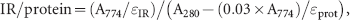Abstract
Given that overexpression of the epidermal growth factor receptor (EGFR) is found in many types of human epithelial cancers, noninvasive molecular imaging of this receptor is of great interest. A number of studies have employed monoclonal antibodies as probes; however, their characteristic long half-life in the bloodstream has encouraged the development of smaller probes. In this study, an anti-EGFR nanobody-based probe was developed and tested in comparison with cetuximab for application in optical molecular imaging. To this aim, the anti-EGFR nanobody 7D12 and cetuximab were conjugated to the near-infrared fluorophore IRDye800CW. 7D12-IR allowed the visualization of tumors as early as 30 minutes postinjection, whereas with cetuximab-IR, no signal above background was observed at the tumor site. Quantification of the IR-conjugated proteins in the tumors revealed ≈ 17% of injected dose per gram 2 hours after injection of 7D12-IR, which was significantly higher than the tumor uptake obtained 24 hours after injection of cetuximab-IR. This difference is associated with the superior penetration and distribution of 7D12-IR within the tumor. These results demonstrate that this anti-EGFR nanobody conjugated to the NIR fluorophore has excellent properties for rapid preclinical optical imaging, which holds promise for its future use as a complementary diagnostic tool in humans.
MOLECULAR IMAGING of cell surface receptors has become an important research area in oncology, contributing to the development of novel diagnostic tools that will ultimately help clinicians in the identification, delineation, and/or characterization of tumors. The epidermal growth factor receptor (EGFR) is a well-investigated transmembrane receptor that is known to be overexpressed in many types of human epithelial cancers, such as lung, head and neck, colorectal, and breast carcinomas.1–3 Several studies have reported on the development of tools for noninvasive detection of EGFR expression, mostly dedicated to nuclear imaging4,5 and based on the application of the anti-EGFR monoclonal antibody (mAb) cetuximab (Erbitux, ImClone Systems Incorporated, New York, NY).4,6–8 Although promising results have been obtained with intact mAbs in nuclear imaging applications, the characteristic slow clearance of these molecules from the bloodstream and their accumulation in the liver have stimulated further research. Smaller probes have been developed, based on antibody fragments or domains, that are expected to be cleared more rapidly, decreasing the exposure of normal tissues to radioactivity and enabling more rapid tumor imaging.9,10
Nanobodies 11 are the smallest functional antigen binding fragments derived from the naturally occurring heavy-chain-only antibodies that were first identified in the blood of llamas. 12 Nanobodies are also referred to as the variable domain of the heavy-chain of heavy-chain-only antibodies (VHHs). 13 Nanobodies have several interesting properties when considered for the development of probes for molecular imaging: (1) they are approximately 15 kDa, which is a tenth of the molecular weight of mAbs, so they have relatively short half-lives in the bloodstream 14 ; (2) although very small, nanobodies can bind very specifically to their targets, with affinities in the low nanomolar range 15 ; and (3) they are generally stable to temperature variations and chemical modifications, 16 enabling the conjugation of reporter moieties for imaging applications. Three studies have already reported promising results on the use of nanobodies as radioactive probes in nuclear imaging, allowing the visualization of tumor xenografts early after administration of the probes.17–19 The possibility of noninvasively detecting tumors and metastasis deeply located in the body (by means of whole-body scans), combined with the prospect of radiotherapy, makes nuclear medicine very powerful. Yet with some of the characteristics of nuclear imaging in mind (eg, limited resolution, ionizing radiation, short shelf life of probes owing to radioactive decay, and high costs), the development of nanobody-based probes for a complementary imaging modality has captured our interest.
Advances in optical imaging technologies 20 have stimulated the development of near-infrared (NIR) fluorescent targeted probes for in vivo optical molecular imaging. NIR fluorophores are excited at wavelengths between 700 and 1,000 nm, a range that allows deeper tissue penetration compared to visible light owing to reduced light absorption from tissue components and almost negligible autofluorescence.21,22 Besides not involving ionizing radiation, these NIR targeted probes are relatively easy to produce, and the technology required for imaging is relatively inexpensive. Furthermore, this imaging modality can be repetitively used to noninvasively determine the expression of molecular targets at different time points, facilitating diagnosis and/or assessing treatment response, or can even be employed in image-guided surgeries.23,24
In this study, the anti-EGFR nanobody 7D12 18 was conjugated to the NIR fluorophore IRDye800CW (IR), and 7D12-IR was evaluated as a probe for preclinical optical molecular imaging, in comparison with a control nanobody R216,25 and the anti-EGFR mAb cetuximab. In vitro, 7D12-IR was shown to bind specifically and with high affinity to human EGFR. In vivo, 7D12-IR allowed clear visualization of human tumor xenografts at much earlier time points after probe administration than cetuximab-IR, whereas the control R2-IR showed no tumor accumulation. Furthermore, the administration of 7D12-IR led to a relatively high tumor uptake that was correlated with a superior penetration and distribution of this probe within the tumor, compared to cetuximab-IR. These results highlight the potential of these nanobody-based probes for optical molecular imaging applications.
Materials and Methods
Nanobodies and Monoclonal Antibody
The nanobody 7D12 was previously described by Gainkam and colleagues and by Roovers and colleagues,18,26 and the nanobody R2 was described by Dolk and colleagues and Frenken and colleagues.16,25 Induction of protein expression and purification of nanobodies from the periplasmic space of Escherichia coli were performed as described by Roovers and colleagues. 27 The anti-EGFR mAb cetuximab was obtained from the VU University Medical Center (Amsterdam, the Netherlands).
Conjugation of the NIR Fluorophore IRDye800CW
The NIR fluorophore IRDye800CW (IR) was purchased as an N-hydroxysuccinimide (NHS) ester (LI-COR Biosciences, Lincoln, NE). The conjugation was performed with a fourfold molar excess of IR to the moles of protein, and the reaction mixture was incubated for 2 hours at room temperature. After conjugation, the nonconjugated IR was removed using two Zeba Spin Desalting columns (Thermo Fisher Scientific, Perbio Science Nederland B.V., Etten-Leur, the Netherlands) per protein in two sequential steps.
