Abstract
Although it has been well established that induction of growth-associated protein-43 (GAP-43) during development coincides with axonal outgrowth and early synapse formation, the existence of neuronal plasticity and neurite outgrowth in the adult central nervous system after injuries is more controversial. To visualize the processes of neuronal injury and repair in living animals, we generated reporter mice for bioluminescence and fluorescence imaging bearing the luc (luciferase) and gfp (green fluorescent protein) reporter genes under the control of the murine GAP-43 promoter. Reporter functionality was first observed during the development of transgenic embryos. Using in vivo bioluminescence and fluorescence imaging, we visualized induction of the GAP-43 signals from live embryos starting at E10.5, as well as neuronal responses to brain and peripheral nerve injuries (the signals peaked at 14 days postinjury). Moreover, three-dimensional analysis of the GAP-43 bioluminescent signal confirmed that it originated from brain structures affected by ischemic injury. The analysis of fluorescence signal at cellular level revealed colocalization between endogenous protein and the GAP-43-driven gfp transgene. Taken together, our results suggest that the GAP-43-luc/gfp reporter mouse represents a valid model system for real-time analysis of neurite outgrowth and the capacity of the adult nervous system to regenerate after injuries.
GROWTH-ASSOCIATED PROTEIN-43 (GAP-43; also known as B50, F1, neuromodulin, pp46, and P-57) is a neuron-specific phosphoprotein involved in neurite growth and plasticity-associated processes.1,2 GAP-43 is synthesized at high levels during axonal elongation and initial formation of synaptic connections in neuronal development. 3 Although, normally, its expression is mostly silent in the adult central nervous system (CNS), GAP-43 is strongly upregulated in adult injured neurons as part of the regenerative processes, as described in peripheral nerve/spinal cord 4 and brain injuries.5–8 Interestingly, deregulation of GAP-43 has also been observed in some neurodegenerative disorders, such as Alzheimer disease (AD),9,10 multiple sclerosis (MS), 11 and amyotrophic lateral sclerosis (ALS).12–14
Previous findings suggest that upregulation of GAP-43 may represent a suitable marker for the assessment of axonal damage and regenerative response in mature CNS neurons. 8 However, the exact spatial and temporal dynamics of the GAP-43 induction in the injured brain remain unclear, as well as the extent to which compensatory sprouting occurs after different types of brain injury and/or pathologies. Because the capacity of the adult brain to regenerate is a requirement for later functional recovery following injuries including stroke, there is a need for better understanding of the molecular mechanisms that modulate neuronal proregenerative signals. Recent work from our laboratory and others suggests that promoter-based bioluminescence is one of the best suited imaging strategies to study real-time events originating from deep brain structures in acute and chronic animal models of disease.15–19
To visualize neuronal responses to injury and repair in real time, we generated a transgenic mouse carrying the reporter genes luc (luciferase) and gfp (green fluorescent protein) under the transcriptional control of the murine GAP-43 neuron-specific promoter. We describe here a novel transgenic tool for in vivo bioluminescence and fluorescence imaging of neurite outgrowth during development and the neuronal responses to injury and repair in the adult nervous system.
Material and Methods
Generation of Transgenic Mice and Genotyping
The Nhe1/Sal1 1.7 kb fragment corresponding to the luciferase reporter gene (luc2) from pGL4 (Promega, Madison, WI) and the Sal1/Not1 0.7 kb fragment corresponding to the Aequorea coerulescens green fluorescent protein (AcGFP) reporter from pAcGFP1 (BD Biosciences, Mississauga, ON) were both inserted into the internal ribosome entry site (IRES) vector (BD Biosciences). A 10 kb fragment containing the murine GAP-43 promoter 20 was cloned into the pIRES-LUC2-AcGFP recombinant vector. The integrity of the final construct was verified by sequencing. The GAP-43-luciferase-GFP transgene was isolated as a Spe1/Cla1 fragment and microinjected into the male pronucleus of fertilized C57BL/6 oocytes. Transgenic mice were generated in the Transgenic and Knockout Facility of the Research Center of the Centre Hospitalier de l'Université Laval (CHUL). Transgenic animals were genotyped by polymerase chain reaction (PCR) detection of the luciferase reporter gene as previously described (primers 5′-GGCGCAGTAGGCAAGGTGGT-3’ and 5′-CAGCAGGATGCTCTCCAGTTC-3′).15,18
Surgical Procedures
Experimental Ischemia
Unilateral transcient focal cerebral ischemia was induced as previously described15,21,22 by intraluminal filament occlusion of the left middle cerebral artery for 1 hour followed by a reperfusion period. The body temperature was maintained at 37°C with a heating pad for 1 week after ischemia.
