Abstract
Bioluminescence imaging (BLI) of luciferase reporters provides a cost-effective and sensitive means to image biological processes. However, transport of luciferase substrates across the cell membrane does affect BLI readout intensity from intact living cells. To investigate the effect of ATP-binding cassette (ABC) transporters on BLI readout, we generated click beetle (cLuc), firefly (fLuc), Renilla (rLuc), and Gaussia (gLuc) luciferase HEK-293 reporter cells that overexpressed different ABC transporters (ABCB1, ABCC1, and ABCG2). In vitro studies showed a significant BLI intensity decrease in intact cells compared to cell lysates, when ABCG2 was overexpressed in HEK-293/cLuc, fLuc, and rLuc cells. Selective ABC transporter inhibitors were also applied. Inhibition of ABCG2 activity increased the BLI intensity more than two-fold in HEK-293/cLuc, fLuc, and rLuc cells; inhibition of ABCB1 elevated the BLI intensity two-fold only in HEK-293/rLuc cells. BLI of xenografts derived from HEK-293/ABC transporter/luciferase reporter cells confirmed the results of inhibitor treatment in vivo. These findings demonstrate that coelenterazine-based rLuc-BLI intensity can be modulated by ABCB1 and ABCG2. ABCG2 modulates
THE ATP-BINDING CASSETTE (ABC) TRANSPORTERS are one of the largest families of active transport molecules. 1 There are 48 ABC genes in the human genome, and they are dispersed mostly on different chromosomes. 2 The functional protein typically contains two nucleotide-binding folds and two transmembrane domains. The transporters can be grouped into seven subfamilies (A–G) based on the conservation of amino acid sequence in nucleotide-binding folds. ATP binding and hydrolysis provide an energy-requiring mechanism for ABC transporters to efflux specific compounds across the membrane or to flip them from the inner to the outer leaf of the membranes.1,3,4 ABC transporters can serve a wide variety of cellular roles. They regulate local transport across the blood-brain, blood–cerebrospinal fluid, and blood-testis barriers and the placenta. 5 In the liver, gastrointestinal tract, and kidney, ABC transporters protect the organism through the excretion of toxins. 6 ABC transporters also play an active role in the immune system by transporting peptides into the endoplasmic reticulum that are identified as antigens by class I human leukocyte antigen (HLA) molecules.7,8 Furthermore, they play physiologic roles in cellular lipid transport and homeostasis. 9
From a clinical perspective, multidrug resistance (MDR) involves increased expression of members of the ABC transporter family. The most extensively characterized MDR genes include ABCB1 (also known as MDR1 or P-glycoprotein), ABCC1 (also known as MRP1), and ABCG2 (also known as BCRP or MXR). 10 ABC transporter inhibition remains an attractive potential adjuvant to drug resistance in chemotherapy.11,12 ABC transporters have emerged as an important new field of investigation in the regulation of stem cell (SC) biology. For example, the “side population” phenotype in fluorescence-activated cell sorting (FACS) analysis profiles can be identified on the basis of ABC transporter expression and used as an important marker in the analysis and isolation of SCs. 13
Bioluminescence imaging (BLI) exploits the emission of visible photons at specific wavelengths based on energy-dependent reactions catalyzed by luciferases. Luciferases comprise a family of proteins that emit detectable photons during an enzymatic reaction with specific substrates in the presence of oxygen. Luciferases from the firefly Photinus pyralis (fLuc) and the click beetle (cLuc) catalyze the conversion of luciferin to oxyluciferin and simultaneously produce CO2, phosphate, and light.14,15 The Renilla luciferase (rLuc) and Gaussia luciferase (gLuc) genes were cloned from Renilla reniformis, the sea pansy, and Gaussia princeps, the marine copepod, respectively. Both use coelenterazine as a substrate, which is converted to coelenteramide, CO2, and light. In contrast to fLuc and cLuc, rLuc and gLuc do not require adenosine triphosphate (ATP) to metabolize coelenterazine.16,17 BLI has been widely used as a noninvasive method to evaluate gene expression, signal transduction, protein–protein interaction, receptor activation, and several molecular events. 18 BLI, as well as fluorescence imaging, is simpler, less expensive, more convenient, and more user friendly than other imaging modalities. Compared to fluorescence imaging, BLI is independent of an activation light source and has much higher sensitivity and lower background but requires substrate delivery. 19
The effects of ABC transporters on the efflux of established imaging agents (radiotracers for nuclear imaging and substrates for BLI) have been reported recently. For example, retention of the radiotracers 64Cu-ATSM and 64Cu-PTSM in human and murine tumors is influenced by ABCB1 expression.