Characterization of IR-Conjugated Proteins
Gel permeation chromatography (GPC) was carried out on a Waters Aliance system (Waters, Milford, MA) with a Waters 2695 separating module and a Waters photodiode array detector set to record absorbance at 280 nm (protein) and 774 nm (IR) wavelengths. The column employed was a Superdex 75 10/300GL (GE Healthcare Europe GmbH, Munich, Germany), the elution buffer was phosphate-buffered saline (PBS), and data were analyzed with Empower 2 software (Waters).
SDS-PAGE gel electrophoresis was used to size-separate purified proteins (1 mg) and IR-conjugated proteins (0.5 μg) on a 15% polyacrylamide gel. Subsequently, proteins were stained with Coomassie Brilliant Blue solution (SERVA Electrophoresis GmbH, Heidelberg, Germany), and the gel was imaged on an Odyssey scanner (LI-COR Biosciences) using the 700 nm detector for the Coomassie stain and the 800 nm detector for the IR detection.
Absorbance of the samples (in PBS) was determined at 280 nm (A280) and 774 nm (A774) with a NanoDrop spectrophotometer (NanoDrop Technologies, Wilmington, DE). The degree of IR conjugation, that is, IR/protein molar ratio, was calculated with the formula
where the molar extinction coefficient of IR (ɛIR) is 240,000 M−1 cm−1 and the molar extinction coefficients for the proteins (ɛprot) are 33,920, 30,940, and 210,000 M−1 cm−1 for 7D12, R2, and cetuximab, respectively.
The stability of the IR-conjugated probes was assessed by storing aliquots of the IR-conjugated proteins at 37°C in 100% fetal bovine serum (FBS) (for up to 1 week) or at 4°C in PBS (for up to 14 weeks). The integrity of the IR-conjugated proteins and the ratio of free to conjugated IR was evaluated by SDS-PAGE.
Cell Lines and Culture Conditions
Three murine fibroblast cell lines were used: the NIH 3T3 cell line expressing murine EGFR from American Type Culture Collection (ATCC, Manassas, VA) (Cat. No. CRL-1658), the NIH 3T3 clone 2.2 selected for low endogenous EGFR expression, 28 and the HER14 cell line derived from NIH 33 fibroblasts stably expressing approximately 105 copies of the human EGFR on the cell surface. 28 In addition, three human tumor cell lines were employed: the human head and neck squamous cell carcinoma cell line UM-SCC-14C (developed by Dr. T.E. Carey, Ann Arbor, MI; abbreviated as 14C), the A431 human epidermoid carcinoma obtained from ATCC (Cat. No. CRL-1555), and the HeLa human cervical carcinoma cell line from ATCC (Cat. No. CCL-2). These cell lines were grown in Dulbecco's Modified Eagle's Medium (DMEM) (Invitrogen, Breda, the Netherlands) supplemented with 7.5% (v/v) FBS, 100 IU/mL penicillin, 100 μg/mL streptomycin, and 2 mM L-glutamine at 37°C in a humidified atmosphere containing 5% CO2. Tests for Mycoplasma infection were regularly performed, and cells were consistently found to be Mycoplasma free.
Binding Studies Performed with Living Cells
One day after seeding 8,000 cells per well in 96-well plates (NUNC AS, Roskilde, Denmark), plates were placed at 4°C, the medium was removed, and cells were washed with binding medium (DMEM supplemented with 25 mM Hepes and 1% BSA, at pH 7.2). Thereafter, IR-conjugated or unconjugated proteins diluted in binding medium were added to the cells in a concentration range of 0.2 to 50 nM and incubated on a rocker for 2 hours at 4°C. Then cells were washed twice with binding medium and fixed with 4% paraformaldehyde. For detection of 7D12 and R2, a mouse anti-myc antibody (Upstate, Millipore, Amsterdam, the Netherlands) was used, followed by incubation with an IR-conjugated donkey antimouse antibody (LI-COR Biosciences). For detection of cetuximab, a similar protocol was used with a rabbit antihuman IgG (Dakocytomation B.V., Heverlee, Belgium) as the primary antibody and an IR-conjugated goat antirabbit as the secondary antibody (LI-COR Biosciences). In the case of the IR-conjugated proteins, bound proteins were directly detected with the Odyssey scanner (800 nm channel). Cells were also incubated with the nucleic acid stain TO-PRO-3 (Invitrogen) for normalization of cell number, and TO-PRO-3 signal was detected with the 700 nm channel of the Odyssey scanner. Ratios of fluorescence intensities (800/700 nm) were plotted (in triplicate or quadruplicate ± SEM) versus the concentrations, and the resulting saturation curves were used to determine the apparent affinity of 7D12-IR and cetuximab-IR.
Competition studies were performed with 7D12-IR in the presence of epidermal growth factor (EGF) in equimolar or 10- or 100-fold molar excess, or with cetuximab preincubation, followed by 7D12-IR or cetuximab-IR incubation, as detailed in the text.
Near-infrared Microscopy
One day after seeding half a million 14C cells on gelatin-coated coverslips, cells were incubated with 10 nM of 7D12-IR, R2-IR, or cetuximab-IR in complete culture medium at 4°C for 2 hours. Then unbound protein was washed away at 4°C, and cells were fixed with 4% paraformaldehyde. Coverslips were mounted on microscope slides, and dried samples were stored at −20°C. Samples were imaged with an inverted microscope (TE2000, Nikon Europe), from which the Hg-excitation appendage was removed. Excitation was achieved by focusing light from a 760 nm high-power AlGaAs light-emitting diode (LED) (SMB760-1100-03-I, Epitex Inc. Japan) into a selected objective (60 × amplification) of the microscope. Excitation and IR fluorescence were separated using a dedicated dichroic filter set (set 41037, Chroma Technology, Germany). Images were recorded with an EMCCD camera (Ixon+, Andor, Ireland), and a shutter was used to synchronize the excitation-illumination with the recording interval to minimize bleaching.
