Abstract
Current methods of intraoperative tumor margin detection using palpation and visual inspection frequently result in incomplete resections, which is an important problem in surgical oncology. Therefore, real-time visualization of cancer cells is needed to increase the number of patients with a complete tumor resection. For this purpose, near-infrared fluorescence (NIRF) imaging is a promising technique. Here we describe a novel, handheld, intraoperative NIRF camera system equipped with a 690 nm laser; we validated its utility in detecting and guiding resection of cancer tissues in two syngeneic rat models. The camera system was calibrated using an activated cathepsin-sensing probe (ProSense, VisEn Medical, Woburn, MA). Fluorescence intensity was strongly correlated with increased activated-probe concentration (R 2 = .997). During the intraoperative experiments, a camera exposure time of 10 ms was used, which provided the optimal tumor to background ratio. Primary mammary tumors (n = 20 tumors) were successfully resected under direct fluorescence guidance. The tumor to background ratio was 2.34 using ProSense680 at 10 ms camera exposure time. The background fluorescence of abdominal organs, in particular liver and kidney, was high, thereby limiting the ability to detect peritoneal metastases with cathepsin-sensing probes in these regions. In conclusion, we demonstrated the technical performance of this new camera system and its intraoperative utility in guiding resection of tumors.
Optical imaging using near-infrared fluorescence (NIRF) light has recently emerged as a promising technique to visualize cancer cells during surgery.5–9 The advantages of NIRF light (700–900 nm) include high tissue penetration up to several centimeters deep, low autofluorescence providing a sufficient signal to noise ratio, the current availability of NIRF probes and labels for conjugation to target tumor-specific molecules, and the insensitivity of human eyes to near-infrared wavelengths providing no interference with the surgical field. 6 Nonetheless, only a small number of fixed-geometry or handheld fluorescence imaging systems have been developed with surgical oncology in mind.10–15 These systems employ either a laser light source10,13 or light-emitting diodes (LEDs)11,12 for NIRF excitation. Gutowski and colleagues assessed the technique of intraoperative immunophotodetection using a prototype device (BFP Electronique, Marvejols, France). 10 In this device, a cooled laser diode emitted light at 649 nm through a fiberoptic output. Ke and colleagues used a custom-built imaging system for intraoperative tumor detection, which employed two laser diodes at 660 nm (0.7 mW/cm 2 ) and 785 nm (1.6 mW/cm 2 ). 13 Images were enhanced using an image intensifier tube. Kirsch and colleagues described the use of a handheld device (Siemens Medical Solutions, New York, NY), for which they described the molar detection limit. 14 The Photodynamic Eye (PDE; Hamamatsu Photonics, Hamamatsu, Japan) uses LEDs and is designed for the detection of the clinically available probe indocyanine green; it has been used in several clinical studies on lymph node mapping12,16,17 and tumor imaging. 9 The system provides 1 mW/cm 2 of 760 nm excitation light. The FLARE imaging system (Dr. J.V. Frangioni, Boston, MA) has been used in preclinical18–20 and clinical studies. 15 This system employs high-power, cooled LEDs at 670 nm (4 mW/cm 2 ) and 760 nm (14 mW/cm 2 ) and also uses spectrally separate white light LEDs to illuminate the surgical field. This three-channel approach has the great advantage of displaying the NIRF signal in direct relation to the anatomic landmarks by superimposing the NIRF image on the visible light image. However, the FLARE is significantly larger than other handheld systems.
All systems use charge-coupled devices (CCDs) for signal registration. As the quantum efficiency of CCDs tends to diminish significantly in the NIR spectrum, the choice for a specific CCD is, besides excitation light and quality of optics, of importance for the detection limits of an imaging system. However, technical details such as illuminating power, detection capabilities of the camera, and resolving power are not clearly described for all of the above-described camera systems. To determine an imaging system's contribution to the field of NIRF image-guided surgery, a system needs to be analyzed in a well-structured way for its technical capabilities (ie, near-infrared excitation light production, detection limits) and its practical use (ie, ergonomics, sterility).
