Abstract
Near-infrared (NIR) fluorophores are the focus of extensive research for combined molecular imaging and hyperthermia. In this study, we showed that the cyanine dye IR820 has optical and thermal generation properties similar to those of indocyanine green (ICG) but with improved in vitro and in vivo stability. The fluorescent emission of IR820 has a lower quantum yield than ICG but less dependence of the emission peak location on concentration. IR820 demonstrated degradation half-times approximately double those of ICG under all temperature and light conditions in aqueous solution. In hyperthermia applications, IR820 generated lower peak temperatures than ICG (4–9%) after 3-minute laser exposure. However, there was no significant difference in hyperthermia cytotoxicity, with both dyes causing significant cell growth inhibition at concentrations ≥ 5 μM. Fluorescent images of cells with 10 μM IR820 were similar to ICG images. In rats, IR820 resulted in a significantly more intense fluorescence signal and significantly higher organ dye content than for ICG 24 hours after intravenous dye administration (p < .05). Our study shows that IR820 is a feasible agent in experimental models of imaging and hyperthermia and could be an alternative to ICG when greater stability, longer image collection times, or more predictable peak locations are desirable.
IN VIVO MOLECULAR IMAGING allows not only visualization of organs and tissues but also characterization and measurement of biologic processes at the cellular and molecular levels that are associated with specific diseases. This information can improve the accuracy of a diagnosis, provide better assessment of the severity of disease, and even monitor the response to therapy. Imaging modalities such as magnetic resonance imaging (MRI) and x-ray computed tomography (CT) are generally considered anatomic modalities and not molecular imaging technologies. 1 Nuclear medicine techniques such as positron emission tomography (PET) and single-photon emission computed tomography (SPECT) are common clinical molecular imaging tools. Numerous PET and SPECT imaging agents have been developed for various imaging purposes, 2 but the instrumentation is large, complex, and expensive relative to optical imaging. Optical imaging yields high spatial resolution, can be done in real time, is inexpensive and robust, and can be made portable. Fiberoptics makes optical imaging ideal for incorporation into an endoscope for use in noninvasive or minimally invasive procedures. However, optical imaging suffers from the fact that compared to nuclear photons, light is highly scattered and absorbed by tissue.
Near-infrared (NIR) light between the wavelengths of 700 to 900 nm propagates deeply through tissue owing to minimal deoxyribonucleic acid (DNA) and water absorption 3 and scattering, thus providing a unique approach for molecularly based in vivo imaging. 4 With the development of tumor-specific contrast agents, the technology has a potential for clinical translation for various noninvasive imaging applications. Coupling molecular imaging with therapy can allow for verification and possibly quantification of drug delivered to a specific tissue or cellular site. Dyes that absorb energy in the NIR region will release heat following exposure to the appropriate wavelength of light and can kill cancer cells. 5 With an NIR dye incorporated into a drug delivery vehicle, therapy can be image guided because the drug will not be activated by the laser until the drug has reached its intended target.
Indocyanine green (ICG) is a tricarbocyanine-type dye with NIR-absorbing properties (peak absorption around 800 nm) and little absorption in the visible range. It is approved as a diagnostic aid for blood volume determination, cardiac output, liver function, visualization of retinal and choroidal vasculature, and photodynamic therapy.6,7 The principal advantages include absorption maximum near the isosbestic point of hemoglobin and oxyhemoglobin, confinement to the vascular compartment by binding to plasma proteins, and very low toxicity. ICG has also been proposed for breast cancer imaging. 8 Its ability to generate heat on exposure to NIR illumination has been known for a number of years. 9 In cancer diagnostics and therapeutics, ICG could be used as both an imaging dye and a hyperthermia agent. First, imaging of the fluorescent dye could be used to confirm appropriate targeting of the tumor area. Second, the fact that ICG is thermosensitive would allow for the induction of a hyperthermia effect by irradiating the tumor region with a laser light at the appropriate wavelength. This localized hyperthermia treatment modality can be used in isolation or in combination with chemotherapy. The latter option provides increased cell killing owing to a synergistic combination of the cytotoxic effects of chemotherapy and the increase in temperature, as reported previously by our group. 10 Despite the potential for imaging and therapy applications in cancer, ICG delivery and selectivity continue to be obstacles, particularly owing to the limited stability of ICG in aqueous solution 6 and its short plasma residence time of approximately 3.5 minutes. 11 This limits the in vivo therapeutic window for hyperthermia, imaging, or a combination of both.
Several approaches have been used to create formulations of ICG with improved stability. The primary mechanism of ICG degradation is through the formation of ICG aggregates in solution. 12 Stabilizers such as poly-(α,β)-DL-aspartic acid sodium salt (PASP) can be added to ICG to increase the stability of the dye in solution by preventing the formation of weakly fluorescing ICG aggregates through noncovalent interactions.13,14 However, the addition of a highly hygroscopic and viscous stabilizer complicates the preparation procedure, and the presence of the stabilizer can create difficulties when incorporating the dye into delivery vehicles such as nanoparticles or liposomes for targeted therapies. PASP use has been limited to in vitro research, and its toxicity in potential in vivo applications has not been fully evaluated. Another approach to stabilization of ICG is its incorporation into drug delivery vehicles. Saxena and colleagues reported enhanced ICG stability by polymer encapsulation, 15 and Mordon and colleagues were able to deliver ICG to blood vessels by encapsulating it into heat-sensitive liposomes. 16 Drug delivery vehicles can also be a good alternative to the free dye for combination therapy. Our group recently reported dual encapsulation of ICG and the chemotherapy agent doxorubicin into polylactic co-glycolic acid (PLGA) nanoparticles for dual chemotherapy and hyperthermia applications in cancer cells.17,18
An alternative to modified ICG formulations is the use of other NIR dyes with similar optical and thermal generation characteristics but improved stability profiles. An example of an NIR dye that has potential for imaging and hyperthermia applications and is commercially available is IR820. The chemical structure of IR820 is comparable to that of ICG, with the presence of a chlorobenzene ring that is expected to increase molecular stability. Figure 1 illustrates the chemical structures of ICG (see Figure 1A) and IR820 (see Figure 1B).

