Abstract
One limitation of HSV1-tk reporter positron emission tomography (PET) with nucleoside analogues is the high background radioactivity in the intestine. We hypothesized that endogenous expression of thymidine kinase in bacterial flora could phosphorylate and trap such radiotracers, contributing to the high radioactivity levels in the bowel, and therefore explored different strategies to increase fecal elimination of radiotracer. Intestinal radioactivity was assessed by in vivo microPET imaging and ex vivo tissue sampling following intravenous injection of 18F-FEAU, 124I-FIAU, or 18F-FHBG in a germ-free mouse strain. We also explored the use of an osmotic laxative agent and/or a 100% enzymatically hydrolyzed liquid diet. No significant differences in intestinal radioactivity were observed between germ-free and normal mice. 18F-FHBG-derived intestinal radioactivity levels were higher than those of 18F-FEAU and 124I-FIAU; the intestine to blood ratio was more than 20-fold higher for 18F-FHBG than for 18F-FEAU and 124I-FIAU. The combination of Peptamen and Nulytely lowered intestinal radioactivity levels and increased (2.2-fold) the HSV1-tk transduced xenograft to intestine ratio for 18F-FEAU. Intestinal bacteria in germ-free mice do not contribute to the high intestinal levels of radioactivity following injection of radionucleoside analogues. The combination of Peptamen and Nulytely increased radiotracer elimination by increasing bowel motility without inducing dehydration.
MONITORING ADOPTIVE CELL-BASED THERAPIES and viral vector tracking in gene therapy are challenging, yet they are currently the most clinically relevant applications of reporter gene imaging. The herpes simplex virus type 1 thymidine kinase gene (HSV1-tk), either wild type or the sr39tk mutant, currently is the most commonly used radiotracer-based reporter gene used in positron emission tomography (PET) studies.
1
HSV1-tk gene expression can be effectively imaged using radiolabeled substrates that are selectively phosphorylated by the HSV1-TK enzyme and the phosphorylated substrate is unable to cross the cell membrane; thus, the phosphorylated substrate is essentially trapped within the HSV1-tk transduced cell.
2
The two most commonly used substrates are [124I]-2′-fluoro-1-h-
The characteristics of a good substrate for reportergene imaging studies in vivo include not only high selectivity and sensitivity for the gene product but also a favorable pharmacokinetic clearance profile. The level of background radioactivity must be low and not obscure the specific signal in the anatomic site of interest. The most notable levels of background radioactivity following injection of radiolabeled nucleosides are observed in the abdomen owing to both renal and hepatobiliary clearance of such agents. High levels of radioactivity are initially seen throughout the renal tract, but at later times (1–4 hours), they are largely confined to the bladder.4, 5 The hepatobiliary-intestinal clearance presents a greater problem, resulting in a high and heterogeneous pattern of background radioactivity throughout the abdomen3, 5, 6 that remains high over several hours, often obscuring specific structures of interest. 7
Nimmagadda and colleagues demonstrated in dogs that the time-activity profile of 18F-FIAU in the gallbladder begins to increase 10 minutes after intravenous (IV) injection and continues to increase over 60 minutes as the tracer is cleared from the blood. 8 Yaghoubi and colleagues reported that background radioactivity in the intestine in humans following IV injection of 18F-FHBG was high (almost 10%ID/g) 25 minutes after injection, with a further slight increase over time. 9 Several other HSV1-tk probes have been developed and tested both in vitro and in vivo, and their advantages and disadvantages have been extensively discussed in the literature.4, 10 18F-FHBG, owing to its lipophilic structure, has been associated with higher hepatobiliary secretion and higher intestinal background activity compared to 18F-FEAU. 11 However, to our knowledge, no studies have investigated mechanisms other than hepato-biliary clearance to explain the rapid and diffuse uptake of the radioactivity throughout the whole intestine. Also, no strategy has been suggested in the literature to reduce the abdominal background, particularly the intestinal component.
