Abstract
Multimodal bioluminescence (BLI) and single-photon emission computed tomography/computed tomography (SPECT/CT) imaging were investigated as means to monitor somatostatin receptor subtype 2 (SST2)-positive neuroendocrine tumors as both a subcutaneously implanted and a liver metastasis animal model in mice and rats. Ultimately, such a model will be of use for studying SST2-targeted peptide receptor radionuclide therapy (PRRT). CA20948 cells were transfected with a green fluorescent protein/luciferase plasmid construct. Cells were inoculated subcutaneously in the shoulder of nude mice: nontransfected cells in the left shoulder and transfected cells in the right shoulder. BLI, SPECT/CT imaging, biodistribution analysis, and ex vivo autoradiography of the tumors were performed. BLI and SPECT/CT imaging were also performed on an intrahepatic tumor model in the rat. Caliper volume measurement of transfected tumors could be correlated with BLI measurements (2 = .76). SPECT/CT imaging showed high levels of accumulation of 111In-DTPA-octreotide in control and transfected tumors, which was confirmed by biodistribution analysis and autoradiography. Subcapsular inoculation of transfected cells in rat liver resulted in an intrahepatic tumor, which could be visualized by both SPECT/CT and BLI. Transfection of CA20948 tumor cells did not alter the growth properties of the cell line or the expression of SST2. Transfected tumors could be clearly visualized by BLI and SPECT/CT imaging. The transfected SST2-positive tumor cell line could represent a novel preclinical model for tumor monitoring in studies that aim at further optimizing PRRT for neuroendocrine tumors.
VISUALIZATION of somatostatin receptor–overexpressing neuroendocrine tumors by radiolabeled somatostatin analogues is being applied clinically for tumor detection, staging, and selection of therapy. 1 Equipping these peptides with therapeutic radionuclides for peptide receptor radionuclide therapy (PRRT) rendered promising results in preclinical 2 and clinical studies. 3 Kwekkeboom and colleagues demonstrated an increase in overall survival ranging from 40 to 72 months from diagnosis, with only minor side effects in comparison with historical controls in a clinical trial using 177Lu-DOTA-tate for treatment of 310 patients with gastroenteropancreatic neuroendocrine tumor (GEPNET). 4 Patients with liver metastases of GEPNETs have a median overall survival of 2 to 4 years and hardly benefit from classic therapeutic options such as surgery and chemotherapy, making PRRT an interesting new treatment option.
To further improve PRRT, new approaches, such as locoregional administration of PRRT and PRRT in combination with chemotherapy, are being tested in preclinical tumor models and in the clinic.5–7 For this, we developed a somatostatin receptor subtype 2 (SST2)-positive rat pancreatic tumor of acinar origin, CA20948. 8 For the various preclinical evaluations, these tumors can be grown either subcutaneously or as a liver metastasis model on implantation of subcapsulary tumor in the liver. To monitor tumor size and receptor expression in these models in a sensitive and quantitative way, we aimed to apply nuclear imaging in combination with bioluminescence imaging (BLI). For nuclear imaging, single-photon emission computed tomography (SPECT) is used for molecular imaging of receptor status and computed tomography (CT) for three-dimensional anatomy imaging. To allow for BLI, CA20948 pancreatic tumor cells were stably transfected with a green fluorescent protein (GFP)/luciferase plasmid construct. Tumor models based on these tumor cells in mice and rats were characterized by multimodal BLI and SPECT/CT imaging using 111In-DTPA-octreotide (OctreoScan, Covidien, Petten, the Netherlands).
Methods
Plasmid and Tranfection
The firefly luceriferase gene luc+ was cloned as a ≈ 1.7 kb Nhe1-Xba1 fragment in the Nhe1 site of a pIRES2-EGFP vector, enabling the expression of a single bicistronic transcript that encodes both luciferase and GFP. CA20948 cells were transfected with the construct using lipofectamin (Invitrogen) and cultured under geneticin (G418) selection. The G418 concentration for selecting transfected tumor cells was based on the G418 concentration killing almost all CA20948-wt (wild type) cells in an in vitro cytotoxicity test (SRB [Sulphorodamine B], Sigma-Aldrich) of CA20948-wt cells treated for 24 hours at different concentrations of G418. GFP-positive clones were isolated, and a clone with high GFP/luciferase expression was selected and characterized in vitro for expression of SST2.