Chemical structures of indocyanine green (A) and IR820 (B).
Several recent studies have focused on in vivo applications of IR820 dye or conjugates of the dye with other moieties. Prajapati and colleagues reported the use of IR820 as a blood pool contrast agent to image tissue injuries and tumors in mice. 19 This study showed an estimated plasma clearance half-life of 35 hours for IR820, which is much longer than the plasma half-life of ICG. Pandey and colleagues conjugated IR820 with a photodynamic therapy drug and studied the potential of the conjugate for dual imaging and thermotherapy in mice. 20 Masotti and colleagues recently described the conjugation of IR820 with polyethylenimine for DNA binding applications and in vivo imaging. 21 Although these reports indicate that IR820 has potential for in vivo applications, a need remains to provide a detailed in vitro characterization of IR820 regarding its thermal generation and optical properties. An additional concern is that the mesochlorobenzene ring that provides increased stability to the structure may also be a source of increased toxicity. To our knowledge, studies of toxicity of IR820 on cellular systems are absent from the literature despite the fact that they are an important prelude to in vivo applications.
The aims of this study were to determine the feasibility of using IR820 as a substitute for ICG in imaging and photothermotherapy applications, to perform a general characterization of IR820 that can serve as a foundation for these applications, and to provide a preliminary in vitro toxicity assessment. We performed an in vitro comparison of the two dyes with respect to their optical properties, thermal generation properties, stability profiles, and toxicity effects on cellular systems. Additionally, we directly compared the in vivo imaging characteristics of ICG and IR820 in a rat model.
Materials and Methods
Chemicals
ICG (molecular weight [MW] 775 Da, IR-125) was obtained from Fisher Scientific (Pittsburgh, PA). IR820 (MW 849.47 Da), methanol, formaldehyde, and dimethyl sulfoxide (DMSO) were purchased from Sigma (St. Louis, MO).
Physicochemical Characterization
Absorbance Studies
Dye solutions were prepared in deionized water or in methanol and samples were placed in 1.5 mL polystyrene cuvettes. Samples were diluted to the linear absorption range (< 3 AU), and absorption spectra from 200 to 900 nm were recorded using a CaryWinUV spectrophotometer (Varian/Agilent Technologies, Basel, Switzerland). Automatic blank correction was performed using the appropriate solvent (water or methanol). The instrument was allowed to warm up for 30 minutes before measurements, and operating conditions were kept constant for all experiments.
Fluorescent Spectrum Characterization
We prepared ICG and IR820 solutions in deionized water at 20 μg/mL and then performed serial dilutions to 0.078 μg/mL. The fluorescence spectrum of each sample was measured using a Fluorolog-3 spectrofluorometer (Jobin Yvon Horiba, Edison, NJ) in steady-state mode. The device was allowed to warm up for 30 minutes and then calibrated following the manufacturer's specifications for both the xenon lamp source and the expected Raman spectrum of water. Samples were placed in 4.5 mL polystyrene cuvettes and measured immediately after preparation. For ICG and IR820 samples, we measured emission after excitation at 785 nm because this wavelength is typically used in ICG imaging systems. For IR820, we performed an additional set of measurements in which we used the peak absorption wavelength for IR820 in water as the excitation wavelength. We recorded spectral emission readings up to 900 nm, in 1 nm intervals. Deionized water was used as a blank for background correction. The samples were handled under reduced lighting conditions at all times to avoid bleaching.
Effects of Temperature and Light on Stability in Aqueous Solution
To characterize the optical stability of ICG and IR820 in aqueous solution, we prepared solutions of ICG and IR820 as described above and selected a concentration in the linear range, specifically 0.625 μg/mL. Each sample (ICG or IR820) was aliquoted into five borosilicate glass containers, and each container was assigned to one of five groups: (1) room temperature with constant direct light, (2) room temperature covered with foil, (3) room temperature in the dark, (4) refrigerator, or (5) covered water bath at 42°C. A sample from each container was taken and measured at 24-hour intervals, following the same calibration and measurement procedure. Measurements of fluorescent intensity at the peak emission were done in triplicate. The instrument operating conditions were kept constant throughout the 4-day period. Data analysis followed the procedure described by Saxena and colleagues in their study of ICG degradation in aqueous solution. 6 Briefly, data were plotted as percent peak fluorescence remaining versus time in hours and fit to a pseudo–first-order kinetics model to obtain the observed degradation rate constant kobs. From this, the corresponding degradation half-time was calculated as t1/2 = ln(2)/kobs.
Stability in Different Solvents under Constant Light and Temperature Conditions
The stability of ICG and IR820 dissolved in DMSO or methanol over a period of 4 days was studied by selecting a concentration in the linear range, specifically 0.312 μg/mL, and placing a sample of each solution (ICG or IR820) at the chosen concentration in a borosilicate glass container in the refrigerator at 4°C in the dark. Samples from each container were measured at 24-hour intervals, following the same measurement procedure detailed above but using methanol or DMSO as the calibration blank as appropriate. Measurements were done in triplicate. The instrument operating conditions were kept constant throughout the 4-day period.
Heat Generation Properties
An NIR laser module source (RLDH808-1200-5, Roithner Laserthchnik Gmbh, Vienna, Austria) emitting at 808 nm with an output power of 1.0 watts was used for the heat generation studies. Details are provided in our previous work. 10 Briefly, the laser module was fixed to a clamp to direct NIR light from above. A heated stage insert (World Precision Instruments Inc, Sarasota, FL) was placed on a mobile stage positioned directly below the laser and used to keep a 96-well plate containing dye samples at a baseline temperature of 37°C. The laser, when operating, was enclosed in a box to prevent external emission of the NIR light and to avoid air circulation that would affect the temperature of the sample. The mobile stage (holding the heated insert) had an extension arm to the outside. The arm was used to move the location of the well plates, thereby allowing individual wells to be exposed to the NIR energy in succession without opening the box.