Similarities between HSV1-tk and prokaryotic tk genes have been described. Bettegowda and colleagues demonstrated that endogenous bacterial thymidine kinases are sufficiently similar to that of viral thymidine kinases to permit bacteria to be imaged in mammalian hosts using the same radiolabeled nucleoside analogues. 12 Prokaryotic tk genes in a wide range of bacteria allowed the detection and imaging of these bacteria with 125I-FIAU in experimentally created infections. This approach has also been successfully tested for visualizing musculoskeletal bacterial infections using 124I-FIAU PET–computed tomography (CT) in a small number of patients. 13 We previously reported that a probiotic bacterial strain of Escherichia coli Nissle 1917, which endogenously expresses HSV1-TK, trafficked to a subcutaneous (SC) breast tumor xenograft following IV administration and that the tumor-targeting bacteria could be visualized by 18F-FEAU and 124I-FIAU microPET. Given that the SC tumors were located in the shoulders of the host mice, the renal-hepatobiliary clearance of the radiotracers did not interfere with measurements of the tumor uptake. Furthermore, it was also shown that the level of tumor radioactivity could be used as an indicator of the number of bacteria present in the target tissue. 14
The role of commensal intestinal flora in the gut and the clearance of radiolabeled nucleoside analogues from the body have not been investigated previously. This commensal population, composed of anaerobic, aerobic, and facultative aerobic bacteria, represents a heterogeneous microbial ecosystem containing approximately 1014 bacteria, forming a natural protective barrier and exerting numerous protective, structural, and metabolic effects on the intestinal epithelium.15, 16
In this study, we tested the hypothesis that radionucleoside analogues can translocate from the bloodstream to the intestinal mucosa, where they can be taken up, phosphorylated, and trapped by the commensal intestinal flora contributing in this manner to the high and early intestinal background observed in radionucleoside imaging. We determined whether abdominal background activities of three different radionucleoside analogues (18F-FEAU, 18F-FHBG, and 124I-FIAU) are substantially different in germ-free mice raised in a sterile vivarium environment compared to animals with normal bacterial flora raised in a normal vivarium environment. We evaluated organ-specific activities by both in vivo microPET imaging and by ex vivo tissue sampling. In addition, we evaluated different strategies to reduce the intestinal background by increasing intestinal motility and radiotracer fecal excretion in nude mice bearing an HSV1-TK-expressing xenograft.
Materials and Methods
18F-FEAU, 18F-FHBG, and 124I-FIAU Syntheses
18F was produced on the Memorial Sloan-Kettering Cancer Center (MSKCC) cyclotron. 18F-FEAU was synthesized by coupling the radiolabeled fluoro sugar with the silylated pyrimidine derivatives following a procedure previously reported by Serganova and colleagues. 17 The specific activity of the 18F-FEAU was ≈ 37 GBq/μmol (≈ 1 Ci/μmol), and the radiochemical purity was > 95% after purification by high-performance liquid chromatography (HPLC). The product was formulated in 0.9 NaCl containing 5% ethanol and terminally sterilized by filtration.
18F-FHBG was synthesized using 18F in the form of potassium fluoride in aqueous solution as previously described. 18 The specific activity of the final product was ≈ 70 GBq/μmol (≈ 1.9 Ci/μmol), and radiochemical purity was > 96% by HPLC.
124I-FIAU was synthesized by reacting the precursor of 5-trimethyl-stannyl-1-(2-deoxy-2-fluoro-β-
Cell Lines
The RG2 rat glioma cell line was obtained from American Type Culture Collection (Manassas, VA). RG2 cells were transduced with the recombinant replication-deficient STK retrovirus containing the NeoR gene and HSV1-tk gene. The transduced cell line, RG2-tk, has been characterized previously. 19
Experimental Groups of Animals
Animal studies were performed in compliance with all applicable policies, procedures, and regulatory requirements of the Institutional Animal Care and Use Committee, the Research Animal Resource Center of MSKCC, and the National Institutes of Health (NIH) Guide for the Care and Use of Laboratory Animals.
Three groups of five germ-free (defined as a gnotobiote— free of all demonstrable microbial association) Swiss Webster mice 6 to 8 weeks old raised in a sterile environment (Taconic Farms, Inc., Germantown, NY) were shipped in a sterile container with sterile food and kept under sterile conditions until imaging. Experiments were conducted on 3 consecutive days after shipment. Each day a single cage of five animals was carefully removed using sterile technique to preserve the sterility of the container and of the remaining cages. Fecal pellets were collected in each cage and microbiologically evaluated to confirm the germ-free status of the animals. Three groups (n = 5 per group) of bacteria-associated Swiss Webster mice 6 to 8 weeks old (Taconic Farms, Inc.) raised in a normal vivarium environment were used as controls. Fecal pellets were also collected from the control animals and evaluated for bacteria in the same manner.
