Abstract
To enrich our understanding of the mechanism of tumor lymphatic metastasis, we developed a model system for tracking metastatic tumor cells in the lymphatic system with cellular magnetic resonance imaging (MRI) in live mice to observe the interaction between tumor cells and the lymphatic system. Nude mice were inoculated subcutaneously with superparamagnetic iron oxide (SPIO)-labeled and unlabeled LOVO cells in the foot pad, groin, or axillary area. Serial 7 T MRI of the tumors and surrounding regions was performed in the following 2 weeks. After imaging, tumor tissues and regional lymph nodes were collected and subjected to immunohistologic analysis. T2/T2*-weighted MRIs showed the primary tumor growth and the draining lymphatic architecture, as well as the SPIO-labeled tumor cells metastasized into the regional lymph node at 8 days. MRIs also revealed information on sentinel lymph node mapping with high-resolution anatomic information. Histologic findings confirmed the in vivo MRI results and revealed lymphangiogenesis, angiogenesis, infiltration of macrophages, and expression of vascular endothelial growth factor C in tumor and draining lymph nodes as well. This technology provides a powerful tool for tracking SPIO-labeled cancer cells in the lymphatics by cellular MRI. There was a close relationship between tumor lymphatic metastasis and lymphangiogenesis.
COLORECTAL CANCER (CRC) remains a major health problem worldwide because of its high prevalence, as well as its morbidity and high mortality. 1 Lymph node metastasis is the hallmark of CRC progression, is considered one of the most important determinants of treatment, and predicts prognosis in patients.2,3 Tumor cells dissociate from the tumor mass, invade into the lymphatic channel, travel in the lymphatic system, and then go on to form secondary tumors within the lymph node or spread to distant organs. 4 Thus, a thorough understanding of how invading tumor cells interact with the host lymphatics is critical to dig into the fundamental mechanical aspects of tumor lymphatic metastasis. A promising approach to the further understanding of interaction between invading tumor cells and host lymphatics is to directly visualize tumor cells and the lymphatic system simultaneously in vivo.
Several methods for localizing sentinel lymph nodes (SLNs) are currently in use: peritumoral injections of isosulfan blue dye or a radionuclide-labeled sulfur or albumin colloid.5,6 These methods have limitations. The dye method requires dissection of tissue until the blue-dyed SLN is detected7,8; the radionuclide method is limited by low temporal and spatial resolution. 5 Most importantly, the main lymphatic channels draining the tumor cannot be imaged by lymphoscintigraphy. 5 Lymphography, which has been predominantly limited to the use of blue dye 7 and fluorescently labeled dextrans, 9 can transiently mark lymphatic architecture but fails to provide a durable strategy for in vivo lymphatic visualization. And the blue dye and fluorescently labeled dextrans identify the metastatic tumor foci based on the lack of contrast agent filling into lymph nodes where nodal regions are occupied by metastatic lesions. These technologies cannot indentify the presence of tumor cells directly. Engineered tumor cells, which stably express an optical reporter gene such as firefly luciferase or green fluorenscent protein (GFP), enable repetitive detection of primary tumors and disseminated lesion in small animals. 10 However, due to light absorption and scattering by tissues, this method does not allow three-dimensional localization of the signal. Therefore, there is an urgent need to develop a noninvasive imaging modality to track metastatic tumor cells in lymphatics with high-resolution anatomic information while allowing repeated analysis at multiple time points.
Magnetic resonance imaging (MRI) provides excellent soft tissue contrast with high spatial resolution and unlimited depth penetration and allows for three-dimensional whole-body or organ imaging and presentation of data in any desired anatomic plane.11–13 Thus, MRI offers the potential for clearer delineation of both tumor draining lymph nodes and lymphatic vessels with high-resolution anatomic information. The development of contrast agents such as particles of superparamagnetic iron oxide (SPIO) extended the use of MRI to cellular imaging.14–16 SPIO with an extremely high T2 relaxation rate at low fields appears as hypointense spots on a bright background on T2/T2*-weighted MRIs, which facilitates labeled cell detection and localization. 17 Recently, it has been demonstrated that clustering of multiple SPIO nanoparticles with amphiphilic polymers can greatly improve T2 relaxivity,15,18 and low-molecular-weight amphiphilic polyethylenimine (PEI)-2k-covered SPIO nanoparticles can effectively label cells with good biocompatibility. 15 Cellular MRI can noninvasively acquire information on temporal changes of SPIO-labeled cell location for longitudinal studies linked to high-resolution anatomic information by excellent contrast against the background of host tissue. But, to date, cellular MRI has not been used for tracking tumor cell in lymphatics.