Xenograft Mouse Model
Female athymic nude mice weighing 20 to 25 g (7–8 weeks of age) were obtained from Harlan Nederland (Horst, the Netherlands). Mice were housed in sterile cages under standard conditions (24°C, 60% relative humidity, 12-hour light/dark cycles) and provided with water and food ad libitum. The experiments were performed according to national regulations and approved by the local animal experiments ethical committee. A subcutaneous tumor was induced by inoculating 1 million cells of the A431 human epidermoid carcinoma cell line at the right and left hind legs. One week after tumor cell inoculation, tumors were approximately 100 to 200 mm3 and mice were randomly assigned to the different groups (as described below).
In Vivo Studies
Mice bearing A431 tumor xenografts at the hind legs were divided into three main groups: one group was injected with 25 μg of R2-IR, the second group with 25 μg of 7D12-IR, and the third group with 100 μg of cetuximab-IR. The dose of cetuximab-IR was selected based on previous studies (M. Stigter-van Walsum, personal communication, 2010), and the dose of IR-conjugated nanobodies was determined to be the optimal dose after preliminary studies employing 50, 25, and 10 μg of 7D12-IR.
For the imaging study, mice (four per group) were anesthetized with isofluorane and imaged before and right after the injection of the probes, as well as at 15 and 30 minutes and 1, 2, 3, 4, 5, 6, 24, 48, and 72 hours postinjection. Each image took less than a minute to be acquired. Images were obtained with two mice at a time, using the IVIS Lumina system with indocyanine green filter sets (Caliper Life Science, Hopkinton, MA), and data were analyzed with the Living Image software from Xenogen version 3.2 (Caliper LS). Regions of interest (ROI) were drawn around the tumor areas and in normal tissues, and the values of average fluorescence radiance (p/s/cm2/sr) from these ROI were used to calculate tumor to normal tissue (T/N) ratios, which were plotted in time; values represent the mean of eight tumors ± SEM.
For the biodistribution study, mice (five per group) were sacrificed by cervical dislocation at 2 and 24 hours postinjection. Tumors and organs were collected, weighed, snap-frozen in liquid nitrogen, and stored at −80°C until use. Tumors and organs were disrupted with the TissueLyser II system (Qiagen, Venlo, the Netherlands) using precooled Eppendorf holders, 5 mm stainless steel beads, and RIPA buffer supplemented with protease inhibitors. Thereafter, the quantity of each probe in each organ was determined by extrapolation of the unknown fluorescence values (Odyssey scanner) from the respective calibration curves using the software Prism 5 (GraphPad Software Inc., La Jolla, CA). These values were subsequently used to calculate tumor to organ ratios.
The EGFR binding specificity of 7D12-IR was assessed in vivo by means of a preinjection of unconjugated cetuximab (1 mg, intraperitoneally) 48 hours prior to the injection of cetuximab-IR or 7D12-IR (five mice per group, at doses described above). Then, 2 hours after the injection of 7D12-IR or 24 hours after the injection of cetuximab-IR, mice were sacrificed and tumors were collected for quantification of the 7D12-IR and cetuximab-IR present in the tumors.
Immunohistochemistry
Frozen A431 tumors were cryosectioned (5 μm thickness) with a Leica cryostat (Leica Microsystems B.V., Rijswijk, the Netherlands). Sections were fixed with acetone, and nonspecific immunoglobulin binding was blocked by incubating the sections with 2% normal rabbit serum (Dakocytomation B.V.) in PBS with 1% BSA. Sections were then incubated with the primary antibody, a rabbit antihuman IgG antibody (Dakocytomation B.V., Cat. No. A0424), followed by incubation with the secondary antibody, a goat antirabbit antibody coupled to horseradish peroxidase (HRP) (Dakocytomation B.V.). After washing, the sections were incubated with the substrate for the HRP chromagen reaction, diaminobenzidine (DAB) diluted in a ready-to-use DAB solution buffer (Sigma-Aldrich, Zwijndrecht, the Netherlands) until color developed. Nuclei were counterstained by incubation with hematoxylin solution (Sigma-Aldrich). Finally, sections were mounted with Kaiser's glycerol gelatin (Merck, Haarlem, the Netherlands) and sealed with a coverslip. The slides were visualized with a Nikon microscope (Nikon Instruments Europe B.V., Badhoevedorp, the Netherlands). Additionally, a number of frozen tumors collected 2 hours after injection of R2-IR and 7D12-IR and 24 hours after injection with cetuximab-IR were cryosectioned and immediately mounted on coverslips after fixation with acetone for imaging with the Odyssey scanner.
Statistical Analysis
Statistical analyses were performed using Prism 5 using analysis-based one-way analysis of variance with Bonferroni correction for multiple comparisons. A p value < .05 was considered significant.
Results
Characterization of the IRDye800CW-Conjugated Probes
To evaluate the potential of nanobodies as probes for optical molecular imaging of tumors, two different nanobodies were used: 7D12 and R2. 7D12 is a nanobody that binds specifically to the ectodomain of the EGFR. 18 It is also known to compete with EGF and with the mAb cetuximab for binding to EGFR.18,29 The nanobody R2 is directed against the copper-containing azo-dye RR616,30 and was herein employed as a non-EGFR binding (or negative) control. A direct comparison was made between 7D12 and the mAb cetuximab to determine the value of this small nanobody probe with respect to the conventional antibody format for molecular imaging applications. Both nanobodies and cetuximab were conjugated to the fluorophore IRDye800CW (IR); the IR-conjugated proteins are referred to as 7D12-IR, R2-IR, and cetuximab-IR.