Three- to 4-week-old athymic nu/nu mice (CrTac:NCr-Foxn1nn, Taconic Farms, Inc.) were used in a second set of experiments involving tumor implantation. Twenty mice, divided into four groups (control, Nulytely [Braintree Laboratories, Braintree, MA], Peptamen [Nestlé Nutrition, Vevey, Switzerland], combination of Peptamen and Nulytely; n = 5/group) were studied with two tumor xenografts produced in each animal. Transduced RG2 HSV1-tk cells (test) and wild-type RG2 cells (control) were removed by trypsinization and washed in phosphate-buffered saline (PBS), and 1.0 × 106 cells (resuspended in 50 μL Matrigel) were implanted subcutaneously into the right and left shoulders, respectively, of each animal in all four groups. Mice were monitored for tumor growth by daily measurements of the tumor size and of the animal's weight. Animals were studied when the SC xenografts reached a diameter of 12 to 15 mm, 14 to 21 days after SC implantation of the RG2 and RG2-HSV1-tk cells. The in vivo growth rate and macroscopic appearance were similar in the HSV1-tk transduced and wild-type SC xenografts.
All animal procedures were performed under inhalation of 2% isoflurane anesthesia. After the studies, all animals were sacrificed by CO2 inhalation.
Different Strategies to Increase Radionucleoside Excretion
We compared three strategies to increase intestinal motility and reduce fecal mass relative to those for the standard diet. To reduce the intestinal content, we used a complete peptide-based elemental nutritional diet (Peptamen) formulated for the dietary management of patients with inflammatory bowel disease, pancreatic insufficiency, short bowel syndrome, radiation enteritis, and chronic diarrhea. Peptamen provides high-energy nutrition (10 kcal/mL; caloric distribution in % of kcal: protein, 16%; carbohydrate, 51%; fat, 33%) without formation of feces. A vanilla-flavored Peptamen (enzymatically hydrolyzed whey protein) was chosen to improve the taste of the solution to stimulate feeding. A ≈ 1:2 dilution in water was placed in a regular 450 mL water bottle.
To increase bowel motility and obtain faster excretion of the radiotracer, an isosmotic laxative agent (Nulytely; PEG-3350, sodium chloride, sodium bicarbonate, and potassium chloride for oral administration) was used. Nulytely is given orally to patients for bowel cleansing prior to colonoscopy. According to the vendor, Nulytely is solubilized in water to obtain a solution containing PEG-3350 31.3 mmol/L, sodium 65 mmol/L, chloride 53 mmol/L, bicarbonate 17 mmol/L, and potassium 5 mmol/L. The final product was placed in a cage water bottle.
The control group was fed with a standard diet and water ad libitum for 21 days after tumor implantation. For the Nulytely group, regular solid food and water were provided for the first 18 days after tumor implantation; solid food was then discontinued and Nulytely was substituted 36 hours before imaging and sacrifice. For the Peptamen group, a regular diet was provided for 7 days after tumor implantation and then a Peptamen diet for 14 days prior to imaging. The fourth “combination” group received a normal diet for 7 days after tumor implantation followed by Peptamen for 14 days and Nulytely for 36 hours before imaging and sacrifice. Animals determined to be dehydrated were administered fluids intraperitoneally as described in the institutional guidelines for maintaining fluid homeostasis.
MicroPET Imaging
18F-FEAU
Five germ-free Swiss Webster mice and five bacteria-associated Swiss Webster mice were injected retro-orbitally with 9.25 MBq (250 μCi) of 18F-FEAU and imaged sequentially. A separate cohort of 20 nude mice divided into four groups (control, Nulytely, Peptamen, combination of Peptamen and Nulytely) were injected retro-orbitally with 9.25 MBq (250 μCi) of 18F-FEAU. MicroPET imaging was performed 2 hours after tracer administration using a 10-minute list-mode acquisition.
