Abstract
[11C]Acetate (ACT) positron emission tomography/computed tomography (PET/CT) is useful in the detection of hepatocellular carcinoma (HCC). This study aimed to evaluate whether [18F]fluoroacetate (FAC) could be an alternative analogue of [11C]ACT for the diagnosis of HCC. [18F]FAC was synthesized using the precursor t-butyl 2-(methanesulfonyloxy)ethanoate. Five volunteer patients with known HCC were recruited after consent. Whole-body [18F]FAC PET/CT was performed at 20 minutes and 1 hour postinjection and compared to [11C]ACT PET/CT at 20 minutes postinjection to assess biodistribution and tumor uptake characteristics. Qualitative and semiquantitative analyses were performed with statistical correlations on the physiologic organs of accumulation and HCC lesions for both tracers. [18F]FAC was obtained with 99% radiochemical purity, and the reaction yield was 16.0% with 1-hour synthesis time. The biodistribution of [18F]FAC on PET/CT was significantly different from that of [11C]ACT (p < .05) by the lack of preferential uptake in any specific organ, particularly the pancreas, resembling the pattern of blood-pool retention although partly metabolized via the bowel. There was no significant defluorination, and none of the [11C]ACT-avid HCC lesions showed increased [18F]FAC activity. These were different from the results reported on other species. [18F]FAC may not be a potential alternative tracer for [11C]ACT in PET/CT evaluation of HCC in human subjects.
IN RECENT YEARS, [11C]acetate (ACT) positron emission tomography (PET) was found useful in various research and clinical applications, including urologic,1,2 liver, 3 and lung 4 cancers. However, for PET centers that do not have on-site cyclotrons, the use of [11C]ACT is limited by the short radioactive half-life (20 minutes) of [11C]. Even with a high-yield synthesis, only a limited number of patients can be studied from a single-batch production. Therefore, acetate analogues labeled with positron emitters with a longer half-life are needed. One of the potential radiopharmaceuticals proposed in the literature is [18F]fluoroacetate (FAC),5–9 which has a longer half-life of 110 minutes. The purpose of this study was to (1) assess the radiolabeling efficiency and quality of [18F]FAC, (2) compare the biodistribution pattern of [18F]FAC and [11C]ACT PET in human subjects, and (3) evaluate the feasibility of [18F]FAC in the diagnosis of hepatocellular carcinoma (HCC) already documented clinically and shown to have strong avidity for [11C]ACT on PET imaging.
Materials and Methods
General Preparation
Unless otherwise stated, all chemicals were purchased from Sigma-Aldrich and BDH and were not further purified. [18O]H2O was supplied by Marshall Isotopes Ltd. Radio thin-layer chromatography was performed on Whatman silica gel 60 F-254 plates. 1H and 13C nuclear magnetic resonance (NMR) spectra were recorded on a Bruker 300 MHz spectrometer.
Chemical Synthesis
Synthesis of Ethyl 2-(methanesulfonyloxy)ethanoate
Ethyl glycolate (1.0 g, 9.6 mmol) and methanesulfonyl chloride (0.8 mL, 10.3 mmol) were added to a 100 mL round-bottomed flask with methylene chloride (10 mL). The solution was cooled to 0°C, and triethylamine (0.8 mL, 5.7 mmol) was added dropwise with stirring. The resulting solution was stirred at room temperature for 80 minutes and extracted with ice-cold HCl (1.0 M, 15 mL) and then with water twice (2 × 15 mL). The organic layer was isolated and dried with anhydrous magnesium sulfate. The solution was concentrated, and the desired product was purified by silica gel column chromatography using ethyl acetate and hexane at a ratio of 3:7. The product was isolated as a colorless liquid (0.93 g, 53% yield). The NMR spectra data were: 1H NMR (CDCl3, 300 MHz): δH 1.29 (t, 3H, CH2
Synthesis of t-Butyl 2-(methanesulfonyloxy)ethanoate
Silver mesylate (1.44 g, 7.07 mmol) was added to a solution of bromoacetic acid t-butyl ester (1.0 g, 5.13 mmol) in acetonitrile (20 mL) at room temperature. The reaction mixture was stirred for 40 hours. The reaction flask was covered by aluminum foil to protect it from light during stirring. At the end of reaction, the precipitate formed was removed by filtration and the solvent was evaporated off by a rotary evaporator. The product was isolated from the crude mixture by silica gel column chromatography using ethyl acetate and petroleum ether at a ratio of 1:9 as white crystals (0.89 g, 74.4%). The melting point was 36.5 to 37.5°C. The NMR spectra data were: 1H NMR (CDCl3, 300 MHz): δH 1.47 (s, 9H, t-Bu), 3.18 (s, 3H, SC
Radiosynthesis of [18F]FAC
[18F]FAC was synthesized by modifying Richter and colleagues′ methodology8,9 with an automatic synthesizer (F121R) manufactured by Sumitomo Heavy Industries (SHI), Japan. No-carrier-added (NCA) aqueous [18F]fluoride was produced in the SHI CYPRIS cyclotron (HM12s, 12.0 MeV) via the 18O[p,n]18F nuclear reaction on 18O-enriched (> 97% purity) water loaded inside a silver target bombarded with a target current of 50 μA for a 1-hour period. The [18F]fluoride ion (53.2 GBq) produced was trapped in a Sep-Pak light Accell plus QMA cartridge (Waters) and then eluted to the reaction vessel by using Kryptofix/potassium carbonate solution in anhydrous acetonitrile (0.9 mL). After the addition of 0.6 mL anhydrous acetonitrile, water was azeotropically removed from this mixture by heating to 100°C. Then a precursor solution of t-butyl 2-(methanesulfonyloxy) ethanoate (3.4 mg) 8 in acetonitrile (1.0 mL) was added to the dried NCA [18F]fluoride. The reaction vessel was sealed and the mixture inside was heated at 100°C for 10 minutes. After cooling, dilute hydrochloric acid (1 M, 0.56 mL) was added and the mixture was heated at 100°C for another 10 minutes. The final pH of the reaction solution was adjusted to alkaline by adding sodium hydroxide (2 M, 0.63 mL) followed by dilution with water (14 mL). This solution was allowed to pass through two anion exchange cartridges connected in series (Alltech Maxi-clean SAX, 600 mg). The product [18F]FAC trapped in the cartridges was rinsed with water (2 × 12 mL), dried by nitrogen flushing, and then eluted by sodium bicarbonate solution (8.4%, 3 mL). The eluant was then passed through two alumina N plus Sep-Pak cartridges (Waters) for removal of unreacted [18F]fluoride and a vented filter (0.22 μm) before entering the collection vial.