Recent studies have suggested that lymphangiogenesis actively contributes to tumor metastasis, and elevated expression of vascular endothelial growth factor C (VEGF-C) plays a central role in lymphangiogenesis.2,19 Also, tumor-associated macrophages promote tumor invasion, growth, metastasis, and angiogenesis. In this study, we tracked subcutaneous SPIO-labeled tumor cells in mice by cellular MRI and studied lymphangiogenesis and VEGF-C expression as well as infiltration of macrophages at an early stage of tumor metastasis. We hoped that tracking SPIO-labeled tumor cells in the lymphatic architecture would provide information on the interaction between invading tumor cells and the host lymphatic system and that investigating lymphangiogenesis and the expression level of the lymphangiogenesis factors VEGF-C in tumor and micrometastases at an early stage of tumor metastasis would enrich our understanding of the mechanism of tumor lymphatic metastases.
Materials and Methods
Cells
Human CRC LOVO cells (a kind gift from the Department of Oncology, West China Hospital, Sichuan University) were cultured in Dulbecco's Modified Eagle's Medium (DMEM) supplemented with 5% heat-inactivated fetal bovine serum at 37°C with 5% CO2.
Synthesis of Amphiphilic PEI/SPIO Nanocomposites
Synthesis and characterization of SPIO nanoparticles, magnetization and T2 relaxivity studies, and in vitro cytotoxicity testing were done as previously described.15,20 Briefly, 6 nm SPIO nanoparticles were synthesized using iron (III) acetylacetonate as precursors in benzyl ether at high temperatures. The crude products were purified through ethanol wash and dispersed in hexane. Amphiphilic PEI was synthesized through alkylation of branched PEI-2k with 1-iodododecane. Amphiphilic PEI/SPIO nanocomposites were obtained by transferring SPIO nanoparticles into the aqueous phase with the help of amphiphilic PEI.
SPIO Nanocomposite Labeling and Prussian Blue Staining
LOVO cells at the midexponential dividing stage with an 80% confluence were incubated for 4 hours in culture medium that included SPIO nanocomposites that were diluted with cell culture medium to a final concentration of 7 μg(Fe)/mL. 18
For Prussian blue staining, the cells were cultivated for 24 hours in 24-well plates on glass coverslips. Cells were washed thrice with phosphate-buffered saline (0.01 mol/L, pH 7.4) and subsequently fixed with prechilled methanol and acetone at − 20°C for 5 minutes. For Prussian blue staining, the fixed cells were incubated with 10% potassium ferrocyanide for 5 minutes and 10% potassium ferrocyanide in 20% hydrochloric acid for 30 minutes and counterstained with nuclear fast red. FE-Pro labeling efficiency was determined by manual counting of Prussian blue–stained and unstained cells using a microscope. The images were processed by Adobe Photoshop 7.0 (San Jose, CA). Cells were considered Prussian blue positive if cytoplasm was blue or brown (DAB enhanced) stained. The percentage of labeled cells was determined from the average of 5 to 10 high-powered fields. 21
Cell Metabolism Assay In Vitro
The effect of SPIO labeling on the cell viability was tested by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) methods as described. 15 LOVO cells were plated into 96-well plates at a density of 5 × 103/well and allowed to adhere overnight before adding serial diluted SPIO nanoparticles. After 4 hours, MTT reagent (Sigma) was added at an amount of 0.5 mg per well and incubated for another 4 hours. The medium was discarded, and 200 mL dimethyl sulfoxide (DMSO) was added for each well. Absorbance of samples was measured, including blanks, at 570 nm in a Microplate Reader (model 550, BIO-RAD, Hercules, CA). A linear relationship between cell viability and absorbance could be established.