Here we describe the Fluobeam (Fluoptics, Grenoble, France), a novel, handheld, intraoperative fluorescence camera system. The minimal detection limits, resolving power, and intraoperative utility are addressed in primary breast cancer and metastatic colorectal cancer in two syngeneic rat models.
Material and Methods
NIRF Camera System
The Fluobeam system is composed of class 3B continuous-wave laser (100 mW) emitting at 690 nm (Power Technology, Alexander, AR) and power is supplied by a mains supply box (Mascot, Norway) continuously delivering a 9 V output current with a maximum of 4.5 A. The light from the excitation source is scattered using a diffuser to produce a homogeneous lightened field (6−8 cm diameter, field homogeneity > 30%) with an illumination power of 2.6 mW/cm 2 . The animal is placed under the laser and illuminated by white light (Photonic Optics, China) filtered with a band-pass filter (350–650 nm), providing an irradiance of 7 × 10 3 lx at the animal level. The fluorescence signal is collected through a long-pass filter (> 700 nm; OD value at 690 nm is 3.5) by a digital 12-bit CCD camera (PCO Imaging, Kelheim, Germany) using a fixed-focus objective (Schneider Optische Werke GmbH, Bad Kreuznach, Germany). Camera exposure times are adjustable between 1 and 1,000 ms. Camera exposure times of 10 and 20 ms provided the best contrast between tumor and background during surgical exploration for ProSense680 and ProSense750 (VisEn Medical, Woburn, MA), respectively, and were used during both the in vitro and the in vivo part of this study. The spatial resolution of the Fluobeam is 0.17 mm/pixel at focus point. The resolving power of the system is 2.52 line pairs per millimeter, as determined using the 1951 USAF resolution test chart. The laser, the filtered white light illumination, and the camera are suspended on a multiangle adjustable arm that is positioned 20 cm above the point of focus, allowing sufficient space for surgical maneuvers (Figure 1). The system is operated by a desktop computer and the fluorescence signal is displayed in real time on the computer screen.
NIRF Probe
The NIRF probes ProSense680 and ProSense750, with peak absorbance of 680 and 750 nm, respectively, were used for fluorescent imaging. ProSense is an autoquenched fluorescent probe that converts from a nonfluorescent to a fluorescent state by proteolytic activation of lysosomal cysteine or serine proteases such as cathepsin B. 21

The Fluobeam intraoperative near-infrared camera system.
Calibration of Camera System
For calibration of the Fluobeam camera system, 2 nmol ProSense (150 μL) was activated with 100 μL 0.25% trypsin–ethylenediaminetetraacetic acid (EDTA) at 37°C for 1 hour. 14 The cleaved probe was diluted to a concentration of 128 nM and diluted 10 times on the ratio 1:2 in phosphate-buffered saline (PBS) and aliquoted into a 96-well plate (Greiner Bio-one, Alphen aan den Rijn, The Netherlands, #655090, suitable for fluorescence measurements). Subsequently, the samples were imaged with the Fluobeam at various camera exposure times. The experiment was performed for both ProSense680 and ProSense750. PBS was used as a negative control. To confirm the accuracy of dilution, the same samples were measured using the Odyssey NIRF scanning device (LI-COR Biosciences, Lincoln, NE).
Cell Line Experiments
The syngeneic MCR86 breast cancer rat cell line and the syngeneic CC531 colorectal cancer cell line were used for cell line experiments.22,23 Tumor cells were cultured in RPMI 1640 supplemented with 2 mM
To determine the minimal number of tumor cells that could be detected, MCR86 and CC531 tumor cells were cultured in T75 culture flasks up to three-quarter confluence and incubated in 20 mL medium with 10 nM ProSense680. After 24 hours' incubation with ProSense680, cells were washed in PBS, harvested as described above, washed in medium, and adjusted to a suspension containing 1 × 10 6 tumor cells per milliliter. This suspension was diluted seven times on the ratio 1:2 in medium and aliquoted in 1.5 mL tubes. Tubes were centrifuged at 13,000 rpm for 5 minutes to generate cell pellets. Subsequently, the cell pellets were imaged with the camera system at 10 ms camera exposure time. Tumor cells incubated with medium only served as a negative control. This experiment was repeated three times for both the MCR86 and CC531 cell lines.