The final product, [18F]FAC, obtained had a radioactivity of 8.51 GBq and radiochemical purity of 99%. The reaction yield was 16.0% end of synthesis (EOS) with a 1-hour synthesis time. For radio–high-performance liquid chromatography (HPLC), the retention time (Rt) of FAC was 6.0 minutes using a 4.6 × 250 mm Allsphere ODS-2 column, a ultraviolet (UV) detector (210 nm, Agilent 1200), and a radiochromatography monitor (Bioscan). The mobile phase was 0.01 M sodium dihydrogen phosphate solution with concentrated HCl (0.03%, v/v) moving at a flow rate of 0.5 mL/min. The identity of the product was verified by spiking with sodium fluoroacetate standard, 10 and the quantity was calculated by using a standard calibration curve. The radio–thin-layer chromatography (TLC) data were: 1. t-butyl [18F]FAC, retention factor (Rf) = 0.69 (ethyl acetate:PE 1:1), 2. [18F]FAC, Rf = 0.18 (acetonitrile:water 95:5).
Radiosynthesis of [11C]ACT
[11C]ACT was synthesized using a SHI C100 automatic synthesizer. [11C]CO2 produced in the SHI CYPRIS cyclotron (HM12s, 12.0 MeV) was bubbled into an ethereal solution of methylmagnesium bromide (0.2 M, 2 mL) at a flow rate of 30 mL/min for 3 minutes. Water (0.2 mL) was added, and ether was evaporated under a stream of nitrogen gas. After cooling down, phosphoric acid (10%, 0.6 mL) was added, and the reaction mixture was heated to 80°C to hydrolyze the intermediate. The hydrolyzed mixture was diluted with water (2.5 mL). This solution was transferred to the collection vial containing normal saline (6 mL) via two Alltech Maxi-clean cartridges, IC-Ag and IC-H, and a 0.22 μm vented filter. The pH was adjusted to 8 by the addition of sodium bicarbonate solution (8.4%, 1.2 mL). This isotonic, sterile, and nonpyrogenic solution was ready for injection. The radiochemical percentage purity was determined by HPLC using an Allsphere ODS-2 column 4.6 × 250 mm; mobile phase was NaH2PO4 (0.01M) acidified with concentrated HCl (0.03% v/v), and the flow rate was 1.0 mL/min. The [11C]ACT was monitored with a UV detector (210 nm, Agilent 1200) and a radiochromatography diode detector (Bioscan). The retention time was 4.1 minutes. The identity of the product was verified by spiking the sample 10 (sodium acetate standard), and the quantity was calculated using a standard calibration curve.
PET/Computed Tomographic Imaging
Five patients with known HCC were referred from an affiliated academic institution after ethics committee approval for a 2-day protocol of [11C]ACT and [18F]FAC PET/computed tomography (CT) within 1 week. Both informed and written consent with regard to the purpose and nature of this study and possible side effects of the tracer materials, procedures, and schedules of follow-up were obtained before PET/CT scanning. All five patients were male with a mean age of 51 years (range 20–82 years) and four were hepatitis B carriers.
Intravenous 560 to 590 MBq [11C]ACT was given and limited whole-body PET/CT (Siemens HR Biograph 40 LSO) from the skull base to the upper thigh was performed 20 minutes after [11C]ACT injection. Data acquisition began with CT (with no contrast) at 130 kV, 110 to 115 mA, 2 mm pitch, and 1-second tube rotation, followed by PET with 2-minute emission acquisition at 21.6 cm axial field of view per position. The images were reconstructed by means of TrueX (Siemens) technique at 21 subsets and 3 iterations with a 168 × 168 matrix for PET and a 512 × 512 matrix for CT.