20
Two articles also reported that ABC transporters modulated BLI readout through efflux of its substrates, coelenterazine (ABCB1) and
In addition, we initiated a comparative study of ABC transporter expression and BLI in a mouse embryonic stem cell (ESC) line (mES-J1) and a mouse embryonic fibroblast cell line (NIH/3T3). We showed significant differences in the levels of ABC transporter expression in these cells that correspond to the differences in fLuc and rLuc BLI readout that was observed. This report points out several confounding factors in quantitative or semiquantitative BLI related to the active efflux of luciferase substrates, particularly when SCs are involved.
Materials and Methods
Cell Lines
HEK-293 cell lines overexpressing ABCB1, ABCC1, and ABCG2 transporters (HEK-293/ABC transporter), as well as HEK-293 mock cells (control cells stably transfected with empty vector), were generously provided by Drs. Susan Bates and Robert Robey (National Cancer Institute).
23
These cells were cultured in Eagle's Minimum Essential Medium (MEM) (Invitrogen, Carlsbad, CA) supplemented with 10% fetal bovine serum (Hyclone, Logan, UT) and 2 mg/mL Geneticin (Invitrogen). The mouse ESC line, mES-J1, generously provided by Dr. Mark Tomishima (Memorial Sloan-Kettering Cancer Center [MSKCC]), was cultured in the conditional medium (830 mL knockout Dulbecco's Modified Eagle's Medium [Invitrogen], 150 mL fetal bovine serum [Hyclone], 10 mL MEM nonessential amino acids [Invitrogen], 10 mL
Reverse Transcription–Polymerase Chain Reaction Analysis
Total ribonucleic acid (RNA) was extracted from cells using an RNeasy Mini Kit (Qiagen, Valencia, CA) and treated with ribonuclease-free DNase I (AmBion, Austin, TX) before complementary deoxyribonucleic acid (cDNA) synthesis (Stratagene, La Jolla, CA). Specific primers for mouse ABCB1, ABCC1, ABCG2, and β-actin (ACTB) genes for polymerase chain reaction (PCR) amplification were as follows:
Plasmid Construction and Virus Packaging
We developed a dual-reporter system in an SFG retrovirus backbone; the SFG plasmid was originally developed to transduce bone marrow cells for transplantation and was derived from Moloney murine leukemia virus. 