Abstract
Positron emission tomography (PET) for in vivo monitoring of phosphatidylserine externalization and glucose metabolism can potentially provide early predictors of outcome of cardioprotective therapies after myocardial infarction. We performed serial [68Ga]annexin A5 PET (annexin-PET) and [18F]fluorodeoxyglucose PET (FDG-PET) after myocardial infarction to determine the time of peak phosphatidylserine externalization in relation to impaired glucose metabolism in infracted tissue. Annexin- and FDG-PET recordings were obtained in female (C57BL6/N) mice on days 1 to 4 after ligation of the left anterior descending (LAD) artery. [68Ga]annexin A5 uptake (%ID/g) in the LAD artery territory increased from 1.7 ± 1.1 on day 1 to 5.0 ± 3.3 on day 2 and then declined to 2.0 ± 1.4 on day 3 (p = .047 vs day 2) and 1.6 ± 1.4 on day 4 (p = .014 vs day 2). These results matched apoptosis rates as estimated by autoradiography and fluorescein staining. FDG uptake (%ID/g) declined from 28 ± 14 on day 1 to 14 ± 3.5 on day 4 (p < .0001 vs day 1). Whereas FDG-PET revealed continuous loss of cell viability after permanent LAD artery occlusion, annexin-PET indicated peak phosphatidylserine expression at day 2, which might be the optimal time point for therapy monitoring.
APOPTOTIC CARDIOMYOCYTES have been identified in hypoxic and ischemic tissue following acute myocardial infarction. 1 In recent years, it has been demonstrated that novel therapies, for example, with erythropoietin or parathyroid hormone, can attenuate ischemic cardiomyopathy after myocardial infarction. Rescue of tissue at risk is obtained through enhancement of neovascularization and stem cell migration, as well as through improved cell survival via antiapoptotic effects.2–6 Preclinical testing of such novel therapies in small-animal models can be facilitated through serial noninvasive monitoring of tissue survival. 7 Positron emission tomography (PET) enables serial in vivo molecular imaging of myocardial survival and viability and lends itself to translation into large-animal models or humans using clinical PET/computed tomography (CT). 8 It is well established that [18F]fluorodeoxyglucose (FDG)-PET gives quantitative information about the viability and function of damaged myocardium in vivo. 8 More recently, single-photon emission computed tomography (SPECT) and PET studies have indicated that uptake of radiolabeled annexin A5 can serve as a marker for measuring levels of phosphatidylserine (PS) externalization in the outer plasma membrane in both animal and human tissues.9–11 This is based on the affinity of annexin A5 for the plasma membrane PS, which is presented during apoptosis and, to some degree, during other forms of cell death.10,12 Annexin A5 is an anticoagulant protein that binds with high affinity to externalized PS and forms a two-dimensional molecule network through protein–protein interactions on the surface of cells at risk. 13
PET studies using radiolabeled annexin A5 have the potential to reveal and quantify the extent of PS externalization in myocardial infarction, thus providing a potential early marker to predict the outcome of cardioprotective therapies. However, this application requires more detailed knowledge about the time course of PS externalization, in association with changes in glucose metabolism, as an indicator of myocardial viability. To address this issue, we performed pairs of PET acquisitions with [68Ga]annexin A5 and FDG in mice for 4 days following an irreversible ligation of the left anterior descending (LAD) artery. We sought to identify the time of peak PS levels in comparison with impaired glucose metabolism as the optimal time point for future application of a PET method for monitoring cardioprotective therapies.
Materials and Methods
Animal Model of Coronary Occlusion
Experimental protocols were approved by the regional governmental commission of animal protection (Regierung von Oberbayern, Germany) and were performed in accordance with the Guide for the Care and Use of Laboratory Animals published by the US National Institutes of Health. 14
Female mice (C57BL6/N) weighing 18 to 25 g were used in all experiments. Mice were anesthetized with 1.5% isoflurane delivered in 0.15 L O2/min and were maintained in this condition by mechanical ventilation. Body temperature was monitored continuously with a rectal temperature probe and maintained within an appropriate range using a heating lamp. Left thoracotomy was performed, and the heart was exposed. A small needle was used to place a suture (8-0 Prolene, Ethicon, Somerville, NJ) about the proximal portion of the LAD artery, which was then ligated. Occlusion of the LAD artery inducing a myocardial infarction was verified by visual inspection (paling of the tissue). The chest was then closed, and the animals were taken back to their cages to recover.
In Vivo PET Imaging
Annexin A5 was labeled with 68Ga by applying a kit-like labeling technique, yielding a specific activity of 20 to 45 GBq/mmol, as described recently by our group. 15 [68Ga]Annexin A5 and FDG imaging was performed on a dedicated small-animal PET scanner (Inveon Dedicated PET, Preclinical Solutions, Siemens Healthcare Molecular Imaging, Knoxville, TN) on day 1 (24 hours; n = 9), day 2 (48 hours; n = 5), day 3 (72 hours; n = 10), and day 4 (96 hours; n = 9) after LAD artery occlusion. For scanning, animals were anesthetized with 1.5% isoflurane delivered in 0.15 L O2/min, received an intravenous catheter into the tail vein, and were placed in the aperture of the tomograph. First, 15 MBq [68Ga]annexin A5 was injected in a volume of 150 μL, plus 50 μL saline for flushing the catheter. On injection, a three-dimensional dynamic list-mode acquisition lasting 90 minutes was initiated, followed by a transmission scan with an external 57Co point source for subsequent scatter and attenuation correction. Next, 20 MBq FDG was injected in a volume of 100 μL, plus 50 μL for flushing the catheter. On injection, a list-mode PET acquisition lasting 30 minutes was acquired, after which time, a second attenuation scan was conducted (Figure 1).