After IR conjugation, proteins were characterized by GPC (Figure 1A). The peaks detected at 280 nm (protein, red line) and 774 nm (IR, green line) present overlapping retention times, confirming the conjugation of IR to the proteins. In each chromatogram, an extra peak is observed, which corresponds to free IR (at ≈ 50 minutes retention time). Although separation columns were used to remove unconjugated IR, a small percentage (lower than 7.5% of the IR-coupled fraction) was still detected in the GPC data analysis (Table 1). Furthermore, the random conjugation of IR to the proteins resulted in different degrees of IR conjugation for the different proteins, as indicated by the IR to protein molar ratios listed in Table 1, although the IR conjugation was revealed to be reproducible for each protein (data not shown). The IR-conjugated probes were also analyzed by gel electrophoresis (Figure 1B). For each protein, the band corresponding to the IR-conjugated protein observed in the gel (in green) migrated slightly more slowly, that is, had a slightly higher molecular weight, compared to the purified protein (in red). These results show the successful conjugation of the IR fluorophore to R2, 7D12, and both the light and heavy chains of cetuximab.

Characterization of IR-conjugated proteins. A, Samples of R2-IR, 7D12-IR, and cetuximab-IR were analyzed by gel permeation chromatography using a detector set to record absorbance at 280 nm (protein) and 774 nm (IR) wavelengths. B, Samples of purified and IR-conjugated proteins were size-separated by gel electrophoresis. Subsequently, proteins were stained with Coomassie Brilliant Blue. Red shows the Coomassie stain, and green corresponds to the IR signal (molecular weight: R2 and 7D12, approximately 15 kDa; cetuximab, ≈ 150 kDa with heavy and light chains of 50 kDa and 25 kDa, respectively; IRDye800CW-NHS, 1,066 Da).
Characterization of IR-Conjugated Probes by Gel Permeation Chromatography

Peaks were observed at different retention times according to the molecular weight of the proteins, and the areas under the curve for the peaks obtained at 774 nm (IR absorbance) are listed in the table. “Area Protein” corresponds to IR-conjugated and “Area Free” to the free IR fluorophore present in the samples. From those values, the percentage of free IR was calculated. Absorbance values of the samples at 280 and 774 nm were used to calculate the degree of IR conjugation, that is, the IR to protein (IR/protein) molar ratio, using the molar extinction coefficients and the formula described in the Materials and Methods.
Characterization of 7D12-IR Binding to EGFR
To characterize 7D12-IR binding to EGFR, binding studies were performed with several cell lines. It was clearly demonstrated that 7D12-IR binds specifically to human EGFR as the results presented for the two human EGFR-negative cell lines (NIH 3T3 and 3T3 2.2) showed no difference between 7D12-IR and R2-IR (Figure 2A). Furthermore, values plotted for the human tumor cell lines incubated with 7D12-IR were proportional to their EGFR expression levels, that is, A431 > 14C > HeLa, which strengthens EGFR specificity. The incubation of these cell lines with cetuximab-IR led to similar results, where the values obtained when bound to human EGFR are approximately 2.8-fold higher than the values obtained with 7D12-IR (consequence of the different degree of IR conjugation for the different proteins; see Figure 2A). As for the control nanobody, no binding of R2-IR was observed for any of the six cell lines evaluated, which confirms that R2-IR is a suitable negative control.

7D12-IR binds to human epidermal growth factor receptor (EGFR) and competes with epidermal growth factor (EGF) and cetuximab for binding to EGFR. A, Six cell lines were used for binding studies on living cells: three are murine fibroblasts (NIH 3T3, 3T3 2.2, HER14) and three are human tumor cell lines expressing different levels of EGFR (A431 > 14C > HeLa). Ratios of fluorescence intensities 800 nm/700 nm (ie, IR-bound protein/TO-PRO3 stain for correction cell number) in triplicate are plotted ± SEM, obtained after incubation with probes at 12.5 nM concentration. B, 14C cells were incubated with various concentrations of R2-IR, 7D12-IR, 7D12, cetuximab-IR, and cetuximab. Bound IR-conjugated proteins were directly detected, whereas cell-bound 7D12 and cetuximab (ie, unconjugated proteins) were detected through a primary antibody followed by an IR-conjugated secondary antibody. The graph shows fluorescence intensities (800 nm) in triplicate ± SEM, with increasing concentrations of the purified and IR-conjugated proteins. Apparent affinities are indicated, KD, in nM. C, Images taken with a microscope equipped with an NIR filter set for IR detection. Cells were incubated with the probes at 10 nM concentration (scale bars = 10 μm). D, 7D12-IR at 25 nM concentration was incubated with 14C cells in the presence of equimolar and 10- and 100-fold molar excess of EGF. The results in triplicate are presented as percentage relative to the fluorescence intensity obtained when cells were incubated with 7D12-IR alone. E, 14C cells were preincubated with unconjugated cetuximab at 4°C for 1 hour, followed by incubation with 7D12-IR or cetuximab-IR (positive control) in equimolar concentrations. The results in triplicate are presented as percentage relative to the fluorescence intensity obtained when no preincubation was performed.
To determine whether the IR conjugation had any detrimental effect on the binding properties of7D12, binding assays were also used to determine the apparent affinity (KD) ofboth IR-conjugated and unconjugated proteins for binding to cells presenting EGFR in its most natural environment (ie, avoid fixation or the use of cells in suspension). The apparent affinity determined for 7D12-IR was 2 nM and for cetuximab-IR was 0.5 nM (Figure 2B). The values of apparent affinity obtained for 7D12 and 7D12-IR were very similar, indicating that the conjugation of the fluorophore did not have a detrimental effect on its binding affinity. As for cetuximab and cetuximab-IR, a larger difference was observed in the apparent affinities, although the values obtained were still in the low nanomolar range.
