Abstract
Glioblastoma multiforme (GBM), the most aggressive brain malignancy, is characterized by extensive cellular proliferation, angiogenesis, and single-cell infiltration into the brain. We have previously shown that a xenograft model based on serial xenotransplantation of human biopsy spheroids in immunodeficient rodents maintains the genotype and phenotype of the original patient tumor. The present work further extends this model for optical assessment of tumor engraftment and growth using bioluminescence imaging (BLI). A method for successful lentiviral transduction of the firefly luciferase gene into multicellular spheroids was developed and implemented to generate optically active patient tumor cells. Luciferase-expressing spheroids were injected into the brains of immunodeficient mice. BLI photon counts and tumor volumes from magnetic resonance imaging (MRI) were correlated. Luciferase-expressing tumors recapitulated the histopathologic hallmarks of human GBMs and showed proliferation rates and microvessel density counts similar to those of wild-type xenografts. Moreover, we detected widespread invasion of luciferase-positive tumor cells in the mouse brains. Herein we describe a novel optically active model of GBM that closely mimics human pathology with respect to invasion, angiogenesis, and proliferation indices. The model may thus be routinely used for the assessment of novel anti-GBM therapeutic approaches implementing well-established and cost-effective optical imaging strategies.
GLIOBLASTOMA MULTIFORME (GBM) represents the most common and aggressive form of malignant brain tumors, classified as grade IV glioma by the World Health Organization (WHO).1,2 GBMs are highly invasive and vascularized tumors, further characterized by focal necroses and microvascular proliferations. Despite advances in neurosurgery, radiation, and chemotherapy, inherent resistance to existing therapeutic modalities results in a poor patient outcome.3,4
A lack of suitable animal models is a major limitation in translational GBM research. Commonly used preclinical rodent models for evaluating the efficacy of anticancer agents include nude mice bearing subcutaneous or intracranial human tumor xenografts established using immortalized cell lines.5,6 The advantages of subcutaneous xenograft models include their practical utility for high-throughput testing, reproducible growth rates, and ease of monitoring using simple caliper measurements. However, the genotypical and phenotypical differences, when compared to patient lesions, make them less useful for translational research. 7 Thus, a number of intracranial GBM xenograft models have been established. These include xenografts established from syngeneic rodent cell lines, 8 human cell lines grown in serum-supplemented media, 9 and stem-like cancer cells grown in serum-free media. 10
Rodent GBM cell lines applied at the orthotopic site in immunocompetent rodents are employed to develop syngeneic models. These models are well placed to evaluate immunotherapeutics, which may be studied only in the presence of intact host immunofunctions. 8 However, these models have clear limitations in assessing the efficacy of therapeutic approaches that target human GBM cells due to species-specific barriers and the fact that they do not reflect the complete genetic alterations seen in human GBMs.11–13
Owing to clonal selection in vitro, immortalized human GBM cell lines also fail to recapitulate the typical growth pattern of human GBMs in situ, even when placed in an orthotopic setting. Usually, cell line-based xenografts develop as vascularized lesions that lack the diffuse single-cell invasion seen in human GBMs.11,14 However, following transplantation of stem-like glioma cells grown as neurospheres, an extensive infiltration to the normal brain may be observed. 15
A large number of genetically engineered animal models (GEMs) have been developed (for a review, see Hambardzumyan and colleagues 16 ) to mimic human GBM characteristics in vivo. Even though GEMs have provided important insight into genetic alterations that initiate and drive glioma development, they fail to reflect the cellular heterogeneity of human GBMs.
To overcome some of the limitations associated with human cell line–based GBM xenografts and the difficulties associated with GEMs, several models based on intracerebral injection of solid explants have been established.15,17–19
We have previously reported that human GBM biopsy specimens form spheroids when cultured on soft agar in vitro 20 and when serially xenotransplanted into the brains of nude rodents closely recapitulate human GBM pathology, including extensive infiltration into the brain parenchyma, angiogenesis, and necrosis.21–23 As such, this model is clinically relevant and well suited for application in the assessment of novel therapeutic strategies, representing a useful alternative to traditional in vitro propagated cell lines.24–26
To facilitate noninvasive in vivo monitoring of tumor progression over time, newly emerging animal models of disease should ideally support longitudinal imaging strategies.27–30 Therefore, we exposed human GBM biopsy spheroids to lentiviral vectors containing the firefly luciferase coding sequence in an effort to combine a highly sensitive optical imaging modality with a clinically relevant GBM animal model. To overcome the limitations with virus penetration into the three-dimensional (3D) spheroid core,31–33 we developed a new combined dissociation/transduction protocol of 3D cultures to obtain uniform, long-term luciferase expression in GBM spheroids. Orthotopic implantation of transduced, optically active tissue facilitates noninvasive, high-throughput longitudinal bioluminescence imaging (BLI) of intracranial tumor growth.
In the present study, we sought to develop and validate an optically active, luciferase-expressing, human biopsy–based GBM mouse model and found it to be similar in major aspects, such as growth pattern, proliferation rates, and microvessel density, to the already established and well-described GBM biopsy spheroid model.