Using the same imaging and acquisition protocols, [18F]FAC PET/CT was performed at 20 minutes and 1 hour, respectively, after intravenous [18F]FAC injection (430–490 MBq). Vital signs and pulse oximetry were regularly monitored before, during, and after [18F]FAC injection. Attenuation correction and reconstruction parameters were the same as those specified for [11C]ACT. Patients′ physical status was followed for 7 days and at 1 month after the procedure.
Tracer Biodistribution and Uptake Characteristics in HCC
The biodistribution of tracers was assessed by observation and semiquantitative calculation of the mean standard uptake value (SUVmean) of the regions of interest (ROI) within major visceral organs (physiologic ROI) on [18F]FAC-PET. The tracer uptake characteristics of primary and metastatic HCC lesions were assessed by visual inspection and SUVmean within lesion ROI (pathologic ROI).
Physiologic Biodistribution
The physiologic ROI of major visceral organs except the bowel were drawn automatically on CT using a “region-growing” algorithm based on the similarity of the neighborhood by the commercial software MIMContouring version 4.1.8. Bone ROI was drawn in the body of L3. The ROI generated from CT were copied onto the 20-minute and 1-hour [18F]FAC-PET images and the [11C]ACT-PET images. Bowel ROI was drawn in the area of highest activity on 20-minute [18F]FAC-PET using a “PET edge” technique (based on the second derivative computation method) by MIMContouring 4.1.8 and copied onto the 1-hour [18F]FAC-PET and [11C]ACT-PET images. So for each organ, the same ROI was applied to all three sets of PET images and the SUVmean within each ROI was calculated. The two sets of physiologic ROI on [18F]FAC-PET were compared visually and semiquantitatively (SUVmean) to examine for any change in biodistribution and degree of defluorination for each patient. The two sets of physiologic ROI on [18F]FAC-PET were also compared to those on [11C]ACT-PET. Specific care was taken to avoid the primary liver tumors and metastatic lesions, so drawing of physiologic ROIs was on the nontumor part of organ or tissue structures.
Tracer Uptake Characteristics in HCC Lesions
Based on the [11C]ACT-PET images, the locations of the primary HCC lesions and metastases were identified by three independent nuclear medicine physicians in consensus. Pathologic ROI were drawn on the [11C]ACT-PET images automatically by PET edge of MIMContouring 4.1.8 and copied onto the two sets of [18F]FAC-PET images. The SUVmean within each ROI on three sets of PET images was calculated.
Statistical analysis was performed using chi-square and paired Student t-tests (95% confidence level considered significant).
Results
Radiopharmaceutical Yield and Quality Assurance
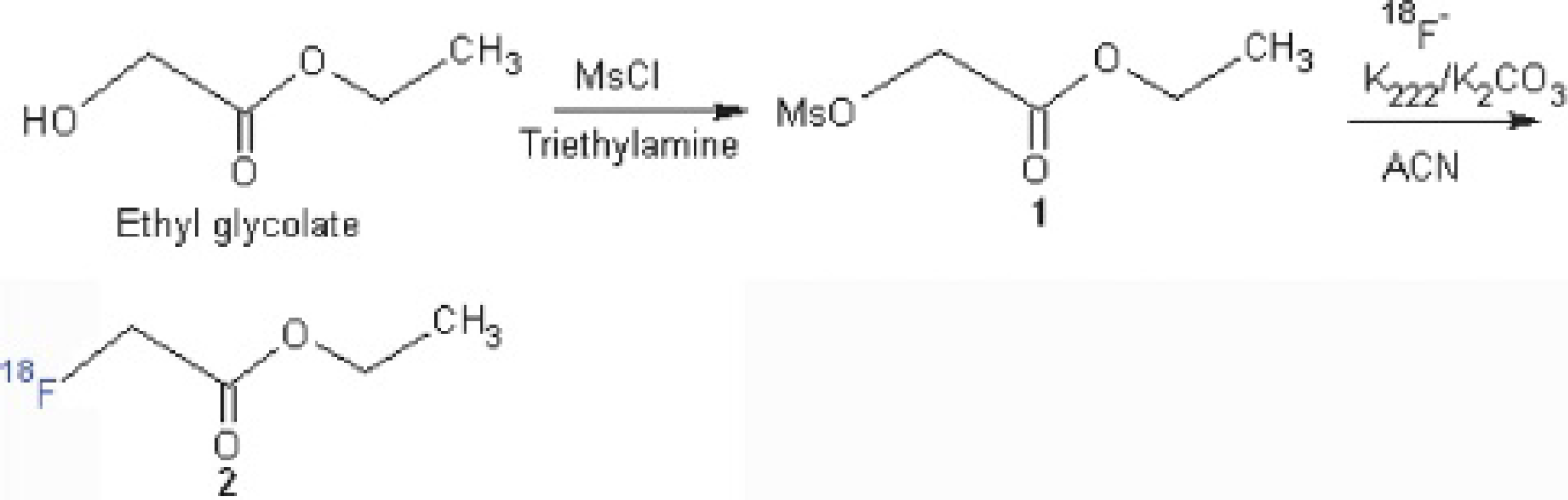
[18F]FAC has been synthesized by several research groups using the precursor compound ethyl 2-(methanesulfonyloxy) ethanoate.6–8 In this study, we prepared this compound by heating ethyl glycolate with methanesulfonyl chloride in methylene chloride (Figure 1). The precursor compound and NCA dried [18F]fluoride were then heated at 100°C for 20 minutes to give a radioactive intermediate ethyl [18F]FAC, which was analyzed by radio-HPLC using an Allsphere ODS-2 column 4.6 × 250 mm. The mobile phase was acetonitrile and water (1:1). The identity of ethyl [18F]FAC was verified by spiking with the standard compound (cold ethyl fluoroacetate). 10 Multiple trials to hydrolyze this compound using 1 M NaOH7,8 or 1 M KOH 6 gave a mixture of highly polar radioactive products, which had the same retention time as the solvent in HPLC analysis. No conceivable amount of [18F]FAC was isolated. The real reason for failed synthesis under this scheme was not entirely certain but was speculated to be related to the presence of two very acidic methylene protons adjacent to the carboxyl carbon and [18F] atom in ethyl [18F]FAC. These two acidic protons were more likely to be removed than the desired reaction in the basic medium provided by the hydrolysis step. The anionic species thus formed was highly reactive and more likely to give rise to many side products.