24 This vector contained a constitutively expressed luciferase gene and enhanced green fluorescent protein (EGFP) separated by an internal ribosome entry site (IRES) (Figure 1A). Four dual-reporter constructs were generated by cloning four different luciferases, cLuc, 25 fLuc, 26 rLuc, and gLuc, into the vector, respectively. The reporter construct was transiently transfected into the H29 virus packaging cell line 27 by Lipofectamine 2000 (Invitrogen) according to the manufacturer's instructions. To generate HEK-293/ABC transporter cells with four different BLI reporters, supernatant from the packaging cells was applied to HEK-293 cells overexpressing ABCB1, ABCC1, and ABCG2 transporters and HEK-293 mock cells with 8 μg/mL polybrene (Sigma, St. Louis, MO). Two rounds of FACS were performed to select stable transduced EGFP-positive reporter cells.
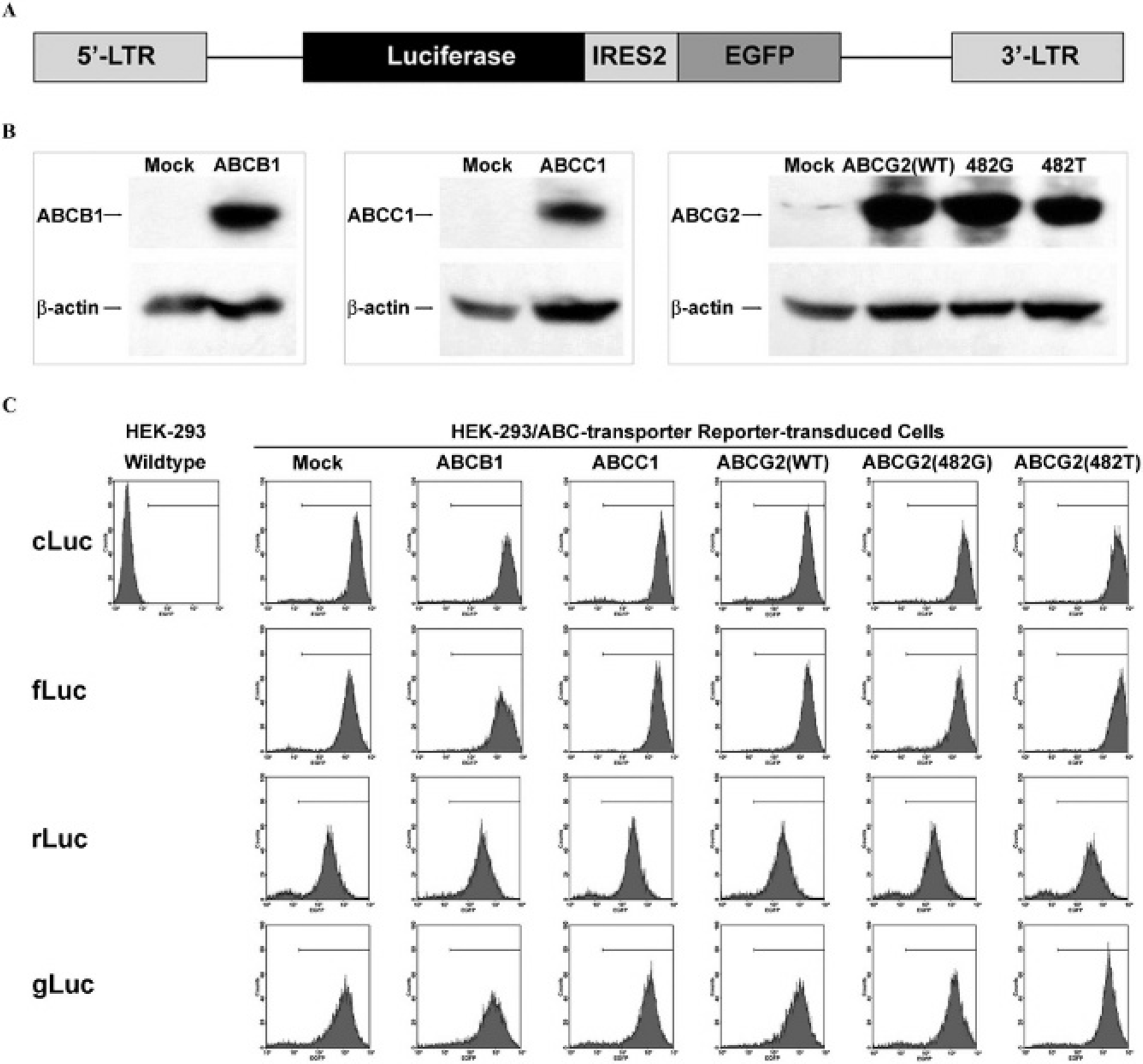
Generation of HEK-293 reporter cells with ABC transporter overexpression (HEK-293/ABC transporter reporter cells). A, Structure of the dual-reporter system in an SFG backbone. Luciferase and enhanced green fluorescent protein (EGFP) were constitutively expressed. EGFP was used as a selection marker. B, Validation of ABC transporter overexpression in stably transfected HEK-293/ABC transporter cells by Western blotting. β-Actin was used as a loading control. C, FACS analysis of the stably transduced HEK-293/ABC transporter reporter cells. Mock cells, empty vector stably transfected HEK-293 cells, were used as a negative control. ABCG2(WT), wild-type ABCG2; ABCG2(482G) and ABCG2(482T), two ABCG2 mutants with point mutations at amino acid 482 (R→G, ABCG2(482G) and R→T, ABCG2(482T)). cLuc, fLuc, rLuc, and gLuc: HEK-293/ABC transporter cells transduced with cLuc-IRES2-EGFP, fLuc-IRES2-EGFP, rLuc-IRES2-EGFP, and gLuc-IRES2-EGFP, respectively. LTR = long terminal repeat.
Western Blot Analysis
Total protein (10 μg) from HEK-293/ABC transporter cells was run on 4 to 12% polyacrylamide gel electrophoresis (Invitrogen) and electrophoretically transferred to a polyvinylidene fluoride (PVDF) membrane. Mouse anti-ABCB1 monoclonal antibody (Abcam, Cambridge, MA) (1:1,000) and mouse anti-ABCC1/ABCG2 monoclonal antibodies (Kamiya Biomedical, Seattle, WA) were used as primary antibodies. After binding a horseradish peroxidase–antimouse-IgG antibody (1:5,000) (Vector Labs, Burlingame, CA), Western blot was visualized using the SuperSignal West Pico Substrate (Thermo Scientific, Rockford, IL). The stripped membrane was reprobed by mouse anti-β-actin monoclonal antibody (Sigma) (1:10,000).