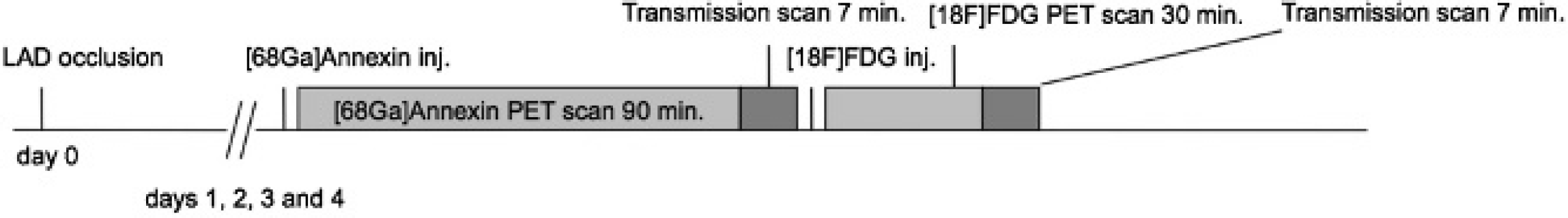
Schematic illustration of the study protocol. LAD = left anterior descending artery.
All data were processed with an Inveon Acquisition Workplace (Siemens Medical Solutions, Knoxville, TN). The [68Ga]annexin A5 list-mode scan was dynamically framed (3 × 600 s, 2 × 1,800 s) and iteratively reconstructed using ordered-subsets expectation maximization (OSEM) 3D (four iterations) and MAP (32 iterations) image reconstruction algorithms. The final images consisted of a 256 × 256 matrix (159 slices, thickness 0.796 mm, resulting in a voxel size of 0.15 μL) with a zoom factor of 100% and a β of 0.15. The FDG data were statically framed and likewise reconstructed with the OSEM 3D (4 iterations) and fastMAP (32 iterations) image reconstruction algorithms. All data were normalized and corrected for random coincidences, dead time, and decay, as well as attenuation.
PET Image Analysis
An Inveon Research Workplace (Siemens Medical Solutions) was used for PET image analyses. [68Ga]Annexin A5 images were automatically fused (automated volumetric fusion algorithm) with the FDG images for anatomic landmarking and proper delineation of the infarcted area; there was no suggestion of body movement between the two acquisitions. Standard volumes of interest (VOI) were then drawn around the region of most intense [68Ga]annexin A5 accumulation. Correct VOI placement was visually verified in axial, coronal, and sagittal projections. The mean radioactivity concentration within the VOI was then quantified as the percentage of the injected dose per gram (%ID/g).
For quantification of the FDG uptake, VOI were placed in the center of the infarcted area. A VOI of the same size was drawn in healthy myocardium contralateral to the infarct. Proper VOI placement was again visually verified in axial, coronal, and sagittal projections, and the mean radioactivity within the ROI was expressed as %ID/g.
Calculation of the Area at Risk
According to a previously described procedure, 16 a volumetric sampling algorithm (MunichHeart Cardiac Analysis Package, Technical University, Munich, Germany) was used to create polar maps of the mouse left ventricular FDG uptake. A standardized ROI was then placed into the center of the septum and set to 100% to normalize the polar map intensities. For the proper delineation of the area at risk, a 60% threshold was set to differentiate between infarcted and noninfarcted myocardium. The size of the area at risk was then expressed as a percentage of the entire left ventricular myocardium area in the polar projection.
[68Ga]Annexin A5 Autoradiography
Autoradiography ex vivo was performed in mice on days 1,2, 3, and 4 after myocardial infarction. After induction of anesthesia as above, 15 MBq of [68Ga]annexin A5 was injected into the tail vein. Mice were killed by cervical dislocation at 90 minutes after tracer injection; the heart was excised, rinsed with cold physiologic saline solution, and then frozen in Tissue-Tek (Embedding Medium for Frozen Tissue Specimens, Sakura Finetek USA, Torrance, CA). Hearts were then cut using a cryostat set at −20°C into serial short axis sections (25 μm thickness each), which were thaw-mounted on glass slides. Within 30 minutes (i.e., 120 minutes after tracer injection), the slides were placed on an autoradiographic imaging plate (Fujifilm MS Imaging Plates in a Fujifilm BAS Cassette 2 2025, Fujifilm Europe, Düsseldorf, Germany) for 24 hours. The exposure was quantified using the Fujifilm FLA-5100 apparatus (Fujifilm Europe).
Specific Binding of [68Ga]Annexin A5
To verify the specific binding, a group of mice (n = 4) was pretreated with 100 μL of unlabeled annexin A5 (corresponding to a mass of 0.03 mg) at 30 minutes before a [68Ga]annexin A5 PET scan, which was conducted on day 2 (48 hours) after LAD artery occlusion and followed by an FDG scan, as above. Image acquisition and evaluation were performed as described above.
Histology
Another group of mice was sacrificed on day 1 (n = 2), day 2 (n = 3), day 3 (n = 2), and day 4 (n = 3) after LAD artery occlusion. The hearts were excised, rinsed with cold physiologic saline solution, and fixed by immersion in formaldehyde (3.7%) at 4°C for a period of 24 hours. The hearts were then embedded in paraffin and cut into 4 mm thick tissue sections, which were then fixated on glass slides and deparaffinized.