We prepared 2.5, 5, and 10 μM solutions of ICG and IR820. Each sample was added to 3 wells in a 96-well plate sitting on the heated stage insert and excited with the 808 nm laser at a power of 1,440 J/cm2. The temperature inside the well was measured with a thermocouple for the 180-second laser exposure, and the three well values for a given dye were averaged.
Cellular Experiments
Cytotoxicity Experiments
The cytotoxic effect of ICG or IR820 with and without laser treatment was studied in SKOV-3, MES-SA, and Dx5 cancer cells acquired from American Type Culture Collection (ATCC, Manassas, VA). The cells were cultured in McCoy's 5A medium supplemented with 10% fetal bovine serum (FBS) and 1% penicillin in a humidified incubator at 37°C and 5% CO2 and seeded into two 96-well plates, labeled plate A and plate B, at 5,000 to 8,000 cells per well. The plates were incubated overnight to allow for cell attachment.
On day 0, 24 hours after the initial cell seeding, the cells in plate A were fixed and a sulforodamine B (SRB) assay was performed using a TECAN plate reader (TECAN Systems Inc, San Jose, CA) at a wavelength of 530 nm to determine the control growth of untreated cells. The absorbance values of the wells containing the cells were background subtracted (from wells containing medium only) and averaged to give T0, defined as the initial amount of cells on day 0. Thus, plate A establishes a reference for the number of cells at the same time the experimental treatment is introduced in plate B. On day 0, at the same time plate A was fixed to provide the T0 value, plate B was treated with IR820 or ICG (final concentration of 0–10 μM). A final concentration of 0 μM indicates that no dye was added to the wells. Next, the cells in plate B were incubated at 37°C for approximately 2 hours, after which half of the wells in plate B were subjected to laser treatment for 1 minute. For each concentration of dye (0–10 μM), half of the wells received laser treatment. Thus, for the control cells where no dye was added, half of the cells were exposed to laser. This provided a laser, no-dye control group. Plate B was returned to a 37°C incubator for another 24 hours.
On day 1 (ie, 48 hours after initial cell seeding and 24 hours after laser treatment), the cell protein amount was measured by the SRB assay. Potential crosstalk between wells was eliminated by not using adjacent wells for cells that were exposed to laser treatment, thus ensuring that applying laser treatment to one well would not cause a temperature increase in another well. The SRB absorbance values were background subtracted to give T (absorbance of the treated cells) and C (absorbance of control cells not exposed to laser). All measurements were done in four wells per treatment.
The following formula was used to calculate the net cell growth: if T ≥ T0, net cell growth = (T – T0)/(C – T0); if T < T0, then net cell growth (or cell death) = (T – T0)/T0. T0 is determined from SRB measurements in plate A and represents the number of cells at the time treatment was initiated. C is determined from SRB measurements in plate B for the wells that were not exposed to either laser or dye, and T is the absorbance of the treated wells (laser treatment, dye treatment, or both). Net growth was plotted against dye concentration to show the toxicity effects as described by Monks and colleagues. 22 Statistical analysis (α = 0.05) was performed using a t-test to compare (1) treatment values with the control value and (2) laser treatment versus no treatment for the same concentration of dye (0–10 μM).
In Vitro Cell Imaging
SKOV-3, MES-SA, and Dx5 cell lines were purchased from ATCC and cultured following ATCC specifications with 10% FBS-supplemented McCoy's 5A medium in a 37°C humidified incubator at 5% CO2 level. Cells were seeded at a density of 4 × 104 cells per well (SKOV-3) or 1 × 105 cells per well (MES-SA, Dx5) in a 24-well plate over poly-D-lysine precoated glass coverslips placed in each well and incubated overnight to reach confluence. The medium was removed and replaced with 0.5 mL of 10 μM IR820 or ICG solution and incubated for 30 minutes at 37°C without light exposure. After incubation, cells were washed three times with Dulbecco's Phosphate Buffered Saline (DPBS) and fixed with 4% formaldehyde for 15 minutes at 37°C, followed by washing three times with DPBS. The coverslips were then removed and mounted on precleaned glass microslides with PROLONG antifade reagent/mounting medium mixture. Then the specimens were observed using a fluorescent microscope (Olympus IX81, Olympus, Tokyo, Japan) with a 60× water immersion objective and images were recorded with a charge-coupled device (CCD) camera. The same filter set (λex = 775 nm and λem = 845 nm) was used for imaging of both fluorescence dyes. The CCD camera settings and exposure times were the same for both dyes for a given cell line (1,000 ms for SKOV-3 and 4,000 ms for MES-SA and Dx5). Green pseudocolor was added using EZ-C1 software (Nikon, Melville, NY).
In Vivo Imaging Studies
An imaging system consisting of a Sanyo DL 7140-201S laser (80 mW, 785 nm) and a Retiga 1300 CCD camera (QImaging, Surrey, BC) was used. The entire setup was covered by BK5 blackout material. The power at the imaging plane ranged from 0.1 to 0.5 mW. An equimolar solution of ICG or IR820 was prepared based on a dose of 0.24 mg/kg of body weight 13 and an injection volume of 0.25 mL. Wistar rats (160–224 g, 9 weeks old) were purchased from Harlan (Indianapolis, IN), kept under standard housing conditions, and fed ad libitum. All protocols followed the regulations of the Institutional Animal Care and Use Committee. Rats were randomly assigned to groups. On the day of the experiment, the animals were first anesthetized by inhalation of isoflurane in a closed chamber and then injected intraperitoneally with a 40 mg/kg dose of pentobarbital. The chest and abdomen were shaved, and the animal was placed in a supine position facing the imaging setup.