Animal Model
Thirty-six immunodeficient male BALB/c nu/nu mice (5–7 weeks old) were used to establish the lymphatic metastasis model. The animals were purchased from Animal Center of West China, Sichuan University, and all the animal experiments were performed in accordance with the policies of the Animal Ethics Committee of the university.
For experiments in vivo, six animals were used in each group; the six groups were animals bearing tumors in the right hindlimb foot pad, right groin area, or right axillary area injected with SPIO-labeled LOVO cells or unlabeled LOVO cells. To establish the lymphatic metastasis model, nude mice were anesthetized with chloral hydrate injection for all procedures. The right hindlimb foot pad, right groin area, or right axillary area was sterilized with alcohol and slowly subcutaneously injected with 0.1 mL of labeled or unlabeled LOVO cell suspension at a concentration of 3 × 107 cells/mL over a 2-minute period. Then 7 T MRI was performed to monitor the inoculation areas and surrounding regions in the following 2 weeks. Animals were sacrificed by CO2 inhalation when labeled tumor cells metastasized into regional lymph nodes. Then tumor tissues and regional lymph nodes were collected and subjected to immunohistologic analysis. The other mice were continually monitored by 7 T MRI until SPIO-labeled tumor cells could not be detected. For each condition, there were three animals in each group.
In Vivo MRI Protocol
Nude mice were imaged with a Bruker BioSpec 7.0 T magnetic resonance microimaging system using a Bruker rat brain coil located over the tumor and regional lymph nodes after inoculation. T2- and T2*-weighted imaging were performed daily postinjection (PI) before the micrometastasis was detected. The other mice were continually monitored by 7 T MRI until SPIO-labeled tumor cells could not be detected. Mice were anesthetized with 1.5 to 2% isoflurane by nosecone, and their vital signs were monitored in a real-time manner. MRI was performed using a TurboRARE T2 sequence (repetition time/echo time [TR/TE] = 3,000/45 ms, number of excitations [NEX] = 1; 256 × 256 matrix; 27 × 27 × 27 mm3 field of view [FOV]) and an MGE-T2* sequence (TR/TE = 500/5 ms, NEX = 8; 384 × 384 matrix; 67 × 67 × 500 μm resolution).
Immunohistochemisry and Prussian Blue Staining
Based on the MRI findings, we collected tumors and regional lymph nodes in which tumor micrometastasis were detected. All the tissue samples were fixed, embedded in paraffin, and then subjected to immunohistologic analysis. Consecutive slides were subjected to hematoxylin and eosin, Prussian blue, CD31, lymphatic vessel endothelial hyaluronan receptor (LYVE-1), VEGF-C, cell keratin 20 (CK20), or CD68 staining.
Statistical Analysis
All data are expressed as mean ± SE. Analysis for significant differences was performed using the Student t-test or one-way analysis of variance. A p value < .05 was considered significant.
Results
Amphiphilic PEI/SPIO Nanocomposites and SPIO-Labeled LOVO Cell Viability In Vitro
The SPIO nanocomposites were about 60 to 70 nm (peak) in diameter and were strongly positively charged with zeta potentials in the range of 50 to 65 mV, with a high T2 relaxivity of 345 Fe mM−1 s−1 (3 T).20,22 The accumulation of the SPIO particles in LOVO cells was rapid, and labeling efficiency was 90%. MTT assay results showed that SPIO-labeled cells have similar metabolic activities compared to unlabeled cells (Table 1).
Cell Viability and Labeling Efficiency

Activity values are mean ± SD; n = 4.
MRI of Tumor Formation
After subcutaneous inoculation with labeled or unlabeled LOVO cells in the right hindlimb foot pad, right groin area, or right axillary area, T2/T2*-weighted MRI was performed daily to monitor tumor formation. MRI demonstrated hypointense regions where SPIO-labeled tumor cells were located, and then the low signal increased gradually and sped up from 5 days PI (Figure 1 and Figure 2). The increase in signal of the peripheral parts was more rapid than that of central parts of the tumor with tumor growth; therefore, the tumors were unable to be distinguished from surrounding tissues from 14 days PI (see Figure 2). The time of tumor formation in different regions and different groups was similar.