For the verification of apoptosis, terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) technique was applied using the In Situ Cell Death Detection Kit (Roche Diagnostics GmbH, Roche Applied Science, Mannheim, Germany) according to the manufacturer's instructions. For counterstaining, VECTASHIELD Mounting Medium (Vector Labs, Burlingame, CA) with 4′,6-diamidino-2-phenylindole (DAPI), a fluorescent stain that binds to adenosine-thymidine-rich regions of deoxyribonucleic acid (DNA), was used according to the manufacturer's instructions. The slides were then analyzed with a fluorescence microscope at a magnification of 40 X, at a wavelength of 515 to 565 nm for apoptotic cells and 450 to 500 nm for all counterstained cells in the field of view (FOV). For each section, cell numbers in five FOVs were counted within the infarction, and the apoptosis ratio was calculated as the number of apoptotic cells divided by the total number of all cells in each FOV.
Statistical Analysis
For modeling the end point PS expression, we performed one-way analysis of variance (ANOVA) using orthogonal polynomial contrasts, which allow modeling nonlinear relationships between the factor time after ischemic injury and PS levels. In the same way, we modeled the end points FDG uptake in the area at risk, size of infarction, and apoptosis ratio in fluorescein staining. For PS expression, we performed a sensitivity analysis, wherein the size of the myocardial infarction was an additional factor in the ANOVA model. The end points were transformed by adequately selected powers to satisfy the normality and homoscedasticity assumptions of the ANOVA model. Graphically based residual analyses were performed to check whether the assumptions of the ANOVA models were reasonably fulfilled. Furthermore, we used the Dunnett t-test for testing results on a reference day against the other 3 days, employing the Bonferroni-Holm procedure to correct for multiple testing. All statistical tests were performed two-sided, and an (adjusted) p value < .05 was considered statistically significant. All statistical analyses were performed with the software package R (version 2.12.1, The R Foundation for Statistical Computing, Vienna, Austria) and SigmaPlot 11.0 (Systat Software, Inc., Chicago, IL).
Results
In Vivo Quantification of FDG Uptake and PS Expression
LAD artery occlusion was successful, and the size of the area at risk could be automatically delineated in all animals as described above. Mean areas at risk (expressed as a percentage of the left ventricular myocardium 6 standard deviation) were 17 ± 5.0% on day 1, 13 ± 4.8% on day 2, 14 ± 4.7% on day 3, and 16 ± 2.9% on day 4.
FDG uptake in the area at risk declined from 28 ± 14 %ID/g on day 1, to 16 ± 5.0 %ID/g on day 2, to 12 ± 5.8 %ID/g on day 3 and remained stable on day 4 with 14 ± 3.5 %ID/g, as depicted in Figure 2A, which corresponds to a significant decline (p < .0001 day 1 vs days 2, 3, and 4). The FDG uptake in the control region on day 1 (57 ± 14 %ID/g) was considerably elevated relative to that in infarct and remained stable for 4 days.

FDG PET in vivo imaging and [68Ga]annexin A5 in vivo imaging. A, Decrease in myocardial FDG uptake over time after occlusion of the murine left anterior descending (LAD) artery. Persistently high myocardial FDG uptake was observed in a reference volume of interest (VOI) in healthy myocardium. B, Peak of phosphatidylserine expression on day 2 after occlusion of the LAD artery. Persistently lower phosphatidylserine expression was observed in a reference VOI in healthy myocardium.
Three-dimensional image fusion of FDG with the [68Ga]annexin A5 emission images was performed to allow proper attribution of the latter tracer to the area of myocardial infarction (Figure 3). [68Ga]Annexin A5 uptake was initially 1.7 ± 1.0 %ID/g on day 1 after ischemic injury, peaked at 5.0 ± 3.3 %ID/g on day 2 (p = .20 vs day 1), and was significantly lower on days 3 (2.0 ± 1.4 %ID/g; p = .047) and 4 (1.6 ± 1.4 %ID/g; p = .01) relative to day 2 (Figure 2B). The annexin A5 uptake in the control region was considerably lower than in the infarcted region (1.2 ± 0.3 %ID/g on day 2).