Reaction scheme for synthesis of ethyl [18F]FAC from ethyl glycolate.
We then switched to using another precursor compound, t-butyl 2-(methanesulfonyloxy)ethanoate. This compound was shown to work better as a labeling precursor than the ethyl derivatives. 9 We prepared this precursor compound from a reaction between bromoacetic acid t-butyl ester and silver mesylate stirring at room temperature for 40 hours, which took a longer reaction time than using the corresponding iodoacetic acid derivative as starting material (Figure 2). The purified product was then reacted with dried NCA [18F]fluoride at 100°C for 10 minutes in an SHI F121R synthesizer. The reaction mixture was analyzed by radio-TLC analysis following the conditions suggested in the literature. 9

Reaction scheme for synthesis of [18F]FAC from bromoacetic acid t-butyl ester.
The desired radioactive intermediate, t-butyl [18F]FAC, was the only radioactive compound obtained and the Rf value was 0.69, consistent with the value reported by Richter and colleagues. 9 This reaction mixture was further analyzed by radio-HPLC, using the same column, mobile phase, and flow rate as used for ethyl [18F]FAC. The retention time was 5.2 minutes. The t-butyl-protecting group in this radioactive intermediate could be removed under acidic conditions. 9 Finally, acid hydrolysis to remove the t-butyl group gave the desired product [18F]FAC with 19.25 ± 3.94% yield (decay corrected) (n = 3). The identity was confirmed by injection of a spiked sample 10 in HPLC analysis (Figure 3). The difference in the retention time (ΔRt) between UV and radiochromatography channels was 0.259 minutes, which was consistent with the acceptable range of ΔRt in our good practice for HPLC in the laboratory. This reaction was reliable and reproducible. No further experiments were carried out to improve the percentage yield.

The retention time difference (ΔRt) between the [18F]FAC (Rt = 5.976 minutes) and the spiked sodium fluoroacetate standard (Rt = 5.717 minutes) is 5.976 to 5.717 = 0.259 minutes, within the acceptable range of ΔRt. The peaks at Rt 4.776 minutes correspond to the excipients sodium bicarbonate and sodium chloride.
The UV channel (210 nm) of the analytical HPLC showed no mass peak corresponding to sodium fluoroacetate. The detection limit (LOD) and quantitation limit (LOQ) 11 of our HPLC was determined to be 0.0025 mg/mL (signal to noise ratio [S/N] = 5) and 0.009 mg/mL (S/N = 14.9) for sodium fluoroacetate. With the LOQ value obtained, the maximum amount of the cold compound sodium fluoroacetate present in the product solution was determined to be 0.027 mg (0.2318 μmol). Taking the [18F]FAC activity reported in the experimental section for calculation, the minimum specific activity of [18F]FAC obtained was 36.7 GBq/μmol (at EOS), which was about the same order of magnitude as the value (74–129.5 GBq/mmol) reported by Ponde and colleagues. 8
Monitoring the Safety of Radiotracer Administration
In the literature, a lethal dose of sodium fluoroacetate in human beings was reported to be 2 to 10 mg/kg body weight. 12 The maximum amount of sodium fluoroacetate injected to the patient from our preparations was the product of LOQ and total volume of the final production, which was about 0.027 mg (0.00054 mg/kg, body weight = 50 kg), significantly less than the lethal dose value. Nishii and colleagues reported that from a pharmacologic perspective, diagnostic dosages of [18F]FAC are nontoxic to primates and, therefore, likely to be safe when administered to human patients for PET imaging. 5
The vital statistics of the volunteers, including blood pressure, pulse, respiratory rate, body temperature, and oxygen saturation parameters, were stable and within normal ranges before, during, and 4 hours after [18F]FAC injection. Follow-up monitoring at 2 and 7 days after [18F]FAC PET/CT showed that all patients were well, with no signs of adverse reaction (such as convulsion, bradycardia, or hypothermia) or discomfort. Blood tests at patients′ institution did not reveal a significant change in essential organ functions. Patients were alive with no specific symptoms on final follow-up at 1 month after this study.