To image cell binding of these IR-conjugated proteins at the microscopic level, a new wide-field microscope was specially adapted to operate in the NIR range of the spectrum. As could be expected from the above-mentioned results obtained with R2-IR, the control nanobody did not show any cell binding, whereas the pattern of fluorescence observed with either 7D12-IR or cetuximab-IR was similar (Figure 2C).
To assess if 7D12-IR remained antagonistic, a competition assay was performed with 7D12-IR in the presence of EGF. Increasing concentrations of EGF were able to reduce the binding of 7D12-IR: a 10-fold molar excess of EGF was able to reduce ≈ 90% of 7D12-IR bound to cells (Figure 2D). An additional assay was performed to assess the capacity of 7D12-IR to bind to cells after these were preincubated with cetuximab. The decreased percentage of bound 7D12-IR detected after preincubation of equimolar amounts of cetuximab (Figure 2E) confirms that 7D12-IR competes with cetuximab for binding to EGFR. These observations are in agreement with former studies performed with 7D12 29 and, together with the determined apparent affinities, indicate that the IR conjugation had no detrimental effect on EGFR binding capacity or on the specificity of the 7D12 nanobody.
EGFR-Targeted In Vivo Optical Molecular Imaging
To evaluate the potential of 7D12-IR as a probe for optical molecular imaging, mice bearing A431 human tumor xenografts at the hind legs (red arrows in Figure 3A) were intravenously injected in the tail vein with 7D12-IR and imaged at several time points postinjection. In parallel, the nonbinding nanobody R2-IR was used as a negative control and the anti-EGFR mAb cetuximab-IR was evaluated for comparison. Images taken as early as 30 minutes postinjection clearly showed IR fluorescence at the tumors in the case of 7D12-IR, where the accumulation of fluorescence was proportional to the size of the tumors, whereas no signal was detectable at the tumor sites for either R2-IR or cetuximab-IR (see Figure 3A). The clearest images, showing higher fluorescence at the tumors with better contrast, were obtained 2 hours postinjection of 7D12-IR and in the case of cetuximab-IR only at 24 hours postinjection. At 5 and 24 hours postinjection of7D12-IR, the fluorescence of the tumors had decreased to half and one-fourth of the intensity detected at 30 minutes postinjection, respectively. In contrast, in mice injected with cetuximab-IR, the average fluorescence intensity of the tumors was still increasing after 5 hours postinjection. No specific accumulation of R2-IR at the tumor sites was observed, which is in agreement with the in vitro data that showed no specific cell binding of this irrelevant nanobody to any of the cell lines employed (see Figure 2A). For both nanobodies, kidneys were clearly delineated at early time points (green arrows, see Figure 3A), which is expected from their small molecular weight, well below the threshold of kidney glomerular filtration (ie, < 50 kDa). In contrast, mice injected with cetuximab-IR presented clear fluorescence in the abdominal area, likely owing to liver accumulation of cetuximab (expected IgG metabolism).
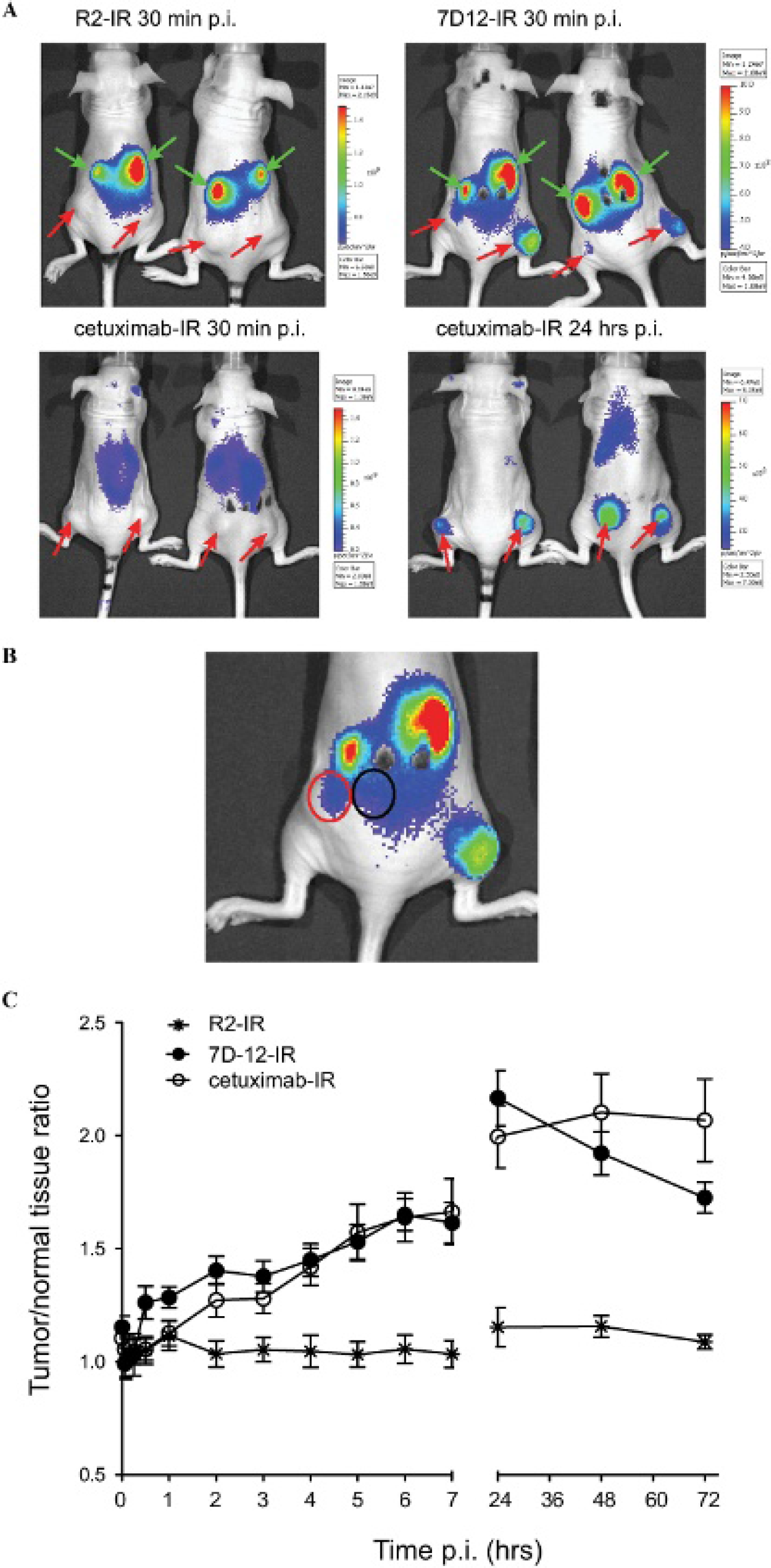
In vivo optical molecular imaging. Athymic nude mice bearing A431 human tumor xenografts at both hind legs were intravenously injected with 7D12-IR, R2-IR, or cetuximab-IR and imaged under anesthesia at several time points postinjection. A, Images of mice taken with the optical imaging system at 30 minutes and 24 hours postinjection (p.i.), in which tumors are indicated with red arrows and kidneys with green arrows. B, For all of the images obtained, regions of interest were drawn around the tumor areas and in normal tissues (exemplified with red and black line, respectively), and the corresponding values of average fluorescence radiance (p/s/cm2/sr) were used to calculate tumor to normal tissue ratios. C, These values are plotted in time postinjection.