An image of the rat was obtained under white light illumination to determine the position of the target and to focus the camera before dye injection. Then the laser was turned on and another image was taken prior to dye injection to establish a background. Laser current was set to 60 mA. After the white and background images were obtained, ICG or IR820 dye was injected through the tail vein using a three-way stopcock system connected to two syringes, one containing the dye and another containing saline solution to flush the tail vein after injection. The camera started recording immediately before the injection, and 10-second exposure images of the abdomen were recorded in series using QCapture Pro software (QImaging) for at least 40 minutes or until the rat regained consciousness. An image focused on the chest was also taken before removing the animal from the setup on the day of the injection. Twenty-four hours later, the rat was imaged again (abdomen and chest) using the same imaging setup under the same operating conditions. The protocol was completed in six rats, three per dye. Additionally, organs were carefully dissected and extracted after the 24-hour imaging protocol was completed in the intact animal. The lungs, kidneys, and liver were then placed in black-coated Petri dishes and imaged using the same imaging setup.
Images were processed in Matlab (MathWorks, Natick, MA) to select a background-subtracted region of interest and calculate the normalized intensity, R, as the ratio of total pixel intensity (pixel index) in the region of interest to the area of the signal in μm 2 . To create the final figures, dye images were superimposed with their corresponding white image using Matlab. Statistical analysis was performed to compare average ratios for each dye (t-test, 95% confidence level).
Quantitative measurements were performed by dye extraction in DMSO following the procedure detailed by Saxena and colleagues for ICG. 15 Briefly, the organs were homogenized and then incubated for 4 hours in DMSO. After 4 hours, the samples were centrifuged at 4,500 rpm for 5 minutes, the supernatant was collected, and sample emission was measured in a spectrofluorometer after 785 nm excitation. Dye mass in micrograms obtained from this measurement was normalized to homogenized organ mass in grams, and statistical analysis was performed to determine significant differences in dye content (t-test at 95% confidence level). Dye mass was determined from a standard calibration curve prepared by adding known concentrations of dye to homogenized tissue samples of nontreated animals.
Results
Physicochemical Characterization
Absorbance Studies
Figure 2 shows the absorbance profiles of IR820 and ICG in methanol and deionized water. In methanol, the peak absorptions occur at 820 nm for IR820 and 779 nm for ICG. In water, the absorption maximum of IR820 experiences a significant hypsochromic shift and is located at 691 nm, whereas the ICG peak absorption is found at 784 nm.

Absorbance spectra of IR820 and indocyanine green (ICG) in deionized water (solid line) and methanol (dashed line).
Fluorescent Spectrum Characterization
Figure 3 shows curves of fluorescence emission intensity versus wavelength for different ICG (see Figure 3A) and IR820 (see Figure 3B) concentrations in deionized water after excitation at 785 nm. Both dyes showed quenching for concentrations above 1.25 μg/mL. Our results show that IR820 has a linear range similar to that of ICG. From these curves, we can also see that for a given concentration, IR820 has approximately 10 times lower fluorescent yield than ICG after excitation at 785 nm.

Fluorescence emission spectra of (A) indocyanine green and (B) IR820 solutions.
Table 1 shows peak emission location for different concentrations of ICG and IR820. For ICG, the peak emission wavelength changes with concentration, so more dilute samples show a shift of the emission peak toward shorter wavelengths. The shift in peak emission wavelength with changes in concentration is much smaller for IR820 samples, which maintained a predictable peak location between 823 and 822 nm.
Emission Peak Location for Aqueous Solutions of ICG and IR820

ICG = indocyanine green.
In addition to comparing the fluorescent spectrum of IR820 after excitation at 785 nm with ICG, we investigated the fluorescence properties of IR820 on excitation at its 691 nm peak absorption in water. The profiles are shown in Figure 4 and are similar to those obtained for IR820 aqueous solutions after excitation at 785 nm.

IR820 fluorescent emission spectrum after excitation at 691 nm.
Quenching occurs for concentrations above 1.25 μg/mL, and within the linear range, the IR820 peak emission is located at 822 to 823 nm. Fluorescence intensities are lower than those of comparable concentrations of ICG. However, a slight increase in fluorescence emission of approximately 13 to 16% was observed when compared to the IR820 aqueous solutions excited at 785 nm.
Effects of Temperature ana Light on Stability in Aqueous Solution
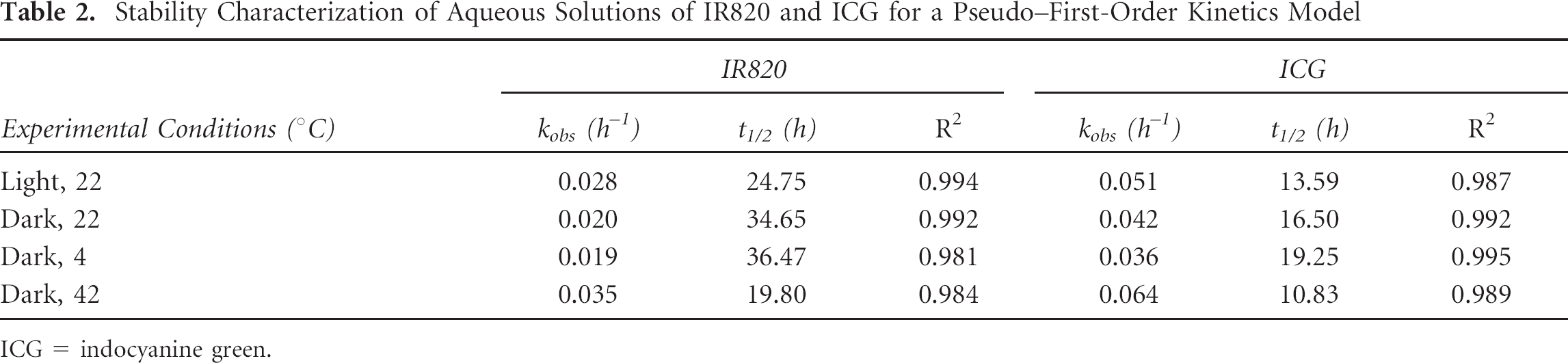
The degradation kinetics of IR820 and ICG are shown in Figure 5 (different temperatures) and Figure 6 (different lighting levels). The degradation profiles of both dyes follow pseudo–first-order kinetics with exponential decay. The fluorescence intensity of the remaining IR820 was consistently higher than that of ICG for all temperature and light conditions. This was reflected in degradation half-time values that were roughly double those of ICG, as shown in Table 2. Covering the solutions with foil protected them from degradation in a manner comparable to keeping them in the dark. Direct light exposure and high temperatures had similar degradation consequences for both dyes, with marked loss of peak fluorescence and loss of smooth spectral features by day 4.