In Vitro BLI
HEK-293/ABC transporter reporter cells were seeded in triplicate into two separate 96-well plates (1 × 104 cells/well). After 24 hours, potassium D-luciferin (Caliper Life Science, Hopkinton, MA) was added to cLuc and fLuc reporter cells on one plate (final concentration 150 μg/mL), or coelenterazine (Biotium, Hayward, CA) was added to rLuc and gLuc reporter cells (final concentration of 470 nM). A Biospace system (Biospace Lab, Paris, France) was used to measure bioluminescence intensity for these in vitro studies. Cells in the second, duplicated plate were lysed with lysis buffer (Promega, Madison, WI) (20 μL/well) for 10 minutes at room temperature. BLI from the cell extracts was determined by the above procedure. For the ABC transporter inhibition assay, HEK-293/ABC-transporter reporter cells were preincubated for 30 minutes in the absence or presence of Reversin 121 (ABCB1 inhibitor [Sigma], 1.5 μg/mL), MK-571 (ABCC1 inhibitor [Sigma], 1.5 μM), and fumitremorgin C (FTC) (ABCG2 inhibitor, 1 μM, a generous gift from Drs. Bates and Robey). Photo Acquisition version 2.7 and M3Vision software (Biospace Lab) were used to acquire and analyze BLI data. Light intensities of the regions of interest were expressed as total flux (photons per second).
In Vivo BLI
Animal protocols were approved by the Animal Care and Use Committee at MSKCC. All animal procedures were performed under 2.5% isoflurane anesthesia; 1 × 106 HEK-293/ABC transporter reporter cells were suspended with Matrigel (BD Bioscience, San Jose, CA) and injected subcutaneously in 8-week-old female BALB/cAnNCr-nu/nu mice. Four xenografts were inoculated in each mouse, and the locations of the xenografts were paired (mock vs ABC transporter reporter cells) in both shoulders and thighs for BLI convenience. These animals were injected retro-orbitally with potassium
Statistics
Each cellular assay was performed in triplicate, and xenograft studies were repeated in five animals for each HEK-293/ABC transporter reporter cell line. Data were presented as mean ± SE. A t-test was used to determine significance, and values of p < .05 were considered significant.
Results
The effects of ABC transporters on luciferase-based BLI intensity have not been fully elucidated. We investigated whether BLI intensity could be enzyme (luciferase)-dependent and substrate-dependent when a constitutive reporter is expressed at comparable levels. In a retroviral plasmid, SFG vector, 24 we developed a dual-reporter system with a bicistronic cassette (eg, luciferase-IRES2-EGFP) (see Figure 1A). The dual-reporter constructs allow both bioluminescence and fluorescence constitutive reporter imaging. Four different luciferases, cLuc, fLuc, rLuc, and gLuc, were cloned into the bicistronic cassette. HEK-293 cells stably transfected to overexpress ABC transporters (ABCB1, ABCC1, ABCG2, and two ABCG2 mutants with point mutations at amino acid 482 (R→G, ABCG2(482G) and R→T, ABCG2(482T)) 11 were used in this study. Overexpression of ABCB1, ABCC1, and ABCG2 (wild type and mutants) in HEK-293/ABC transporter cells was confirmed by Western blotting (Figure 1B). Then these five cell lines, plus HEK-293 mock cells, were stably transduced with the four different dual-reporter constructs (cLuc/fLuc/rLuc/gLuc-IRES2-EGFP). Stably transduced reporter cells were selected by FACS based on comparable level of EGFP expression (Figure 1C).
To investigate the effect of ABC transporters on luciferase-based BLI, we examined the BLI intensity from HEK-293/ABC transporter reporter cells. These in vitro studies showed that the BLI intensity of intact cells, compared to cell lysates, decreased significantly when ABCG2 was overexpressed in HEK-293 cLuc, fLuc, and rLuc reporter cells (Figure 2). The normalized BLI intensity ratio of living cells to cell lysates was 0.11, 0.23, and 0.25 in HEK-293/ABCG2/cLuc, fLuc, and rLuc cells, respectively. No significant BLI change was observed in HEK-293/ABCG2/gLuc cells. Overexpression of ABCB1 transporter reduced the BLI intensity ratio only in HEK-293/ABCB1/rLuc cells by 51%. ABCC1 overexpression did not affect the BLI intensity ratio in any of the HEK-293/ABCC1/reporter cells (see Figure 2).

Relative bioluminescence imaging (BLI) intensity of HEK-293 reporter cells with and without ABC transporter overexpression. Reporter cells were seeded into duplicate 96-well plates for BLI intensity measurements in intact cells and cell lysates. Relative BLI intensity was determined by calculating the BLI intensity ratio of intact cells divided by that from cell lysates and then normalized to the value obtained from mock cells. Mock refers to empty vector stably transfected HEK-293 cells without ABC transporter overexpression; the cell to lysate BLI intensity ratio of the mock reporter cells (prior to normalization) was as follows: cLuc, 0.095 ± 0.003; fLuc, 0.085 ± 0.005; rLuc, 0.059 ± 0.002; gLuc, 0.919 ± 0.015. Error bar indicates standard error. *p < .05.