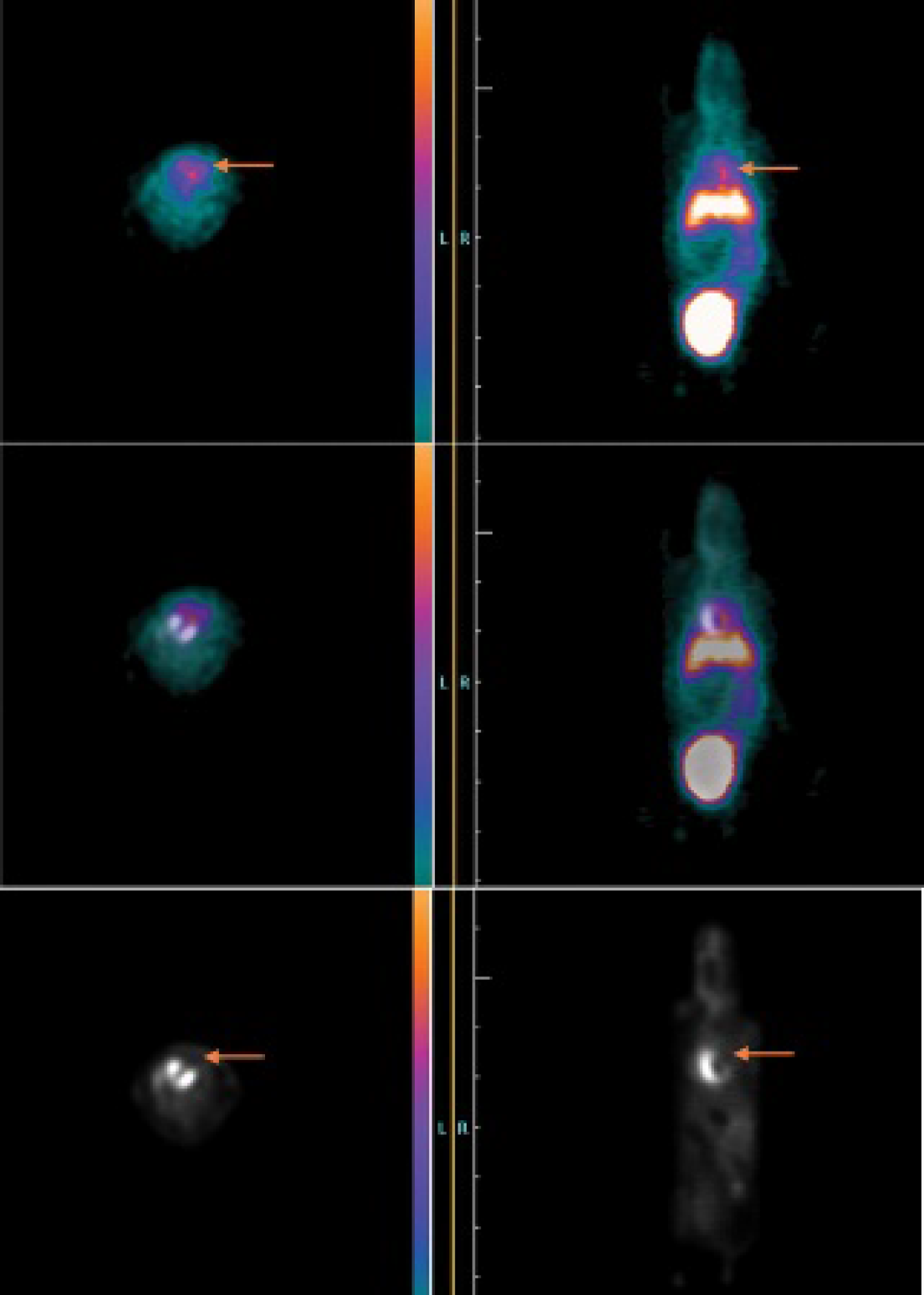
Combined in vivo imaging of apoptosis and viability. The upper image shows axial (left) and coronal (right) slices of the [68Ga]annexin A5 PET scan on day 2 after LAD occlusion. Elevated annexin A5 uptake can be observed in the area at risk, as indicated by the red arrows. Furthermore, high physiologic [68Ga]annexin A5 uptake can be observed in the liver. The lower image shows axial (left) and coronal (right) slices of the FDG-PET scan on day 2 after LAD artery occlusion. Decreased glucose metabolism can be observed in the anterior left ventricular wall and the apex (as indicated by the orange arrows) as a consequence of myocardial infarction. By means of three-dimensional image fusion of the FDG and the [68Ga]annexin A5 PET scan (middle image), [68Ga]annexin A5 uptake can be attributed to the infarcted area.
Histologic Fluorescein Staining
Fluorescein staining gave excellent delineation of the infarction, as shown in representative images for days 1 to 4 (Figure 4). A representative fusion image is shown in Figure 4C. The percentage of apoptotic cells peaked on day 2 (52 ± 2.7%) and declined on days 3 (39 ± 2.1%) and 4 (38 ± 5.2%) (Figure 5), in good accordance with the results of [68Ga]annexin A5 PET imaging, which showed a similar dynamic in the observed time frame (see Figure 2A).

Histologic fluorescein staining. A, Representative field of view enables clear delineation of the infarcted area at an original magnification of 10, where apoptotic cells fluoresce. B, Representative fields of view at an original magnification of 40 × at 1, 2, 3, and 4 days after ischemic injury, where apoptotic cells fluoresce. Peak apoptosis occurred at day 2. C, Fluorescein staining, counterstaining, and fusion of both images for the assessment of the percentage of apoptotic cells in the area at risk.
