Abstract
The present study demonstrates the targeting of ultrasound contrast agents to human xenograft tumors by exploiting the overexpression of the glycolipid Gb3 in neovasculature. To this end, microbubbles were functionalized with a natural Gb3 ligand, the B subunit of the Shiga toxin (STxB). The targeting of Gb3-expressing tumor cells by STxB microbubbles was first shown by flow cytometry and fluorescence microscopy. A significantly higher proportion of STxB microbubbles were associated with Gb3-expressing tumor cells compared to cells in which Gb3 expression was inhibited. Moreover, ultrasonic imaging of culture plates showed a 12 dB contrast enhancement in average backscattered acoustic intensity on the surface of Gb3-expressing cells compared to Gb3-negative cells. Also, a 18 dB contrast enhancement was found in favor of STxB microbubbles compared to unspecific microbubbles. Microbubble signal intensity in subcutaneous tumors in mice was more than twice as high after the injection of STxB-functionalized microbubbles compared to the injection of unspecific microbubbles. These in vitro and in vivo experiments demonstrated that STxB-functionalized microbubbles bind specifically to cells expressing the Gb3 glycolipid. The cell-binding moieties of toxins thus appear as a new group of ligands for angiogenesis imaging with ultrasound.
Microbubbles can also be functionalized by antibodies or receptor ligands. 7 The contrast agent thus acquires an affinity for specific cells, based on antigen expression in normal or pathologic tissues. Microbubbles are constrained to the blood vessel lumen and therefore target sites that are present on the accessible surface of vascular endothelial cells, 8 immune cells, 9 or blood clots. 10
Given that tumor neovascularization is involved in the transition to malignancy, new blood vessels are of particular interest for cancer imaging 11 and possibly therapy. 12 Microbubbles have been successfully targeted to tumor angiogenesis via vascular endothelial growth factor (VEGF) receptors 13 and αvβ3-integrin. 14 These targeted ultrasound contrast agents were used to image implanted brain tumors 14 and breast tumors, among others. 13
Glycolipids are another class of markers for the vascular endothelium of tumors. Indeed, almost all human and animal tumors are characterized by aberrant glycolipid expression. 15 The glycosphingolipid globotriaosylceramide (Gb3 or CD77) is highly expressed on several solid tumors, such as ovarian, 16 testis, 17 breast, 18 and colon tumors,19,20 and on lymphomas. 21 Of particular interest here, Gb3 was also detected in high amounts on tumor neovasculature.22–24
Shiga toxin and the related verotoxins are natural Gb3 ligands. Shiga toxin is produced by
One pentamer of STxB binds up to 15 Gb3 molecules and demonstrates a high affinity for cells, with dissociation constant values in the nanomolar range. 27 STxB was also shown to be a very stable protein. 28 Moreover, the low immunogenicity of STxB has been demonstrated in mice, and in humans, it was found that patients infected by Shiga toxin-producing bacteria do not develop an antibody response against the B subunit. 29
Given the expression of Gb3 by tumor cells and the capacity of STxB to cross tissue barriers and to escape from extra- and intracellular inactivation, exploiting the protein as a tumor delivery tool has been suggested. 29 STxB targets spontaneous and xenograft tumors in mice 30 and primary human tumoral enterocytes.20,24 Therapeutic compositions on the basis of STxB are currently being developed. 31 STxB also targets tumor vasculature in mice.31–34
We here explored the possibility of using STxB to functionalize microbubbles for ultrasound contrast imaging of tumor angiogenesis. The specific interaction between Gb3-expressing cells and STxB microbubbles was demonstrated in vitro using immunofluorescence and ultrasound imaging. The STxB microbubble agent was also used in a tumor xenograft model in mice.
Materials and Methods
Cell Culture and Animal Model
For in vitro experiments, colon adenoarcinoma HT29 cells were grown in Dulbecco's Modified Eagle's Medium (DMEM) supplemented with 10% (vol/vol) fetal bovine serum (FBS), 100 units/mL penicillin, 10 μg/mL streptomicin, 4 mM glutamine, and 5 mM sodium pyruvate. For inhibition of Gb3 synthesis, HT29 cells were cultured for 6 days with DL-PPMP (DL-threo-1-phenyl-2-palmitoyl-amino-3-morpholino-1-propanol) at a final concentration of 5 μM. The day before experiments, HT29 cells were seeded in Opticell plates. In these culture dishes, a sterile chamber is confined by two membranes that are treated for optimal cell adhesion. Importantly, these membranes are transparent to both light and ultrasound.