Materials and Methods
Tumor Tissue Culture
A human GBM tumor biopsy was obtained during surgery (patient 3). The collection of tumor tissue was approved by the regional ethical committee at Haukeland University Hospital (Bergen, Norway), and the patient gave written informed consent to the use of biopsy tissue for research. Specimens were transferred to test tubes containing complete growth medium, that is, Dulbecco's Modified Eagle's Medium (Sigma, St. Louis, MO), containing 10% fetal bovine serum supplemented with nonessential amino acids, 100 U/mL penicillin/streptomycin, and 400 mM L-glutamine (all from Cambrex, East Rutherford, NJ), and spheroids were prepared as previously described. 20 Briefly, tissue samples were minced into fragments of approximately 0.5 mm in size and placed into 75 cm2 tissue culture flasks (Nunc, Roskilde, Denmark) base-coated with 0.75% agar (Difco, Detroit, MI). Spheroids were maintained in complete growth medium in a standard tissue culture incubator with 5% CO2 and 100% relative humidity at 37°C, and the medium was changed once a week. GBM biopsy-based spheroids were expanded via serial transplantation into immunocompromised rodents, resulting in phenotypically identical (highly invasive and highly angiogenic) GBMs in all xenografts. 21 Array comparative genomic hybridization confirmed the same genetic aberrations present in the primary biopsy derived from patient 3 and in resulting xenografts as determined previously. 23
Preparation of Lentiviral Vector Supernatants
The 293T cell line was used for transient lentiviral vector production. The lentiviral luciferase vector pLenti6-luc2 (500 ng), packaging plasmid pCMV-R8.74psPAX2 (5850 ng), and envelope plasmid VSV-G/pMD2.G (650 ng) were cotransfected into 293T cells. Virus was harvested, filtered, and stored at −80°C.
Transduction of Tumor Material
GBM spheroids were dissociated using the Neuronal Tissue Dissociation Kit (Miltenyi, Bergisch-Gladbach, Germany) according to the manufacturer's protocol and resuspended in cell culture growth medium supplemented with 0.06% methylcellulose at a concentration of 4 × 104 cells/mL. A methylcellulose stock of 1.2% was prepared according to Korff and Augustin. 34 One hundred microliters of the cell suspension was distributed on a 96-well plate with a U-shaped bottom (Nunc, Roskilde, Denmark), and then 100 μL of the firefly luciferase gene-encoding viral supernatant was added and the plate was centrifuged for 2,250 rpm for 90 minutes in a plate centrifuge. Spheroids were incubated for 1 week before in vitro imaging and implantation. The medium was changed every other day.
Generation of Xenograft Models
Xenograft studies were performed with either male or female NOD/SCID mice (6–8 weeks old) bred and maintained in an isolated facility within a specific pathogen-free environment. Animals (N = 12) were anesthetised by intraperitoneal administration of ketamine (10 mg/kg)/midazolam (5 mg/kg). The heads of the recipient mice were secured in a stereotactic frame (David Kopf, Tujunga, CA). Local analgesia/anesthesia (bupivacaine, 8 mg/kg) was applied for 3 minutes before a short longitudinal incision was made. A burr hole was prepared 0.5 mm posterior to the bregma and 2.5 mm to the right of the sagittal suture using a microdrill. Ten luciferase-expressing or wild-type GBM spheroids were implanted into the cerebral cortex of mice (n = 7 and 5, respectively). The spheroids were slowly injected, using a Hamilton syringe (Hamilton, Bonaduz, Switzerland), at 2 mm depth below the brain surface. The syringe was left in place for 5 minutes prior to withdrawal. The skin was closed with an Ethilon 3-0 suture (Ethicon, Somerville, NJ). After surgery, the animals were allowed to recover in an incubator set at 35°C before being returned to their cages. The surgical procedures were performed in accordance with the Norwegian Animal Protection Act, and local ethical committee approval was granted. The tumors were allowed to grow for 2 to 3 months. Animals were sacrificed using CO2 at the onset of disease symptoms, such as weight and activity loss, and the brains were removed. The tumors were excised and placed in culture as described above. New spheroids were generated and serially transplanted into new nude rodents.
Magnetic Resonance Imaging
Magnetic resonance images (MRIs) were acquired using a Bruker Biospin MRI GmbH Pharmascan 7 T small-animal magnetic resonance scanner (Bruker Corporation, Ettingen, Germany) with a 23 mm inner diameter mouse head linear volume Bruker Biospin MRI HmpH RF coil and an animal bed for mouse brain applications (Bruker Corporation). Animals (n = 7) were anesthetized with 1 to 2% isoflurane mixed with 50% air and 50% O2 and placed in a prone position in a cradle containing a heating pad at 37°C. Respiration and body temperature were monitored throughout the MRI procedure. The MRI sequences used included axial T2-weighted scans (repetition time [TR] = 4.2 s, echo time [TE] = 35.22 ms; measuring method: rapid acquisition with relaxation enhancement [RARE], number of averages: 6, number of repetitions: 1, number of echo images: 1, slice thickness: 1 mm, field of view [FOV]: 2 cm, matrix size: 256 × 256, 16 slices) and T1-weighted scans (TR = 0.7 s, TE = 7.5 ms; measuring method: RARE, number of averages: 3, number of repetition: 1, number of echo images: 1, slice thickness: 1 mm, FOV: 2 cm, matrix size: 256 × 256, 16 slices) following subcutaneous delivery of the contrast agent 0.5 mmol/mL Omniscan (gadodiamide, 287 mg/mL, GE Healthcare, Little Chalfont, UK).