Sciatic Nerve Transection Model
As previously described, 17 the left sciatic nerve was completely transected using sharp scissors proximal to the branching of the tibial and common peroneal nerves under 2% isoflurane in 100% oxygen (2 L/min) anesthesia. All experimental procedures were approved by the Laval University Animal Care Ethics Committee and are in accordance with The Guide to the Care and Use of Experimental Animals of the Canadian Council on Animal Care.
In Vivo Bioluminescence Imaging
As previously described,15,17,18 the images were gathered using the IVIS 200 Imaging System (CaliperLS, Alameda, CA). The luciferase substrate
Ex Vivo Fluorescence
Taking advantage of the dual-reporter system, E14.5 embryos that were still light permissive were imaged on the same imaging system using a fluorescent excitation source. Ex vivo fluorescence images were acquired by 1-second exposure time using the GFP excitation/emission filters set (IVIS 200 Imaging System). 23
Tissue Collection and Immunofluorescence
Animals were anaesthetized by a chloral hydrate intraperitoneal injection (150 mg/kg, solution 10 mg/mL) and then transcardially perfused with 30 mL of 0.9% saline, followed by 4% paraformaldehyde (PFA) at pH 7.4 dissolved in phosphate-buffered saline (PBS). Tissue samples were then postfixed overnight in 4% PFA and equilibrated in PBS/30% sucrose for 48 hours. Tissues were cut with a microtome (25 µm thick) and stored at −20°C in antifreeze solution (5.75 mM sodium phosphate monobasic, 19.25 mM sodium phosphate dibasic, 30% ethylene glycol, 20% glycerol). Tissue sections from antifreeze solution were washed in PBS, mounted on microscopic glass slides, and then dried for 2 hours under vacuum. Sections were blocked in 1% bovine serum albumin/PBS and incubated overnight at room temperature using primary antibodies, mouse monoclonal antineuronal nuclei NeuN 1:500 (Millipore Chemicon), 1:750 rabbit polyclonal anti-Iba1 (Wako), 1:250 goat polyclonal anti-GAP-43 (Santa Cruz Biotechnology), 1:500 mouse monoclonal anti-GAP-43 (Sigma), 1:500 rabbit polyclonal anti–glial fibrillary acidic protein (GFAP; DakoCytomation), and 1:500 mouse monoclonal anti-GFP (Invitrogen) or 1:250 rabbit polyclonal anti-GFP (Millipore Chemicon). After PBS washes, sections were incubated in corresponding fluorescent secondary antibody (Invitrogen), rewashed, and covered with Fluoromount G medium (Electron Microscopy Sciences, Hatfield, PA).
Results
To visualize neuronal response to injury and repair in live animals, we generated a transgenic mouse bearing a dual bicistronic reporter system (firefly luciferase [luc] and GFP) under the transcriptional control of the murine GAP-43 promoter. A 10 kb fragment containing the mouse GAP-43-promoting sequences 20 was cloned into the bicistronic construct containing promoter-less luciferase PGL4 (1.7 kb) and AcGFP (0.7 kb) reporters (Figure 1A), with those two genes located on either side of the IRES and followed by the SV40 polyadenylation signal. We previously demonstrated that the dual (fluorescence/bioluminescence) reporter system represents an optimal and valid approach to studying processes in living animals. 18 The main advantage comes from the fact that optimal microscopic resolution can be achieved using fluorescent signal, whereas optimal bioluminescence spectral emission of firefly luciferase (mostly above 620 nm) is best suited for live imaging of the deep brain structures. The transgenic mice were genotyped using PCR with primers aiming at a segment of the luc transgene (Figure 1B). Three transgenic founders were obtained using this construct. To determine the functionality of the transgenic mice, the animals were screened for bioluminescence responsiveness of the reporter during development, following ischemia and peripheral nerve injury. Further analyses were focused on the line with the strongest signal induction and the appropriate GAP-43-driven transgene expression pattern.