Data collected with optical imaging, from 0 minutes up to 72 hours postinjection, were analyzed, and ROI, drawn around the tumor areas and in normal tissues, were used to determine T/N ratios. Here it should be noted that the proximity of the tumors to the kidneys was taken into account; that is, the ROI of normal tissues were drawn as close to the kidneys as were the ROI of the tumors (as depicted in Figure 3B with black and red lines, respectively). R2-IR presented T/N ratios over time of approximately 1 (Figure 3C), which confirms that R2 does not specifically accumulate at the tumors. Although moderate differences were observed between 7D12-IR and cetuximab-IR, it is interesting to note that 7D12-IR presented higher T/N ratios than cetuximab-IR at early time points (until 3 hours postinjection), whereas later than 24 hours postinjection, cetuximab-IR presented higher T/N ratios than 7D12-IR. These observations reflect the effect of the different half-lives of these IR-conjugated proteins in the bloodstream.
Biodistribution and Tumor Uptake
The observation of the clearest images at 2 and 24 hours postinjection of 7D12-IR and cetuximab-IR, respectively, led to a biodistribution study in which A431 tumors and several organs were collected at these time points after injection of the probes. After lysis and homogenization of the collected tumors and organs, the IR-conjugated proteins were quantified based on the fluorescence intensities, by extrapolation from calibration curves obtained with serial dilutions of the IR-conjugated proteins. To support this assay, the stability of the IR-conjugated proteins was assessed in PBS at 4°C for up to 100 days and in serum at 37°C for up to 7 days. The integrity of these IR-conjugated proteins was confirmed by means of gel electrophoresis (ie, the ratio of free to conjugated IR remained constant, and no visible breakdown of protein was observed).
As could be expected from the different molecular weights of the probes, at 2 hours postinjection, a large percentage of the injected dose was detected in the kidneys and in the urine of mice that were injected with the IR-conjugated nanobodies, whereas in the case of mice injected with the IR-conjugated mAb, the highest percentage of injected dose was detected in the liver (Figure 4A). The results obtained from the organs collected 24 hours postinjection suggest that the IR-conjugated nanobodies (and/or IR alone) are to some extent retained in the kidneys, with a small percentage still being excreted in the urine, whereas only trace amounts were detected in the blood 2 hours postinjection and even less at 24 hours postinjection. In contrast, IR-cetuximab was still well detectable in the blood 24 hours postinjection; it was highly present in the liver and also detected in well-perfused organs (spleen and lung; Figure 4B). IR fluorescence was also detected in the urine of mice injected with IR-cetuximab, both at 2 and 24 hours postinjection, which corresponded to free IR. Importantly, quantification of the IR-conjugated proteins in the tumors revealed ≈ 17% injected dose per gram (ID/g) as tumor uptake 2 hours after injection of 7D12-IR, whereas 24 hours after injection of cetuximab-IR, ≈ 10% ID/g of tumor was detected (the difference was statistically significant, p < .05). As for the control R2-IR, its tumor uptake at 2 hours postinjection resembles the percentage of R2-IR detected in well-perfused organs (see Figure 4A), indicating no preferential accumulation of this irrelevant nanobody in the tumor, as already concluded by the T/N ratios over time determined with the imaging experiment (see Figure 3C). Most importantly, these values indicate that the anti-EGFR nanobody 7D12-IR accumulates in the tumors earlier and to a greater extent than cetuximab-IR, highlighting the value of this small probe for optical imaging applications.

Biodistribution study and tumor to organ ratios. At 2 and 24 hours after intravenous injection of 7D12-IR, R2-IR, or cetuximab-IR, mice were sacrificed and their tumors and organs were collected for quantification of IR-conjugated proteins. Values are presented as percentage of injected dose (%ID) per gram of tissue/tumor ± SEM (five mice per group) (A) 2 and (B) 24 hours postinjection (p.i.). From the values obtained for tumors and organs or tissues, ratios of tumor to organs were calculated and plotted in these graphs for (C) 2 and (D) 24 hours postinjection time points.