MRI-Based Tumor Volume Calculations
Tumor volumes were calculated based on T2-weighted MRIs. Tumor area was estimated by manually drawn regions of interest on slices where tumor was visible using ImageJ software (National Institutes of Health, Bethesda, MD). Tumor volume was calculated as the sum of tumor areas from each slice with visible tumor and multiplied by the slice thickness.
Bioluminescence Imaging
In vitro imaging of GBM spheroids was performed following incubation of spheroids with 1.6 μg/μL of D-luciferin (Biosynth, Staad, Switzerland). Image acquisition was performed 10 minutes later. For in vivo imaging, animals (n = 7 using an Optix MX2 Small Animal Molecular Imager [ART Inc., Saint-Laurent, QC] and n = 7 using an IVIS Spectrum scanner [Caliper Life Science, PerkinElmer Company, Hopkinton, MA]) were depilated and scanned following intraperitoneal delivery of 150 mg/kg of D-luciferin. Images were recorded using the Optix MX2 and analyzed using Optix OptiView version 2.02 (ART Inc.). Alternatively, images were recorded using the IVIS Spectrum scanner and analyzed using Live Imaging 3.2 software (Caliper Life Science) to obtain higher throughput and a shorter acquisition time. 35 For ex vivo imaging, brains (n = 6) were placed on sterile plastic petri dishes right after the mouse was sacrificed, and 25 mg/mL of D-luciferin was added topically right before ex vivo imaging.
Preparation of Brain Sections
Mouse brains (n = 8) were removed, rinsed twice in Dulbecco's phosphate-buffered saline, and fixed in 4% neutral-buffered formalin for 1 to 2 days. Fixed brains were embedded in paraffin, and 5 mm thick sections were prepared. For the evaluation of tissue histology, routine hematoxylin and eosin (H&E) staining was performed.
Histologic Examination and Immunohistochemistry
Paraffin-embedded, formalin-fixed tissue sections from mouse brains were dewaxed with xylene and rehydrated according to standard histologic procedures. For epitope retrieval, sections were boiled for 20 minutes in 10 mM citrate buffer, pH 6.0. Sections were rinsed in Tris buffered saline (TBS)-Tween and blocked in 5% serum in TBS for 1 hour followed by incubation in room temperature for 1 hour with the following primary antibodies: antihuman Ki-67 (Millipore, Billerica, MA), dilution 1:100; mouse antirodent nestin, clone rat-401 (Millipore), dilution 1:200; goat antiluciferase polyclonal antibody (pAb) (Promega, Fitchburg, WI), dilution 1:50; and goat antihuman allograft inflammatory factor 1 (AIF-1) (LifeSpan BioSciences Inc., Seattle, WA), dilution 1:100 in TBS containing 1% serum. Biotinylated goat antimouse or horse antigoat secondary antibodies (Vector Laboratories, Burlingame, CA), dilution 1:200 in TBS/1% serum, were applied for 1 hour at room temperature followed by ABC complex (Vectastain ABC kit, Vector Laboratories) incubation for 30 minutes. Positive signal was developed with diaminobenzidine (DAKO, Glostrup, Denmark) according to the manufacturer's instructions. Sections were counterstained with hematoxylin to visualize nuclei. For assessment of proliferation indices (percentage of Ki-67-positive cells), fractions of labeled tumor cells were assessed in 10 microscopic high-power fields per section using X40 magnification. Microvessel density based on criteria previously described 36 was assessed using rodent-specific antinestin staining. Representative images of H&E or anti–firefly luciferase–stained slides were captured using a Nikon Eclipse E600 light microscope at X10 and/or X20 magnification.
Immunofluorescence Staining
Migrating cells were double-stained with human-specific mouse antinestin, clone 10C2 (Millipore), dilution 1:200, and antiluciferase pAb, dilution 1:50. Sections were incubated with primary antibodies overnight at 4°C. Fluorescein isothiocyanate–labeled rabbit antimouse antibodies (Millipore), dilution 1:200, or biotinylated antigoat IgG (H+L) (Vector Laboratories), dilution 1:200, and ExtrAvidin-Cy3 (Sigma), dilution 1:200, antibodies were used as secondary reagents added for 1 hour at room temperature. Sections were mounted with Vectashield mounting medium (Vector Laboratories) containing 4′-6-diamidino-2-phenylindole (DAPI) for visualizing nuclei. Tissues were examined by laser-scanning confocal microscopy (LSM 510 META, Zeiss, Jena, Germany) at X40 and/or X63 magnification.
Statistical Methods
Statistical analysis using mixed-model linear test analysis (SPSS, IBM Corporation, Armonk, NY) and unpaired t-test (GraphPad Prism version 5.00 for Windows, GraphPad, San Diego, CA) was implemented. GraphPad Prism version 5.00 for Windows was used to correlate the logarithmic values of tumor volumes at each time point with corresponding logarithmic values of bioluminescence signals for each tumor-bearing mouse.