In Vivo Bioluminescence and Fluorescence Imaging of the GAP-43 Signal in Neuronal Development
Because GAP-43 is strongly upregulated during neuronal development, we first assessed the functionality of the reporter by following the GAP-43 gene promoter activation by imaging transgenic embryos. Normally, the endogenous GAP-43 messenger ribonucleic acid (mRNA) is first detected at E8.5, whereas the protein is first observed from E9.5 in the ventral neural tube and cranial ganglia, becoming more widespread by day E12.5. 24 After birth, by postnatal day 7, the GAP-43 levels slowly decrease until adulthood. 25 To validate our reporter expression during development, we imaged at various time points the nontransgenic C57BL/6 female mice carrying transgenic embryos. The bioluminescent GAP-43 signal was first detectable at E10.5 (Figure 2A) and significantly increased later at E12.5 (Figure 2B) and E14.5 (Figure 2C), at which time point, the mice were sacrificed for ex vivo and histologic analysis of the embryos. Because of the small blood pool in the embryo, bioluminescent signal from the embryo was unstable and dropped quickly once luciferin supply from maternal circulation stopped. Alternatively, E14.5 embryos are still somewhat transparent and light permissive, so excitation with a fluorescent source and acquisition with the GFP filters allowed detection of a strong florescent reporter activity arising from the embryonic brain (Figure 2D). Importantly, immunofluorescence analysis of the developing brain at E14.5 revealed colocalization of the endogenous GAP-43 protein and the GFP reporter signals (Figure 2, E–H), clearly suggesting that our transgenic reporter was following the GAP-43 developmental expression pattern as described by Biffo and colleagues and Vanselow and colleagues.24,25
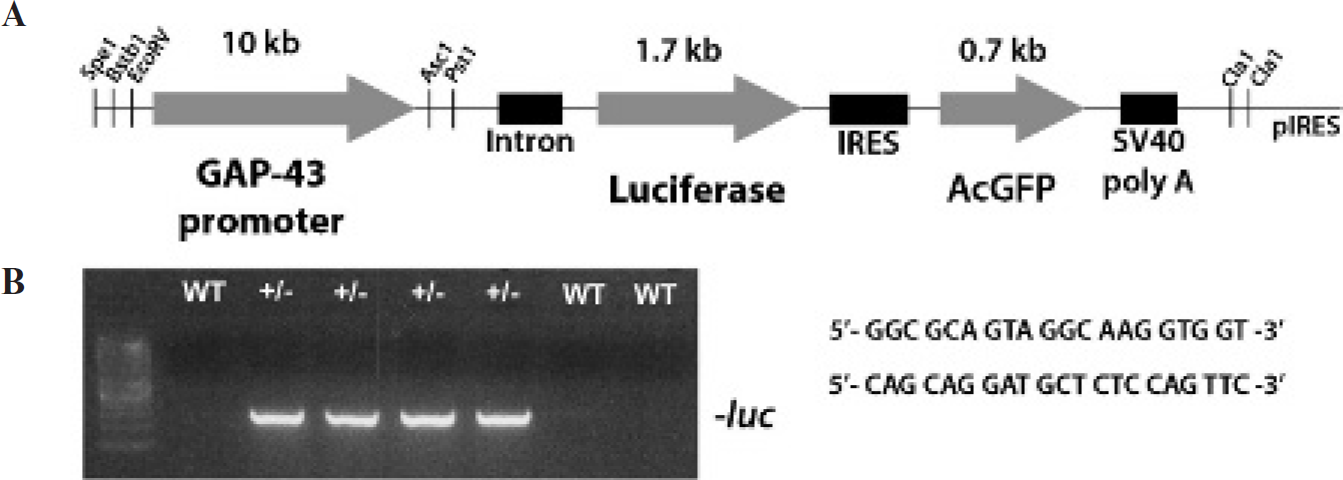
Generation of GAP-43-luc/gfp transgenic mice. A, Schematic representation of the DNA construct used to generate the GAP-43-luc/gfp transgenic mice. A 10 kb fragment containing the murine GAP-43 promoter was inserted in the recombinant vector containing the luciferase and the green fluorescent protein reporter genes located on either side of the internal ribosome entry site (IRES). The SV40 polyadenylation signal (SV40 poly A) is located downstream of the reporters genes. B, Genotyping of transgenic mice by polymerase chain reaction with primers designed to amplify a segment from the luciferase reporter gene.