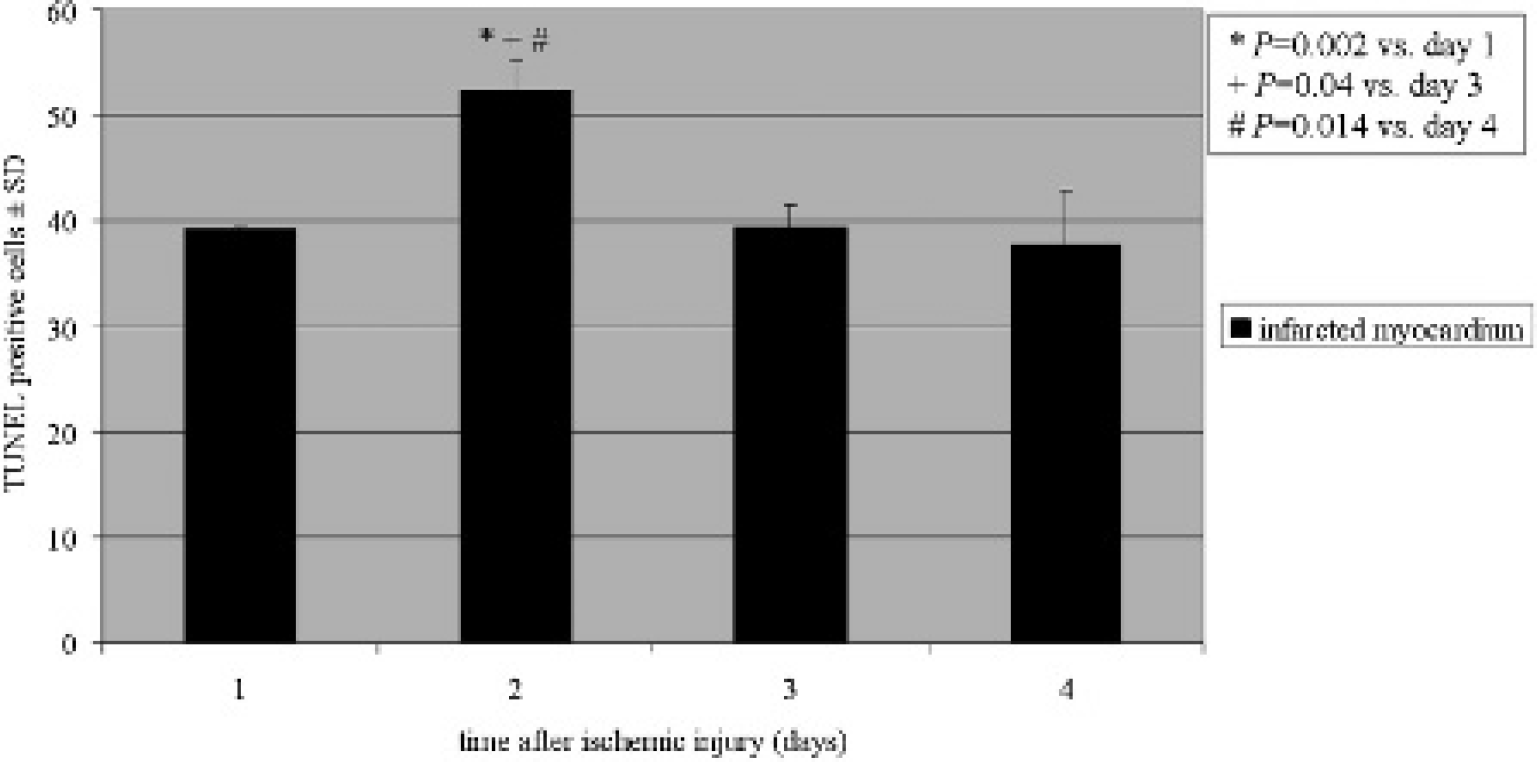
Histologic fluorescein staining. Time course of apoptosis (as measured as fraction of apoptotic cells compared to all cells in the field of view) after occlusion of the murine left anterior descending artery.
Specificity of Tracer Uptake
In the blocking study, pretreatment with 100 μL (corresponding to a mass of 0.03 mg) of nonlabeled annexin A5 30 minutes before tracer injection reduced the mean in vivo [68Ga]annexin A5 binding on day 2 from 3.1 ± 0.4 %ID/g in the unblocked condition (n = 4) to 2.1 ± 0.7 %ID/g in the blocked condition (n = 6; p = .04; Figure 6), indicating the magnitude of specific PS binding in the infarcted region.
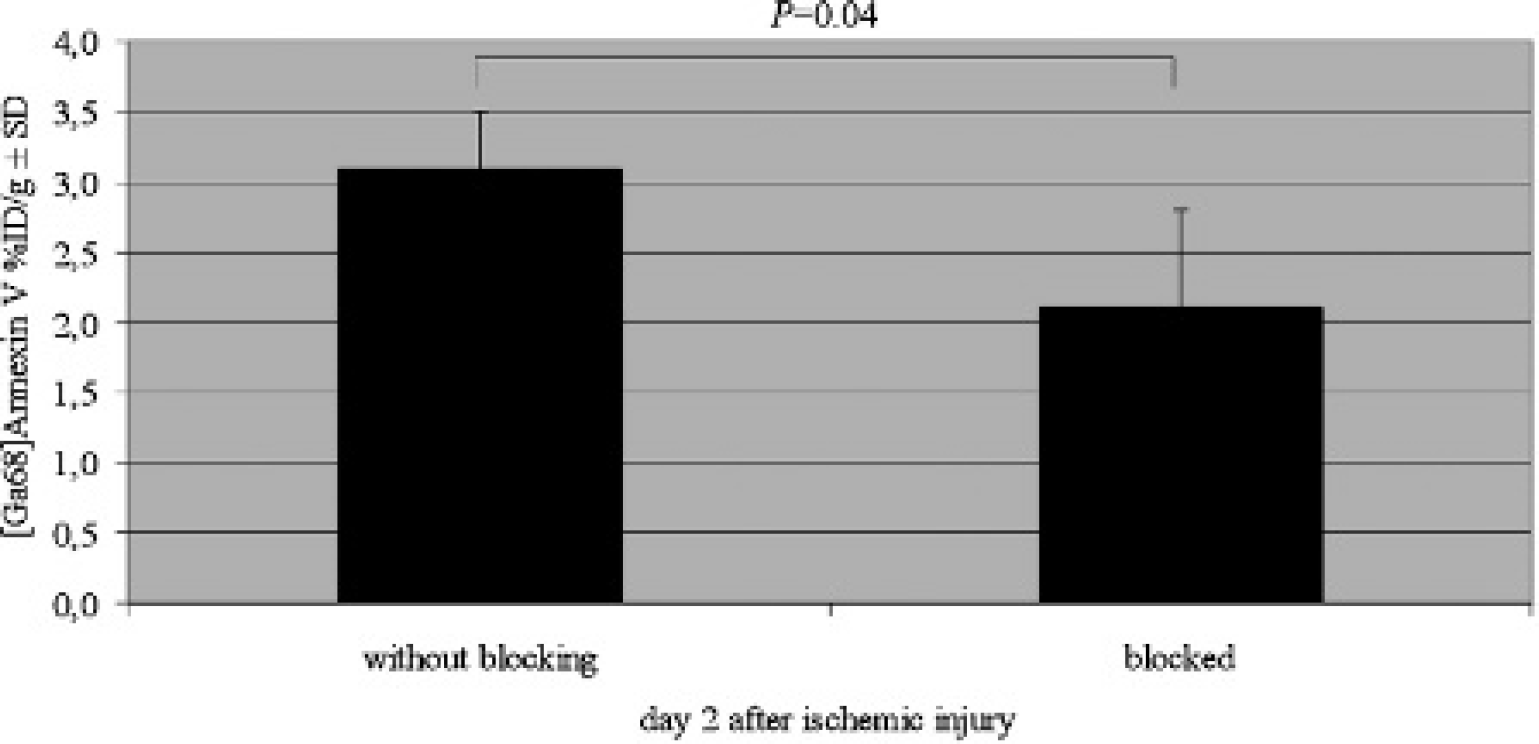