A, Magnetic resonance images (MRIs) of labeled and unlabeled tumor cell implantation in the foot pad for 7 days PI demonstrating hypointense regions (white arrows) where SPIO-labeled tumor cells were located. There were no hypointense regions in the primary tumors in the control group. B, MRIs of regional lymph nodes (small white arrows) and the tumor (big white arrow) in the foot pad inoculation group at 12 days PI.
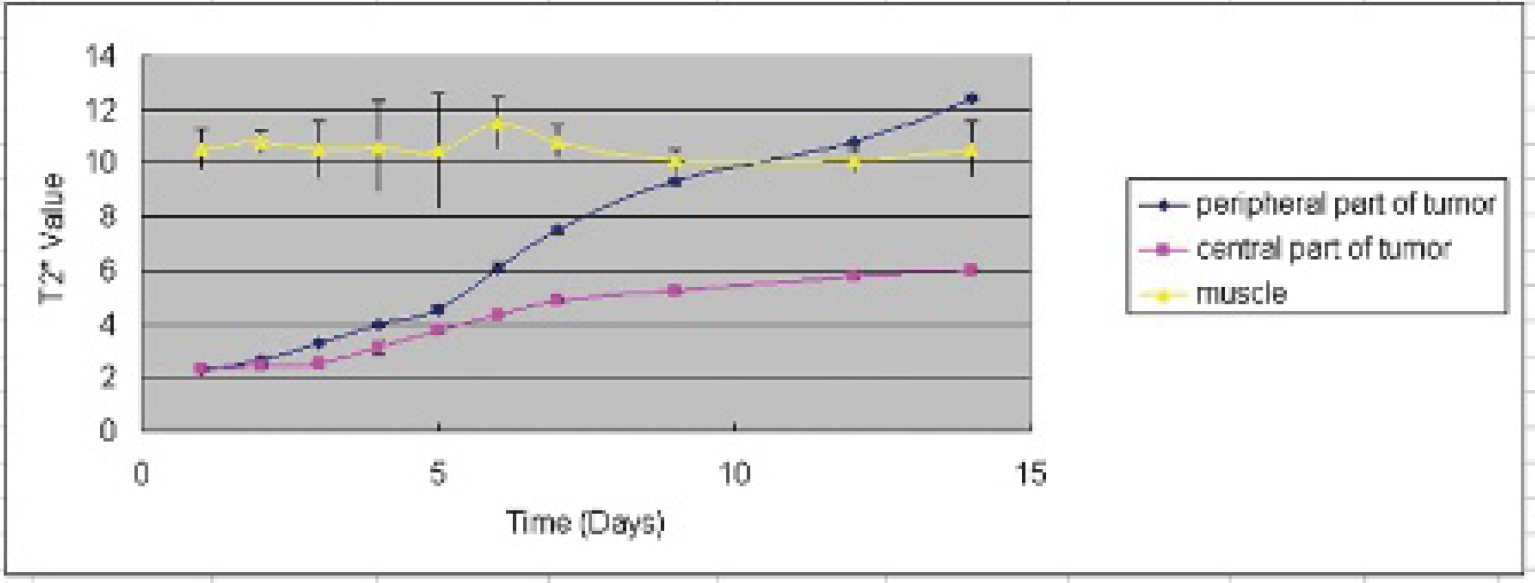
T2* values of tumors in the study group 2 weeks PI.