Results
Selection of Highly Bioluminescent GBM Spheroids
A new transduction protocol was employed to transduce 3D GBM cell cultures with recombinant lentiviruses containing the firefly luciferase gene. Tumor spheroids were dissociated and transduced as described in Materials and Methods. BLI signal was recorded in vitro (Figure 1) 1 week postinfection. Spheroids that showed the highest photon counts/mm2/s values were selected for intracranial implantation.

Glioblastoma multiforme (GBM) patient spheroids transduced with firefly luciferase–encoding lentiviruses are highly luminescent and may be imaged in vitro using bioluminescence imaging. GBM spheroids (4 × 103 cells each) were transduced with 100 μL of the firefly luciferase gene-encoding viral supernatant and imaged 7 days postinfection. Spheroids were selected for intracranial implantation (arrowheads) based on the highest in vitro bioluminescence signal obtained 10 minutes following incubation with 1.6 μg/μL D-luciferin.
Quantitative Assessment of Tumor Growth
Tumor growth was noninvasively monitored over time using BLI and MRI. Both imaging modalities facilitated qualitative (Figure 2A) and quantitative (Figure 2, B–E) assessment of tumor progression. A time course assessment of T2-weighted and T1-weighted MRIs post–contrast administration from a representative tumor-bearing animal demonstrated the growth of a luciferase-expressing xenograft tumor.

Growth of intracerebral human glioblastoma multiforme biopsy xenografts assessed using bioluminescence (BLI) and magnetic resonance imaging (MRI). Anatomic localization of the xenografts and tumor volumes were monitored weekly with T2-weighted and T1-weighted MRI sequences post–contrast administration (arrowheads). Luciferase activity was also monitored weekly with BLI. Images show tumor growth in a representative animal using both imaging modalities (A). Logarithmic BLI signal for each individual mouse (n = 7) and logarithmic mean BLI signal ± SEM were plotted over time (B, C, respectively). Logarithmic tumor volume obtained by T2-weighted MRI for each mouse (n = 7) and logarithmic mean tumor volume ± SEM were plotted over time (D, E, respectively).
Corresponding BLIs from the same animal revealed an intense BLI signal (see Figure 2A). Bioluminescence values and tumor volumes (as estimated from T2-weighted MRIs) at each time point were quantified. Both revealed a steady increase over time, concordant with progressive tumor growth (see Figure 2, B–E).
Correlation between Bioluminescence and Corresponding Tumor Volume
T2-weighted MRIs were used to estimate tumor volume. Logarithmic values of tumor volumes at each time point were compared to corresponding logarithmic values of bioluminescence signals for each tumor-bearing mouse. A substantial correlation (Spearman correlation, r = .890, p < .001) between bioluminescence signal and MRI tumor volume (Figure 3) was demonstrated.

Bioluminescence imaging (BLI) signal correlates with magnetic resonance imaging (MRI)-based volumetric calculations. Relationship between the logarithmic BLI signals versus the logarithmic T2-weighted MRI-based tumor volumes as defined by regression analysis. A substantial correlation (Spearman correlation, r = .890, p < .001) was seen.
Bioluminescence Signal of GBM Xenograft Tumors Ex Vivo
The brains from six mice were removed and exposed to luciferin for 10 minutes prior to optical imaging to confirm tumor location and determine bioluminescence signal in the ex vivo setting (Figure 4A). Mouse 5 was not assessed for ex vivo BLI as it required humane euthanization due to disease-related illness. Signal assessed ex vivo was plotted together with the in vivo signal collected immediately prior to sacrificing animals (on week 8) (Figure 4B). Figure 4 demonstrates similarly high sensitivity of BLI in vivo when compared to ex vivo signal.

Ex vivo and in vivo signals are similar in luminescent glioblastoma multiforme (GBM) xenografts. Ex vivo image of a representative mouse brain obtained 8 weeks postimplantation of transduced GBM spheroids (A). Logarithmic bioluminescence signal obtained in vivo and ex vivo for each mouse on the day of sacrifice (n = 6) (B).
Detection of GBM Invading Tumor Cells
Diffuse infiltration into the brain parenchyma was observed in the peripheral tumor areas (Figure 5, A and B, arrowheads). A low-power image of a luciferase-expressing xenograft revealed robust, uniform expression of the luciferase transgene throughout the tumor mass and extensive spread of tumor cells over the corpus callosum (Figure 5C, arrowhead). GBM cells expressing firefly luciferase seen in the corpus callosum (Figure 5, D and E, arrowheads) were also found in the subventricular zone (Figure 5F, arrowhead). Luciferase/human-specific nestin costaining confirmed that these luciferase-positive invading cells originated from the implanted human GBM biopsy spheroids (Figure 5, G and H, arrowheads).