The HBCx-12 tumor specimen that was used in vivo was obtained with consent from a breast cancer patient by surgical resection of the tumor. The tumor sample was established as xenograft by subcutaneous implantation of a tumor fragment into the scapular area of nude mice and sequentially transplanted. HBCx-12 fragments of 30 to 60 mm3 were grafted subcutaneously in 3- to 5-week-old Swiss nude mice under anesthesia. Care and housing of mice are in accordance with the guidelines of the French Ethical Committees and Direction des Services Vétérinaires-Protection et Santé Animale (Paris, France).
Preparation of STB-Functionalized Fluorescent Microbubbles
The STxB-Cys variant carrying an additional Cys at its C-terminus was coupled to biotin by incubating a molar excess of a polyethylene glycol-maleimido modified biotin at room temperature with STxB-Cys overnight in phosphate-buffered saline (PBS). After dialysis, the protein was conjugated on primary amines to
Flow Cytometry Analysis of Microbubble Binding to HT29 Cells
HT29 cells (5 × 10 5 ) treated or not with DL-PPMP, were washed two times in PBS supplemented with 3% FBS, incubated for 30 minutes at 4°C with 10 μL of the stock solution of microbubbles, diluted in 100 μL of PBS-3% FBS, washed three times with PBS-3% FBS, and finally resuspended in 500 μL of the same buffer. Cells were then analyzed by flow cytometry with a FACSCalibur flow cytometer (BD Biosciences, San Jose, CA) in the FL1H channel for Alexa Fluor 488 fluorochrome detection.
Fluorescent and Ultrasound Imaging of STxB Microbubbles on Tumor Cells In Vitro
HT29 cells grown in Opticell plates were incubated for 30 minutes at 4°C in 10 mL DMEM-3% FBS containing 200 μL of STxB microbubble or biotin microbubble stock solutions. The cells were placed on top for optimized binding of floating microbubbles. The cells were then washed three times with DMEM-3% FBS. For all subsequent steps, the Opticell plates were inverted to place the cells at the bottom, thereby decreasing contributions from unspecific binding.
For simultaneous observation of microbubbles and cell membranes, HT29 cells were incubated for 1 minute at 4°C with the membrane dye FM4-64 at a final concentration of 5 μg/mL in PBS. The cells were then immediately observed by epifluorescence microscopy.
When devoted to ultrasound imaging, Opticell plates were controlled by epifluorescence microscopy for microbubble binding to cells, without membrane labeling. The plates were then transferred into a water bath and imaged using a 8 MHz linear array of transducers (128 elements) driven by an ultrasound scanner (Supersonic Imagine, Aix-en-Provence, France), with cells facing the bottom surface (Figure 1). Unattached microbubbles that were left after washing floated under the top membrane. Thanks to the axial (along depth axis) resolution of the imaging system, these microbubbles could be resolved from the bottom membrane. Several B-mode images were recorded. All analysis was performed linearly on a multielement radiofrequency signal using

Experimental setup to measure the acoustic reflection of microbubbles attached to cells in an Opticell dish.
Imaging Microbubble Localization onto Tumors In Vivo
Mice bearing HBCx-12 human breast xenograft were anesthetized by intraperitoneal injection of xylazine (10 mg/kg) and ketamine (100 mg/kg). The stock solution of STxB microbubbles was injected in the retro-orbital sinus of the mouse as a bolus of 100 μL (
Immunohistochemistry of the Expression of Gb3 in Tumor Neovessels
Nude mice bearing HBCx-12 xenograft were injected intravenously with 100 μg of STxB-Cy3 (3.34 mg/kg). Tumors were collected 24 hours postinjection and incubated 3 hours at room temperature in a solution of paraformaldehyde (4% paraformaldehyde in an aqueous solution containing 10 mM PBS, 0.1 mM CaCl2, and 1.0 mM MgCl2). Tumors were stored in sucrose 30%, and 5 μm frozen tumor sections were incubated with monoclonal anti-CD34 (MEC 14.7 clone, Santa Cruz Biotechnology, Santa Cruz, CA) and secondary donkey antirat IgG coupled to Alexa Fluor 488 (Molecular Probes, Eugene, OR). 4,6-Diamidino-2-phenylindole (DAPI) was used to stain nuclei. Sections were observed under a confocal microscope (TCS SP2, Leica).