Tracer Biodistribution
Analysis of the biodistribution data for the [11C]ACT-PET and two [18F]FAC-PET studies for the patients is shown in Figure 4. The SUVmean of the nontumor parts of major organ ROI of each patient was calculated and averaged for all five patients. Each data set was composed of three histogram bars plotted and compared per organ basis. There was no significant difference in SUVmean between the two sets of [18F]FAC PET at 20 minutes and 1 hour in all patients (p values for all patients > .05). The SUVmean of [11C]ACT for each major organ was, however, statistically very different from those of [18F]FAC (p values for all patients < .05). Table 1 tabulated the biodistribution data in terms of the averaged SUVmean ± standard deviation for each major organ (nontumor part) of the five human subjects.
[18F]FAC and [11C]ACT Biodistribution of the Five Human Subjects

[11C]ACT = [11C]acetate; [18F]FAC = [18F]fluoroacetate.
Data are expressed as the averaged SUVmean ± standard deviation of all patients.

Comparison of the biodistribution of 20- and 60-minute [18F]fluoroacetate (FAC) to 20-minute [11C]acetate (ACT) in the nontumor parts of the major organs of five human subjects with known primary and metastatic hepatocellular carcinoma disease. LV = left ventricle.
The results showed that there was rapid clearance of [11C]ACT tracer from the major vascular structures at 20 minutes, leaving very little activity in the left ventricle (LV) (SUVmean = 1.04 ± 0.17), aortic arch (SUVmean = 1.05 ± 0.11), abdominal aorta (SUVmean = 1.19 ± 0.05), and abdominal vena cava (SUVmean = 1.19 ± 0.06). Radiotracer accumulation of [11C]ACT is highest in the pancreas (SUVmean = 7.51 ± 2.64), significantly higher than in other organs, such as the lung, myocardium, liver, spleen, kidney, adrenal gland, bowel, bone, prostate, and urinary bladder (all p values < .05). With [18F]FAC PET, although the major vascular tracer concentration decreased slightly from 20 minutes to 1 hour (LV chamber = 11.0 ± 1.2%, aortic arch = 11.4 ± 2.0%, abdominal aorta = 9.2 ± 0.9%, abdominal vena cava = 10.5 ± 1.6%), there was still relatively high vascular tracer activity retention at 1 hour (SUVmean in LV chamber = 2.40 ± 0.57, aortic arch = 2.33 ± 0.52, abdominal aorta = 2.05 ± 0.34, abdominal vena cava = 2.04 ± 0.34). Furthermore, the decrease in [18F]FAC activity for all five patients from 20 minutes to 1 hour in major organs such as the myocardium (10.6 ± 2.9%), liver (9.1 ± 1.7%), spleen (9.9 ± 2.6%), pancreas (9.8 ± 4.5%), kidney (7.1 ± 0.9%), and prostate (10.4 ± 2.2%) also followed a very slow clearance pattern similar to that of the large vessels (all p values > .05). There was no significant uptake in the lungs and adrenal glands on both sets of [18F]FAC-PET. Bone/marrow uptake of [18F]FAC was also mild, although its metabolic intensity increased slightly over time (SUVmean = 1.31 ± 0.08 and 1.38 ± 0.06 at 20 minutes and 1 hour, respectively), probably owing to a small amount of free [18F]fluoride bone uptake as a result of slight defluorination in 1 hour of study. There was very minimal urinary [18F]FAC activity in the kidneys or urinary bladder, as shown by a slight increase in [18F]FAC uptake in the urinary bladder with time (SUVmean = 1.30 ± 0.18 and 1.56 ± 0.30 at 20 minutes and 1 hour, respectively). [18F]FAC bowel activity was the highest among the nonvascular organs at both 20 minutes and 1 hour (SUVmean = 2.44 ± 0.06 and 2.64 ± 0.15 at 20 minutes and 1 hour, respectively), suggesting that some excretion was via the bowel.
Tracer Uptake Characteristics in HCC
The patient and tracer uptake characteristics of the five patients are summarized in Table 2. In all of the [11C]ACT-avid primary and metastatic HCC lesions, none (except three of the metastatic lesions in patient 5; Table 3) were shown to have [18F]FAC-avid pathology on the 20-minute or 1-hour [18F]FAC-PET.
Patient and Tracer Characteristics

[11C]ACT = [11C]acetate; [18F]FAC = [18F]fluoroacetate; HBV = hepatitis B virus; HCC = hepatocellular carcinoma.
Specific Data for Patient 5 Showing the Differences in the Tracer Characteristics (SUVmean) of the Metastatic HCC Lesions and an Angiomyolipoma

[11C]ACT = [11C]acetate; [18F]FAC = [18F]fluoroacetate; HCC = hepatocellular carcinoma; SUVmean = mean standard uptake value.