18F-FHBG
Five germ-free Swiss Webster mice and five bacteria-associated Swiss Webster mice were injected retro-orbitally with 9.25 MBq (250 μCi) of 18F-FHBG. MicroPET imaging was performed 2 hours after tracer administration using a 10-minute list-mode acquisition.
124I-FIAU
Five germ-free Swiss Webster mice and five bacteria-associated Swiss Webster mice were injected retro-orbitally with 29.6 MBq (800 μCi) of 124I-FIAU. MicroPET imaging was performed 2 and 24 hours after tracer administration using 10-minute and 30-minute list-mode acquisitions, respectively.
Germ-free animals were injected and handled under sterile conditions. The preanesthesia box, nose cone, and microPET table were rinsed with 100% ethanol, and measures to avoid contamination were implemented. After tracer administration and before imaging, animals were allowed to recover from anesthesia in their sterile cages and thus to maintain their germ-free status. Imaging was done using a Focus 120 microPET dedicated small-animal PET scanner (Concorde Microsystems, Inc., Knoxville, TN). Mice were maintained under 2% isoflurane anesthesia with an oxygen flow rate of 2 L/min during the entire scanning period. Three-dimensional list-mode data were acquired using an energy window of 350 to 700 keV for 18F and 410 to 580 keV for 124I and a coincidence timing window of 6 nanoseconds. Data were sorted into two-dimensional histograms by Fourier rebinning, reconstructed, and normalized as previously described. 14
MicroPET image analysis was performed using ASIPro software (Concorde Microsystems, Inc.). For each microPET scan, the intestine was identified and regions of interest (ROIs) were manually drawn in the anterior abdominal compartment. ROIs were also placed over the RG2 wild-type and RG2 HSV1-TK xenografts. Reconstructed images were parameterized in terms of %ID/g and the maximum and total voxel values were recorded for each tissue; the tumor to organ radioactivity concentrations ratios were also calculated.
Radioactivity Quantification of Tissue Samples
Euthanized mice were rinsed with 100% ethanol before tissue removal. Large and small intestines were removed “en bloc” with their fecal content and liver, gallbladder, kidneys, heart, skeletal muscle, and blood were sampled and weighed before radioactivity measurement. Tissue harvesting of germ-free mice was performed under sterile surgical conditions and organs were collected in sterile scintillation vials. 18F-FEAU-, 18F-FHBG-, and 124I-FIAU-derived radioactivity (%ID/g) in the tissue samples was measured in a Wizard 1470 gamma counter (Perkin-Elmer, Waltham, MA), and the tissue to blood ratios were calculated. The correlation between microPET and scintillation counter radioactivity values was assessed.
Bacterial Evaluation of Intestinal Samples
Microbiologic analysis was performed on the fecal pellets (collected from the cage before injection and imaging) and on both small and large intestinal contents from germ-free and control mice. Postmortem intestinal samples were collected under sterile conditions. The fecal pellets and intestinal samples were homogenized in 1 mL PBS and dilutions (1:10, 1:100, and 1:1,000) were plated on blood agar plates.
Statistics
One-way analyses of variance were applied using Prism software (GraphPad Software, San Diego, CA) to determine the statistical significance among the experimental groups. Statistical significance was set at p < .05. Results were reported as mean ± SD.
Results
Comparison of Radiotracer Distributions and Clearance in Germ-Free and Control Mice
18F-FEAU and 18F-FHBG radioactivity (%ID/g) was imaged/measured in different organs/tissues of germ-free and control mice 2 hours after IV administration, whereas 124I-FIAU was imaged/measured at 24 hours in the same animal cohorts (Figure 1). The absence of enteric bacterial growth from fecal pellets and intestinal samples from the germ-free mice compared to control mice (Figure 2A) confirmed the germ-free status of these mice during the 18F-FEAU and 18F-FHBG study.

Ex vivo biodistribution of 18F-FEAU, 18F-FHBG, and 124I-FIAU in germ-free and control mice. Ex vivo biodistribution (%ID/g) (left panel) and normalized to blood data (right panel) in mice following injection of (A) 18F-FEAU, (B) 18F-FHBG, and (C) 124I-FIAU in germ-free and control mice. No statistically significant difference in large intestine radioactivity (%ID/g) was observed between the germ-free and control animals. Radioactivity measured in the small intestine was slightly lower in germ-free animals than controls for each radiotracer, although not statistically significant. 18F-FHBG was associated with higher large intestine radioactivity (2.9-fold compared to 18F-FEAU and 18-fold compared to 124I-FIAU, p < .001). The lowest levels were observed with 124I-FIAU at 24 hours. The intestine to blood ratio for 18F-FHBG (201 ± 37) was very high compared to that for 18F-FEAU (6.9 ± 4.5) at 2 hours and 124I-FIAU (6.4 ± 2.3) at 24 hours.