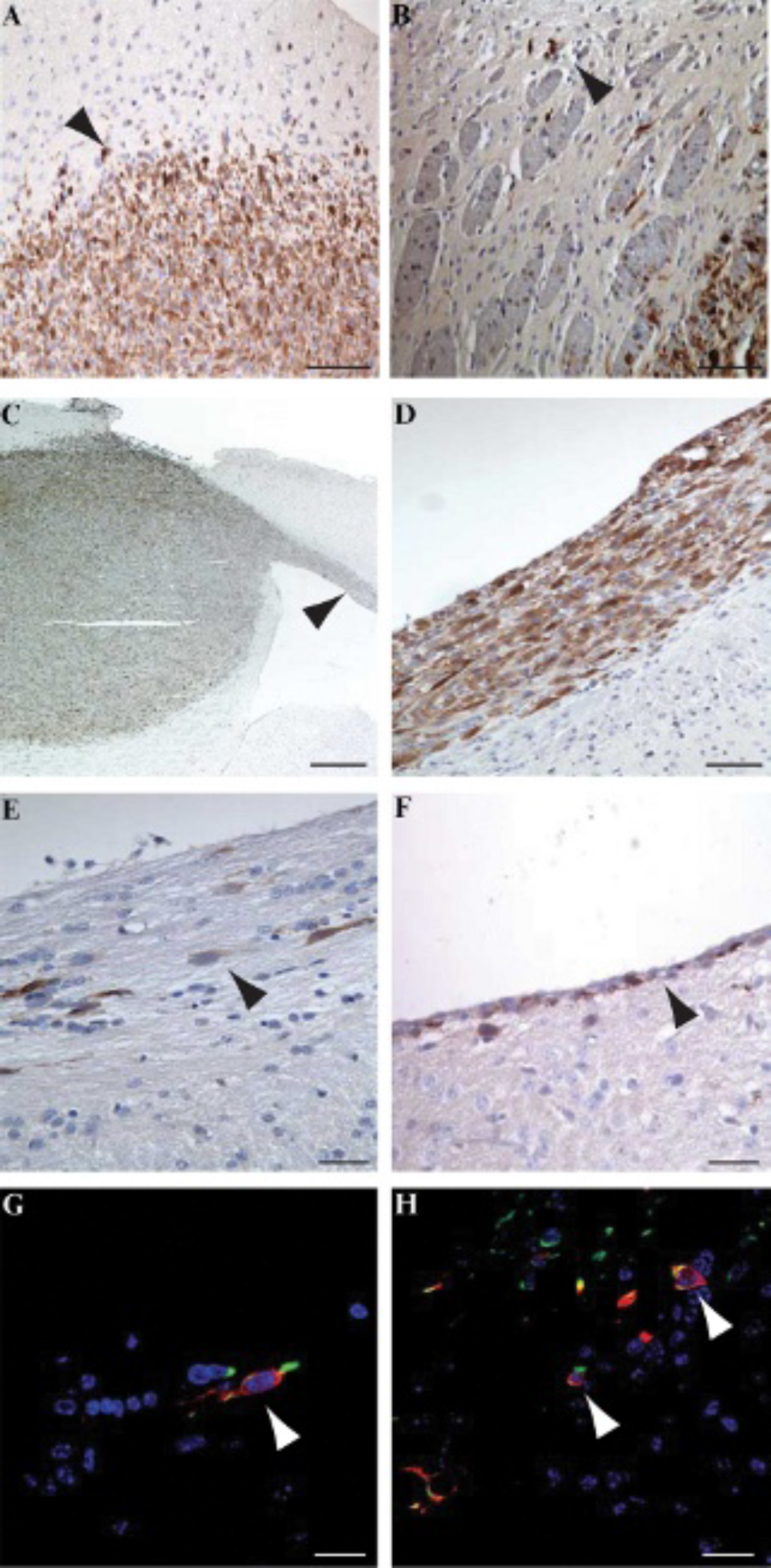
Luciferase-expressing human glioblastoma multiforme cells infiltrate the mouse brain. Locally invasive cells close to the tumor mass (A, arrowhead; B, arrowhead). A low-power view of a luciferase-expressing xenograft shows robust transgene expression throughout the tumor mass (C) and extensive infiltration over the corpus callosum (C, arrowhead). A higher-power view of the corpus callosum (D), contralateral side of the corpus callosum (E, arrowhead), and periventricular cell layer (F, arrowhead) shows luciferase-expressing tumor cells. Invasive tumor cells were detected in ventricles following fluorescent double-staining with human-specific nestin and antiluciferase antibodies (G and H, arrowheads). Scale bars = 1,000 μm (C, X4 magnification), 100 μm (A–B, X20 magnification), 50 μm (E–H, X40 magnification).
H&E Staining
Xenograft lesions generated from luciferase-expressing spheroids (Figure 6, A, C, E, and G) showed the same histologic characteristics as wild-type xenografts (Figure 6 B, D, F, and H), recapitulating the histologic hallmarks of human GBM. Similar to patient GBMs, we observed diffuse infiltrative single-cell spread (see Figure 6, A and B), the presence of large central necrotic foci with palisading (see Figure 6, C and D) and multiple smaller areas of necrosis (see Figure 6, E and F), and abnormally dilated, irregularly shaped vessels (see Figure 6, G and H).

Bioluminescent glioblastoma multiforme biopsy xenografts maintain the histologic hallmarks of grade IV human glioma. Hematoxylin-eosin staining of wild-type xenografts (A, C, E, G) and biolumiescence-active tumors (B, D, F, H) was performed (n = 3 and 5, respectively). Diffuse infiltrative growth (A, B), large areas of necrosis (C, D), multiple small necrotic foci (E, F), and dilated, abnormally shaped vessels (G, H) were identified in wild-type and luciferase-expressing tumors. Scale bars = 250 μm (X10 magnification, A-B), 100 μm (X20 magnification, C–H).