Using the quantification of the IR-conjugated proteins in the organs and tumors, tumor to organ ratios were calculated. Interestingly, 2 hours after injection of the IR-conjugated probes, ratios of tumor to different organs and tissues were the highest for 7D12-IR, with, for instance, a tumor to blood ratio of approximately 20 (Figure 4C). As for cetuximab-IR or R2-IR, considerably lower ratios were found. Although the content of 7D12-IR decreased in the tumors and organs at 24 hours, compared to the amount at 2 hours postinjection, and the ratios obtained for cetuximab-IR increased moderately, ratios of tumor to organs or tissues remained the highest for 7D12-IR compared to cetuximab-IR (Figure 4D).
EGFR Specificity In Vivo and Tumor Distribution
To confirm that 7D12-IR and cetuximab-IR bind to the same epitope on EGFR in vivo, as has been observed in vitro (see Figure 2E), a preblocking experiment was performed that consisted of a preinjection of a high dose of cetuximab 48 hours prior to the injection of 7D12-IR or cetuximab-IR, in an attempt to block all of the available binding sites of the A431 tumors (ie, EGFR). The quantification of tumor uptake at 24 hours postinjection of cetuximab-IR resulted in a reduction of approximately 70% (Figure 5A), compared to tumor uptake of cetuximab-IR without preinjection of cetuximab. This confirms that cetuximab-IR and cetuximab bind to the same epitope on EGFR in vivo. In contrast, the quantification of 7D12-IR 2 hours after injection resulted in a reduction of less than 30% in tumor uptake, compared to the tumor uptake obtained when no preinjection of cetuximab had been applied beforehand.

Epidermal growth factor receptor (EGFR) specificity of 7D12-IR in vivo. A, Mice bearing A431 tumors were injected intraperitoneally with 1 mg cetuximab 48 hours prior to the intravenous injection of 7D12-IR or cetuximab-IR, at doses described previously. At 2 hours postinjection (p.i.) of 7D12-IR and 24 hours postinjection of cetuximab-IR, mice were sacrificed, tumors were collected, and tumor uptake was determined for each IR-conjugated protein. Values plotted are percentages of reduction of tumor uptake relative to the uptake obtained without preinjection of cetuximab. B, Sections of tumors obtained from mice that were injected with R2-IR or 7D12-IR at 2 hours postinjection, or cetuximab-IR at 24 hours postinjection imaged with the Odyssey scanner to detect IR fluorescence and to assess the distribution of these probes (scale bars = 100 μm). C, Immunohistochemistry (IHC) for detection of cetuximab on tumor sections obtained from mice injected with cetuximab-IR and the same sections ex vivo incubated with cetuximab as a control for maximum binding of cetuximab (cetuximab is shown colored in brown; scale bars = 100 μm).
To investigate whether the difficulty in preblocking 7D12-IR binding in vivo was due to a different distribution of the probes within the tumors, sections of tumors obtained from mice 2 hours after injection of 7D12-IR or R2-IR and 24 hours after injection of cetuximab-IR were imaged with the Odyssey scanner. It is interesting to see that the IR fluorescence of 7D12-IR is homogeneously distributed throughout the tumors, whereas in the case of R2-IR and cetuximab-IR, the fluorescence seems to be confined to the periphery and, in the latter case, in the core of the tumor (Figure 5B). Given that these images correspond to IR fluorescence and there is a possibility that a mixture of free IR and cetuximab-IR is present in the tumors obtained 24 hours postinjection, immunohistochemistry (IHC) was performed to stain tumor sections for detection of cetuximab. As a positive control, the tumor sections were also incubated ex vivo with cetuximab and were subjected to the same protocol for IHC. In fact, cetuximab was detected irregularly at the stroma, likely surrounding blood vessels, whereas in the positive control, cetuximab was homogeneously distributed throughout the tumors (Figure 5C). These observations highlight the limited tumor penetration of cetuximab, which can explain the fact that cetuximab only partially blocked 7D12-IR binding in vivo (see Figure 5A), and therefore support the superior capacity of 7D12-IR to penetrate and distribute within tumors.
Discussion
Noninvasive assessment of expression levels of cell surface receptors has become an important research area in oncology, contributing to the development of novel diagnostic tools that will, in due course, help clinicians in the identification and characterization of tumors and in the rational prescription of novel targeted therapies.31,32 EGFR has been widely accepted as a relevant target for cancer therapy, and despite some controversy on the value of the detection of EGFR expression to predict the response to anti-EGFR therapies,33,34 the development of molecular imaging tools for noninvasive detection of EGFR could contribute to the diagnosis of many cancers overexpressing EGFR and to treatment monitoring. Recent advances in optical imaging technologies have stimulated the development of NIR fluorescent probes directed to EGFR for noninvasive optical molecular imaging applications.35–41
In this study, the antagonistic anti-EGFR nanobody 7D12 was conjugated to an NIR fluorophore and evaluated as a probe for optical molecular imaging. 7D12-IR specifically bound human EGFR, and although this could be considered as a drawback for in vivo testing in a mouse model owing to a lack of binding to normal tissues (ie, murine EGFR), it was clearly shown that 7D12-IR can distinguish between different expression levels of EGFR in vitro: a more than 20-fold difference was observed in values obtained for the different human tumor cell lines (A431 > HeLa), suggesting that 7D12-IR will be able to distinguish between normal and elevated expression of EGFR in tissues (eg, human oral tumor–derived cell lines can express approximately 10-fold higher levels of EGFR per cell compared to normal oral epithelium 42 ).
Given that the fluorophore used here was conjugated by means of an NHS linker, the coupling reaction occurred at random. Therefore, the possibility of affecting the binding properties of a small protein such as 7D12 had to be considered. In vitro results showed that the IR conjugation had no detrimental effect on the binding properties of 7D12 (see Figure 2). However, given that the determination of the apparent affinities of IR-conjugated and unconjugated proteins was performed in two different experimental set-ups, comparisons should be made with caution (also with respect to cetuximab and cetuximab-IR). In this context, ongoing research in our laboratory is focused on the site-directed conjugation of the fluorophore to the nanobody. As 7D12-IR is, in fact, a mixture of IR-conjugated and unconjugated molecules (see Table 1; IR/protein < 1), the results reported here could likely be improved by a new modality of IR conjugation, possibly allowing optical imaging with lower doses of this probe.