Figure 5 depicts the whole-body [18F]FAC-PET at 20 minutes and 1 hour and [11C]ACT-PET at 20 minutes after injection for patient 3. On the [11C]ACT-PET images, a large HCC extensively involving the left lobe and segments V to VIII of the right lobe was intensely avid for [11C]ACT but not [18F]FAC (Figure 6A). Two metastatic bone lesions in the left ilium and left scapula were also discretely [11C]ACT avid with osteolytic changes on CT bone window. However, no hypermetabolic bone lesion was identified on the [18F]FAC PET images (Figure 6B). Figure 7 shows the PET images of patients 1 and 2 with advanced primary and metastatic HCC disease. All lesions were avid only for [11C]ACT, whereas none showed increased activity on both sets of [18F]FAC-PET images.
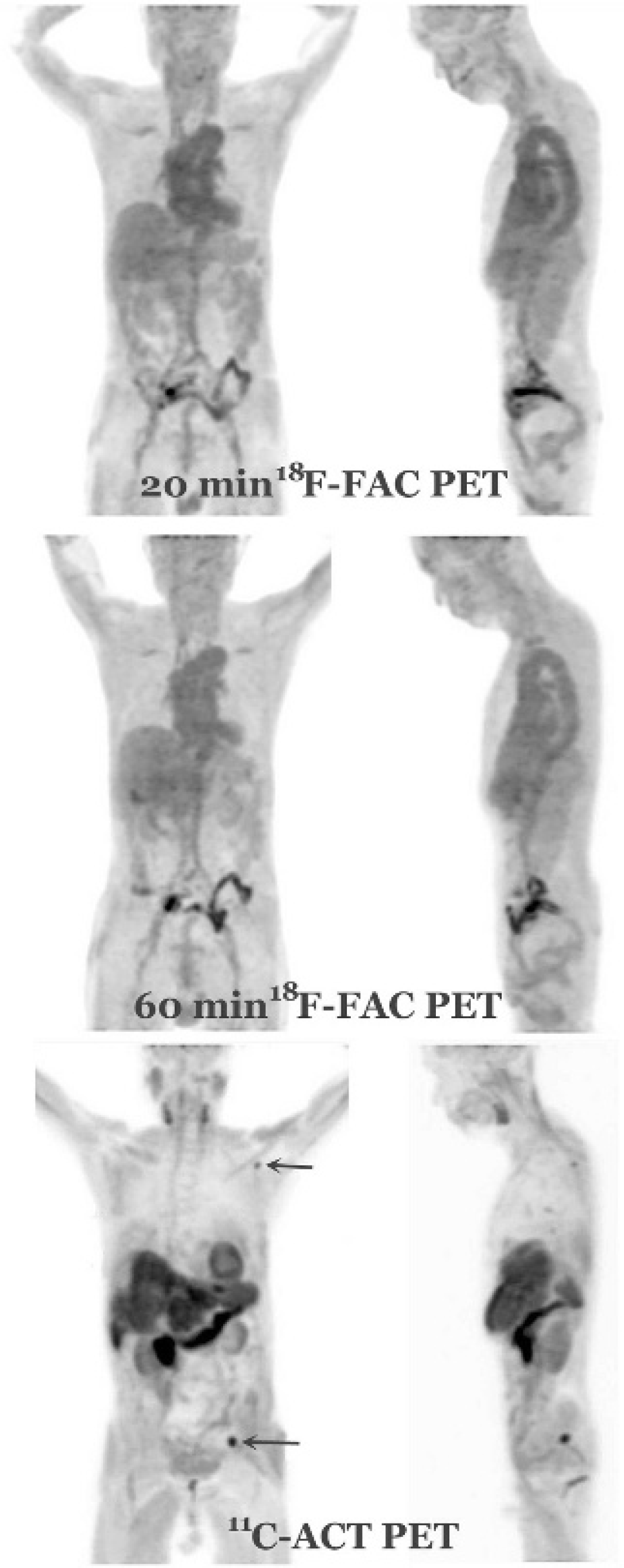
Twenty- and 60-minute [18F]fluoroacetate (FAC) and 20-minute [11C]acetate (ACT) whole-body PET images for patient 3, who has a large primary hepatocellular carcinoma in the liver and two metastatic bone lesions (arrows).