Internalization Experiments
Twenty-four hours before the experiment, 106 CA20948 cells were plated per well in six-well plates. The cells were then washed with 2 mL phosphate-buffered saline (PBS, 37°C) and incubated in 1 mL incubation medium (RPMI 01640 medium [Gibco BRL] supplemented with 1% bovine serum and 20 mM Hepes) containing 111In-DOTA-octreotate at a concentration of 10−9 M. To determine nonspecific internalization, cells were incubated with an excess of unlabeled peptide (10−6 M octreotate). Cellular uptake was stopped by removing medium from the cells, followed by washing twice with 2 mL PBS. To discriminate between internalized and noninternalized (surface bound) fraction, intact cells were incubated with 1 mL 20 mM sodium acetate (pH 5) as previously described. 9
Fluorescence Microscopy
Cells grown on fibronectin-coated glass coverslips were analyzed by fluorescence microscopy using a Zeiss Axiovert 100M microscope and an ORCA-II-ER camera (Hamamatsu).
Animals
NMRI Nu/Nu mice (body weight ≈ 27 g) were used. Lewis rats (body weight around 275 g) were used. Animals were kept under standard laboratory conditions (12 hours light/12 hours dark) and given a standard laboratory diet and water ad libitum. During SPECT/CT imaging and BLI, animals were placed on a heated bed (38°C) and kept under isoflurane anesthesia (2.5% at 0.6 L/min). The animal studies conformed with the Animal Welfare Committee requirements of our institution and were conducted following generally accepted guidelines.
Tumor Cells
CA20948 is a SST2-positive pancreatic tumor of acinar origin that was originally induced by azaserine and is transplantable in syngeneic Lewis rats or in nude mice. 8 The tumor is maintained in culture as a monolayer in Dulbecco's Modified Eagle's Medium (Gibco, Paisley, UK) supplemented with 10% fetal calf serum. Cells were harvested from stationary cultures with ethylenediaminetetraacetic acid (EDTA).
For tumor induction, 2 × 106 living CA20948 cells in 0.2 mL were injected subcutaneously in the shoulder of nude mice: nontransfected cells in the left shoulder and transfected cells (CA20948-luc) in the right shoulder. Tumor growth was measured by caliper (tumor volume = length × diameter2 × 0.4) and BLI.
For the liver metastasis model, two male Lewis rats (Harlan, Horst, the Netherlands) were used. All surgical and injection procedures used isoflurane/O2 anesthesia and a microsurgery microscope. During surgery, animals were kept warm with a heating pad. After laparotomy of the rats' upper abdomen by a 1.5 cm incision, the main liver lobe was fixated between two swabs and around 100 μL CA20948 tumor cells (1.5 × 106 cells) suspended in Matrigel basement membrane matrix (BD Biosciences, Breda, the Netherlands) were injected subcapsularly through a 27-gauge needle mounted on a 1 mL syringe. The abdomen was then closed by absorbable sutures.
Bioluminescence Imaging
BLI (IVIS 200, Xenogen, Hopkinton, MA) of tumor-bearing animals was performed after intraperitoneal injection of luciferin (0.15 mg/g body weight). During the procedure, animals were anesthetized with isoflurane and kept at 37°C.
SPECT/CT Imaging
SPECT/CT imaging was performed using the NanoSPECT/CT (Bioscan Inc., Washington, DC) 4 hours after intravenous injection of 111In-DTPA-octreotide (30 MBq/rat or mouse). The exact injected activity was determined by measuring the syringe in a dose calibrator before and after injection. Multipinhole mouse and rat collimators with nine pinholes (1.4 mm diameter for mice and 1.5 mm diameter for rats) per head were used with a matrix of 256 × 256 and 24 projections (1 minute per projection). CT was performed at 45 kV. SPECT scans were reconstructed iteratively using InVivoScope software version 1.32 (Bioscan) with medium noise reduction, a voxel size of 0.3 mm3, and standard reconstruction settings.
Tumor Uptake Quantification
Tumor uptake of 111In-DTPA-octreotide was performed by gamma-photon counting of the tumor using a LKB-1282-Compugammasystem (Perkin Elmer, Waltham, MA) 24 hours after injection (counting time was 60 s/tumor). Before counting, the tumor was weighed.