Specific binding of [68Ga] annexin A5. Significantly lower uptake was detected in the group pretreated (blocked) with unlabeled annexin A5 compared to the untreated group (without blocking).
Autoradiography
Representative hearts were prepared and sectioned for autoradiography. At day 1 after ischemic injury, there was only slight [68Ga]annexin uptake, which attained a peak on day 2 and declined on days 3 and 4 (Figure 7). The tracer uptake could be attributed to the infarction in the anterior wall.
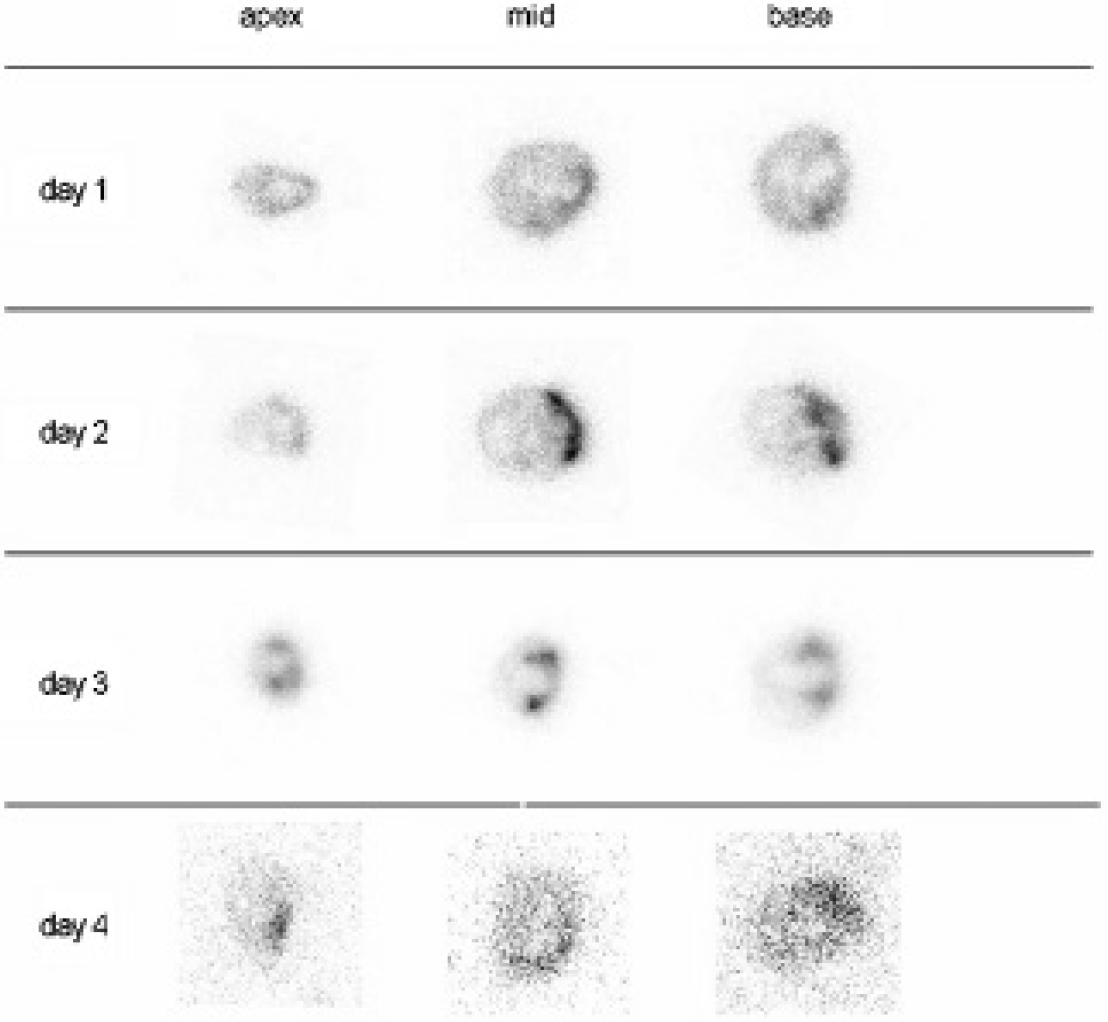
[68Ga]Annexin A5 autoradiography. Selected transaxial sections from the apex to the base at different time points after left anterior descending artery occlusion.