Real-time imaging of GAP-43 induction during neuronal development. Bioluminescent in vivo imaging of GAP-43 activation during development of transgenic embryos at embryonic days E10.5 (A), E12.5 (B), and E14.5 (C). The images represent the same mouse imaged at different time points to follow bioluminescent progression during embryogenesis (A–C). Ex vivo fluorescence imaging of an E14.5 transgenic embryo show a strong green fluorescent protein (GFP) reporter signal from the developing brain. Fluorescence images were acquired using 1-second exposure and the GFP excitation/emission filters. For in vivo and ex vivo images, the scales on the right are the color maps of the source intensity (D). Representative photomicrographs of immunofluorescence staining of E14.5 transgenic brain section showing colocalization of the GFP reporter (green) and the endogenous GAP-43 (red); scale bar 5 100 µm, (E-H). Higher-magnification photomicrographs of (F) GFP staining, (G) GAP-43 staining, and (H) a merge showing both signals originating from the same cells; scale bar 5 50 µm.
GAP-43 Bioluminescent Signal: A Marker of Postinjury Neuronal Plasticity in the Adult Brain
Although mostly silent in the adult neurons, GAP-43 is strongly upregulated following acute neuronal damage. 8 Ischemic injury has been associated with axonal sprouting in the peri-infact cortex as part of the recovery and remapping process and reactivation of the expression of growth-promoting genes, including GAP-43. 8 To assess the basal levels of the endogenous GAP-43 protein expression in normal conditions, we analyzed GAP-43 and GFP immunoreactivities in several nonstroked control brains. In keeping with previous findings,26,27 analysis of the adult brains in normal conditions revealed a low basal level of the endogenous GAP-43 protein presented in fibrillary/punctate formations (Figure S1, A, D, G, and J [online version only]) and low levels of the gfp transgene immunoreactivity. More detailed, high-magnification immunofluorescence analysis of the different brain regions showed colocalization of the endogenous protein and the GAP-43-driven transgene GFP in the CA1 and CA3 pyramidal cells of the hippocampus (we did not observe GFP immunoreactivity in granule cells [Figure 1B-C]) (Figure S1, D–I [online version only]) and in some cortical neurons (Figure S1, J–L [online version only]). In addition, in agreement with previous work, 28 under normal conditions we also observed baseline levels of the endogenous GAP-43 and GFP immunoreactivity in large motoneurons of the spinal cord (Figure S1, M–O [online version only).