18F-FEAU, 18F-FHBG, and 124I-FIAU microPET imaging in germ-free and control mice. A, Pictures of the blood agar plates where homogenized samples of the fecal pellet were placed (1:10 dilutions) to confirm the germ-free status versus control groups of animals. The red-to-brown color shift indicates the presence of metabolic activity, corresponding to the growth of bacteria. Bacterial colonies were visible in the brown (control) plates but not in the plates from the germ-free animals. B, Coronal and axial PET images of germ-free versus control mice at 2 hours after injection of 18F-FEAU and 18F-FHBG and 24 hours after injection of 124I-FIAU. 18F-FHBG is associated with the highest abdominal radioactivity compared to 18F-FEAU and 124I-FIAU; note the higher threshold of the intensity bar (5% ID/g) for 18F-FHBG. No difference is observed in the PET images between germ-free and control animals in each radionucleoside group. C, Comparison of the intestinal uptake of 18F-FEAU, 18F-FHBG, and 124I-FIAU in germ-free versus control mice, calculated from multiple intestinal regions of interest drawn on the whole intestine on the axial plane. Values of the maximum pixel value (maximum %ID/g) and total activity (%ID/g) are illustrated. The highest values were observed for 18F-FHBG; maximum pixel (%ID/g) intestinal background was ≈ 6.7-fold higher than that for 18F-FEAU (p < .05) and more than 50-fold higher than that for 124I-FIAU (p < .001).
It should be noted that all visceral contents were included in the ex vivo measurements of tissue radioactivity.
No differences in organ activities were observed between germ-free and control mice, including the large intestine and its contents: 18F-FHBG (2.38 ± 0.84 vs 2.67 ± 0.58 %ID/g), 18F-FEAU (0.86 ± 0.40 vs 0.83 ± 0.42 %ID/g), and 124I-FIAU (0.15 ± 0.07 vs 0.09 ± 0.07 %ID/g), respectively. Although the radioactivity levels in the small intestine were slightly lower in germ-free than in control animals for each radiotracer, these differences were not statistically significant. The highest radioactivity levels were observed with 18F-FHBG in the large intestine and gallbladder. Normalizing intestinal radioactivity to that in blood yielded intestine to blood ratios of 201 ± 37 for 18F-FHBG and 6.9 ± 4.5 for 18F-FEAU at 2 hours and 6.4 ± 2.3 for 124I-FIAU at 24 hours.
The microPET images were consistent with the tissue sampling results; no difference in the pattern of abdominal and total body radioactivity distributions was observed between germ-free and control animals (Figure 2B). Specifically, quantitative analysis of the anterior abdominal compartment of the microPET images, based on maximum voxel and total radioactivity values (%ID/g), yielded no statistically significant differences between germ-free and control mice (Figure 2C). The highest maximum-voxel and abdominal radioactivity values (%ID/g) were observed for 18F-FHBG; they were 6.7- and 6.6-fold higher than 18F-FEAU (p < .05) and more than 50- and 13-fold higher than 124I-FIAU (p < .001), respectively. Importantly, by setting a higher threshold for 18F-FHBG compared to 18F-FEAU and 124I-FIAU, only the higher activity in the gut was visible (see Figure 2B).