Results
In Vitro Targeting of Gb3-Expressing Tumor Cells by STxB-Conjugated Microbubbles
For convenient fluorescent detection of STxB-biotin binding to the surface of microbubbles and binding of targeted microbubbles to cells, STxB was conjugated to the Alexa Fluor 488 fluorophore. Streptavidinated microbubbles were incubated with Alexa Fluor 488-STxB-biotin, leading to the appearance of a specific fluorescence signal on the microbubble membranes (Figure 2), demonstrating that STxB was attached to the surface of the contrast agent. To demonstrate the Gb3-dependent tumor cell binding of STxB microbubbles, we first performed flow cytometry analysis on human colorectal carcinoma HT29 cells that had been incubated or not with an inhibitor of Gb3 synthesis. As shown in Figure 3, the mean was 10-fold higher on Gb3-expressing cells when compared to inhibitor-treated cells, indicating a specific interaction of STxB microbubbles with the cellular glycolipid receptor.

Fluorescence imaging of STxB microbubbles. Alexa Fluor 488-STxB-biotin was incubated with streptavidin-coated microbubbles. Alexa Fluor 488 labeling on the surface of microbubbles could be detected by epifluorescence microscopy (630× magnification).
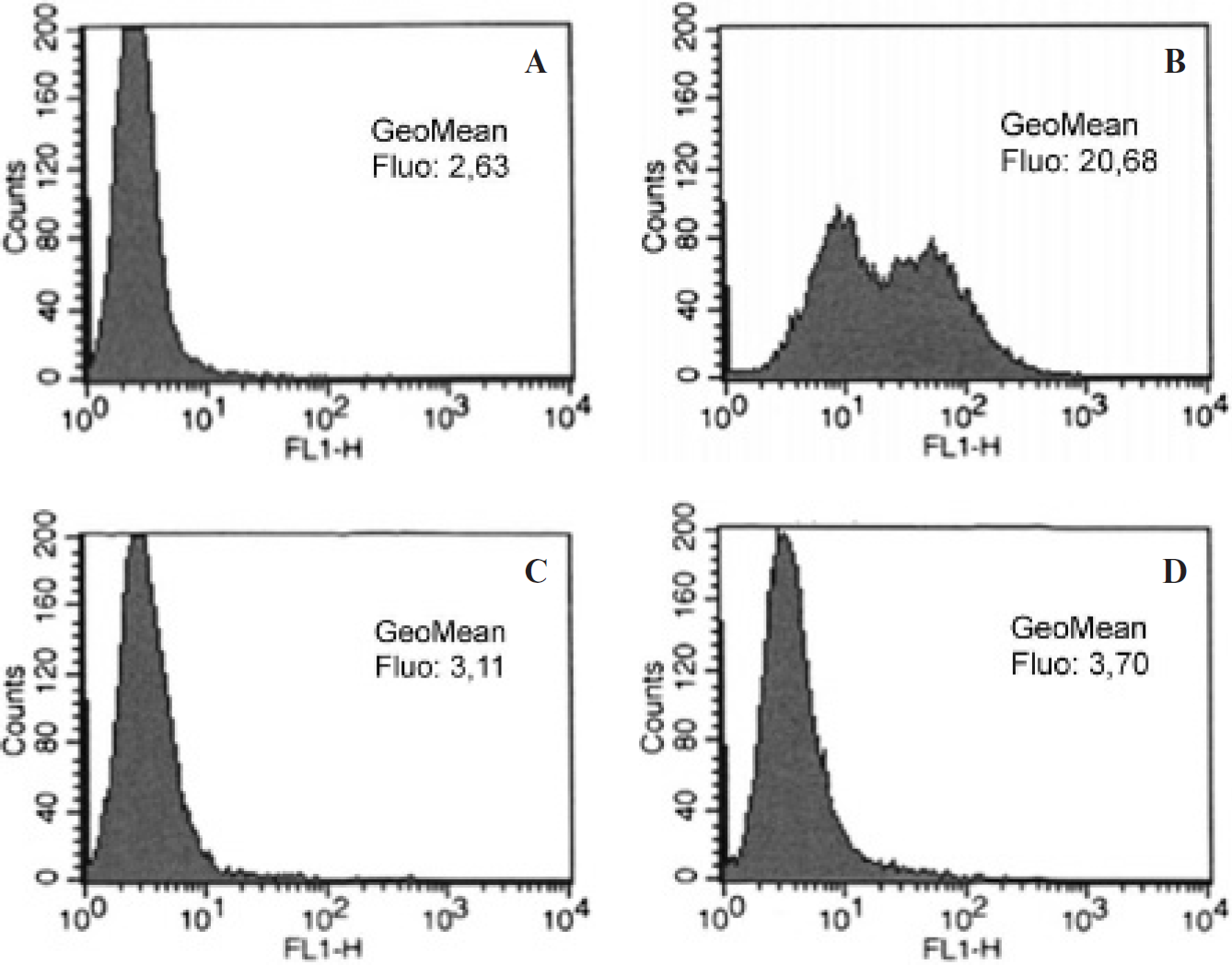
Flow cytometry analysis of STxB microbubble binding to HT29 cells. HT29 cells, nontreated (A, B) or treated with the Gb3 synthesis inhibitor DL-PPMP (C, d), were incubated alone (A, C) or with Alexa Fluor 488-STxB microbubbles (B, D) for 30 minutes at 4°C, washed, and analyzed for microbubbles binding by flow cytometry in the FL1H chanel (Alexa Fluor 488 fluorochrome detection).