The effect of selective ABC transporter inhibitors (Reversin 121 against ABCB1,
28
MK-571 against ABCC1,
29
and FTC against ABCG230) on BLI intensity from HEK-293/ABC transporter reporter cells was also studied. Compared to vehicle treatment, Reversin 121 elevated BLI intensity 2.11 ± 0.16–fold only in HEK-293/ABCB1/rLuc reporter cells, whereas FTC increased BLI intensity more than 2-fold in HEK-293/ABCG2/cLuc, fLuc, and rLuc reporter cells (Figure 3). These results indicate that ABCG2 expression modulates BLI signal intensity generated from both

Normalized bioluminescence imaging (BLI) intensity of HEK-293 reporter cells with and without ABC transporter overexpression treated by selective ABC transporter inhibitors. Relative BLI intensity was determined by calculating the BLI intensity ratio of intact cells over cell lysates, and then normalized to the value obtained from corresponding vehicle-treated cells. Reversin 121, ABCB1 inhibitor; MK-571, ABCC1 inhibitor; fumitremorgin C (FTC), ABCG2 inhibitor. Error bar indicates standard error. *p < .05.
BLI of xenografts derived from HEK-293/ABC transporter reporter cells confirmed the results of the in vitro inhibitor treatment studies. Animals bearing subcutaneous HEK-293/ABC transporter reporter xenografts were imaged sequentially after Reversin 121, MK-571, FTC, and corresponding vehicle treatment. Reversin 121 had little or no effect on cLuc- or fLuc-based BLI intensity with
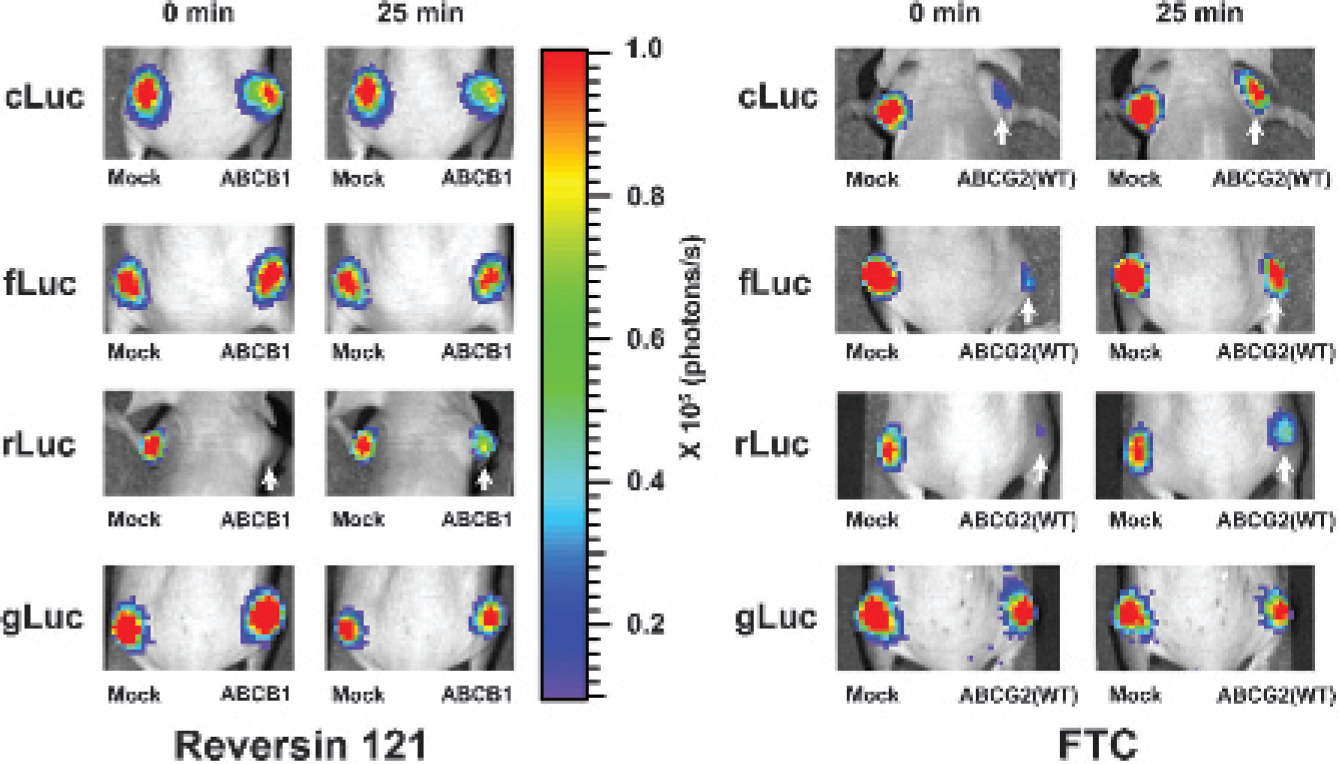
Effects of specific ABC transporter inhibitors on bioluminescence imaging (BLI) intensity from xenografts derived from HEK-293 reporter cells with and without ABC transporter overexpression. Reporter cells were subcutaneously implanted into the nude mice. After inhibitor or vehicle injection, sequential imaging was performed. Representative images of the same animal are shown. The pseudocolor bar shows BLI intensity (photons/cm2/s/seradian). The arrows point to xenografts with enhanced BLI intensity after inhibitor treatment. Reversin 121, ABCB1 inhibitor; fumitremorgin C (FTC), ABCG2 inhibitor.