Next, to determine the functionality of the GAP-43-luc/gfp reporter mice, we subjected our transgenic mice to 1-hour transient middle cerebral artery occlusion (MCAO). Mice were longitudinally imaged over a period of 7 weeks. The imaging protocol was initiated shortly before stroke (baseline level measurements), and the same animals were longitudinally imaged at 24 hours, 72 hours, and 5, 7, 10, 14, 21, 28, and up to 50 days after the initial ischemic injury. Although preinjury mice showed a very low bioluminescence baseline level (mean 1.74 × 10e4 p/s) (Figure 3, A, E, and I), ischemic brain damage induced a marked and progressive increase in the intensity of a reporter signal (Figure 3, B–D, F–H, and J–L), reaching its peak expression around 2 weeks after the lesion (mean 1.52 × 10e5 p/s). The GAP-43 signal then slowly decreased but remained above the baseline levels for up to 7 weeks postinjury (Figure 3M). To determine whether photons detected by charge-coupled device camera after cerebral ischemia were emitted from the appropriate brain regions, we performed spectral imaging and a 3D reconstruction of the recorded signals using DLIT algorithms (see Material and Methods). As revealed in Figure 3F, the 3D signal reconstruction of the bioluminescent sources revealed that the GAP-43 signals were indeed arising from the areas surrounding the brain structures normally affected by MCAO, including parts of the frontal and parietal cortex, striatum, and hippocampus. These findings were further validated by immunohistochemistry analysis. Importantly, recorded signal distribution was consistent with our previous results obtained by in vivo imaging of different reporter mice following transient MCAO.15,18
To analyze GAP-43 signal induction at the cellular level, brains were collected at 1 week and 2 weeks after ischemia and were stained for immunofluorescent analysis. Focusing on the peri-infarct area of the cortex, we first observed coexpression of the GFP reporter and the endogenous neuron-specific GAP-43 (Figure 4, A–C). The GFP immunoreactivity also colocalized with the neuronal marker NeuN (Figure 4, D–F), further confirming the neuronal source of the bioluminescence signal. The neuronal specificity of the transgene expression was then assessed by analysis of the GFP expression in other cell types known to be highly responsive to brain damage, such as astrocytes and microglia. Histologic analysis of the peri-infarct region showed no colocalization of the GFP with the microglial marker Iba1 (Figure 4, G–I) or the astrocyte marker GFAP (Figure 4, J–L). In addition to analysis of the peri-infarct regions in the cortex, expression of the gfp transgene was also assessed in the hippocampus known to be involved in injury-induced plasticity in adult brain. Although no GFP signal was observed in control brains (Figure 5A), we first observed the upregulation of the GAP-43-driven GFP in the ipsilateral dentate gyrus at 1 week postinjury (Figure 5C). Two weeks after MCAO, the GFP immunoreactivity was markedly increased in both the ipsilateral and the contralateral hippocampus (Figure 5, D and E). Further analysis revealed that the majority of the GFP-positive cells were also expressing endogenous GAP-43 protein (Figure 5, F–H).
The functionality of the GAP-43-luc/gfp reporter mice was also assessed in the well-established model of peripheral nerve injury, a sciatic nerve axotomy. Previous studies have demonstrated a marked increase in the GAP-43 mRNA level following peripheral nerve injury starting after 2 days and returning to control levels by 28 days. 29 We subjected transgenic mice to a unilateral sciatic nerve axotomy and longitudinally analyzed the response by bioluminescence imaging. Following peripheral nerve injury, GAP-43 is upregulated in damaged axons and in the neuronal cell bodies of damaged axons localized in the spinal cord or for sensory axons in dorsal root ganglia. Imaging analysis revealed that after injury, the signal increased both at the site of the lesion (Figure 6, D and F) and in the spinal cord (Figure 6, C and E) (baseline mean 2.01 × 10e4 p/s in spinal cord) (Figure 6, A, B, and H). Because part of the signal at the site of the lesion may have been associated with the tissue reaction owing to a surgical procedure, we quantified upregulation of the GAP-43 bioluminescent signals arising from the spinal cord. The signal detected in the spinal cord reached its peak at 2 weeks (mean 6.99 × 10e4 p/s in spinal cord) and started to decline after 3 to 4 weeks postinjury (Figure 6H). To confirm the origin and the specificity of the signal for the site of the injury, we proceeded by ex vivo imaging of the spinal cord and sciatic nerves (Figure 6S) (note that the right and undamaged contralateral nerve did not emit any signal). Further, to assess the cell specificity of the signal, we looked by immunofluorescence for the expression of the GFP in the lumbar spinal cord 2 weeks after the lesion. Analysis of the ventral horn area revealed large cells corresponding to motoneuron cell bodies, expressing both the GFP reporter and endogenous GAP-43 protein (Figure 6, U–W).