To directly visualize the Gb3-dependent binding of STxB microbubbles to HT29 cells, the cells were seeded in Opticell plates for observation by epifluorescence microscopy after plasma membrane labeling with FM4-64 dye. As shown in Figure 4, STxB-microbubbles were present in greater numbers on the Gb3-expressing cells compared to cells in which Gb3 expression was inhibited. We counted an average of 1.2 microbubbles per Gb3-expressing cell versus 0.2 per Gb3-negative cell (

Fluorescence imaging of microbubbles binding to HT29 cells. HT29 cells were treated (C, D) or not (A, B) with the Gb3 synthesis inhibitor DL-PPMP and then incubated with STxB microbubbles for 30 minutes at 4°C. After washing, the cells were incubated for 1 minute at 4°C with FM4-64 for plasma membrane labeling and then observed by fluorescence microscopy (B and D: 400× magnification; A and C: 630× magnification).
Taken together, the flow cytometry and immunofluorescence data demonstrated that STxB mediates Gb3-dependent targeting of microbubbles to cells in vitro.
The Opticell plates were also imaged with ultrasound. Even with disruption imaging, echoes from the Opticell plate membranes were detectable, creating a background signal. Both membranes (2 mm apart) could thereby clearly be distinguished. Cells were placed to face the bottom side, thereby separating floating microbubbles from cell-attached ones. In the first pair of controlled testing (Figure 5, A and B), the average intensity of the echo from the bottom surface was 12 dB higher (fourfold increase) for Gb3-expressing cells compared to inhibitor-treated Gb3-negative cells. More STxB bubbles were thus attached to the cells when the molecular target was present. In the second set of controlled testing (Figure 5, C and D), STxB microbubbles displayed a 18 dB higher (eightfold increase) average intensity than biotin microbubbles when measured over the entire plate. Very clearly, STxB microbubbles were more efficiently targeted than control microbubbles to Gb3-expressing cells.