Discussion
Radiolabeled Annexin A5 as a Marker for PS Externalization
The present study was designed as a proof of principle for sequential PET imaging of both myocardial annexin A5 and FDG uptake as surrogate markers for, respectively, PS expression and cell viability in a murine model of myocardial infarction. Furthermore, we aimed to compare the dynamics of annexin A5 and FDG uptake after infarction to determine the optimal time point for in vivo imaging.
PS is translocated to the outer surface of the plasma membrane bilayer) of the cell membrane from its intracellular location via the activation of a nonspecific Ca2+ -dependent lipid scramblase. By means of specific receptor interactions, PS externalization serves as a phagocytosis signal for dying cells.10,17 We found that annexin A5 uptake within the infarcted myocardium peaked on the second day after the infarct, as shown by the concordance of PET and ex vivo autoradiography findings.
Annexin A5 has earlier been evaluated for serial in vivo imaging of apoptosis in SPECT studies with [99mTc]-labeling.18–21 In those studies, PS expression was apparently not restricted to apoptotic cells because annexin A5 binding was also observed in necrotic cell elements. 22 Thus, the peak [68Ga]annexin A5 uptake at day 2 in the present study could indicate the composite of apoptosis and necrosis. However, we found a close correlation between histologic TUNEL staining and [68Ga]annexin A5 PET imaging results. It has been demonstrated that the specificity of TUNEL staining for apoptosis can exceed 87% in certain settings and still reaches 70% when a predominantly necrotic injury is induced, 23 such that we surmise that the preponderance of the annexin A5 signal is due to apoptosis. It has to be noted, however, that other markers may be more specific to apoptosis or necrosis, that is, activated caspase 3 as a marker for apoptosis 24 or several interleukins as markers for inflammation and necrosis. This imperfect specificity presents a possible limitation of our study and should be considered in further studies employing labeled annexin A5 to image cell death. Nonetheless, it has been shown previously that TUNEL staining and antiactivated caspase 3 assays yielded similar results in detecting programmed cell death, 24 albeit in a study using chondrocytes, in a setting not directly comparable to the present myocardial infarct model.
Various cardioprotective therapies, for example, with parathyroid hormone, have been shown to enhance cell survival by attenuating apoptosis. 6 Thus, even though the fraction of apoptotic and necrotic cells cannot be determined in the current study setting, the use of [68Ga]annexin A5 still emerges as a valid marker for preclinical therapy monitoring in murine models of myocardial infarction. The validity of this application is supported by the fact that ischemia-induced PS expression in cardiomyocytes is potentially reversible, thus indicating a return of stressed cells to a healthy state 20 in studies of follow-up design.
In clinical settings, PET often provides several advantages compared to SPECT, such as higher sensitivity, better contrast, and higher spatial resolution,25,26 which need not necessarily hold for modern microSPECT imaging systems. Indeed, in certain settings, the spatial resolution obtained with microSPECT can exceed that which is attainable by microPET, especially in hybrid scanners. 27 However, due to the translational aspect of out research, and in consideration of the future employment of our imaging techniques in clinical settings, we elected to establish our methods in microPET. Furthermore, the short physical half-life of 68Ga, only 68 minutes, is permissive to multitracer PET studies within a single scanning session, as in the present [68Ga]annexin A5 and FDG-PET study. Finally, due to our improved labeling protocol as described above, [68Ga]annexin A5 has become readily available. One possible limitation of [68Ga]annexin A5 arises from its relatively high uptake in the liver, which potentially gives rise to spillover of signal to the adjacent heart in rodent studies. Although a tracer with less liver uptake might present advantages, this potential limitation was obviated in the present study throughout optimization of the spatial resolution of the reconstructed microPET images (≈ 1 mm full width at half maximum), which allowed proper delineation of the myocardial annexin uptake. We demonstrated specific binding of [68Ga]annexin A5 through blocking of the PS binding sites with nonradioactive annexin A5, which significantly lowered the uptake of [68Ga]annexin A5 on day 2 after myocardial infarction. Furthermore, our additional use of autoradiography ex vivo served to attribute the PET tracer uptake to the territory of the occluded LAD. Autoradiography ex vivo has been used in a number of previous studies evaluating radiotracers for PET/SPECT apoptosis imaging, in which good concordance with the results of histology has been reported.18,20,28 In the present study, the [68Ga]annexin A5 uptake ratio by autoradiography ex vivo showed the same temporal dynamics as measured by PET imaging and concurred with the time course of histologic TUNEL staining.
