 hours and (D) 20 hours, (E) 124I-hu1G8 scFv-Fc at 120 hours, and 124I-hu1G8 minibody at (F) 21 hours and (G) 44 hours postinjection. Mice with thyroid gland activity (Th) did not have Lugol solution in the drinking water. Images are scaled to the same threshold. Color scale indicated.
The 124I-hu1G8 minibody had previously been shown to produce high-contrast images of various prostate cancer tumors at 21 hours postinjection. However, the hu1G8 minibody has also been shown to have a prolonged half-life; therefore, we analyzed mice scanned by microPET also at 44 hours postinjection and compared both time points. In mice injected with 124I-hu1G8 minibody and scanned and analyzed at 21 hours postinjection, there was higher tumor radioactivity versus the muscle and liver, but these levels did not reach statistical significance (Figure 3B, left panel). However, in mice analyzed at 44 hours postinjection, there was a preferable increase in tumor radioactivity and a decrease in radioactivity in the nontarget tissues. The tumor radioactivity accumulation was significant over all nontarget tissues (F(4,3) = 9.127, p = .0013). The selected time point of 12 hours postinjection for analyzing mice injected with the 124I-hu1G8 diabody resulted in poorer tumor radioactivity accumulation versus nontarget tissue radioactivity accumulation (Figure 3B, right panel). MicroPET imaging of mice injected with 124I-hu1G8 minibody at 21 hours (Figure 3F) and 44 hours (Figure 3G) postinjection revealed that tumor radioactivity accumulation can produce high-contrast tumor images at both time points; however, radioactivity is markedly cleared from the circulation at 44 hours versus 21 hours. The radioactivity decrease in the nontarget tissues seen on the PET images is in agreement with the ROI analysis (see Figure 3B, left panel).
Comparison of Percent Tumor Activity across Different 124I-hu1G8 Platforms
To directly compare the uptake of the radiolabeled antibodies into the LAPC-9 tumors by our ROI methodology, radioactivity levels in the tumor were proportioned to the cumulative radioactivity in the nontarget organs analyzed. The percent activity in the PSCA-positive tumors was highest in mice injected with the 124I-hu1G8 minibody and scanned at 44 hours postinjection (Figure 4). Thus, the 124I-hu1G8 minibody with the best tumor radioactivity versus nontarget tissues across the different 124I-hu1G8 formats is the superior engineered fragment. In addition, although not significant, the higher percent tumor activity compared to the 168-hour time point of mice injected with 124I-hu1G8 IgG validates the high-contrast PET images observed with the 124I-hu1G8 minibody at 44 hours and serves as a potential immunoPET tracer for evaluating prostate cancer detection at much earlier times between injection and scanning compared to intact antibodies.

Percent tumor activity of selected 124I-hu1G8 antibody formats at high-contrast imaging times.
Discussion
In this report, we explore the imaging properties of five matched antibody formats of the hu1G8 antibody, all of which bind with high affinity to PSCA, to reach a conclusion on the best antibody format and imaging time point for PET imaging in SCID mice bearing PSCA-positive LAPC-9 xenografts. Tumor versus nontarget tissue radioactivity levels were determined by ROI analysis and evaluated with small-animal PET images scaled to one another. Serial scanning of mice allowed for the evaluation of 124I-hu1G8 antibody distribution throughout normal organs and accumulation in the LAPC-9 tumors. Our results point to the 80 kDa 124I-hu1G8 minibody at 44 hours postinjection as the best engineered antibody fragment and time point identified for detecting LAPC-9 prostate cancer tumors.
We have previously explored tumor imaging time points with anti-PSCA 124I-hu1G8 IgG, minibody, and diabody in LAPC-9 xenograft bearing SCID mice14,18,19 and showed that the engineered minibody and diabody fragments could clear rapidly from nontarget tissue and provide high-contrast images of prostate cancer at earlier time points relative to their parental IgG. Although the earlier imaging time points resulted in good visual tumor image quality, the ideal time points for the minibody and the diabody were drastically different. These results are in stark contrast to those of previous studies comparing diabodies and minibodies, which found that tumors in nude mice bearing colorectal xenografts could be imaged at the same 18-hour postinjection time point. 23 In addition, the 124I-hu1G8 minibody has displayed a serum half-life 2-fold and 1.6-fold longer compared to similar minibodies specific to different cancers.24,26 Therefore, it was unknown what the best postinjection imaging time point was for the parental 124I-hu1G8 IgG, a standard that is necessary to determine if a more rapidly clearing hu1G8 format can have comparable tumor uptake and provide high-contrast prostate cancer images at an earlier postinjection time point. The parental 124I-hu1G8 IgG originally showed high blood-pool radioactivity at 72 hours that was reduced by the 168-hour postinjection time point scan. In this study, we compared all the 124I-labeled engineered fragments at serial imaging within a selected time frame or preselected time points that previously provided high-contrast prostate cancer images and used ROI analysis to determine the best hu1G8 antibody format and the best time point for imaging.