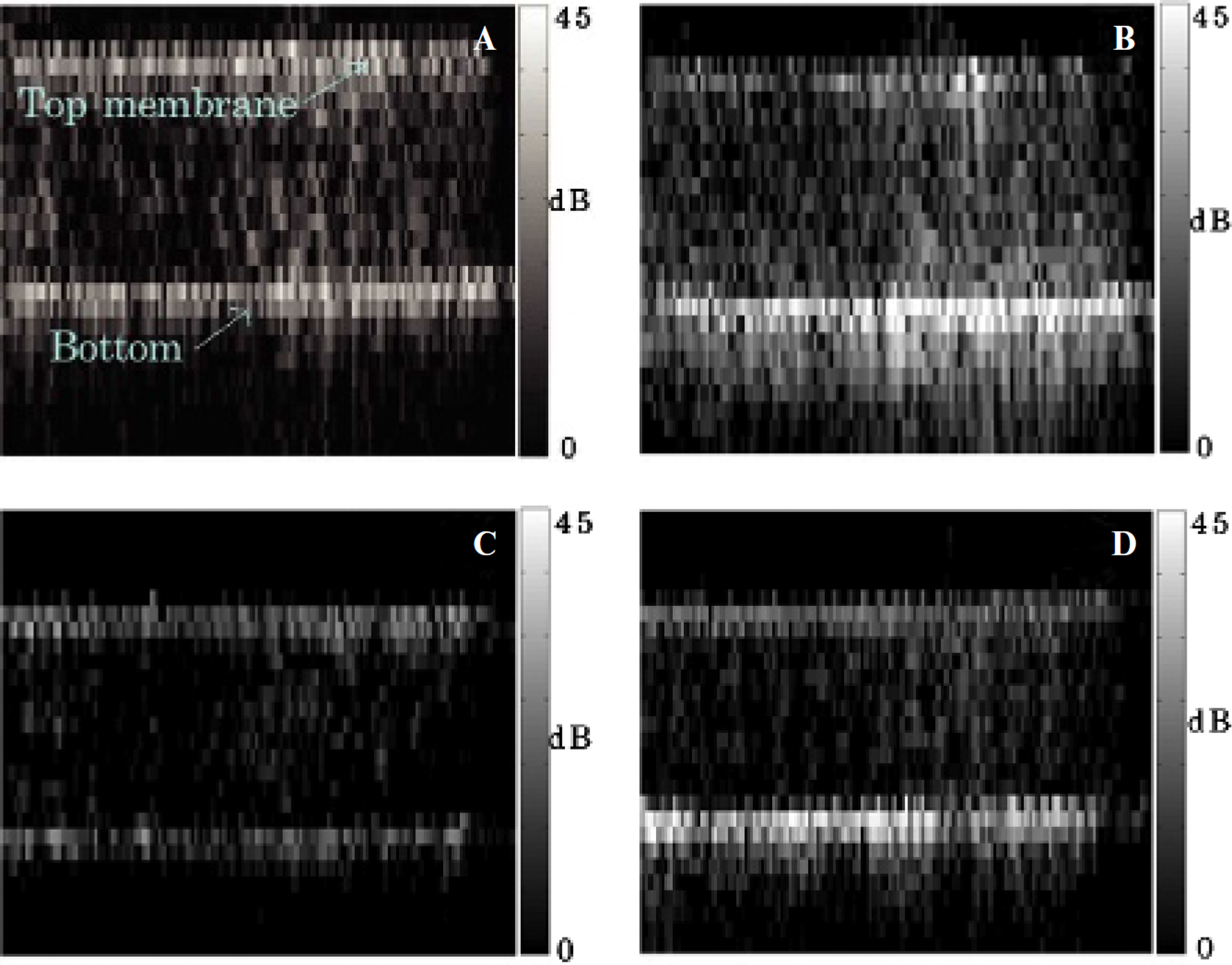
Ultrasound scan of an Opticell dish, of which the bottom surface was covered with HT29 cells. STxB microbubbles on Gb3-negative (A) and Gb3-positive (B) cells; biotin microbubbles (C) and STxB microbubbles (D) on Gb3-expressing cells. Not drawn to scale: the images are 4 cm wide, whereas the vertical distance between the membranes is 2 mm. The images of the membrane were also tilted back to the horizontal position.
In Vivo Targeting of Gb3-Expressing Tumors by STxB-Conjugated Microbubbles
In vivo experiments were performed on human breast cancers (HBCX-12) implanted subcutaneously in mice. As STxB microbubbles are expected to target tumor microvasculature, immunohistochemistry was performed on HBCX-12 tumor sections to confirm Gb3 expression by neovessels. As shown in Figure 6, after in vivo injection of Cy3-STxB, specific labeling could be detected at the level of the neovessels, documenting the Gb3 expression in microvasculature in this tumor model.

Targeting of HBCX-12 xenografted tumor microvasculature by STxB (
For in vivo ultrasound imaging on mice, microbubbles were injected in the retro-orbital sinus, a method previously tested with unspecific microbubbles. In preliminary experiments, it was shown that the agents could readily be identified in the liver. Also, these experiments confirmed that the plane wave harmonic imaging technique was appropriate to reveal the presence of microbubbles both in vitro and in vivo.
At the time of imaging, xenografted tumors were 5 to 10 mm in diameter. In conventional ultrasound imaging, the tumors were identifiable as hypoechoeic masses compared to the underlying tissue. In Figure 7, microbubble harmonic imaging (

Ultrasound imaging of Gb3-positive HBCX-12 tumors. The four tumors on the

Average intensity of the nonlinear microbubbles echoes over the area of the tumor. The difference between the two sets is statistically significant (unpaired
Discussion
Because of the previously described overexpression of the Shiga toxin receptor Gb3 in tumor neovasculature, we hypothesized that STxB-functionalized microbubbles would preferentially accumulate at sites of tumor growth.