Tracking Tumor Cells in Draining Lymphatic System
Every day after PI, 7 T MRI of the tumors and surrounding regions was performed. On day 8, lymphatic network structures (bright) draining from the primary tumor were identified, and metastatic tumor cells (dark) could be imaged in draining lymph nodes simultaneously on T2- and T2*-weighted MRIs (Figure 3 and Figure 4). The excellent soft tissue contrast with high spatial resolution of MRI allowed accurate mapping of the SLN, visualizing the directly connected lymphatic vessel between the tumor and the draining SLN and clear delineation of the afferent and efferent lymphatic vessels of the receiving SLN, as well as detection of metastatic tumor cells in the lymphatic system (see Figure 3 and Figure 4) The size of the lymphatic vessel was 0.2 ± 0.02 mm (see Figure 3). The size of the regional lymph node was 1.4 ± 0.1 mm in diameter with nearly 0.4 ± 0.8 mm2 micrometastasis. At 12 days, the number of enlarged lymph nodes found in subcutaneous area of the abdomen, chest, or back and surrounding regions of the tumor on MRI in the foot pad inoculation group, the groin area inoculation group, and the axillary area inoculation group was 5 ± 1 (n = 3), 5 ± 1 (n = 3), and 6 ± 1 (n = 3), respectively (Figure 1, Figure 5, and Figure 6). The size of the smallest lymph node was 0.8 × 1.2 mm2 (see Figure 5). There was no significant difference in the time of tumor lymph node metastasis in different groups.

Visualization of sentinel lymph node draining from the primary tumor by MRI and immunohistochemistry and Prussian blue staining at 8 days PI in the groin area inoculation group. A, CK20 and Prussian blue staining of the primary tumor. CK20 staining (× 40 original magnification) shows generalized expression in the tumor (brown cells). Prussian blue staining (×40 original magnification) shows the positive tumor cells (arrows) (hematoxylin-eosin [H&E] staining; ×10 original magnification). B, T2- and T2*-weighted images of sentinel lymph node (small white arrow) draining from the primary tumor (white circle). The large white arrow indicates the connected lymphatic vessel between the tumor and the draining sentinel lymph node. The size of micrometastasis (black arrow) was 0.4 × 0.8 mm2 in the sentinel lymph node with a 1.5 mm diameter. C, CK20 and Prussian blue staining of sentinel lymph node. CK20 staining (×10, ×40 original magnification) shows that tumor cells were located mainly in the central parts of the lymph nodes (arrows). Prussian blue staining (×10, ×40 original magnification) shows the positive tumor cells in central parts of the lymph nodes (arrows).

Visualization of the draining lymphatic system by MRI and Prussian blue staining in the groin area inoculation group. A to F, Serial contiguous T2-weighted images of tumor draining the lymphatic system. MRI allowed clear delineation of the afferent (large white arrows) and efferent (white arrowheads) lymphatic vessels of lymph nodes (small white arrows) in which micrometastasis (black arrows) was detected at 8 days PI The size of the lymphatic vessel is 0. 2 mm. T2*-weighted (G) and T2-weighted (C) images were in the same anatomic plane, and T2*-weighted (H) and T2-weighted (F) images were in the same anatomic plane. Prussian blue staining (× 10 original magnification) confirms the in vivo T2*-weighted image (G, H) results and shows positive cells in central parts of the lymph nodes.

The size of the smallest lymph node (arrow) was 0.8 × 1.2 mm2 on MRI. CK20 staining shows that a few tumor cells were in the central parts of lymph nodes (arrows). LYVE-1 staining shows lymphangiogenesis in lymph nodes (arrows). VEGF-C staining show the low expression level of VEGF-C in the lymph node. Scale bars = 50 μm.

Magnetic resonance images of tumor implantation in the axillary area at 12 days PI showing the primary tumor (white arrow, A) and the afferent (large white arrows, C, D) and efferent (white arrowheads, D) lymphatic vessels of lymph nodes (small white arrows, B, C, D).
Immunohistochemical Staining
Animals were sacrificed by CO2 inhalation when labeled tumor cells metastasized into regional lymph nodes. Then tumor tissues and regional lymph nodes were harvested and subjected to immunohistologic analysis according to MRI. Prussian blue staining was positive in central parts of the draining lymph nodes and tumors, and there were lymphangiogenesis, angiogenesis, infiltration of macrophage, and expression of VEGF-C in tumor and SLNs as well (see Figure 3, Figure 5, and Figure 7). A large number of CK20-positive tumor cells were located mainly in the central parts of lymph nodes. In one lymph node, only a few tumor cells were detected, and the VEGF-C level was low (see Figure 5).