Animal Models
EMR86 Breast Cancer Rat Model
The syngeneic transplantable EMR86 breast cancer model originated in a female WAG/Rij rat bearing a subcutaneously implanted estrogen pellet and is related to the MCR86 cell line. This model is developed by our research group. 24 Tumors are induced and maintained only in rats carrying estrogen pellets, whereas tumors transplanted into nonestrogenized animals never grow out. Fresh EMR86 tumor fragments of 1 mm 3 were subcutaneously implanted into the mammary fat pads at four sites in 4- to 6-month-old female WAG/Rij rats (Charles River, the Netherlands). Simultaneously, an estrogen pellet was implanted subcutaneously in the intrascapular region of the neck. The in-house-generated pellets consist of 2 × 3 mm silicone tubes containing 1.5 mg 17β-estradiol on a 1:3 cholesterol/paraffin basis. Tumor volumes were estimated using digital calipers by measuring three orthogonal diameters of the tumor and multiplying this product by pi/6. After 4 weeks, tumors had reached a volume of approximately 1 cm 3 .
CC531 Colorectal Cancer Rat Model
To induce CC531 peritoneal metastases, CC531 cells were harvested as described above, washed three times in PBS, and adjusted to a suspension containing 2 × 10 6 viable (trypan blue exclusion test) tumor cells per milliliter. The peritoneal cavity was inoculated with 2 × 10 6 cells in 6-month-old male WAG/Rij rats (Charles River). 25 Two weeks after inoculation, small metastases of approximately 2 mm originated in the abdominal cavity.
Animal Experiments
All animals were housed in the animal facility of the Leiden University Medical Center. Pellet food and fresh tap water were provided ad libitum. The weight of the animals was followed throughout the experiment to monitor their general health state. Throughout imaging and surgical procedures, the animals were anesthetized with 5% isoflurane for induction and 2% isoflurane for maintenance in oxygen with a flow of 0.8 L/min and placed on an animal bed with an integrated nose mask. The Animal Welfare Committee of the Leiden University Medical Center approved these studies.
Rats were injected intravenously with ProSense (10 nmol per animal) 24 hours before imaging. Before injection, autofluorescence of tumors, surrounding tissue, and abdominal organs was determined. Rats were shaved to reduce absorption of the optical signal by fur. Animals were anesthetized with isoflurane as described above. Ethanol 70% was used as a disinfectant. EMR86 mammary tumors were removed by making an incision ventrally of the tumor and carefully dissecting the tumor with direct guidance of the real-time fluorescent signal. For the detection of CC531 intraperitoneal metastases, a median laparotomy was performed followed by a systematic exploration of the small and large bowel along with the mesentery and peritoneal cavity. Metastases identified clinically or by fluorescence were carefully excised. The fluorescence intensity of tumors and abdominal organs was determined in vivo and ex vivo using the Fluobeam. Excised tumors were snap frozen in isopentane and stored at −80°C or fixed in formalin and embedded in paraffin (FFPE) blocks. Frozen tissue sections of 20 μm or FFPE tumor sections of 4 μm were air-dried and stained with hematoxylin and eosin.
Statistical Analysis
Fluobeam-derived NIRF data were analyzed using the open-source software ImageJ 26 by drawing regions of interest and measuring the fluorescence intensity of the 12-bit images. For determination of detection limits, the fluorescence intensity of test samples was divided by the fluorescence intensity of the negative control. A ratio higher than 2 was considered discriminative. For the animal experiments, regions of interest were drawn at the tumor and at the surrounding tissue within a range of 2 mm of the demarcation line of the tumor and the surrounding tissue. Statistical analysis and generation of graphs were performed using GraphPad Prism software (version 5.01; GraphPad Software, La Jolla, CA). Unless otherwise stated, mean fluorescence intensity and associated standard deviations were reported. Pearson correlation coefficients R 2 were used for the in vitro experiments. Unpaired and paired t-tests were used for testing differences of continuous variables between groups. Statistical tests were two-tailed, and p < .05 was considered significant.