Previous reports have compared different antibody formats to determine a superior fragment size and imaging time point for rapid imaging between antibody administration and subject scan to provide high tumor to nontarget tissue background ratios and crisp visual detection of prostate cancer. A study of a family of matched engineered antibody fragments (IgG, diabody, miniantibody, and scFv), when radiolabeled with 86Y (t1/2 = 14.74 hours), resulted in the “miniantibody” (VH-linker-VL-linker/hinge-IgE CH4; 80 kDa) and the diabody able to produce high-contrast images at 24 hours postinjection in mice bearing minden/RG-1-positive LNCaP xenografts. 20 However, in addition to accumulation in the tumor, the diabody and the miniantibody were detected in the kidneys, liver, and blood. An imaging time course of the miniantibody revealed that the significant nontarget tissue radioactivity accumulation did not decrease from 24 to 48 hours postinjection. This suggests that although the 80 kDa miniantibody is similar in size to the hu1G8 minibody, there are clear differences in nontarget tissue clearance that could be due to differences in antibody isotype (ie, IgE-CH4 vs IgG-CH3). Ultimately, the 124I-hu1G8 minibody may be superior for its ability to provide very high tumor to nontarget tissue ratios and high-contrast images of prostate cancer in xenograft models at 44 hours postinjection due to very low radioactivity in the liver, kidneys, and blood. We do note that the heart is the normal organ evaluated with the slowest washout for all the antibodies evaluated. This may be due to the remaining radiolabeled antibodies in circulation. Although in Schneider and colleagues, LNCaP xenografts were used 20 and in this study we evaluated LAPC-9 xenografts, it would be preferable to evaluate the imaging potential of these 80 kDa formats in the same tumor model of prostate cancer. Unfortunately, LNCaP cells do not express PSCA. However, we have demonstrated high tumor uptake and tumor to blood ratios (LNCaP-PSCA = 9.3 ± 2.3 %ID/g and blood = 3.3 ± 1.6 %ID/g) in stably transfected PSCA-overexpressing LNCaP xenografts at 21 hours postinjection. In addition, LAPC-9 expresses endogenous PSCA at an expression level that is indicative of metastatic disease 28 and has been a benchmark for PSCA expression evaluations.11,12
There has been strong interest in the development of radionuclide detection agents to improve the clinical evaluation of prostate cancer. PET imaging using 18F-FDG frequently falls short in prostate cancer due to low uptake of the tracer. Because 18F-FDG is the most widely used and successful clinical PET tracer for different cancers, there has been continued investment to elucidate a role for FDG-PET in prostate cancer management. However, in the vital area of evaluating metastatic spread, 18F-FDG has reported a low sensitivity when evaluating prostate cancer patients with known sites of metastases. For example, when evaluating the accuracy of PET with 18F-FDG in the detection of osseous and soft tissue metastases of prostate cancer; 18F-FDG showed 65% overall sensitivity and was less sensitive than current bone scintigraphy in the identification of osseous metastases. 7 New radiotracers that use the radioisotope carbon 11 (11C) have shown promise in the clinic. 11C-labeled methionine has shown rapid uptake in 10 minutes postinjection with minimal uptake from the bladder. 34 Other 11C-labeled PET agents include 11C-acetate and 11C-choline, both of which have shown promise in imaging prostate cancer metastases. For example, 11C-choline has been demonstrated to detect recurrences in locoregional prostate cancer. 35 11C-acetate is currently under clinical evaluation. 35 18F-fluorocholine has also been shown to have rapid uptake into prostate cancer cells and has demonstrated that imaging can be performed very early (≈ 1 minute) following intravenous injection. 35 However, 11C has an approximate half-life of 20 minutes and so must be used rapidly after production; thus, it requires a local cyclotron and is therefore not a widely available option.
mAb imaging of prostate cancer continues to galvanize nuclear medicine clinicians, primarily due to the correlation between prostate cancer cell surface phenotypes and advanced-stage prostate cancer and/or metastatic disease.11,12,36 For example, major efforts have been made to evaluate and to determine a clinical utility for ProstaScint, an 111In-labeled murine mAb specific for prostate-specific membrane antigen (PSMA) and used to image prostate cancer patients by single-photon emission computed tomography (SPECT). Although ProstaScint is approved by the Food and Drug Administration (FDA) for imaging soft tissue prostate cancer at the time of diagnosis, the interpretation of ProstaScint images is challenging because of the following factors: the relatively low spatial resolution and low detection efficiency of medium-energy collimators used for 111In photopeaks; nonspecific antibody localization in the normal blood pool, bowel, bone marrow, and prostate gland; and the lack of anatomic information to localize radiotracer accumulation. 37 Moreover, ProstaScint is not approved by the FDA for detecting metastases to the bone at the time of presurgical staging or as an evaluative diagnostic for monitoring of recurrence after local therapy. When ProstaScint was comparatively evaluated against helical CT and 18F-FDG PET (which has poor sensitivity, as we just described) in the detection of metastatic disease, the mAb scan resulted in a lower detection rate than CT or 18F-FDG PET. 38 There have been improvements with efforts with second-generation antibodies such as 111In-labeled J591, which targets the extracellular domain of PSMA (as opposed to ProstaScint, which is specific for an intracellular domain), resulting in the ability to detect soft tissue and bone metastases. 39 However these overall studies culminate in the conclusion that slow-clearing intact mAbs are not favored for the detection of prostate cancer; particularly in areas where diagnostic agents are needed most for successful management of patients with prostate cancer such as, (1) detection of subclinical metastases at the time diagnosis and (2) detection of sites of recurrence during treatment monitoring.