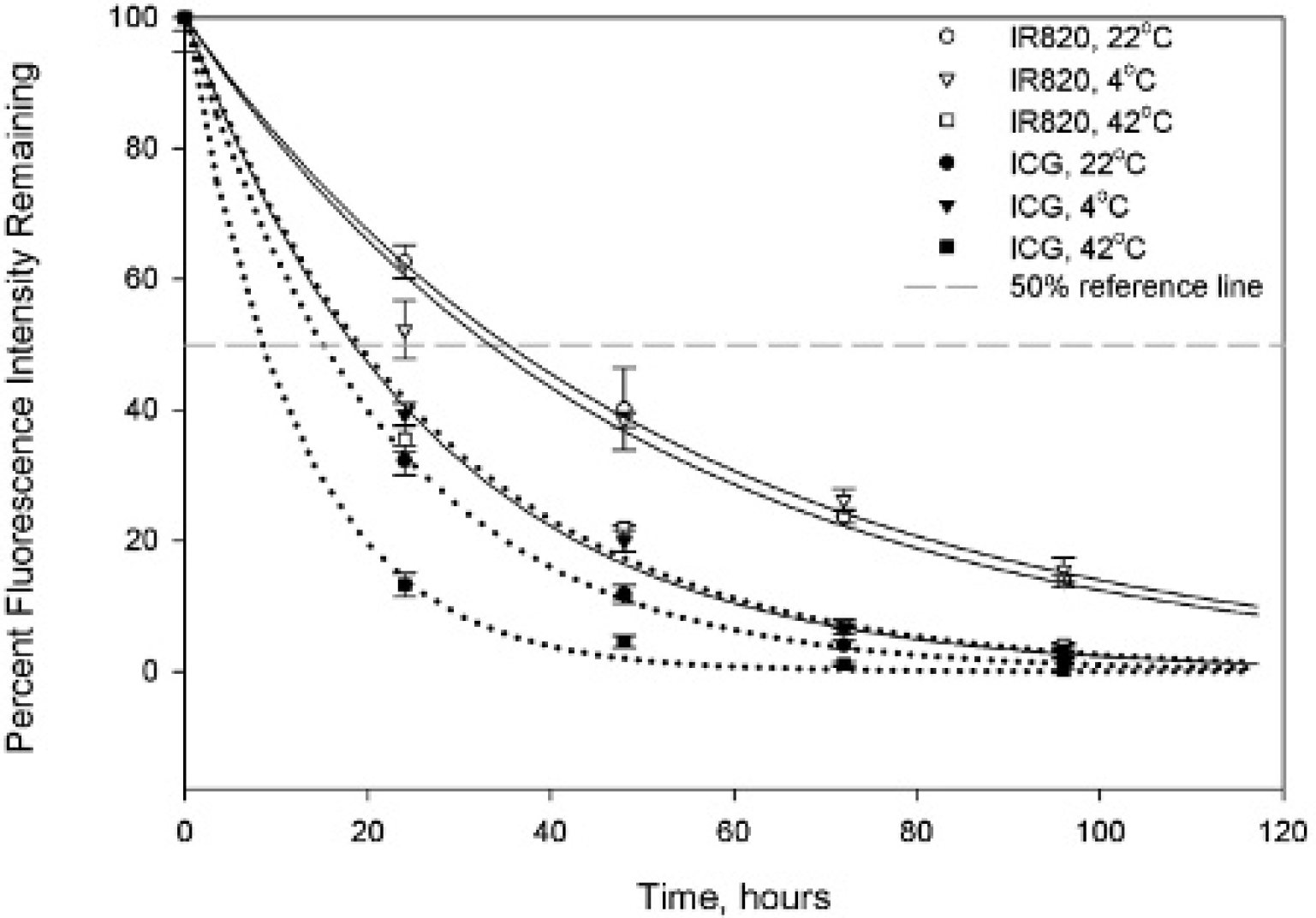
Effect of temperature on degradation of aqueous solutions of IR820 (solid lines) and indocyanine green (ICG; dotted lines). Error bars are ± SD.

Effect of light exposure on degradation of aqueous solutions of IR820 (solid lines) and indocyanine green (ICG; dotted lines) at room temperature. Error bars are ± SD.
Stability Characterization of Aqueous Solutions of IR820 and ICG for a Pseudo–First-Order Kinetics Model
ICG = indocyanine green.
Stability in Different Solvents under Constant Light and Temperature Conditions
ICG and IR820 were both stable in DMSO and methanol over a period of 4 days, with no significant decrease in fluorescence intensity when comparing samples measured on day 1 versus samples measured on day 4 (p > .05, paired t-test).
Hyperthermia Properties
Figure 7 reflects the temperature profile during 180 seconds of laser irradiation for a given dye (see Figure 7 A for IR820 and Figure 7B for ICG). The starting temperature was 37°C. For equivalent concentrations, IR820 produced a significantly smaller peak temperature increase than ICG (4–9% difference in peak temperature, p < .05 for comparisons between equivalent dye concentrations). Samples from the 5 μM stock solutions were tested 3 and 6 days after the initial experiment to investigate the stability of heat generation properties. During this time, the solutions were kept at 4°C in the dark. We followed the peak temperature over the 6-day period as a percentage of the original peak temperature. The hyperthermia properties of both dye solutions when irradiated with 808 nm light were stable over a period of 3 days, with no significant changes in peak temperature generation compared to the initial profile. After 6 days, there was a significant decrease in peak temperature for both dyes (p < .05 compared to the initial peak). Even though the decrease was significant with respect to the initial time point, both dyes still reached peak temperatures higher than 41 °C after 6 days.