Results
Calibration of Camera System
Trypsin-activated ProSense680 and ProSense750 were used to calibrate the Fluobeam camera system (Figure 2A). Fluorescence intensity was linearly correlated with the concentration of activated ProSense680 and ProSense750 (correlation coefficient R 2 = .997 and .997, respectively). The dilution accuracy was confirmed using the Odyssey NIRF scanning device for both ProSense680 (R 2 = .995) and ProSense750 (R 2 = .995). At similar Fluobeam camera exposure times, fluorescence intensity of ProSense680 was on average 2.6 times higher than the fluorescence intensity of ProSense750. This reflects the better matching of the absorbance peak of ProSense680 with the 690 nm emitted by the laser of the Fluobeam. Therefore, ProSense680 was used in subsequent in vitro experiments. Camera exposure time was linearly correlated with fluorescence intensity (R 2 ranged between .997 and 1 for the different concentrations), indicating linearity of the camera system. During the in vivo experiments, the optimal fluorescent signal for tumor identification without saturated pixels was obtained at 10 ms camera exposure time. Therefore, a 10 ms camera exposure time was used during subsequent in vitro experiments using ProSense680. To quantify the sensitivity of the camera system, the minimal detectable concentration of ProSense680 was determined at 10 ms camera exposure time, mimicking the in vivo situation. A signal to background ratio was calculated with PBS as background. A ratio of 2 was used as the cutoff. At these settings, the minimal detectable concentration of trypsin-activated ProSense680 was 9.3 ± 0.1 nM (see Figure 2A). Therefore, a concentration of 10 nM was used in subsequent cell line experiments.

Calibration of the Fluobeam camera system. A, The concentration of trypsin-activated ProSense680 and ProSense750 is plotted against the mean fluorescence intensity at 10 and 20 ms camera exposure times. B, NIRF Fluobeam image showing 1.5 mL tubes containing cell pellets of various amounts of cells of the MCR86 breast cancer cell line after 24 hours' incubation with 10 nM ProSense680. Camera exposure time was 10 ms. C, The number of MCR86 cells incubated with 10 nM ProSense680 is plotted against the total fluorescence intensity at 10 ms camera exposure time.
Cell Line Experiments
To determine the minimal number of tumor cells that could be detected with the Fluobeam and ProSense680, cultured MCR86 and CC531 tumor cells were incubated in medium with 10 nM ProSense680. After 24 hours' incubation, cells were aliquoted in 1.5 mL tubes at various cell concentrations, spun down, and imaged with the Fluobeam (Figure 2B). The total fluorescence of the cell pellets was linearly correlated with the number of cells at 10 ms camera exposure time for both MCR86 (R 2 = .952; Figure 2C) and CC531 (R 2 = .989; data not shown). To determine the minimal detection limit, a signal to background ratio was calculated with medium only as background. A ratio of 2 was used as the cutoff. At 10 ms camera exposure time, the minimal detection limit of the Fluobeam was 20,400 ± 9,200 MCR86 cells and 12,800 ± 2,700 CC531 cells (data not shown). Cell pellets containing 20,000 cells are approximately 0.2 mm 3 , reflecting submillimeter metastases.
Intraoperative Imaging
Primary Breast Cancer
The syngeneic EMR86 breast cancer rat model was used to test the intraoperative application of the Fluobeam camera system. Primary breast tumors were induced in six female rats in the mammary fat pad. Twenty tumors were induced, varying in size from 0.08 to 4.19 cm 3 (mean 0.77 cm 3 ± 1.4). All tumors were successfully detected and resected under direct fluorescence guidance 24 hours after injection with ProSense680 (n = 12 tumors) or ProSense750 (n = 8 tumors; Figure 3). In concordance with the in vitro data, the fluorescent signal of the tumors was higher with ProSense680 (mean 2,552 ± 659.4) than with ProSense750 (mean 1,367 ± 489.9; t = 4.33, p = .0004; see Figure 3B) at a 10 ms camera exposure time. During surgery, an optimal fluorescent contrast between tumor (unsaturated signal) and surrounding mammary fat pad was obtained using a 10 ms camera exposure time for ProSense680 and 20 ms for ProSense750. The signal of ProSense680 was stronger owing to better matching with the 690 nm laser, as discussed above. Although the fluorescence intensity of tumor tissue was significantly higher than the surrounding mammary fat pad with both ProSense680 (paired t = 12.52, p < .0001, n = 12 tumors) and ProSense750 (paired t = 6.29, p = .0004, n = 8 tumors) using these camera exposure times, the tumor to background ratio was significantly higher for ProSense680 (2.34 ± 0.35) when compared to ProSense750 (1.91 ± 0.38; t = 2.53, p = .021; see Figure 3C).