An additional application of radioimmunoimaging is in the development of radioimmunotherapeutic approaches for cancer. Quantitative imaging before therapy allows for the dose estimation of ionizing radiation into the tumor relative to dose-limiting healthy organs. For 124I-labeled mAbs, PET/CT has been shown to reliably quantify radiolabeled macromolecular uptake in vivo in patients with renal cell carcinoma and can have important implications for quantitative pharmacokinetic estimates of macromolecular biodistribution. 40 Taken together with the work demonstrated in this study, 124I-hu1G8 minibody for PET imaging could also provide a sensitive and quantitative approach for determining tumor and normal tissue uptake for potential therapeutic applications.
124I has been the candidate PET radioisotope for the evaluation of prostate cancer imaging with the hu1G8 antibodies because (1) the technique for radioiodination is well established, (2) its physical half-life is compatible with antibodies and engineered fragments, (3) it has rapid clearance from most tissues, and (4) we have not encountered problems with iodination of random tyrosines in the hu1G8 family of antibodies. Despite the positive attributes of 124I labeled to engineered antibody fragments, it has been postulated and demonstrated that 18F-labeled antibodies are more suitable for PET imaging. 41 18F offers the advantages of broad availability, a higher positron yield (nearly 100%, compared to 23% for 124I), and a short half-life, which are better suited for routine clinical use and for the more rapidly clearing engineered antibody fragments. Dynamic PET scans in mice bearing colorectal tumors injected with 18F-labeled diabodies demonstrated rapid tumor uptake by 1 hour and crisp high-contrast tumor images at 4 hours postinjection. 41 However, the kinetics of a minibody was calculated to be too slow with 18F for use as an effective imaging agent. This is likely because the half-life of 18F is too short and will decay while the minibody is washing out from nontarget tissue. For prostate cancer imaging with hu1G8 antibodies, the diabody had unexpectedly high rapid clearance relative to other studied diabodies and may still serve as an effective imaging agent when labeled with 18F.
Some key challenges could arise using the 124I-hu1G8 minibody for imaging prostate cancer in the clinic, such as internalization on receptor binding to PSCA and dehalogenation, resulting in rapid clearance of 124I from prostate cancer cells. These issues can affect PET imaging by resulting in less tumor contrast. However, our results indicate that radioactivity accumulates in the tumor from 21 to 44 hours postinjection with the 124I-labeled hu1G8 minibody, suggesting that dehalogenation may not be a major factor for adequate tumor uptake with this tracer. In this study, we evaluated prostate cancer radioactivity in the tumor and selected nontarget tissues with the goal of comparing different formats in relation to how well prostate tumors were visualized on PET images. However, because of dehalogenation, there was elimination and substantial radioactivity in the bladder in some mice. Because of renal elimination of 124I resulting in accumulation of radioactivity in the bladder, this can potentially make clinical interpretation of 124I-hu1G8 minibody images challenging. However, previous studies with 124I-labeled mAbs have been successfully applied clinically to profile patients with renal cancer where investigators were not hindered by free 124I filtered by the kidneys. 40 Ultimately, renal clearance will have to be taken into account in the clinic. As one of the current unmet needs of prostate cancer is detection of extraprostatic extension of subclinical metastases at the time of presentation, 1 we are currently developing orthotopic LAPC-9 xenograft SCID mouse models of prostate cancer to evaluate 124I-hu1G8 minibody to detect prostate cancer in a more relevant anatomic location. Bladder radioactivity and using ROI to measure targeting will be evaluated to determine how well the 124I-hu1G8 minibody at 44 hours postinjection can detect prostate cancer.
Tumor and normal organ ROI evaluation, in addition to inspection of PET images, supports the development of the 124I-hu1G8 anti-PSCA minibody to evaluate more sophisticated preclinical models of prostate cancer and/or the development of clinical cancer detection strategies.
Footnotes
Acknowledgments
We are grateful for the expert support of Dr. David Stout, Waldemar Ladno, and Judy Edwards at the Crump Institute for Molecular Imaging at UCLA and to Deborah Scollard for reviewing the manuscript.
Financial disclosure of authors: This work was supported by the UCLA SPORE in Prostate Cancer (National Institutes of Health [NIH] CA92131), the UCLA ICMIC (In vivo Centers for Molecular Imaging of Cancer) (NIH CA86306), and Program Project Grant (NIH CA43904). R.E.R. and A.M.W. are members of the UCLA Johnsson Comprehensive Cancer Center (NIH CA16042).
Financial disclosure of reviewers: None reported.