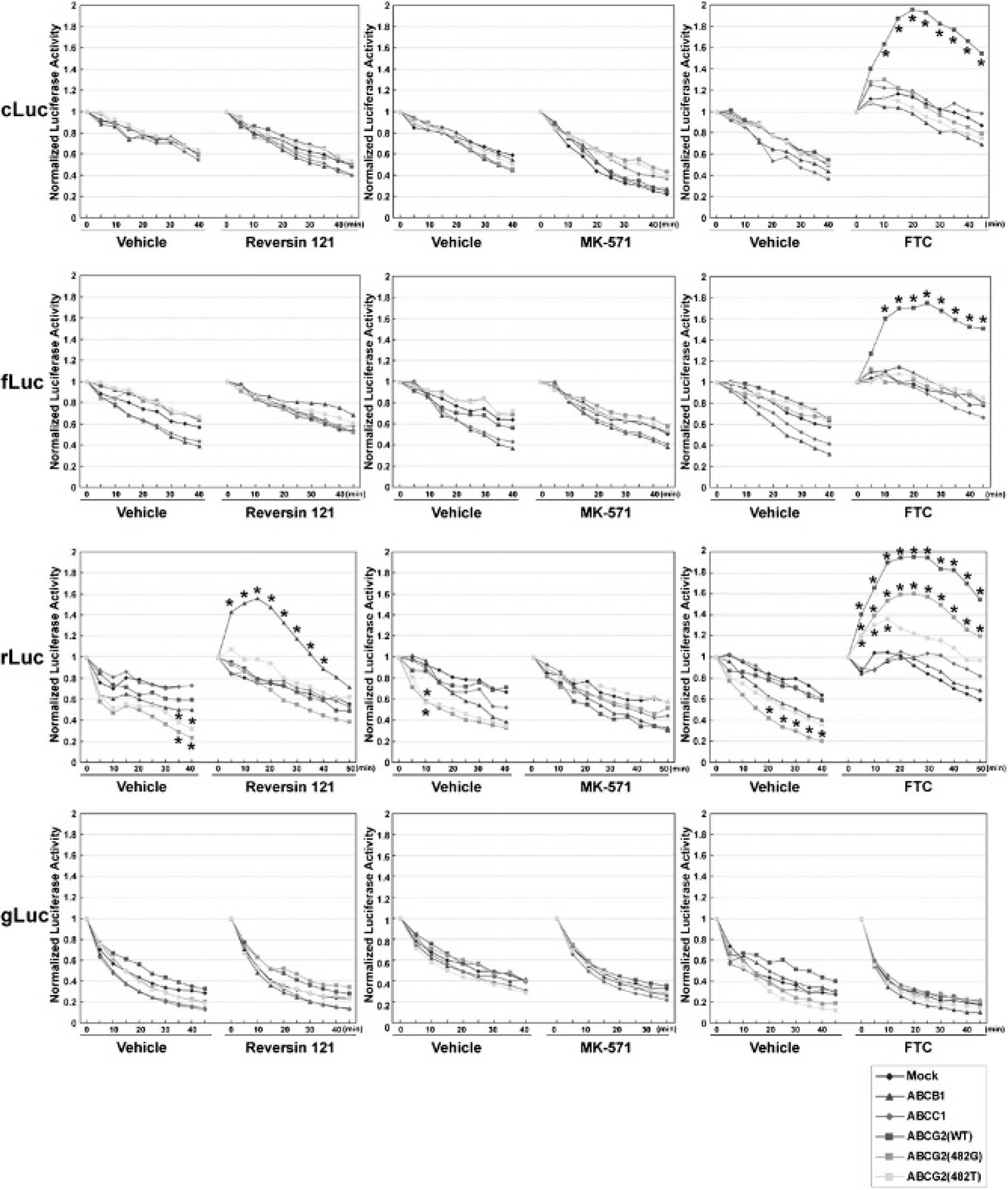
Time course of the normalized bioluminescence imaging (BLI) signal from HEK-293 reporter xenografts with and without ABC transporter overexpression treated with specific ABC transporter inhibitors. The BLI signal immediately after inhibitor or vehicle injection was defined as 1. Data are presented as mean normalized BLI signal from five xenografts in separate animals. *The data with significant differences (p < .05) compared to the normalized luciferase activity profiles of mock cells. Standard errors range from 0.012 to 0.125. Reversin 121, ABCB1 inhibitor; MK-571, ABCC1 inhibitor; fumitremorgin C (FTC), ABCG2 inhibitor.