Ex Vivo Autoradiography
Intratumoral distribution of 111In-DTPA-octreotide was investigated by ex vivo autoradiography. Tumor tissue, isolated 24 hours after injection, was embedded in Tissue Tek (Sakura, Zoeterwoude, Netherlands) and quickly processed for cryosectioning. Sections (10 μm) were mounted on glass slides, which were placed on phosphor imaging screens (Packard Instruments Co., Meriden, CT) for 1 day. The screens were analyzed using a Cyclone phosphor imager and a computer-assisted OptiQuant 03.00 image processing system (Packard Instruments Co.).
Results
Transfected cells were selected by G418 resistance (400 μg/mL; Figure 1) and cloned. A clone with the highest GFP activity (Figure 2) was used for further experiments.

CA2094-wt (wild type) viability in vitro cultured with G418 in different concentrations. The concentration of 400 μg/mL was chosen as the optimal concentration for selecting CA20948-luc cells.

Green fluorescent protein expression in a CA20948-luc clone in vitro (scale bar = 50 μm).
Comparable SST2 receptor binding and internalization of 111In-DOTA-octreotate in the CA20948-wt and CA20948-luc cells were shown in an internalization experiment (Figure 3). Tumor growth of subcutaneous CA20948-luc and CA20948-wt tumor was monitored by caliper measurements and BLI and showed good correlation (R2 = .76) (Figure 4). At day 10 after inoculation, the CA20948-luc tumors on the right shoulder were clearly visible by BLI (Figure 5A), whereas the CA20948 tumors inoculated in the left shoulder were not. Both tumors were visualized by SPECT/CT imaging on intravenously injected 111In-DTPA-octreotide, showing comparable levels of uptake and thus SST2 expression by both the transfected and nontransfected tumors (Figure 5B).

Internalization assay: comparable membrane binding (Membrane) and internalization (Internalized) of 111In-DOTA-octreotate in CA20948-luc and -wt cells with and without blockade with 1,000 times excess octreotate. Data are presented as mean ± SEM.