As proof of principle, in vivo optical molecular imaging was evaluated with the widely employed A431 human tumor xenograft model. To the best of our knowledge, this is the first study in which a direct comparison is made between a nanobody and a conventional antibody in optical molecular imaging. As could be expected from the small molecular weight of 7D12-IR, which allows rapid distribution and fast clearance of unbound molecules, tumors were detected as early as 30 minutes postinjection, with a considerably high tumor uptake at 2 hours postinjection (see Figure 3 and Figure 4). The tumor uptake here obtained is higher than the one reported for 99mTc-7D12 18 and for 68Ga-7D12 (Vosjan and colleagues 43 ). Differences in conjugation efficiency, the nature of the tracer (compared to the fluorophore), and detection time after administration could perhaps explain these variations. Also, similar to what has been reported for mAbs, 44 the administration of high doses of these probes could result in lower tumor uptake when a saturation of the binding sites has been reached (50 mg in the study of Vosjan and colleagues 43 compared to 25 μg in this study).
In contrast to the results obtained in vivo with 7D12-IR, cetuximab-IR needed a longer period of time for accumulation of the probe in the tumors and clearance of unbound cetuximab-IR from the bloodstream. In fact, the already documented “binding site barrier” 45 in which monoclonal antibodies bind near the blood vessels, hampering the distribution throughout tissues, was also observed here by the staining of accumulated cetuximab in tumor sections (IHC; see Figure 5C). Moreover, the limited presence of cetuximab-IR in tumor sections was proven not to be due to the restricted availability of the antigen (ie, EGFR). This was shown by the ex vivo assessment of cetuximab binding to the tumor sections. In fact, it was due to the limited tumor penetration of cetuximab, also in agreement with a previous study. 46 The limited tumor penetration and distribution of cetuximab-IR contributed to a significantly lower tumor uptake at 24 hours postinjection compared to the tumor uptake of 7D12-IR 2 hours postinjection.
Different from what is generally done in preclinical nuclear imaging studies, where biodistribution studies are first carried out at several time points after probe administration and are then followed by single-photon emission computed tomography (SPECT) or positron emission tomography (PET) imaging at one particular time point (approximately 1 hour per scan), the rapid optical imaging technology (one image obtained in less than 1 minute) allowed the reverse order. In the present study, mice were first imaged at several time points after probe administration, and the biodistribution study was then performed at two particular time points (ie, 2 and 24 hours postinjection). Furthermore, unlike other optical imaging studies that assess tissue distribution in intact organs (semiquantitative) ex vivo, here the IR-conjugated probes were quantified in lysed organs. This technique allowed the determination of tumor uptake, which for 7D12-IR was more than fivefold higher of that of R2-IR. Although the results from the biodistribution study are quite comparable for 7D12-IR and R2-IR with respect to normal organs (comparable molecular weights), significant differences are observed in tumor uptake and in T/N ratios between these two nanobodies, showing no accumulation of R2-IR in the tumors. The values for tumor to organ ratios determined by the biodistribution study are higher than the values of T/N ratios determined through the analysis of the ROI (see Figure 3C), but factors such as the deepness of the tumors and the proximity of the tumors to the kidneys could have interfered with the determination of the real average radiance intensity per ROI. These observations reinforce the added value of the novel method here employed for quantification of the % ID/g tissue. However, at the same time, they highlight a limitation of generally used optical imaging systems, that is, quantification of fluorophores. Nevertheless, technological developments have been made, and new systems seem to already circumvent this problem. 47 In addition, the high stability of the IR-conjugated proteins should also be emphasized, and this allowed the use of the same batches of IR-conjugated protein throughout the in vitro and in vivo studies. In the perspective of clinical application, this would be a serious advantage for optical molecular imaging as large batches of probes could be produced and be ready to use for a long period of time.
Another aspect of clinical relevance is the context of the application of optical imaging: whole-body optical imaging is unlikely to be feasible. Nevertheless, the application of this imaging modality in superficial areas of the human body, as well as in hollow organs accessible by means of endoscopes, makes the application of optical imaging in the clinic very promising.48–50 For instance, one can easily foresee the application of optical molecular imaging of the breast: after a suspicious lesion has been detected through (self)-examination or with another imaging modality, independently of women's age and their breast density, molecular optical imaging could add valuable information for diagnosis. Furthermore, it may allow image-guided surgery to arrive at a higher rate of radical resections. 51
As an alternative to nanobodies, even smaller probes have been developed, namely affibodies,52,53 which are also capable of delineation of tumors at 30 minutes to 1 hour postinjection. According to the modeling of Schmidt and Wittrup, very small probes (molecular weight < 15 kDa) require very high binding affinities to allow efficient binding to the target before the rapid removal from the tissues and clearance from the bloodstream. 54 On the other hand, intermediate-size probes have also shown promising results in tumor imaging, for instance, with a single-chain antibody SNAP-tag fusion protein (molecular weight ≈ 45 kDa). 41 These studies highlight the potential of manipulating the affinity, molecular size, and half-life of these probes for imaging applications.
Conclusion
7D12-IR allows the visualization of tumors at much earlier time points than the monoclonal antibody cetuximab-IR, with a superior tumor distribution that leads to considerably high tumor uptake, thereby encouraging further development of nanobody-based probes for optical molecular imaging applications.
Footnotes
Acknowledgments
We would like to thank Dr. David Michael Olive and Dr. Haibiao Gong for interesting discussions.
Financial disclosure of authors: This research was supported by the Center for Translational Molecular Medicine (MAMMOTH project) and by the departments of Pathology and Radiology of the University Medical Center Utrecht.
Financial disclosure of reviewers: None reported.