A, Immunohistologic staining of tumor tissues. LYVE-1 staining shows lymphangiogenesis in the tumor (arrows). CD31 staining shows angiogenesis in the tumor (arrows). CD68 staining shows infiltration of macrophage in the tumor (arrows). VEGF-C staining shows generalized expression of VEGF-C in the tumor (brown). B, Immunohistologic staining of tumor tissues and draining lymph nodes. LYVE-1 staining shows lymphangiogenesis in lymph nodes (arrows). CD31 staining shows angiogenesis in lymph nodes (arrows). CD68 staining shows infiltration of macrophages in lymph nodes (arrows). VEGF-C staining shows generalized expression of VEGF-C in lymph nodes (brown). Scale bars = 50 μm.
Discussion
Lymphatic metastasis is responsible directly or indirectly for the death of patients with CRC. It is one of the most important determinants of CRC treatment and predicts prognosis in patients. 1 Recent results suggest that lymphangiogenesis actively contributes to CRC lymphatic metastasis. 2 Moreover, some studies revealed that expression of lymphangiogenic growth factors leads to formation of lymphatic vessels and results in enhancing lymphatic metastasis. 2 Therefore, it is critical to understand the interaction between invading tumor cells and the host lymphatic system for insights into the fundamental mechanical aspects of CRC lymphatic metastasis. In particular, dynamic evaluation of tumor cells trafficking in the lymphatic system will provide the fundamental information on CRC lymphatic metastasis.
Current lymphatic system imaging modalities, such as lymphography and lymphoscintigraphy, are invasive and cannot allow repeated analysis at multiple time points. In particular, lymphography relies on the lymphatic uptake of moleculars such as blue dye, and fluorescein isothiocyanate (FITC)-dextran can only transiently mark the lymphatic system and fails to provide a durable strategy for in vivo lymphatic visualization.7,9 Lymphoscintigraphy, which can localize SLNs, has low temporal and spatial resolution and fails to adequately image the direct connection of lymphatic vessels to the associated nodes. 5 MRI offers excellent soft tissue contrast with high spatial resolution and unlimited depth penetration to allow for lymphangiography in live mice, 11 which are impossible or extremely difficult to achieve using other imaging methods. In our study, the lymphatic network structure (bright) draining from the primary tumor was clearly identified, and the afferent and efferent lymphatic vessels of the receiving SLN could be clearly delineated and SLNs could be accurately mapped on T2/T2*-weighted MRIs after 8 days PI. This imaging system provides uncompetitive spatial resolution and is sensitive and accurate enough to visualize a lymphatic vessel that is 0.2 ± 0.02 mm in diameter and an SLN that is 1.4 ± 0.1 mm in diameter. Most importantly, visualization of the draining lymphatic system is durable, noninvasive, and linked to high-resolution anatomic information.
The method for cell tracking involves the genetic reporter approach, which is particularly useful for the study of cells at microscopic resolution; however, currently, it does not allow adequate imaging in larger FOVs (eg, at mesoscopic and macroscopic scales) or at increased depths (eg, > 600 mm).23,24 The iron particle labeling method could noninvasively acquire information on temporal changes of SPIO-labeled cell location for longitudinal studies linked to high-resolution anatomic information by excellent contrast against the background of host tissue on MRI, which can be identified in vivo as hypointensities in MRIs as the iron shortens transverse proton TRs. 25 In particular, the spatial and temporal resolution of MRI allows the location of iron-labeled donor cells to be monitored noninvasively over several weeks in vivo. 26 It is important that the labeling procedure allows in vivo detection of a small number of cells and that the incorporation of contrast agents does not affect cell function. 26 In this study, we monitored the fate of SPIO-labeled tumor cells in animals after subcutaneous inoculation and tracked the SPIO-labeled tumor cells′ migration and proliferation in the lymphatic system in live mice by cellular MRI. We observed that tumor cells migrated to the first lymph node that received lymphatic drainage from the primary tumor, survived at the lymph node, and metastasized to the next grade of lymph node as the SPIO-labeled tumor cells appeared as hypointense spots on T2/T2*-weighted MRIs, whereas the draining lymphatic system appeared as hyperintense architecture. More importantly, it provided the opportunity to noninvasively track the tumor cell in the lymphatics and at the same time to clearly visualize the whole draining lymphatic network where the tumor cell transport in the lymphatic channels between the primary tumor and the first lymph node and the afferent and efferent lymphatic vessels of upgraded lymph nodes. Most importantly, this method allows for presentation of data about metastatic tumor cells in lymphatics in any desired anatomic plane, which can detect all lymph node metastasis at one time point.