Heat generation profile of wells containing IR820 (A) or indocyanine green (ICG) (B) when irradiated with near-infrared laser for 3 minutes.
Cellular Experiments
Cytotoxicity Experiments
Figure 8 shows that NIR laser treatment by itself without the addition of any dye to the wells did not cause any changes in cell viability for any of the cell lines. In the presence of ICG or IR820, the cytotoxicity profiles of SKOV-3 (see Figure 8A) and Dx5 (see Figure 8B) were very similar. In these two cell lines, IR820 or ICG without laser treatment did not cause significant decreases in cell viability up to 10 μM, when compared to the control group that received no dye and no laser treatment. Laser treatment of the wells containing IR820 or ICG caused cell growth inhibition at 5 μM dye concentration and resulted in marked cell growth inhibition or cell killing at 10 μM when compared to the control group that received laser treatment only.
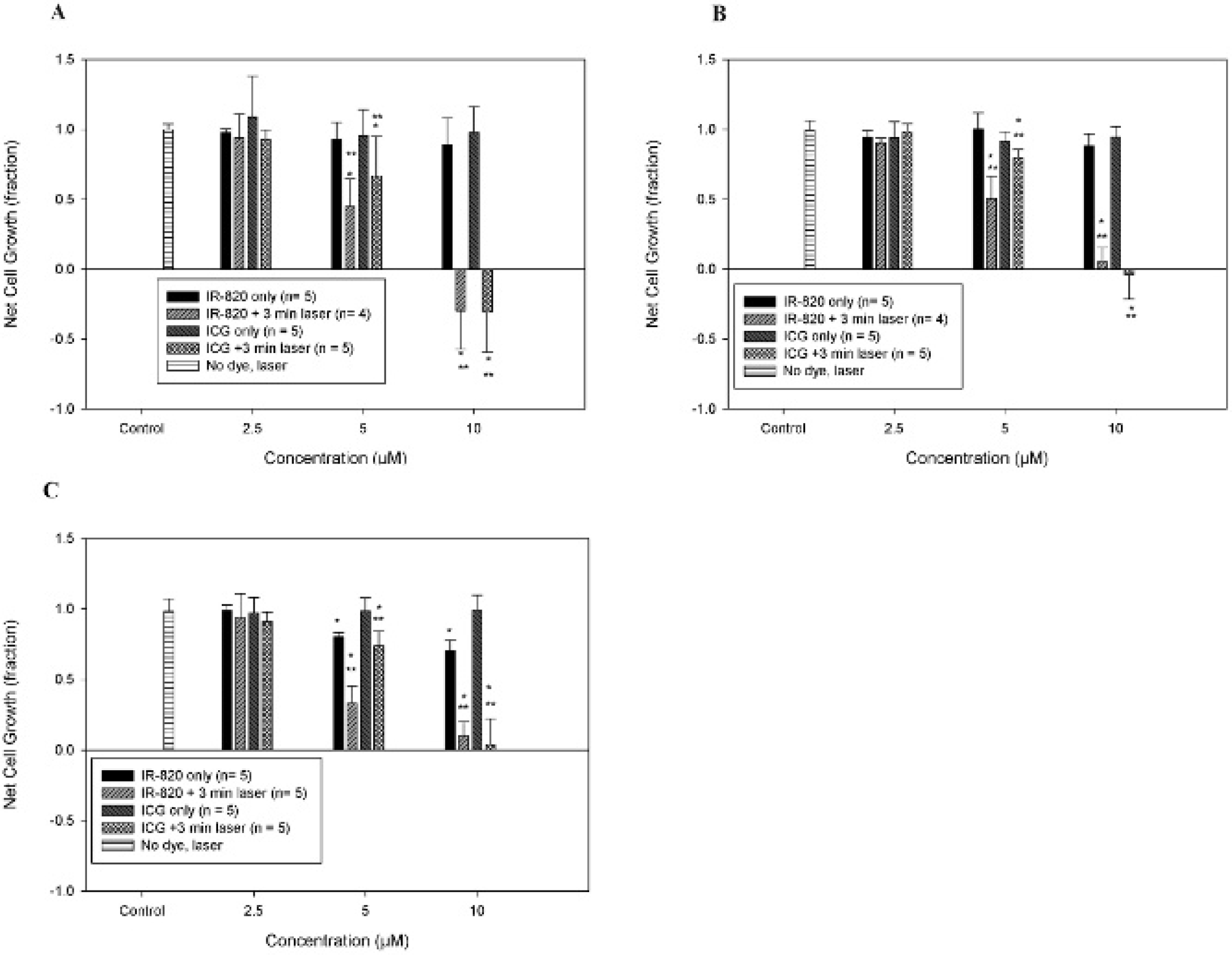
IR820 and indocyanine green (ICG) cytotoxicity and hyperthermia effects in SKOV-3 (A), Dx5 (B), and MES-SA (C) cells. Net cell growth was normalized to a control group that was not exposed to either dye or laser. The results for the group where laser was used in the absence of dye are shown in the graph above. No significant effect of laser exposure per se on net cell growth was observed. *Significant difference (p < .05) versus the control group. **Significant difference (p < .05) versus the same concentration of dye without laser.
In MES-SA cells (see Figure 8C), the response profile was comparable to the other two cell lines, except there was also significant (p < .05) cell growth inhibition on exposure to 5 or 10 μM IR820 dye without laser treatment.
For all cell lines, when comparing the effects of a given dye concentration with and without NIR laser treatment, cell proliferation was significantly lower in the NIR laser treatment group versus the dye-only group for both dyes at 5 and 10 μM.
In Vitro Cell Imaging
Figure 9 shows images of MES-SA, SKOV-3, and Dx5 cells after 30-minute incubation with IR820 or ICG. Image exposure times were well below the 20,000 ms threshold for cellular autofluorescence. Similar dye distributions were observed for all cell lines in the fluorescence microscope images. Both dyes resulted in widespread cytoplasmic fluorescence along with a localized perinuclear signal. The level of fluorescent brightness for both dyes allows for sensitive cell imaging of cancer cell lines.