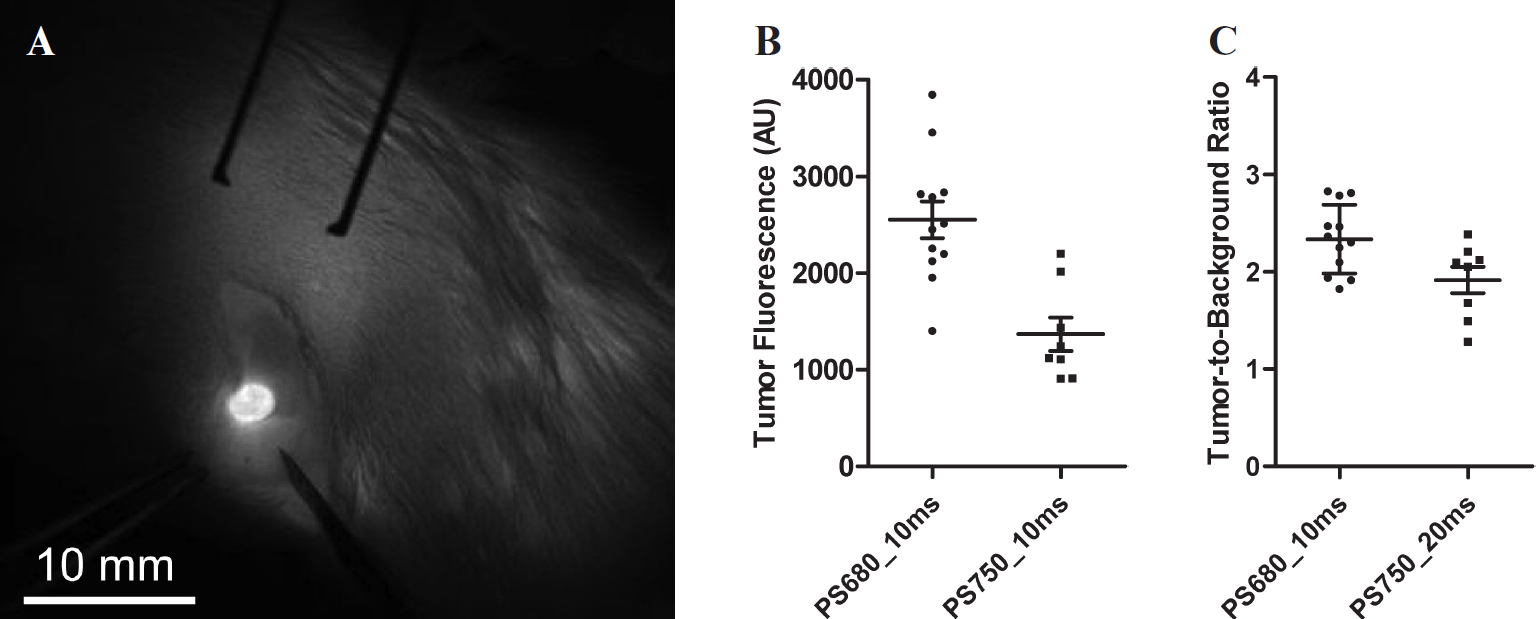
Intraoperative NIRF-guided resection of primary breast cancer using the Fluobeam camera system. A, NIRF image showing the intraoperative view of a 3.5 mm EMR86 breast tumor in a female rat 24 hours after administration of 10 nmol ProSense680. Camera exposure time was 10 ms. B, The mean fluorescence intensity of breast tumors is plotted for ProSense680 (n = 12 tumors, 3 rats) and ProSense750 (n = 8 tumors, 3 rats). Camera exposure time was 10 ms. Horizontal lines represent mean ± SD. The fluorescent signal of the tumors was higher with ProSense680 than with ProSense750 (t = 4.33, p = .0004). C, The tumor to background ratio is plotted for both ProSense680 (n = 12 tumors, 3 rats) and ProSense750 (n = 8 tumors, 3 rats) for the camera exposure times that provided the optimal tumor to background ratio during surgery: 10 ms for ProSense680 and 20 ms for ProSense750. Horizontal lines represent mean ± SD. Tumor to background ratio was significantly higher for ProSense680 when compared to ProSense750 (t = 2.53, p = .021).