Strategies to Reduce 18F-FEAU Background Intestinal Radioactivity Levels
We developed and tested several different bowel-cleansing regimens to increase abdominal clearance of 18F-FEAU during the study, including an osmotic agent (Nulytely), a peptide-based elemental nutrition regimen (Peptamen), and a combination of Peptamen and Nulytely (Figure 3). Only the combination of Peptamen and Nulytely resulted in a significant decrease in large intestine radioactivity levels compared to that in controls (see Figure 3A). Owing to dehydration resulting in a 14 ± 0.5% weight loss in the Nulytely group of animals (see Figure 3C), renal excretion of 18F-FEAU was reduced and high tissue and blood values were measured at 2 hours postinjection. Dehydration occurred in the Nulytely-treated group despite intraperitoneal hydration, whereas only a slight weight loss (6 ± 1%) was noted in the Peptamen plus Nulytely treatment group. No change in animal weight was observed with Peptamen treatment alone. Hydration status and radiotracer retention have to be taken into consideration to meaningfully compare the different bowel-cleansing strategies. Normalizing the 2-hour tissue data to that of blood yielded significantly lower large intestine to blood ratios for the Nulytely (86% decrease; p < .001) and the combination (Peptamen plus Nulytely) treatment groups (65% decrease, p < .005) compared to control animals (see Figure 3B).
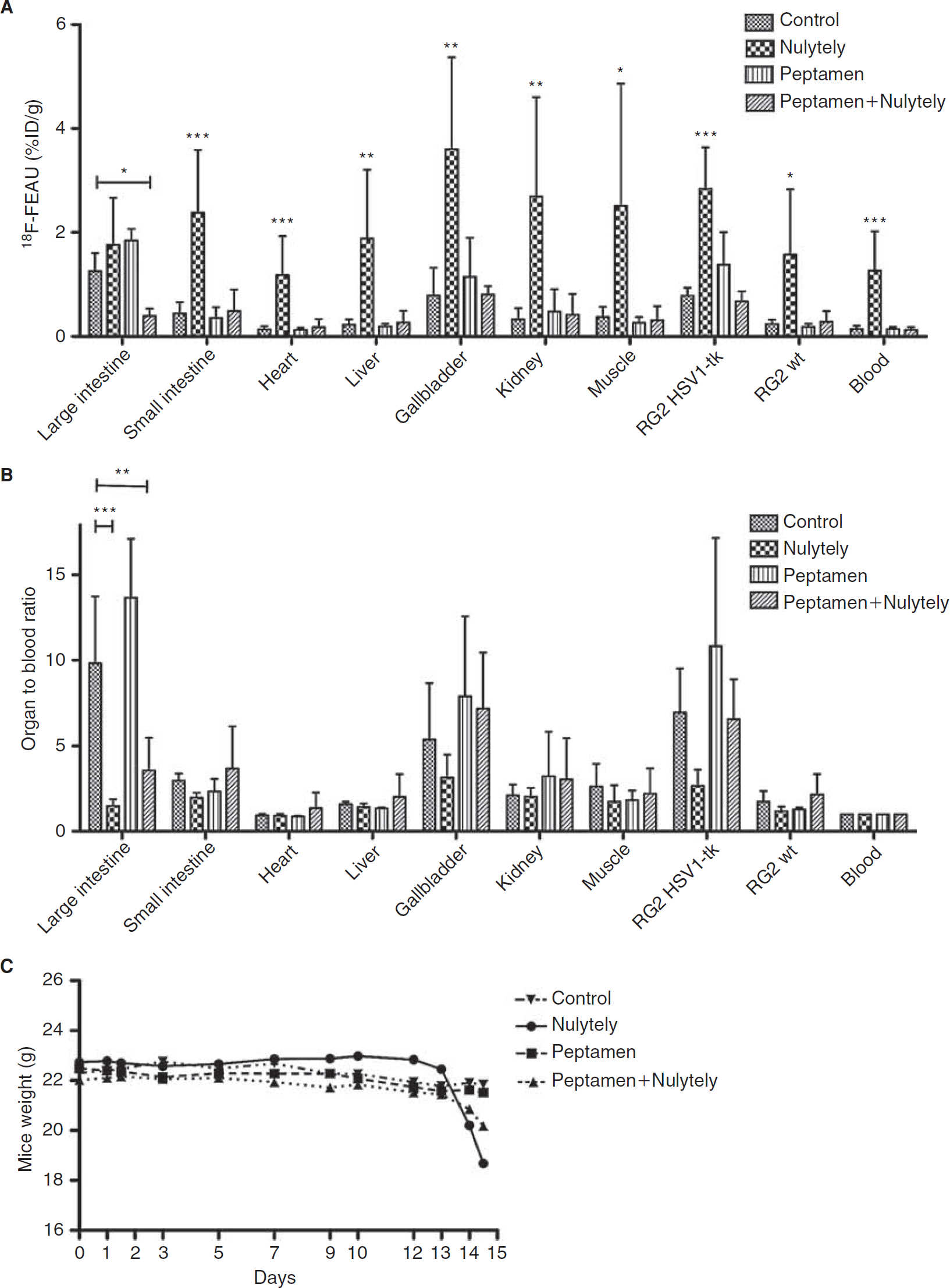
Different strategies in increasing radionucleoside excretion and 18F-FEAU biodistribution. Biodistribution of 18F-FEAU in tumor-bearing nude mice at 2 hours after injection, treated with Nulytely (36 hours), Peptamen (14 days), and a combination of Peptamen (14 days) and Nulytely (36 hours) versus control (regular diet and water ad libitum). Values are expressed in (A) %ID/g and (B) organ-to-blood normalized data.(*p < .05; **p < .01; ***p < .001). Asterisks in (A) indicate the statistically significant difference between the Nulytely group and the control group. The highest values (1.5- to 8-fold higher compared to the control group) were observed in the Nulytely group. The normalization of tissue (organ) radioactivity to that in blood takes into consideration the hydration status of the mice. C, Different intestinal cleansing strategies and their effect on animal weight. A 14 ± 0.5% weight loss was observed after 36 hours of Nulytely treatment alone owing to dehydration and food deprivation. The combination of Nulytely and Peptamen resulted in only 6 ± 1% weight loss during the same period of treatment.
Optimizing HSV1-tk Reporter Imaging by Reducing Intestinal Background Radioactivity