A and D, SKOV-3 cells were incubated for 30 minutes with 10 μM IR820 and indocyanine green (ICG), respectively. B and E, Dx5 cells were incubated for 30 minutes with 10 μM IR820 and ICG, respectively. C and F, MES-SA cells were incubated for 30 minutes with 10 μM IR820 and ICG, respectively. λex = 775 nm and λem = 845 nm. Objective magnification, 60×.
In Vivo Imaging Studies
Figure 10 shows an image of IR820 (see Figure 10A) and ICG (see Figure 10B) signal coming from the rat abdomen 40 minutes after intravenous injection and excitation with a 60 mA laser module. Both dyes accumulate primarily in the liver. As expected from the lower quantum yield that was confirmed in the optical characterization, the normalized intensity of the IR820 image following injection is lower than for the ICG image for an equimolar injected dose. In the abdominal image taken after 24 hours, however, the normalized intensity of IR820 (see Figure 10C) is higher than the corresponding intensity for ICG (see Figure 10D). When the image is obtained from the chest, there is no significant signal from either dye at 40 to 45 minutes after injection. After 24 hours, however, there is a chest signal in IR820 animals, whereas ICG animals show no signal of dye in the chest.

Abdominal images of rats 40 minutes after intravenous injection with IR820 (A) or indocyanine green (ICG) (B) and 24 hours after injection with IR820 (C) or ICG (D). Image ratios indicate signal intensity normalized to area in μm2.
The normalized intensities for IR820 and ICG are shown in Table 3. The quantitative measurements confirm that the abdominal ICG signal is significantly stronger than the abdominal IR820 signal immediately after injection (p < .05), but the opposite is true 24 hours later. When the organs are excised and imaged after 24 hours, as shown in Figure 11, we can see that IR820 is present in the liver, lungs, and kidneys, whereas the ICG signal is limited mostly to the liver. This qualitative observation is confirmed by the normalized intensities, which demonstrate significantly higher values (p < .05) for IR820 compared to ICG in the liver, lungs, and kidneys.
Image Intensity per Area for Different Locations and Time Points after IR820 and ICG Administration

ICG = indocyanine green.
Values are average ± SD.
Significant difference between dye signals, p > .05, for a given location and time point.

Rat liver, lung, and kidney images 24 hours after injection with IR820 or indocyanine green (ICG). Ratios indicate signal intensity normalized to area in μm2.
Quantitative organ dye content data (Table 4) confirm that after 24 hours, the IR820 content (μg/g tissue) is significantly higher (p < .05) than the ICG content in liver and kidneys. For the lungs, the IR820 content was higher than the ICG content, but the difference did not reach statistical significance (p = .066).
Organ Dye Content 24 Hours after Injection

ICG = indocyanine green.
Values shown are average ± SD.
Significant difference, p < .05.
Discussion
Molecular imaging is an attractive, noninvasive diagnostic option, but it has disadvantages, such as poor tissue penetration that prevents deep tissue imaging. Among available probes, NIR fluorophores can be used in a window of maximal tissue penetration and have been the focus of extensive research for the development of optimized optical imaging techniques, with great potential for applications such as endoscopy, guided surgery, and lymphovascular imaging. 23 ICG is especially popular because it has been approved for clinical uses in imaging and diagnostics. Photostable fluorescent probes are an excellent choice to visualize and track complex tissue and cellular level processes. Fluorophores that can generate heat on exposure to NIR laser can also be used in combination imaging-therapy approaches because localized increases in temperature can kill cells or inhibit their growth. ICG has some disadvantages for biologic applications because of its short plasma residence time, instability in aqueous solutions, and concentration-dependent peak emission location. Thus, researchers have shown considerable interest in studying available NIR fluorophores and developing alternate new probes for physiologic systems. In our study, we investigated relevant optical and hyperthermia properties of IR820 versus ICG and compared both dyes in cellular and in vivo imaging applications.
The absorbance profiles of these two NIR probes in water and methanol show that the IR820 peak absorption undergoes a hypsochromic shift when the dye is dissolved in water versus methanol. This blue shift is not observed in ICG, but it has been reported to exist in some heptamethine cyanine dyes, possibly as a result of intramolecular charge transfer or excited-state proton transfer. 24 Considering this property, we expect that the absorption peak for in vivo applications is likely to be located close to the 691 nm peak observed in water, and the higher wavelength methanol peak would be relevant only for in vitro applications in nonphysiologic solvents.
The fluorescent emission profiles of IR820 show a lower quantum yield than ICG for equivalent concentrations. This was observed after 785 nm excitation (to compare with ICG) and after 691 nm excitation (at the optimal absorption peak of IR820 in water). The lower quantum yield after 785 nm excitation is not unexpected given that this wavelength is almost 100 nm away from the absorption peak of IR820 in water. The fact that excitation at the absorption peak wavelength gives higher peak fluorescence intensities, but still lower than those of ICG, indicates that there is also a structural factor contributing to the decreased yield. This is quite possibly related to the intrinsic low quantum yield of the chlorobenzene ring, 25 as well as to the internal heavy atom effect by which the presence of the chlorine moiety typically results in decreased quantum yields and fluorescent lifetimes. 26
An advantage of IR820 fluorescence emission is that the peak location does not seem to have a major dependence on concentration, whereas the ICG peak is more variable and shows a blue shift with decreasing concentration. This predictability of peak location for IR820 could facilitate the design of imaging systems targeting IR820 emission wavelengths by avoiding the peak fluctuation at different concentrations that is observed with ICG. The linear range for both dyes started at 1.25 μg/mL, which is consistent with Saxena and colleagues' reports of nonlinearity for ICG aqueous solutions at concentrations above 2 μg/mL. 6
The stability of ICG and IR820 in methanol and DMSO was excellent over a period of 4 days for both dyes. As expected based on the presence of the mesochlorobenzene ring that confers rigidity to the molecule, IR820 demonstrated higher stability than ICG in aqueous solution under all temperature and light conditions, with degradation half-times approximately double those of ICG for a given experimental condition. The degradation half-times for ICG in this study were consistent with those reported by Saxena and colleagues. 6 Our results showing that IR820 has increased stability compared to ICG are consistent with reports by Lee and colleagues that describe the effect of the structure of cyanine dyes on their fluorescent properties. 27 This research group found that rigidifying the molecular structure of a dye results in enhanced stabilization. Interestingly, keeping the IR820 solution at 4°C in the dark did not result in improved stability over storage of IR820 at room temperature in the dark, whereas there was a difference between the two experimental conditions for ICG. Therefore, light exposure seems to be the most important factor in determining degradation of IR820, rather than temperature, unless temperatures are as extreme as those of a water bath (42°C). At such elevated temperatures, heat collisions increase significantly and the molecules tend to aggregate closer together, leading to increased degradation of the fluorescence properties.