MK-571 had no effect on any of the xenografts (see Figure 5, middle panels). This is consistent with the in vitro result and suggests that ABCC1 does not affect BLI intensity in all HEK-293/ABC transporter reporter cells. In contrast, FTC administration significantly increased BLI intensity in both
To study the effects of ABC transporters expressed in mouse ESCs, we stably transduced a mouse ESC line (mES-J1) and a mouse embryonic fibroblast cell line (NIH/3T3) with the fLuc-IRES2-EGFP and rLuc-IRES2-EGFP reporter constructs (see Figure 1A). In the first series of experiments, BLI signal intensity from intact reporter-transduced mES-J1 and NIH/3T3 cells was compared to lysates from the same reporter cells (Figure 6A). There was a significant difference in the BLI intensity ratio, comparing intact cells to cell lysates. For

A, Relative BLI intensity of mES-J1 and NIH/3T3 reporter cells treated by selective ABC transporter inhibitors. Relative bioluminescence imaging (BLI) intensity is the ratio of BLI intensity from intact cells divided by that from cell lysates. Control, untreated cells; Reversin 121, ABCB1 inhibitor; MK-571, ABCC1 inhibitor; fumitremorgin C (FTC), ABCG2 inhibitor. *Relative BLI intensity from the treated cells has significant difference compared to untreated cells, p < .01. Error bar indicates standard error. B, Endogenous expression of ABC transporters in mES-J1 and NIH/3T3 cells by semiquantitative RT-PCR. β-Actin was used as a loading control.
To explain the discordance of BLI intensity in fLuc and rLuc reporter cells, we first examined the expression level of ABCB1, ABCC1, and ABCG211 in mES-J1 and NIH/3T3 cells by semiquantitative reverse transcription PCR (Figure 6B). Notably, ABCB1 was expressed in both mES-J1 and NIH/3T3 cells, whereas ABCG2 expression was detected only in mES-J1 cells. Then ABC transporter activity in mES-J1 and NIH/3T3 reporter cells was selectively blocked using specific ABC transporter inhibitors to address whether endogenous ABCB1 and ABCG2 expression can affect BLI intensity. As shown in Figure 6A, inhibition of ABCB1 by Reversin 121 increased the relative intensity in coelenterazine-based rLuc-BLI in both cell lines; the intact cell to cell lysate ratio increased 0.058→0.218 in mES-J1-rLuc cells and 0.059→0.259 in NIH/3T3-rLuc cells. Inhibition of ABCG2 by FTC elevated the relative BLI intensity in both fLuc and rLuc mES1-J1 reporter cells but not in NIH/3T3 reporter cells. The increase of 0.142→0.331 in mES-J1-fLuc cells and 0.058→0.259 in mES-J1-rLuc cells was comparable. The absence of an FTC response in NIH/3T3 reporter cells is consistent with undetectable ABCG2 expression in these cells. These data confirm that high endogenous expression of ABCB1 and ABCG2 in mouse ESCs can modulate both coelenterazine-based rLuc-BLI and
Discussion
In this report, we examined the effects of three major ABC transporter proteins (ABCB1, ABCC1, and ABCG2) on luciferase-based BLI. This is the first time that four widely used luciferases, cLuc, fLuc, rLuc, and gLuc, were systemically studied and compared with respect to the effects of ABC transporters on the magnitude of BLI readout.
Our new finding is that coelenterazine, the substrate for rLuc and gLuc, is a substrate ABCG2-mediated efflux. Overexpression of ABCG2 reduced BLI intensity by 75%, and inhibition of ABCG2 transporter activity increased BLI intensity more than two-fold in HEK-293/ABCG2/rLuc reporter cells. In contrast, there was no significant change in BLI intensity with overexpression or inhibition of ABCG2 in HEK-293/ABCG2/gLuc reporter cells. The major difference between rLuc and gLuc is their subcellular localization; rLuc is an intracellular protein and gLuc is a secreted protein, which suggests that coelenterazine-based BLI intensity change is primarily related to an intracellular substrate concentration alteration, mediated by the ABCG2 efflux pump. Additionally, we confirmed the previous observation that coelenterazine is a substrate for the ABCB1 efflux pump. 21 Thus, coelenterazine-based rLuc-BLI intensity could be modulated by several different transporter proteins, including ABCB1 and ABCG2, because different transporters have overlapping substrate specificities.