Real-time imaging of GAP-43 induction following transient middle cerebral artery occlusion (MCAO). In vivo bioluminescence imaging of GAP-43 induction after MCAO shows activation of the GAP-43 promoter up to 7 weeks postischemia. Images represent photographs of the three GAP-43 reporter mice taken at different times after MCAO. Note the occurrence of different scales in various ranges (A–L). Measurement of luciferase activity (photons per second, p/s) at the site of ischemia revealed peak activity around 2 weeks after MCAO followed by a slow decrease in reporter activity at 6 to 8 weeks after the initial stroke. The solid blue line shows the GAP-43 induction after MCAO, whereas the dotted line indicates the control baseline values (mean ± SEM n = 10–18), p < .05* compared to control (M). Three-dimensional (3D) reconstruction of bioluminescent signal 2 weeks after MCAO in GAP-43-luc/gfp mice. Using spectral imaging, DLIT algorithms, and structural images of the mouse, the data were transformed into 3D images. The blue structures represent the areas of the brain with the highest intensity of photon emission. The localization of the highest-intensity area was measured and presented in three axes (x, y, and z; see smaller panels) in millimeters from the skull surface (small panels on the left). The scales on the right are the color maps for source intensity (N–Q).

GAP-43 expression pattern in brain section following middle cerebral artery occlusion (MCAO) in transgenic mice. Immunofluorescent analysis of the ischemic penumbra 2 weeks following the MCAO-induced lesion. Analysis of the immunofluorescence staining revealed colocalization of the green fluorescent protein (GFP) reporter with the endogenous GAP-43 neuron-specific protein (A–C), and with NeuN, a neuron-specific marker (D–F); scale bar = 100 µm. Based on the immunofluorescence analysis of the stroked brain section, the reporter expression was observed only in neuronal cells. Glial markers were assessed, and in the peri-infarct region. GFP was not found in Iba1-positive microglial cells (G–I) or in GFAP-positive astrocytes (J–L); scale bar = 50 µm.

Ischemic injury is associated with the marked green fluorescent protein (GFP) transgene induction in the hippocampus. Immunofluorescent analysis of the hippocampus following the middle cerebral artery occlusion (MCAO)-induced lesion. In the control mice without lesions, the GFP reporter expression was not detectable in the CA1–CA3 regions of the hippocampus or in the dentate gyrus (A); scale bar = 100 µm. Following MCAO, GFP immunoreactivity was first observed at 1 week in the ipsilateral dentate gyrus (C), although it was still absent in the contralateral structures (B). Two weeks after the lesion, GFP was strongly upregulated in both the ipsilateral (D) and the contralateral (E) hippocampus; scale = 100 µm. Immunofluorescence analysis revealed colocalization of the GFP reporter with the endogenous GAP-43 protein (F–H; see white arrows); scale = 50 µm.
Discussion
Here we describe the GAP-43-luc/gfp reporter mouse, a novel transgenic tool for real-time assessment of brain regenerative responses to injuries. Using in vivo bioluminescence and fluorescence imaging, we were able to visualize induction of the GAP-43 bioluminescence signals from the live embryos in utero starting at day E10.5, as well as the responses of the adult neurons to CNS and peripheral nerve injuries. Importantly, in all experimental paradigms, analysis of the fluorescence signal at the cellular level revealed coexpression between endogenous protein and the GAP-43-driven gfp transgene. Taken together, our results suggest that the GAP-43-luc/gfp reporter mouse represents a valid model system for real-time analysis of neurite outgrowth during development and in vivo assessment of a capacity of the adult nervous system to regenerate after injuries.