Breast Tumor Margins
To visualize the fluorescence of tumor margins, excised breast tumors were sectioned, imaged with the Fluobeam, and processed for histopathology. In Figure 4, a typical example of a 4 mm large excised breast tumor is presented. A distinctive difference in fluorescence of the tumor tissue and surrounding mammary fat pad is shown, which is confirmed in a fresh frozen tissue section after hematoxylin and eosin staining.
Metastatic Abdominal Cancer
To assess if a protease-activatable probe could be used in tumor types located in the abdominal cavity, the fluorescence intensity of abdominal organs relevant in cancer surgery was measured with the Fluobeam in three rats before and after injection with ProSense680. Figure 5 demonstrates that liver, kidney, spleen, small bowel, and bladder provide very high background fluorescence after injection of ProSense680. The fluorescence intensity of liver tissue was 69 times higher than in control rats (t = 8.85; p = .003), reflecting the high intrinsic cathepsin B activity of hepatocytes. 27 Also, the signal in the kidney was 32 times higher in the ProSense680 rats, particularly in the renal cortex, owing to the intrinsic cathepsin B activity and the renal clearance of the probe (t = 6.06; p = .009). 28 Similar results were obtained with ProSense750. These results suggest that protease-activatable probes are less useful for image-guided surgery of abdominally located tumors or metastases regardless of the protease activity and associated fluorescence intensity of those tumors.
To test the assumption that the use of ProSense in detecting abdominal tumors is limited, the syngeneic CC531 colorectal cancer rat model was used to induce intraperitoneal tumors. Figure 6 shows a typical example of a rat bearing intraperitoneal CC531 metastases imaged with the Fluobeam after injection with ProSense750. Although small peritoneal metastases could be detected on the mesentery of the small bowel (see Figure 6, A and B), one cannot be sure of detecting metastases in all parts of the bowel and in particular at the liver and kidney owing to the high background fluorescence, as illustrated in Figure 6C.

NIRF imaging of excised primary breast cancer using the Fluobeam camera system. Shown are a color image (A), a NIRF image (B), and a pseudocolored green merge of the two images (C) of a sectioned 4 mm EMR86 breast tumor with surrounding mammary fat pad. The tumor was excised from a rat, which was injected with 10 nmol ProSense680 24 hours prior to imaging. Camera exposure time was 10 ms. D, Hematoxylin-eosin histologic staining of a 20 μm frozen tissue section of the specimen from Figure 3C. Shown is the region indicated by the dashed square (X25 original magnification).

In vivo fluorescence intensity of EMR86 breast tumors and abdominal organs. Fluorescence intensity was measured using the Fluobeam camera system in three rats bearing EMR86 breast tumors before and 24 hours after administration of 10 nmol ProSense680. Camera exposure time was 10 ms. Bars represent mean ± SD. The signal for liver and kidney was saturated at 10 ms camera exposure times (dashed line). The plotted values are extrapolated from the nonsaturated measurements at 5 ms camera exposure time.