Correlation of caliper and bioluminescence imaging measurements (R2 = .76).
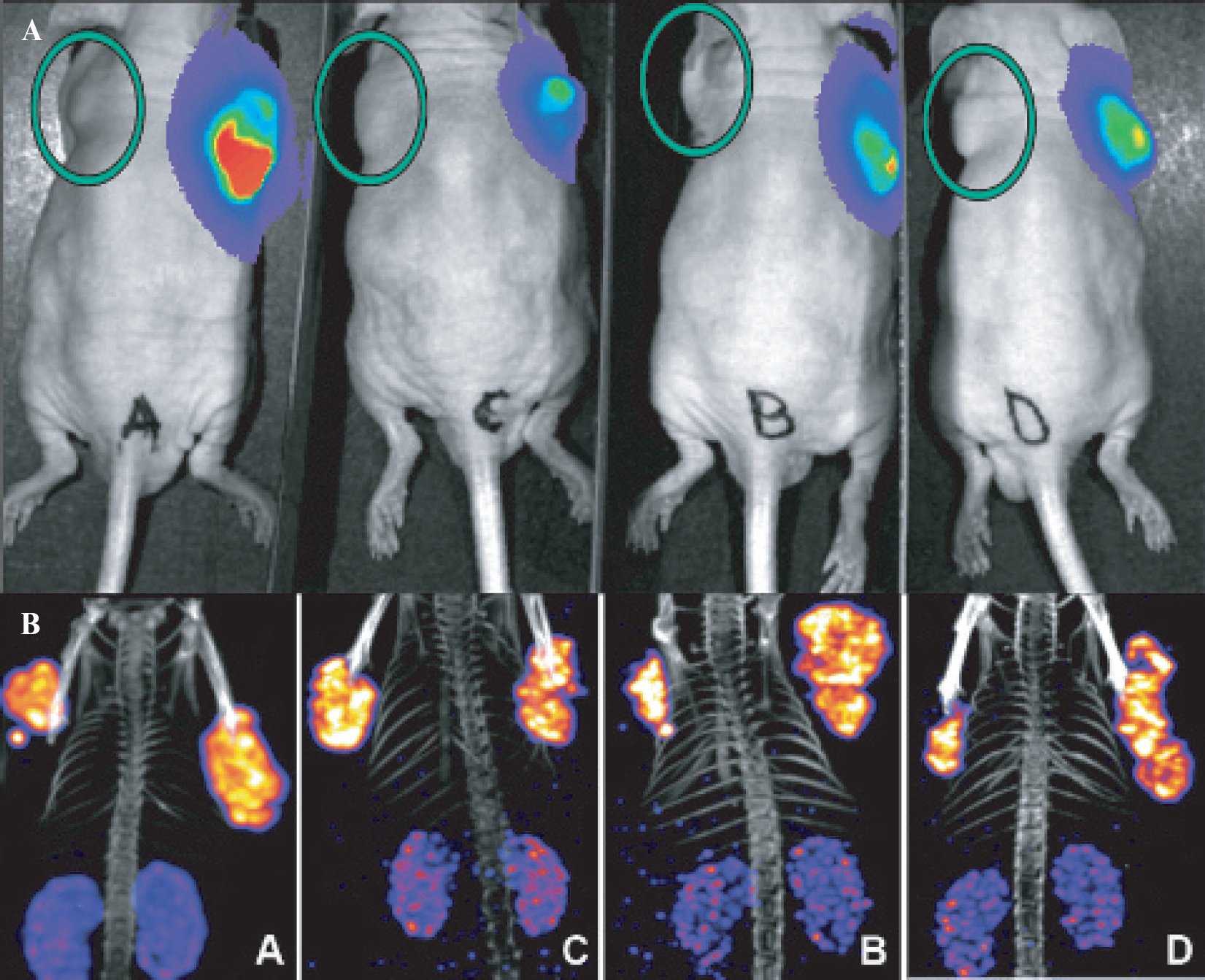
A, Bioluminescence imaging of 4 NMRI Nu/Nu mice 10 minutes after injection of luciferin showing luciferase activity in CA20948-luc tumor (right shoulder). On the left shoulder, the normal CA20948 tumor is visible, but without luciferase activity. The green circles indicate the CA20948-wt tumor. B, 111In-DTPA-octreotide uptake in the CA20948 (left shoulder) and the CA20948-luc (right shoulder) tumors 4 hours after injection.
Further comparison by ex vivo autoradiography of tumor sections (Figure 6) and by radioactivity counting of tumors 24 hours after injection (Figure 7) confirmed that levels of uptake of 111In-DTPA-octreotide were comparable in transfected and nontransfected tumors in vivo.

Ex vivo autoradiography of a CA20948-wt and a CA20948-luc tumor.

Tumor uptake of 111In-DTPA-octreotide in CA20948-wt and CA20948-luc tumors as quantified in a gamma counter 24 hours after injection.
An extra benefit of monitoring tumors by BLI is the possibility of monitoring orthotopically implanted tumors or metastastatic models. In a CA20948-luc liver metastasis rat model, intrahepatic tumor could be clearly visualized by both SPECT/CT (Figure 8A) and BLI (Figure 8B).
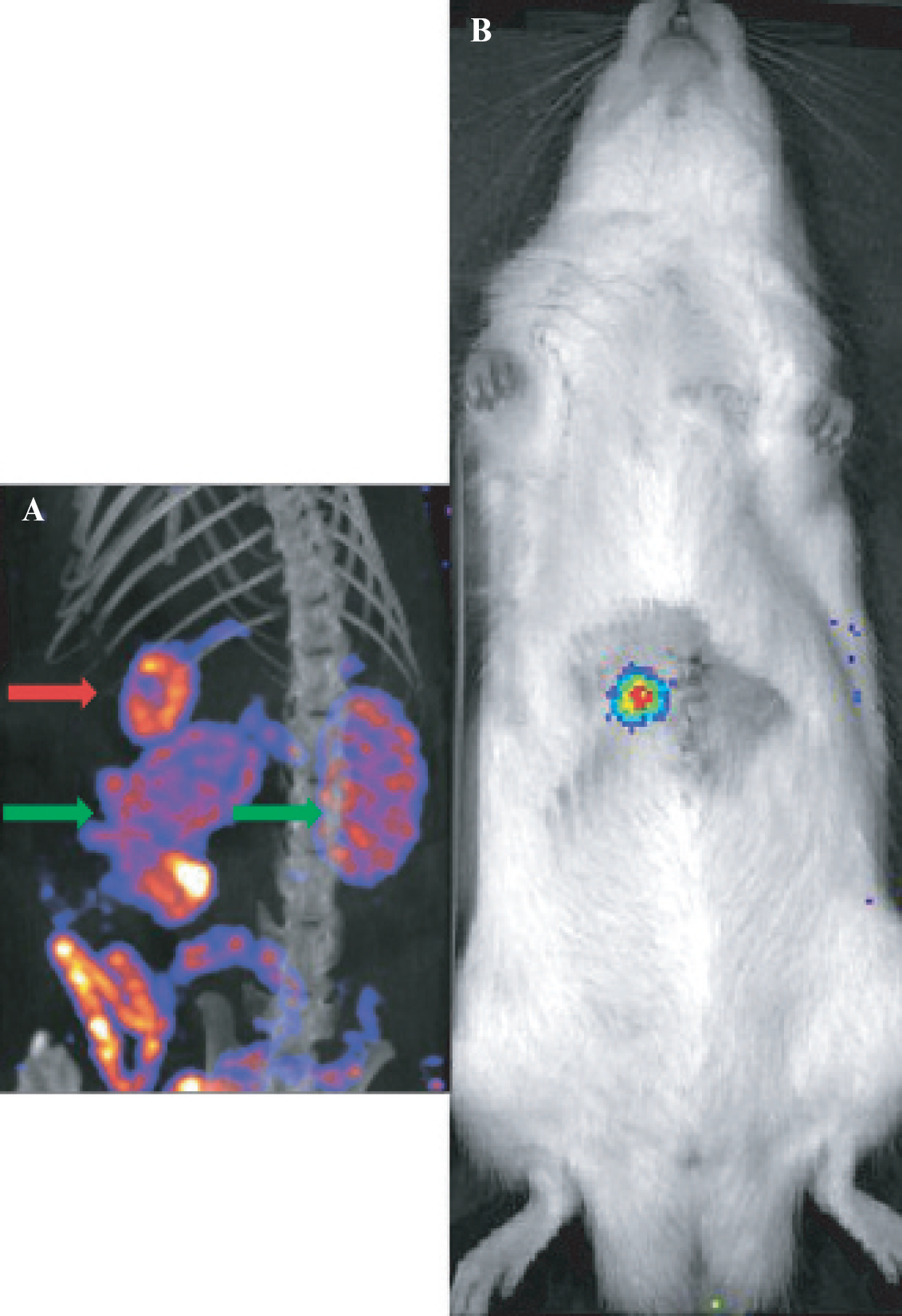
SPECT/CT (A) and bioluminescence (B) images of intrahepatic CA20948-luc tumor. Green arrows indicate the kidneys; the red arrow indicates the tumor.
Discussion and Conclusion
In this study, we were able to demonstrate that luciferase-transfected CA20948 pancreatic tumor cells show SST2 expression and 111In-DTPA-octreotide uptake in vivo comparable to that of nontransfected CA20948. Transfected tumors could be clearly visualized by both BLI and SPECT/CT imaging in mice and rats. BLI measurements of subcutaneous tumors in nude mice showed good correlation with caliper measurements. The use of BLI for measuring the therapeutic effects of PRRT in our animal models, especially in the liver metastasis model, is promising. This approach allows