Proliferation Index
GBM xenograft brain sections were stained for human-specific Ki-67 to determine proliferation indices (Figure 7, A and B). In optically active GBM xenografts expressing luciferase, the mean number of Ki-67-positive nuclei was 41.33 ± 1.35 SEM, and in nontransduced GBM xenografts, the number was 36.70 ± 1.71 SEM per X40 field (Figure 7C). There is no significant difference (p = .431), indicating that neither the lentiviral transduction procedure nor luciferase transgene expression significantly halted tumor growth. Also, these values were well in accordance with proliferation index values seen in high-generation xenografts in the biopsy xenograft model, as previously published. 21
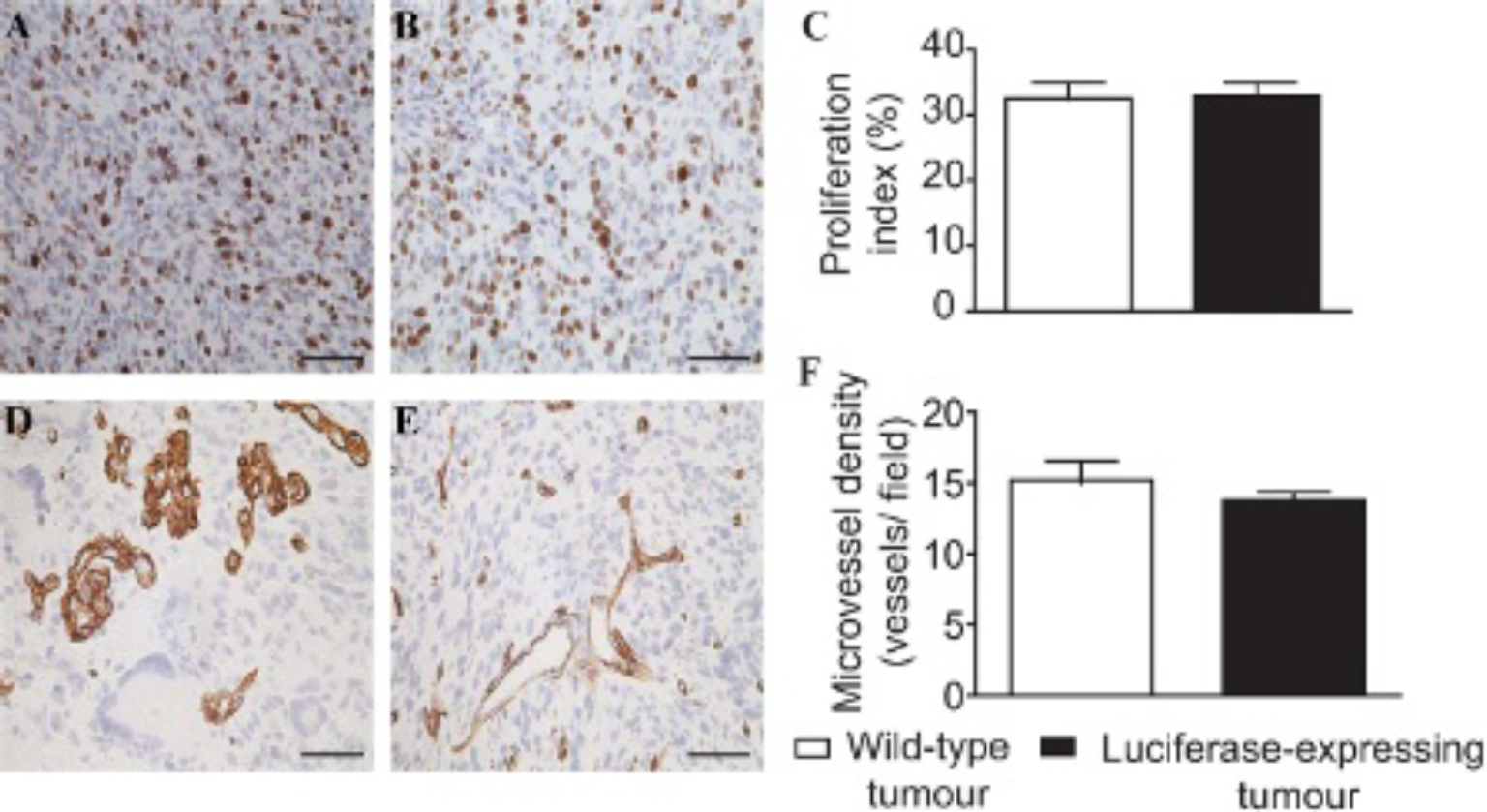
Proliferation rates and tumor microvasculature of bioluminescent xenografts are similar to wild-type xenografts. Ki-67 staining identified proliferating cells in luciferase-expressing (A) and wild-type (B) xenografts (brown staining). Mouse endothelial staining using rodent-specific antinestin antibody was assessed to identify tumor microvasculature of luciferase-expressing (D) and wild-type (E) xenografts (brown staining) (n = 5 and 3, respectively) X40 magnification, scale bars = 50 μm. Proliferation index and microvessel density quantification in the two groups (C, F, respectively). Error bars: mean ± SEM. The mixed-model test was used to calculate p values (p = .431 and .577, respectively).