Regarding hyperthermia applications, IR820 did not generate peak temperatures as high as those of ICG after 3-minute laser exposure. However, at 5 and 10 μM, the IR820 temperatures were still within the hyperthermia range of 40 to 43 °C that is able to selectively impact tumor cell growth. Tumor cells are sensitive to temperatures in this range, whereas the growth of normal cells is not affected until temperatures reach higher levels. 28 Based on our results, a higher concentration of IR820 will be needed to attain the same temperature as ICG in hyperthermia applications. Our cytotoxicity data show that both IR820 and ICG hyperthermia cause cancer cell growth inhibition at 5 μM and marked cell growth inhibition or cell killing at 10 μM, and there is no statistical difference in the effect on net cell growth between IR820 and ICG hyperthermia for a given concentration, indicating that IR820 is a good substitute for ICG in therapeutic applications of hyperthermia. Interestingly, MES-SA cell growth was inhibited starting at 5 μM IR820 concentrations. This is likely due to the fact that MES-SA cells are more sensitive to environmental stressors than the other two cell lines, which are resistant to chemotherapy.
The stability of hyperthermia generation was comparable as well for both dyes over a period of 6 days. This is in contrast to the rapid decay of fluorescence properties in aqueous solution, indicating that nonradiative processes such as heat generation show a different behavior over time compared to radiative processes such as fluorescence.
When used in cell imaging at 10 μM, IR820 provides images that are very similar to those obtained with ICG. The cell images show some areas in the cytoplasm where the fluorescent intensity seems to be higher for both dyes. This may be due to intravesicle storage of the dyes 29 or binding to cytoplasmic proteins such as ligandin. 30 IR820 and ICG cell images had comparable intensities after 30-minute incubation, despite the fact that our spectrofluorometer studies show smaller emission intensities for IR820 compared to ICG at equivalent concentrations. It is possible that over longer incubation periods, IR820 could show an advantage over ICG based on its improved stability in aqueous solution.
In animal studies, the initial IR820 signal in the abdomen was lower than that of ICG, which was expected given the difference in in vitro fluorescent yields, as well as the fact that our imaging system is optimized for ICG imaging with excitation at 785 nm. We would expect that in physiologic systems, IR820 would display an absorption profile similar to its profile in aqueous solution, where the absorption peak is located around 691 nm. After 24 hours, we observed that the IR820 signal in the abdomen became stronger than the ICG signal, and the organ images and quantitative dye content analysis showed that IR820 was present in higher amounts than ICG on the day after injection. This may result in an advantage over ICG for imaging studies by providing longer image collection windows and is consistent with data reported by Prajapati and colleagues. 19 This group estimated a clearance half-life of 35 hours for IR820 in mice with 99% elimination at 8 days and observed that the fluorescence intensity of IR820 increases significantly when bound to serum albumin. 19
Our organ studies showed that both dyes are processed primarily in the liver, but IR820 seems to significantly pool in the kidneys as well. Interestingly, when images were taken from the chest area after 24 hours, there was a signal in the IR820 window, indicating that IR820 may be retained in the lungs in larger quantities than ICG. The lung image data are consistent with this observation and show a significant difference in intensity ratios for IR820 and ICG in the lungs (p < .05). However, lung dye content quantification did not reach statistical significant difference between IR820 and ICG content (p = .066). In one of the ICG experiments, small-point “hot spots” were observed in the lungs 24 hours after injection. This was probably due to the ICG forming aggregates in the blood that were trapped in the lungs. No ICG was detected in the lungs of the other two rats 24 hours after injection. But the failure to reach statistical significance may also be simply due to the inherent individual variability of in vivo studies, which can also be observed in the fluorescence images.
In experimental models of imaging and hyperthermia, IR820 could be an alternative to ICG in applications where greater stability, longer image collection times, or more predictable peak locations are desirable. The presence of the chlorobenzene ring may also make IR820 more amenable to conjugation chemistry compared to ICG and provide opportunities for preparation of conjugate formulations of the dye. Future studies will concentrate on the optimal dye concentration for dual in vivo imaging and hyperthermia treatment, which will depend on dye toxicity, hyperthermia tumor killing effect, plasma clearance, and instrument sensitivity. An additional consideration will be overcoming issues of in vivo attenuation, off-target effects, and the ability to reach appropriate target site concentrations. Potential approaches to solve these problems include targeted formulations using drug-loaded nanoparticles and endoscopic or orthoscopic delivery of light for imaging and inducing hyperthermia.
Footnotes
Acknowledgment
Financial disclosure of authors: This work was conducted using the facilities of the Biomedical Engineering Department at Florida International University and partially funded by the Florida Department of Health (Grant #08-BB-11), the Biomedical Engineering Young Inventor Award from the Wallace H. Coulter Foundation to R.M., the Florida International University Dissertation Year Fellowship to Y.T., and support from National Institutes of Health/National Institute of General Medical Sciences R25 GM061347 to A.F.-F.
Financial disclosure of reviewers: None reported.