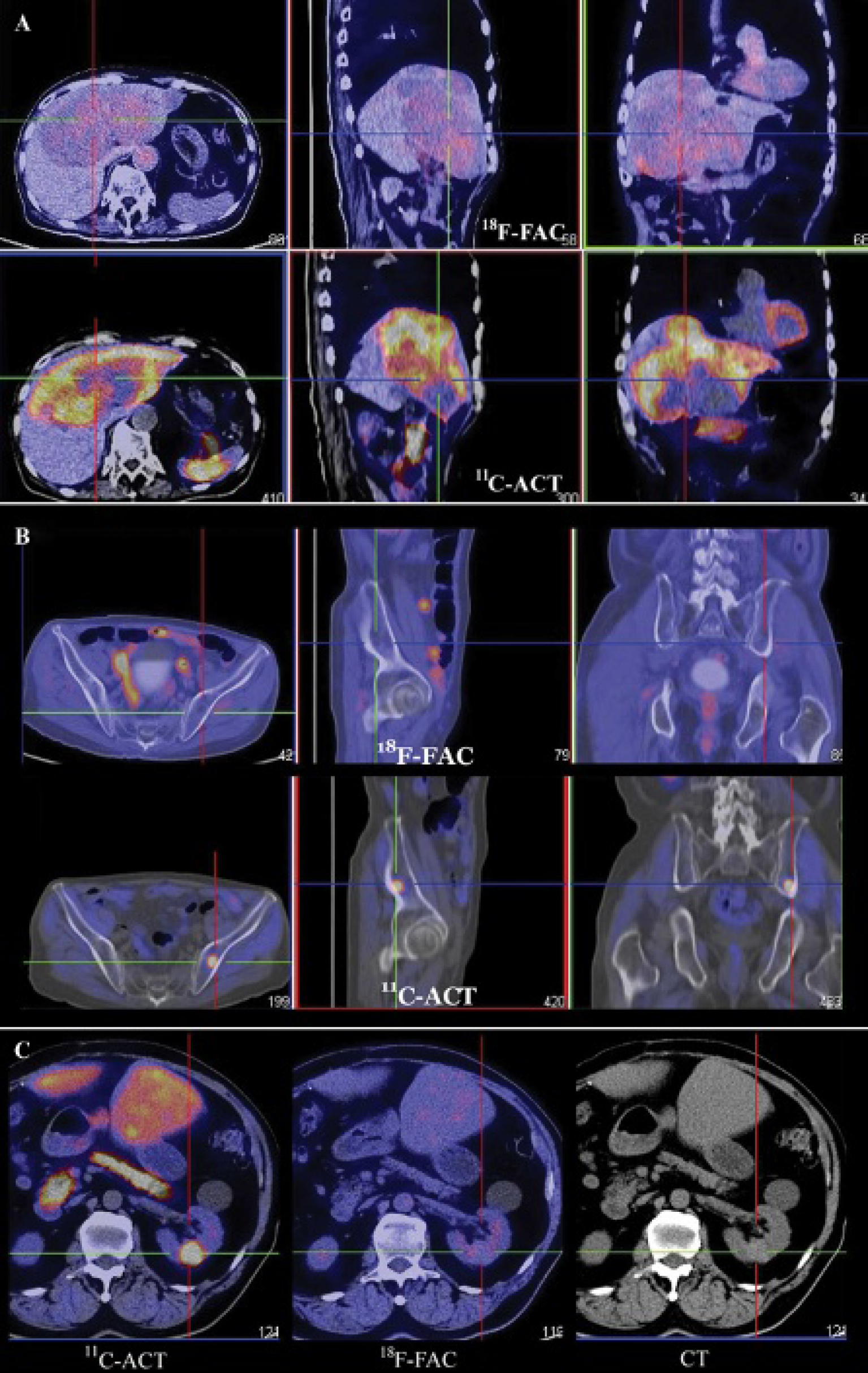
Hybrid PET/CT with transaxial section across the primary hepatocellular carcinoma tumor (A) and a metastatic left iliac lesion (B) of patient 3 and an angiomyolipoma (C) in the left kidney of patient 5. All are markedly hypermetabolic on [11C]acetate (ACT) but negative on [18F]fluoroacetate (FAC) PET images.

Twenty- and 60-min [18F]fluoroacetate (FAC) and 20-min [11C]acetate (ACT) whole-body PET images for patients 1 (upper row) and 2 (middle row) with advanced primary and metastatic hepatocellular carcinoma disease.
Regarding the exception of three metastatic lesions (one in the lung, two in the ribs) in patient 5 (see Table 3) mentioned above, their [18F]FAC activities were very mild. They could be visualized presumably because of the intrinsic low background [18F]FAC activity of the lung and trabeculae marrow. In fact, the [18F]FAC SUV ratios of these three metastatic lesions (with reference to myocardium) at 1 hour were moderately lower than the ratios at 20 minutes (lung lesion decreased 18.6%; two rib lesions decreased 5.7% and 30.1%), indicating that there was even more rapid clearance of [18F]FAC from these three lesions than normal organs. This was not supportive to the increased dual time point uptake ratios usually taken as ancillary evidence of active tracer uptake in malignant tumors.
Patient 5 was also known to have a stable angiomyolipoma of low fat content in the left kidney, which showed markedly increased [11C]ACT metabolism (lesion to kidney SUVmean ratio = 10.44/2.58 = 4.1), in keeping with our previous research data. 13 This lesion showed entirely negative uptake on [18F]FAC PET images (Figure 6C). These findings suggest that the biochemical use of these two tracers is very different whether the neoplasm is benign or malignant.
Discussion
This PET/CT study was designed to evaluate the whole-body biodistribution and tumor uptake characteristics of [18F]FAC in human subjects. The biodistribution of [18F]FAC in humans appeared to be different from that of other species, as reported in the literature.5,8,9,14 There is no sign of hepatobiliary excretion, which is prominent in monkeys5,14 but not in baboons, 8 rats,8,9 or pigs. 14 There is little renal excretion of [18F]FAC, which is significant in monkeys,5,14 baboons, 8 and pigs 14 but not in rats.8,9 No detailed investigation or explanation is offered by the present data in literature, but as proposed by Ponde and colleagues, perhaps there is a “species difference” in relation to the biodistribution. 8 In our study, there was a variable amount of activity in the colon, suggesting that some excretion was likely to be via the bowel, which agreed with the findings of previous research groups.5,8,9,15 On the other hand, the only published case report of [18F]FAC in a human subject 15 was in agreement with our observation that the mode of excretion was predominantly via the bowel and there was no significant urinary activity.