Time Course of Apoptosis after Acute Myocardial Infarction
To our knowledge, there are no previous reports on the time course of PS expression after permanent LAD artery occlusion in mice. Several studies investigated annexin A5 binding in models of myocardial ischemia/reperfusion. For example, Taki and colleagues demonstrated in a rat dual-tracer ex vivo autoradiography study that [99mTc]annexin A5 accumulation was most prominent at 30 to 90 minutes after reperfusion and declined gradually thereafter. 29 Based on this result, Doue and colleagues chose to administer [99mTc]annexin A5 after 20 minutes of left coronary artery occlusion, followed by 30 minutes reperfusion for evaluating antiapoptotic effects after erythropoietin treatment, and elected to assess apoptosis by ex vivo autoradiography. 28 Another study conducted by Dumont and colleagues in 2000 also employed an ischemia/reperfusion model and demonstrated that PS expression increased already after a time interval of ischemia as brief as 15 minutes of ischemia; the authors suggested that annexin might be employed to evaluate cell-death blocking strategies. 30
The ischemia/reperfusion models employed in the above studies constitute an excellent means to mimic the clinical circumstances of myocardial infarction and subsequent revascularization. However, we elected to use a model of permanent ischemia because our predominant aim was to mimic the effects of myocardial infarction and missed or unsuccessful revascularization, which typically leads to left ventricular remodeling and, finally, to ischemic cardiomyopathy.
In our study, we also observed an early increase in tracer uptake on day 1; this is in line with the studies mentioned above, which observed increased tracer uptake on even earlier time points.
Our finding of peak [68Ga]annexin A5 uptake on day 2 after LAD artery occlusion, a time point that was not investigated in the studies described above, might reflect a more prolonged PS externalization and a higher rate of cell death in the absence of reperfusion. However, in contrast to Taki and colleagues′ finding of peak TUNEL-positive cells at 6 to 24 hours after reperfusion, thereafter decreasing until day 3, 29 present in vivo imaging results were in accord with the results of TUNEL staining. Higher apoptosis rates in TUNEL staining corresponded to higher [68Ga]annexin A5 uptake in the area at risk. TUNEL staining is a well-established method to detect apoptosis ex vivo for the purpose of histologic correlation2,18,20,28,31; the results of TUNEL staining corresponded well with annexin A5 uptake in the present study. Our use of TUNEL staining with fluorescein, in contrast to the peroxidase staining used in many studies,2,5,18,20,28 provides superior discrimination between TUNEL-positive and healthy cells due to the high visual contrast between green (apoptotic) and blue (all cells) fluorescence.
Association of Myocardial Apoptosis and Viability
As noted above, the brief half-life of 68Ga was permissive of a dual-tracer protocol entailing FDG-PET in the present study. FDG uptake revealed impaired myocardial glucose metabolism, an indicator of myocardial viability, and was furthermore useful for allocation of [68Ga]annexin A5 uptake to the area at risk through three-dimensional image fusion. Our approach also enabled correlation of apoptosis, as revealed by PS expression, with myocardial viability in the same area. FDG-PET serves for accurate quantitation of infarct sizes in mice after LAD artery occlusion 8 and is indicative of myocardial viability in regions at risk. 32 Our dynamic study revealed that a 50% loss in viability had already occurred on day 1, that is, relative to the reference region in healthy myocardium and an ongoing loss of viability over the course of 4 days after LAD artery occlusion. Relatively low [68Ga]annexin A5 uptake in the presence of high FDG uptake on day 1 was followed by a peak in [68Ga]annexin A5 uptake on day 2, when FDG had further decreased, consistent with an increase in cell death and a fallout of viable cardiomyocytes. Subsequently, both [68Ga]annexin A5 and FDG uptake fall back to low levels as the process of cell demise becomes complete. Given that the peak PS levels are reached on day 2 and that the potential reversibility of PS expression has been demonstrated, 20 annexin A5 might have advantages in predicting the outcome of cardioprotective therapies on the long-term development of ischemic cardiomyopathy compared to FDG, which mainly serves as an indicator for loss of cell viabilty. Although a number of other studies have documented increased annexin A5-uptake as an indicator for apoptosis,18,20,28 the close spatial and temporal relationship of FDG metabolism and annexin A5 accumulation has not previously been evaluated.
Conclusion
In a murine model of myocardial infarction, the time course of both myocardial PS externalization and viability was demonstrated in vivo applying a combined PET imaging protocol with [68Ga]annexin A5 and FDG. We observed an ongoing decline in cell viability until day 4 after permanent LAD artery occlusion, whereas the peak PS expression was identified 2 days after the myocardial infarction, suggesting that this time should be optimal for evaluating cardioprotective therapies. Furthermore, due to the early increase in PS expression in the area at risk and the potential reversibility (compared to FDG uptake) of this process, [68Ga]annexin A5 might be better suited than FDG for the early prediction of the outcome of cardioprotective therapies to prevent ischemic cardiomyopathy in the long term.
Footnotes
Acknowledgments
A substantial part of this work originated from the doctoral thesis of Yordan Vanchev. Furthermore, we would like to thank Dr. Stephan Nekolla, the head of Multimodal Cardiac Imaging at Klinikum Rechts der Isar of the Technical University Munich, for providing the software Munich Heart.
Financial disclosure of authors: Parts of this work were supported by research grants from the Fritz-Bender Foundation and the Else Kroner-Fresenius Foundation.
Financial disclosure of reviewers: None reported.