Flow cytometry experiments demonstrated a stable association of fluorescent STxB-microbubbles with Gb3-expressing cells. On the other hand, inhibition of Gb3 synthesis prevented STxB microbubble accumulation on cells, and control microbubbles also failed to bind cells. Fluorescent imaging of cells seeded in Opticell plates confirmed these results and showed significantly higher amounts of STxB microbubbles bound to the surface of Gb3-expressing cells compared to cells in which Gb3 synthesis was inhibited.
Ultrasound experiments performed on the same Opticell plates showed that bubble-specific signal at the bottom of the plate was significantly higher (12 dB or 4 times higher) for Gb3-expressing cells compared to synthesis inhibitor-treated cells and higher for STxB-functionalized microbubbles (18 dB or 8 times higher) compared to biotin microbubbles.
Finally, in vivo experiments performed on mice xenografted with human breast tumors demonstrated a specific tumor accumulation of STxB microbubbles compared to control microbubbles. These results were in good correlation with immunohistochemistry performed on the same tumor after in vivo bioaccumulation of Cy3-labeled STxB. Indeed, the immunohistochemistry showed a colocalization of STxB with tumor neovessel markers, thus accounting for STxB microbubble targeting to regions of tumor growth.
Altogether, these results demonstrate that STxB microbubbles might be an appropriate tool for investigating tumor angiogenesis. As Shiga toxin has naturally evolved to encounter host defenses, STxB possesses delivery tool properties, such as low immunogenicity and high structural stability in physiologic conditions. Moreover, STxB can be readily produced in and purified from bacteria at gram scales.
In previous studies, we have shown that STxB-functionalized liposomes of smaller size (200 nm) than the microbubbles of the current study (μm scales) are transported inside tumor cells 38 and that they irreversibly associate with the plasma membrane of these cells in a process that depends in a nonlinear manner on Gb3 expression. 39 Notably, the latter points may be of interest for the development of targeted ultrasound approaches for the following reasons: first, an irreversible adhesion may provide the type of stability in time that is required for combining tumor imaging and therapy (see below). Second, the nonlinear binding dependency on Gb3 expression explains at least in part the fact that STxB targets very few tissues in the organism: although many tissues may express Gb3, only in a few cases (notably in tumors and tumor angiogenesis) are threshold levels of Gb3 expression reached that are required for STxB binding.
Gb3 could thus join other well-known biomarkers of angiogenesis, such as VEGF receptor and αvβ3-integrin, and provide complementary information on the disease process, diagnosis, and prognosis. For example, Rychak and colleagues demonstrated selective VEGF receptor-mediated microbubble targeting in mice. 40 Given that VEGF receptor is also used as a target for therapy, the need arises for imaging methods such as STxB-based methodology that are independent from the pathway targeted by the therapy. Thereby, an assessment of changes in tumor angiogenesis in response to treatment could be obtained. Consequently, angiogenesis imaging with STxB microbubbles may improve diagnosis of neoplasia, early detection of metastases, and assessment of novel antiangiogenic therapies. It could also help monitor the effect of STxB-based chemotherapy.32,34
Future investigations will consist in generating a covalent bond between STxB and the microbubbles to improve the blood stability of the conjugate. The experiments will also be performed at higher frequencies to improve the resolution of the ultrasound system. Counting tools will be introduced to link the linear microbubble signal intensity with the number of contrast agent accumulated in the tumor. The binding dynamic of the microbubbles in the tumor will be compared to other organs, such as the liver. STxB microbubbles will also be investigated as a tool for molecular focusing therapy previously described by our researchers 41 and for targeted delivery of antiangiogenic compounds.
Conclusion
Microbubbles that are functionalized by STxB bind specifically to cells expressing Gb3, an angiogenesis marker, both in vitro and in vivo. These targeted contrast agents result in hyperechoic signals in ultrasonic contrast images and could be used for molecular imaging of cancer and to guide therapeutic ultrasound procedures.
Footnotes
Acknowledgments
We thank Bracco Research SA for providing the ultrasound contrast agents. The work was supported by Fondation Pierre-Gilles de Gennes.
Financial disclosure of authors and reviewers: None reported.