Although it has been well established that the induction of GAP-43 during development coincides with axonal outgrowth and early synapse formation, the existence of neuronal plasticity and neurite outgrowth in the adult CNS after injuries is more controversial. Previous reports have demonstrated that brain injury, spinal cord trauma, and/or injury to peripheral nerves are associated with the marked induction of growth and regeneration-promoting genes, including GAP-43.4–8,30 More specifically, after brain ischemia, upregulation of GAP-43 in the peri-infarct zone has been strongly associated with axonal sprouting and related functional recovery.7,8 Furthermore, overexpression of GAP-43 in the adult nervous system has been associated with increased nerve sprouting. 31
Using the GAP-43-luc/gfp reporter mouse, we were able to visualize the spatial and temporal dynamics of the GAP-43 proregenerative signals from the brains of living mice after stroke. The animals were longitudinally imaged over 7 weeks, and the initial pattern of the GAP-43 signal induction correlated with the findings of Carmichael and colleagues (GAP-43 staining substantially increased from day 3 to day 14 after stroke). 8 It is noteworthy that our study was extended to a longer time period and revealed for the first time increased GAP-43 promoter activity even 7 weeks after stroke. Similar temporal dynamics and results were obtained by monitoring the induction of the GAP-43 signal following peripheral nerve injury. The advantage of our in vivo model system is that unlike previous studies on brain repair that were based on single end-point data, the GAP-43 reporter mouse provides a novel powerful analytical tool for understanding real-time and longitudinal neuronal responses to injury. Further, because both luc and gfp reporters are expressed under transcriptional control of the murine GAP-43 promoter, in our transgenic model, we can also trace and identify reporter-emitting cells by detection of in vivo fluorescence and/or GFP staining by immunofluorescence analysis (see Figures 2, 4, and 6). Finally, the efficacy of the dual-reporter approach was demonstrated in our recent study. 18
To date, GAP-43 signal induction has been considered a valid and sensitive biomarker of neuronal sprouting and regenerative response in the mature brain following acute injuries. Interestingly, deregulation of the GAP-43 expression has also been observed in some chronic neurodegenerative disorders.9,10,12–14 Although the biologic and functional significance of the GAP-43 up-regulation in chronic neurologic disorders remains unclear, it is possible that it represents an attempt of damaged neurons to regenerate. Therefore, as an additional step, generation of GAP-43-AD and/or ALS reporter mice would certainly help us better understand the role of GAP-43 in chronic neurodegenerative disorders.