Discussion
A highly promising new development in surgical oncology is image-guided tumor resection using NIRF imaging. The extent of the primary tumor as well as sites of regional disseminated disease can be detected in real time. Using this new information, the surgical procedure can be directly adapted. Moreover, this technique provides a direct assessment of the resection plane after tumor removal to detect any residual cancer tissue, thereby reducing the number of patients with incomplete tumor resections. However, NIRF imaging with tumor-specific probes has not been evaluated in humans. The main target of ProSense, the cysteine protease family (in particular cathepsin B), is upregulated in various human cancers, including breast and colorectal cancer.29–32 In the current preclinical study, we described the performance of a novel handheld, intraoperative NIRF camera system based on a 690 nm laser and demonstrated its utility in detecting and guiding resection of primary and metastatic rat tumors using the cathepsin-activatable probe ProSense. Because the family of proteases is strongly conserved among mammals, it is expected that ProSense will be applicable for cancer patients. 29
The efficacy of NIRF camera systems is determined by the interplay between the type of probe used, probe concentration, tumor size, and camera exposure time. In this study, the cathepsin-activatable probes ProSense680 and ProSense750 were used to test the 690 nm laser–based Fluobeam camera system. Because of the better matching of ProSense680 with the 690 nm laser, the fluorescent signal was significantly higher using ProSense680 (see Figure 2A). During the intraoperative experiments, 10 ms camera exposure time was found to be optimal in terms of tumor to background ratio for ProSense680. When assessed in vitro, the minimal detectable ProSense680 concentration at 10 ms camera exposure time was approximately 10 nM (see Figure 2A). The minimal detectable number of tumor cells with 10 nM ProSense680 at 10 ms camera exposure time was approximately 20,000 for a breast cancer cell line and 13,000 for a colorectal cancer cell line. These cell numbers reflect submillimeter tumor depositions. When these settings were applied in the in vivo experiments, mammary tumors varying in size from 0.08 to 4.19 cm 3 could be detected and subsequently resected under direct fluorescence guidance after administration of 10 nmol ProSense680 with a clear demarcation of tumor margins (see Figure 4). Fluorescence reflectance imaging is not inherently quantitative, and quantification should always be performed relative to control tissue. 5 In this study, a tumor to background ratio of 2.34 was found for ProSense680. Tumor tissue was discriminated from the surrounding tissue by optimal thresholding without any further signal improvement techniques. 33
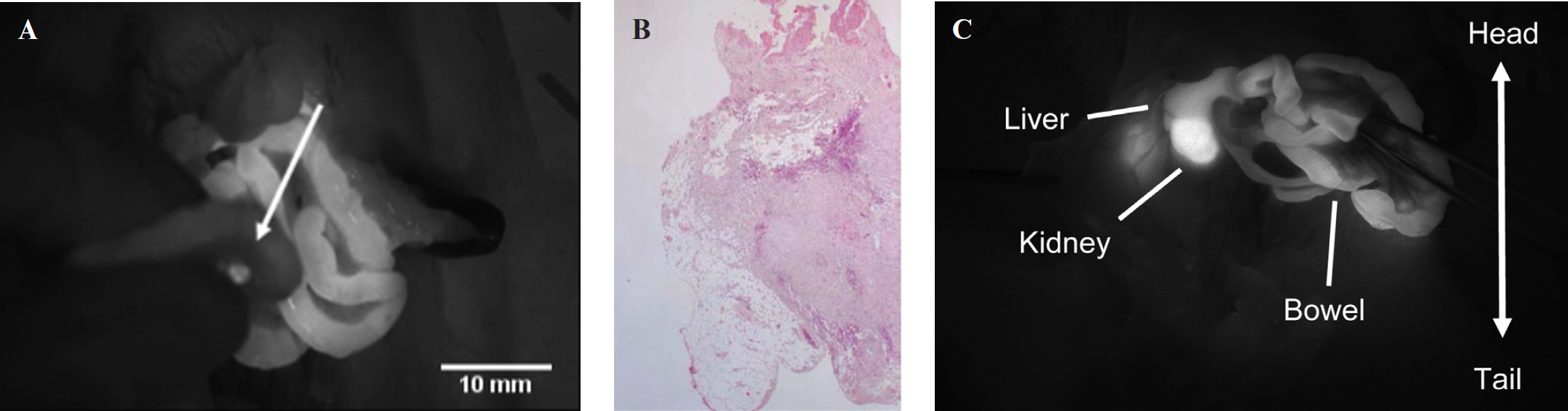
Intraoperative NIRF imaging of colorectal CC531 peritoneal metastases using ProSense and the Fluobeam camera system. A, NIRF image showing a 3 mm fluorescent hotspot (arrow) located on the mesentery of the small bowel in a male rat injected with 10 nmol ProSense750. Camera exposure time was 20 ms. B, A corresponding hematoxylin-eosin–stained, formalin-fixed, paraffin-embedded 4 μm tissue section confirms the presence of CC531 tumor cells (X25 original magnification). C, Intraoperative NIRF image showing the background fluorescence of the liver, kidney, and small bowel in the same animal. Camera exposure time was 20 ms.