In addition, we show that ABCG2 affects
Bioluminescence involves exergonic reactions of molecular oxygen with substrate (luciferin) and enzyme (luciferase), resulting in photons of visible light. 31 Several factors, including the expression level of luciferase, oxygen concentration, presence of magnesium ions, and substrate concentration, determine the BLI intensity. For example, hypoxia in solid tumors could influence oxygen availability for the bioluminescence reaction, leading to an underestimation of the actual number of luciferase-labeled cells during in vivo experiments. 32 Routes of substrate administration33,34 and vascular permeability35,36 also influence BLI dynamics, considering substrate delivery. Here we show that overexpression of ABC transporters also impacts the magnitude of BLI readout and that this mostly occurs through modulation of the intracellular substrate concentration.
The decrease in BLI readout observed in ABC transporter–overexpressed cells is due to a greater efflux of substrate in these cells, which is the only component of several potential confounding factors affecting BLI values. We compared the normalized BLI readout from mock reporter cells and ABC transporter–overexpressed reporter cells under the same culture and imaging condition to avoid differences in luciferase enzyme expression, oxygen concentration, and magnesium ion concentration in both the in vitro and in vivo experiments. Our results showed that blocking ABC transporter activity with selective inhibitors, Reversin 121 for ABCB1 and FTC for ABCG2, increased BLI readout approximately two-fold, which is consistent with the previous literature.21,22 Considering that we inhibited only one type of ABC transporter in each inhibition experiment set and that the inhibition was most likely incomplete at the nontoxic inhibitor concentrations used, we speculate that the ABC transporter effect could be highly significant in some biologic systems.
Given that many therapeutic compounds have been shown to be substrates for ABC efflux transporters and that upregulation of transporter expression can lead to drug resistance and treatment failure, new ABC transporter inhibitors have been sought. A treatment paradigm has been advocated, where transport inhibitors are considered “tumor cell–sensitizing agents.”
37
In this paradigm, drug-resistant cells in a tumor were effectively killed through a drug-combination strategy in which a cytotoxic drug was given along with an ABC transporter inhibitor.
37
Despite initial optimism, the results of early clinical trials using ABC transporter inhibitors have been disappointing.
38
Functional redundancy of this highly conserved transporter superfamily has limited the clinical success of ABC transporter inhibitors to date.
39
High-throughout screens for assaying inhibitors of the full set of ABC transporters are required to obtain more effective inhibitors.
40
Our and others' data demonstrate that both coelenterazine and
Recent progress in SC research has led to the discovery that SCs or progenitor cells (PCs) can be induced to differentiate into a wide range of tissues and potentially can be used for therapeutic applications in regenerative medicine. 41 Monitoring SC/PC trafficking in vivo by BLI is considered a very important new imaging strategy. It has enabled the noninvasive, repetitive assessment of SC/PC location, migration, proliferation, and differentiation within the recipient organism.42,43 However, in some cases, BLI is not always effective for monitoring SCs/PCs. For example, the cytomegalovirus promoter (pCMV), a frequently used promoter to drive constitutive BLI reporter gene expression, is quickly silenced in ESCs by the endogenous epigenetic mechanisms that lead to loss of reporter gene expression over time. 44 Other molecular mechanisms, in addition to pCMV silencing, that result in a loss of or decrease in BLI reporter gene expression or BLI signal intensity in SCs/PCs should be investigated in the future.
High expression of ABC transporter proteins is observed in SCs.
13
For example, hematopoietic SCs express high levels of ABCG2. As the SCs/PCs become lineage committed, expression of the ABCG2 gene is silenced in most committed progenitor and mature blood cells.
45
In this study, we used a mouse ESC line, mES-J1, with high endogenous expression of ABCB1, ABCC1, and ABCG2. Our results show that overexpression of ABC transporters, including ABCB1 and ABCG2, significantly reduced BLI readout intensity for both
Footnotes
Acknowledgments
We thank Drs. Susan Bates, Robert Robey, Mark J. Tomishima, and Vladimir Ponomarev for their generous reagents and cell lines. We thank Dr. Tatiana Beresten and Ms. Katerina Dyomina for technical assistance. We thank the SKI Stem Cell Research Facility and Small Animal Imaging Core for their support.
Financial disclosure of authors: This work was supported by National Institutes of Health (NIH) grants P50 CA86438 and R01 CA102673. The MSKCC Small Animal Imaging Core Facility was supported by NIH Small Animal Imaging Research Program grant R24 CA83084 and NIH Center grant P30 CA08748.
Financial disclosure of reviewers: None reported.