Solid tumor passes through the avascular growth phase, followed by angiogenesis, local tissue invasion, and metastasis. 27 In this study, after subcutaneous inoculation with labeled LOVO cells in the right hindlimb foot pad, right groin area, or right axillary area, T2/T2*-weighted MRI was performed daily to monitor tumor formation. We found hypointense regions where SPIO-labeled tumor cells were located; then the low signal increased gradually and sped up from 5 days PI. The increase in signal of the peripheral parts was more rapid than that of central parts of the tumor; therefore, the tumors were unable to be distinguished from the surrounding tissues from 14 days PI. As the labeled cells proliferate, the constant number of SPIO nanoparticles is merely divided among the daughter cells. Following many cell divisions, the proportion of the cells containing SPIO nanoparticles might become much fewer. 28 The peripheral parts of tumor cells proliferated more quickly than that of central parts of the tumor because tumor growth depends on the availability of nutrient replenishment from adjacent tissues in the avascular growth phase before 5 days PI. Immunohistologic analysis also confirmed angiogenesis in tumors and draining lymph nodes at 8 days PI.
The traditional theory was that tumor cells metastasized to lymph nodes by using preexisting lymphatic vessels; however, recent studies have suggested that lymphangiogenesis actively contributes to tumor metastasis. 2 In particular, tumor cells can also induce lymph node lymphangiogenesis—even before they metastasize—and that metastatic tumor cells continue to induce lymphatic vessel growth within SLNs, theoretically promoting their further metastatic dissemination. Recent evidence indicates that VEGF-C induced lymphangiogenesis-mediated tumor spread and the formation of metastatic disease in the lymph nodes.2,29 Our histology results showed lymphangiogenesis, infiltration of macrophages, and expression of VEGF-C in the primary tumor and regional lymph nodes at 8 days PI, which demonstrates a close relationship between lymphangiogenesis and tumor lymphatic metastasis at an early stage of tumor lymphatic metastasis. Moreover, we found that in one lymph node, there were only a few tumor cells and the VEGF-C level was low. This demonstrates that VEGF-C expression may not be the only factor that drives tumor-induced lymphangiogenesis in lymph nodes.
A limitation of using cellular MRI for tracking tumor cells in the lymphatic system is that the T2* sequence requires 10 minutes or more, so we were not able to see tumor cell migration in the lymphatic vessel in a real-time manner. We will combine MRI with other imaging modalities, such as positron emission tomography or optical imaging, to overcome the current limitation for monitoring trafficking of tumor cells in the lymphatic vessel in our next study.
Although cellular MRI can track tumor cells in the lymphatic system to study the interaction between invading tumor cells and host lymphatics, histologic confirmation of results is still required. Moreover, pathology plays an important role in understanding the molecular mechanism of tumor lymphatic metastasis. Consequently, we will combine cellular MRI with molecular probe technology to visualize the early molecular events of tumor lymphatic metastasis in our next study.
Conclusion
This method provided a powerful approach to study the interaction between invading tumor cells and host lymphatics through tracking tumor cells in the lymphatic system. There is a close relationship between lymphangiogenesis and tumor lymphatic metastasis.
Footnotes
Acknowledgment
Financial disclosure of authors: This project was financially supported by the National Basic Research Program of China (No. 2011CB935800) and the National Natural Science Foundation of China (81130027 and 81071204).
Financial disclosure of reviewers: None reported.