Microvessel Density
As rodent-specific nestin has been shown to be a robust and reliable marker for brain tumor xenograft vessels in mice and rats,37–39 we stained sections for this marker to evaluate microvessel density (Figure 7, D and E). In optically active GBM xenografts expressing luciferase, the mean microvessel density was 13.82 ± 0.54 SEM, and in xenografts with nontransduced explants, 15.21 ± 1.35 SEM per X40 field (Figure 7F). This difference was not significant (p = .577), confirming that neither the in vitro transduction procedure nor transgene expression affected tumor angiogenesis.
Inflammatory Cells
To evaluate inflammatory cell infiltration into the tumor xenografts, we immunostained sections from both groups for AIF-1, expressed by tumor-associated activated microglia/macrophages. Moreover, it has been shown that the presence of AIF-1-positive cells is strongly correlated with malignancy in astrocytomas. 40 Increased numbers of AIF-1-immunoreactive cells were observed in the tumor mass and in areas of infiltrative tumor growth in both xenografts generated from wild-type spheroids (Figure 8, A, C, and E) and those expressing luciferase (Figure 8, B, D, and F). AIF-1-labeled cells with typical stellate microglial morphology were found at the tumor periphery (see Figure 8, A and B), in the tumor center (see Figure 8, C and D), and in the corpus callosum invaded by glioma cells (see Figure 8, E and F). Although the majority of cells labeled with the AIF-1 antibody showed a stellate microglial morphology (Figure 8G, arrows), tissue macrophages often found close to vessels were also immunopositive (Figure 8H, arrows). We employed the area fractions occupied by microglia and macrophages in both experimental groups to evaluate if expression of firefly luciferase induced an enhanced inflammatory response from the host, as was suggested by previous studies. 41 In the tumor core (Figure 8I) and the peripheral areas of the tumors (Figure 8J), there was a trend toward a higher number of inflammatory cells in luciferase-positive xenografts; however, the difference was not significant when compared to wild-type xenografts (p = .404 and .083, respectively, for tumor core and peripheral areas).

Macrophages and microglia accumulate in both luciferase-expressing and wild-type xenografts. Mouse brain sections were stained for allograft inflammatory factor 1 (AIF-1)-expressing cells. Luciferase-expressing tumors (A, D, G) were populated by AIF-1-expressing cells with similar density as in nontransduced tumors (B, E, H) (n = 5 and 3, respectively). The tumor core (A, B), tumor periphery (D, E), and corpus callosum (G, H) are shown. The majority of AIF-1-immunopositive cells displayed stellate microglial morphology (I, arrowheads); however, rounded, phagocytizing cells with macrophage morphology were also found (J, arrowheads). Scale bars = 200 μm (A, B, D, E, G, H, X20 magnification); I and J, 50 μm (X63 magnification). AIF-1-positive cells were quantified in tumor core (C) and tumor periphery (F) areas. Error bars: mean ± SEM. The mixed-model test was used to calculate p values (p = .404 and .083, respectively, for tumor core and peripheral areas).
Discussion
In this report, we present a clinically relevant, human GBM biopsy–based xenograft model that facilitates identification of growing tumors using a facile high-throughput optical imaging strategy. We found that our second-generation optically active GBM biopsy spheroid xenografts are similar in major aspects (such as growth pattern, proliferation rates, and microvessel density) to the already established and well-described wild-type GBM biopsy spheroid xenografts and thus reflect corresponding patient GBM pathology.15,21
BLI is a well-established method for determining brain tumor growth and response to therapy in a quantitative fashion in orthotopic GBM cell line-based rodent xenografts.27,42–45 In particular, BLI is a well-suited method to monitor tumor growth in intracranial GBM xenograft models where simple caliper measurements are not possible. Critically, the main advantages associated with BLI include scanner ease of operation, ability to evaluate several animals in a single image acquisition, minimal processing requirements, and relatively low equipment and running costs when compared to other imaging modalities (eg, MRI). Implementation of BLI in monitoring tumor take rates facilitates exclusion of animals with small tumors (low BLI signal) and appropriate randomization of animals before commencement of treatment protocols. 46 Importantly, the noninvasive nature of BLI allows acquisition of several images from a single animal, thus increasing the reliability and decreasing number of animals required for preclinical drug studies. Good correlation between bioluminescence- and magnetic resonance-based tumor assessment of drug response rates to novel treatments has previously been shown in several studies involving GBM cell line-based xenograft models.9,47 For example, Rehemtulla and colleagues recently reported that a reduction in tumor burden can be accurately (as confirmed by MRI) assessed using a cost-effective BLI technique in a rat xenograft model implanted intracranially with 9L-Luc gliosarcoma cell line and treated with 1,3-bis(2-chloroethil)-1-nitrosourea (BCNU) chemotherapy. 47 Moreover, the utility of BLI for real-time monitoring of GBM response to novel antiangiogenic treatment approaches has been demonstrated by Szentirmai and colleagues. 9 U87MG-LucNeo mouse xenografts were exposed to systemically delivered adenoviral vectors encoding soluble vascular endothelial growth factor (VEGF) receptors. MRI supported BLI data confirming tumor growth inhibition following treatment. In addition, other interesting information may be acquired from BLI assessment. For example Bryant and colleagues implemented the F98-luc cell line-based rat xenograft model to demonstrate the sensitivity of BLI in the assessment of inherent focal necroses within GBMs during late-stage disease. 48 BLI was well correlated with viable tumor volume. A decrease in bioluminescence signal during late-stage tumor development indicated extensive areas of necrosis within the tumor as confirmed by histologic analyses.