Referring to the pioneer data available in the literature, there was indeed strong evidence of species-related biodistribution, toxicity, and excretion differences in regard to nonradioactive fluoroacetate. 16 The differences were attributed to the variation in abundance of the enzyme system associated with the relative ease of conversion of fluoroacetate to fluorocitrate. In fact, these data indicated that even different routes of administrating fluoroacetate in the same species could cause a difference in the severity of toxicity, meaning that different organs may have different susceptibility or tolerance.
Biochemistry and Mechanism
[11C]ACT enters the tricarboxylic acid (TCA) cycle in the mitochondrion after conversion by acetyl coenzyme A (CoA) synthetase to [11C]acetyl CoA. It combines with oxaloacetate to form [11C]citrate, whereby sequential steps of oxidative metabolism within the TCA cycles quickly lead to metabolic clearance of [11C]ACT in the form of [11C]CO2. In normal cells, the rate of de novo lipid synthesis is low because dietary fatty acids are usually abundant. However, in proliferating tumor cells, the glucose-dependent and/or acetate-dependent de novo lipid syntheses are elevated. Reasons for increased lipid use include synthesis of membrane phospholipid, deoxyribonucleic acid (DNA) replication, apoptosis, energy conversion, redox balance in hypoxic states, and signal transduction. [11C]Citrate in the mitochondria, instead of passing down the TCA cycle, is transported to the cytosolic enzyme adenosine triphosphate citrate lyase, which is the key enzyme for convertion of [11C]citrate back to [11C]acetyl CoA and oxaloacetate. Thus, combining with malonyl CoA in the cytosol, [11C]acetyl CoA is the precursor material for lipid synthesis. FAC behaves similarly to ACT initially. It is converted into fluoroacetyl CoA in the TCA cycle and then metabolized by citrate synthase to fluorocitrate. However, different from normal [11C]citrate, fluorocitrate binds strongly with the enzyme aconitase and inhibits further oxidative metabolism in the rest of the TCA cycle. Because of this strong fluorocitrate-aconitase binding and subsequent metabolic blockage, two main effects different from [11C]citrate are noted: impaired clearance of trapped [18F]fluorocitrate from the cells 12 and failure of transport to the cytosol for conversion back to acetyl CoA and lipid synthesis.
The above discussion explains why there is slow clearance of [18F]FAC in the human body. As noted above, the whole-body [18F]FAC PET study, other than some bowel activities, resembles one of “blood-pool” imaging, with retention of activity in the organ or tissue such as myocardium, great vessels, liver, spleen, and other hypervascular or hyperemic structures. The three metastatic HCC lesions of patient 5 that showed minimal [18F]FAC activity are presumably attributed to the same reason that they stand out in a background of low blood-pool activity (lung and bone marrow). In a recent study by Linhe and colleagues evaluating [18F]FAC and [11C]ACT biodistribution in pigs and monkeys, their data also showed that [18F]FAC activity had a much prolonged blood retention even at 2 hours after injection and the kinetics of [18F]FAC in the liver did not seem to bear any resemblance to TCA cycle activity. 14
Our study differs from that of Ponde and colleagues 8 and Matthies and colleagues 15 in using known cases of HCC instead of prostate cancer. Furthermore, we have used [11C]ACT PET/CT as the gold reference to compare the physiologic and pathologic biodistribution with [18F]FAC. Mathies and colleagues, in their short report, did not explain why some of the metastatic bone lesions from prostate primary cancer showed increased [18F]FAC uptake, whereas some did not. 15 It is not certain if these could be attributed to tumor-related biochemical differences or if some lesions were actually of different pathology. However, it is also possible that it could be related to blood-pool concentration differences between a soft tissue nodule (metastatic HCC) and a “hollow” background (trabecular bone), as suggested by the three metastatic lesions discussed above. Our patients in this study had a number of other metastatic HCC sites (bone, lung, adrenal gland, and peritoneum) and a benign angiomyolipoma that were strongly avid for [11C]ACT but not at all for [18F]FAC. From the standpoint of biochemistry, these negative results of [18F]FAC in human subjects appear to be reasonable.
Limitations
Although our preliminary results are not in favor of [18F]FAC in PET imaging of HCC, a small cohort of case studies cannot be a sufficient conclusion or recommendation for ruling out the application of this tracer in other types of tumor. On the other hand, based on the same arguments and observation, researchers potentially using [18F]FAC for investigation of other pathologies in human subjects may give a more careful consideration.
Conclusion
Reliable synthesis of sodium [18F]FAC is possible through the precursor compound t-butyl 2-(methanesulfonyloxy) ethanoate. The biodistribution pattern of [18F]FAC and its negative avidity for primary and secondary HCC suggest that it is visually and semiquantitatively not suitable to be recommended for use as an alternative tracer for HCC in place of [11C]ACT in human subjects. However, this preliminary finding cannot preclude its potential use in other types of neoplasm in which tumor-related biochemical variations may exist.
Footnotes
Acknowledgment
Financial disclosure of authors and reviewers: None reported.