In general, the ratio of the fluorescence intensity between tumor and surrounding tissue determines the applicability of protease-activatable probes in NIRF image-guided cancer surgery. To assess the applicability of the cathepsin-sensing probe in intraperitoneal metastases, the fluorescence intensity of abdominal organs was measured after the administration of ProSense680. In several abdominal organs, the fluorescence intensity was markedly increased after injection of ProSense (see Figure 5). For example, liver, kidney, and intestine cells exploited an extensive cathepsin B activity and were highly fluorescent. 27 Therefore, cathepsin-activatable probes seem less useful for image-guided surgery of abdominally located tumors, even if tumors express high levels of cathepsin B (see Figure 6).
The currently available NIRF camera systems use either a laser13,14 or LED11,12,19 illumination as a photon source for probe excitation. The Fluobeam camera system is laser based, which might possess logistic hurdles in terms of operator safety, although a distance of 18 cm away from the camera head is considered safe. However, LED illumination requires direct cooling at the camera head, causing camera heads to be larger. Conversely, a fiber-guided laser source can be cooled outside the camera head; therefore, the camera head can be smaller and a true handheld system can be created. Direct comparison of various camera systems is necessary to test accuracy and efficacy parameters such as minimal detection limit, excitation power, and signal to noise ratio in a standardized way, as presented here. The advantages of the Fluobeam system include its compact size, high resolving power and spatial resolution, user-friendly and surgeon-oriented mode of operation, and commercial availability. Moreover, the software incorporates the use of a dynamic threshold function, which could enhance the discrimination of tumor and normal tissue. Improvements in the Fluobeam could include the addition of a color video camera for visible light registration, as is already implemented in the LED-based FLARE system. 15
Several new developments in optical imaging, such as fluorescence lifetime imaging, 34 frequency domain imaging, and spatially modulated structured light, 35 have the potential to increase tissue penetration and discriminatory power between fluorophores. These techniques are able to improve localization of an NIRF signal source and improve quantification of the fluorescence signal. These developments are expected to be integrated into intraoperative NIRF imaging devices within the next few years.
In conclusion, the ultimate goal of NIRF imaging is to provide surgical oncologists with a real-time tumor imaging technique to guide surgery for the complete and safe resection of cancer tissue. In this study, we described the technical details and performance of the Fluobeam intraoperative NIRF camera system. By using the Fluobeam and the activatable probe ProSense, we demonstrated that it is possible to resect tumors under fluorescence guidance. If these techniques become available for clinical cancer treatment, surgical oncology will make a major step forward.
Footnotes
Acknowledgments
We want to thank Fluoptics for providing us with the Fluobeam system to perform the above-described experiments and Gabi van Pelt for technical assistance.
Financial disclosure of authors: This study was performed within the framework of the Center for Translational Molecular Medicine (DeCoDe project, grant 03O-101). This study was supported by the Sacha Swarttouw-Hijmans Foundation. J.S.D. Mieog is a MD-medical research trainee funded by the The Netherlands Organisation for Health Research and Development (grant no. 92003526).
Financial disclosure of reviewers: None reported.