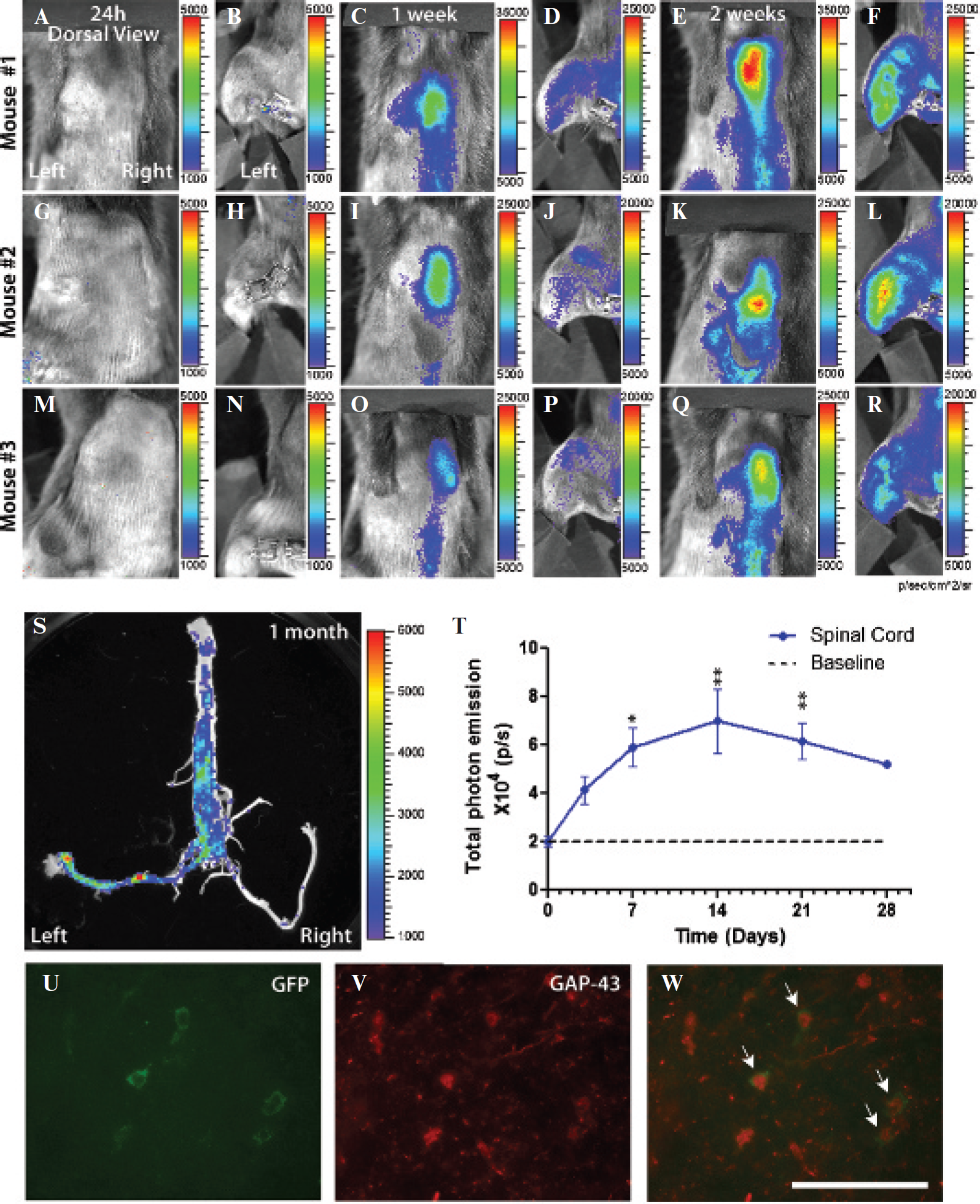
GAP-43 is upregulated in the spinal cord following sciatic nerve injury. Representative bioluminescence in vivo images of the three different GAP-43 reporter mice following a sciatic nerve injury. The same group of animals was followed over 28 days, and the signal was recorded from the spinal cord region and the hindlimb area (A–R). The first images were acquired 24 hours following the lesion (A, B, G, H, M, N). Images acquired 2 weeks following the sciatic nerve cut show strong upregulation of the GAP-43 bioluminescent signal in the spinal cord region (C, I, O) and the hindlimb area (D, J, P). Ex vivo imaging from a dissected spinal cord 1 month after the lesion revealed the specificity of the signal induction for the lesioned side. The left sciatic nerve was cut close to muscle, whereas the right nerve was uncut and used as a contralateral control (S). For in vivo and ex vivo images, the color calibrations at the right are the maps of photon counts. Measurement of luciferase activity (photons per second, p/s) coming from the spinal cord region revealed a marked increase at 2 weeks after the surgical cut followed by a slow decline at 28 days. The solid blue line shows the GAP-43 induction after middle cerebral artery occlusion, whereas the dotted line indicates the control baseline values (mean ± SEM n = 6–8); p < .05* compared to control (T). Immunofluorescence analysis of the imaged spinal cord sections confirmed colocalization of a transgene green fluorescent protein (GFP) (green, U) with an endogenous GAP-43 protein (V, W) (see white arrows); scale bar = 50 µm.
Conclusion
The GAP-43-luc/gfp reporter mouse represents a unique tool for noninvasive in vivo studies of neuronal damage and repair following acute brain injuries and in chronic neurodegenerative disorders. In addition, by using this transgenic tool, it will be possible to visualize the effects of various drugs to prevent damage owing to stroke, spinal cord injury, or neurologic diseases or to promote repair and neuronal regeneration in real time and in a noninvasive fashion.
Footnotes
Acknowledgments
We thank Dr. J.-P. Julien for providing us with the 10 kb GAP-43 murine gene promoter.
Financial disclosure of authors: This work was supported by operating grants from the Canadian Institutes of Health Research (CIHR), No. 93769 and 84355 (CIHR-Neuromuscular Partnership Funds). J.K. is a recipient of a Career Award from the R&D/Health Research Foundation and the CIHR. M.G. has a doctoral scholarship from CIHR-ALS Canada Partnership funds.
Financial disclosure of reviewers: None reported.