for daily monitoring of tumor growth in the same animal for several weeks. Tissue absorption of photons by overlying tissue could be a problem when the tumor is situated deep within the body. In the liver metastasis model used in this study, the tumor is covered by skin, abdominal wall, and liver capsule. The thickness of these structures is not expected to change in time. Emitted photons could be absorbed more and more by the tumor itself during growth of the tumor, making quantitative imaging uncertain. In our experiments, none of this was noticed. Other imaging modalities, such as CT and magnetic resonance imaging, could also be used for tumor size monitoring but are more time consuming. Using BLI, eight mice or four rats can be imaged simultaneously within 10 minutes. Taking these considerations into account, we conclude that BLI can be used in the future in addition to nuclear imaging techniques and measurements of receptor expression levels to follow tumor response to PRRT in a sensitive and quantitative way. This will help optimize PRRT experiments in the liver metastasis model and will prevent unwanted discomfort to the animals and the use of large numbers of animals when using alternative invasive methods. Also, caliper measurements of liver tumor size on surgery could vary considerably between measurements and researchers. BLI is not expected to have this variation when imaging is performed using a standardized imaging protocol. In addition, in PRRT experiments, we often experience central necrotic areas within a tumor that will be taken into account when using caliper measurements but not when using BLI.
The transfected tumor cell line could be a promising new preclinical tool for tumor monitoring in studies that aim at further optimizing PRRT for neuroendocrine tumors, especially when the tumor is not located directly under the skin, as in the case of orthotopically inoculated tumors or in a liver metastasis model.
Footnotes
Acknowledgment
Financial disclosure of authors and reviewers: None reported.