Nevertheless, all of these advantages notwithstanding, cell line-based GBM models, which support the application of BLI in GBM preclinical studies, generally do not reflect fundamental characteristics of human GBM histology, genetics, and gene expression profiles. Therefore, to fully exploit BLI in a clinically relevant GBM rodent model, we developed a new transduction protocol for 3D tissue spheroids. We successfully exposed human biopsy-based GBM spheroids to luciferase gene-encoding lentiviral vectors. In vitro-selected spheroids (see Figure 1) were implanted intracerebrally in immunodeficent NOD/SCID mice. As we have seen, the growth of intracerebral GBM biopsy-based lesions may be detected early postimplantation using BLI (Figure S1, online version only). We used a combined approach using MRI to anatomically evaluate the tumor location and to perform volumetric measurements and BLI to detect tumor engraftment and to follow progression (see Figure 2). Substantial correlation (Spearman correlation, r = .890, p < .001) was seen between the two imaging modalities (see Figure 3). However, when correlating BLI with tumor volume, we have to consider a few limitations of this imaging strategy.
As shown by Jost and colleagues, at early time points following tumor implantation, false bioluminescence signal can be generated by the presence of intraparenchymal hemorrhages and extensive hydrocephalus at the site of injection. 46 A lack of correlation with MRI-determined tumor volumes has also been shown at very late stages of tumor growth, when bioluminescence signal is lost due to the presence of hemorrhagic and necrotic tissue, whereas tumor volumes continue to increase, which is evident on MRI. 46
Therefore, new in vivo GBM models where tumor growth is monitored by BLI should ideally include absolute quantification of tumor volumes, anatomic localization of tumor, and assessment of tumor phenotype and heterogeneity confirmed by MRI (as demonstrated here) or computed tomography (CT). Histologic analysis may also be implemented postmortem. Routine implementation of the following steps may help prevent some of the limitations of BLI in clinically relevant GBM models:
Use of a carefully standardized intracerebral site of implantation
A stable luciferase expression of the spheroid xenograft system
In vivo expansion of the tissue material to maintain histologic traits of the original patient tumor
Generation of a tumor volume/luminescent signal correlation plot for each GBM spheroid–based line developed
Analysis of the exponential phase of tumor growth by bioluminescent signal quantification
Assessment of xenograft volumes
In this way, maintenance of preclinical systems representative of the clinical situation should result.
Within the current study, we performed immunohistochemical analyses to evaluate if efficient transduction of high–generation, biopsy-based GBM spheroids changes tumor histology or tissue phenotype. Proliferation index as assessed by Ki-67 staining indicated that lentiviral transduction of GBM biopsy–based spheroids with luciferase-encoding vectors had no significant effect on tumor proliferative potential (p = .431). Moreover, rodent-specific nestin antibody staining to detect endothelial cells37–39 and microvessel density showed that lentiviral transduction did not have significant effects on tumor angiogenesis (p = .577) (see Figure 7). Luciferase-positive GBM cells were detected in the brain parenchyma, corpus callosum, and subventricular zone, demonstrating the infiltrative nature of this optically active GBM biopsy-based model (see Figure 5).
To investigate whether luciferase expression by the xenografts resulted in increased inflammatory cell numbers, we quantified AIF-1-immunopositive cells (tissue macrophages and microglia) in both groups. However, we detected a trend toward more AIF-1-expressing cells in the tumor core and periphery; these differences were not significant (p = .404 and .083, respectively) (see Figure 8). Although this is a finding to consider, engraftment times and survival figures did not seem to be overtly different between the groups, and we did not experience tumor rejection events. Indeed, a previous study addressed this issue and concluded that firefly luciferase transgene–induced antitumor immunity only plays a role in immunocompetent BALB/c but not in immunodefficient nu/nu mice, 41 in line with our data from NOD/SCID mice.
Taken together, our data demonstrate that in the current model system, lentiviral transduction of biopsy-based GBM spheroids and luciferase transgene expression does not have a significant effect on tumor histology or antitumor immunity.
Conclusion
We have validated a novel, optically active, and clinically relevant model of GBM. This second-generation biopsy-based GBM orthograft model is likely to have future utility in assessing new anti-GBM therapies where tumor cells may be detected early postimplantation using BLI and tumor progression may be assessed in high-throughput manner.
Footnotes
Acknowledgments
We gratefully acknowledge Prof. William Gallagher (University College Dublin) for providing luciferase (pleni6-luc2) lentiviral vector.
Financial disclosure of authors: Funding is acknowledged from the Higher Education Authority Program in Research in Third Level Institution (National Biophotonic Imaging Platform Ireland) and Science Foundation Ireland in the contex of the Moleculor Therapeutic for Cancer Ireland Strategic Research Cluster.
Financial disclosure of reviewers: None reported.