The visualization of the RG2 HSV1-tk xenografts with 18F-FEAU relative to background intestinal radioactivity was optimized in the animals treated with the combination of Peptamen and Nulytely compared to the control and the other treatment groups (Figure 4). The measured 18F-FEAU radioactivity profile comparing the different treatment groups, using either microPET or tissue-sampling measurements of RG2 HSV1-tk xenograft and large intestine radioactivity, was similar (see Figure 4). Intestinal radioactivity measured by microPET (Figure 5) and by tissue sampling in animals treated with the combination of Peptamen and Nulytely was significantly lower (62% and 69% decrease, respectively) compared to control and the other treatment groups.

Quantitative PET analysis and tissue sampling data. Comparison of microPET data (left panel) and tissue sampling data (right panel) for intestinal and tumor (HSV1-TK) 18F-FEAU-derived radioactivity (%ID/g) in nude mice bearing HSV1-TK transduced and wild-type RG2 xenografts, treated with different bowel cleansing agents. In the left panel, the values of maximum (%ID/g) for (A) tumor RG2 (HSV1-TK), (B) intestine (calculated by drawing multiple intestinal on the whole intestine on the axial plane), and (C) the calculated tumor to large intestine ratio are graphed. Similar graphs are presented for the tissue sampling results (D), (E), (F). Significant differences between treatment groups are indicated (*p < .05; **p < .01; ***p < .001).

Different strategies and their effect in mice. Coronal and axial 18F-FEAU PET images of nude mice bearing a RG2 HSV1-TK xenograft in the right shoulder (circle) and RG2 wild-type xenografts in the left shoulder (dotted circle). Coronal images are shown through the midabdomen. Axial images are shown for a section through the abdomen indicated by the orange dotted line. Note that the high intestinal background observed with the laxative agent alone (Nulytely) is due to dehydration and high radioactivity retention.
The tumor to intestine ratios measured by microPET and by tissue sampling (see Figure 4C) were similar and significantly higher (2.2- [p < .01] and 2.5-fold [p < .05], respectively) for the Peptamen plus Nulytely combination treatment compared to the controls. No significant differences were observed between the other treatment groups and the controls. Importantly, the HSV1-tk transduced to wild-type tumor ratios did not change significantly between treatment groups and control animals.
Discussion
In the current study, we investigated whether normal enteric bacterial flora contributes to the intestinal accumulation of radiolabeled nucleoside analogues that are commonly used to image HSV1-tk expression. It has been shown that bacteria can transport and phosphorylate nucleoside analogues.12–14 It has also been shown that radiolabeled nucleoside analogues can be used to image bacterial infection and to image bacteria that target tumors.14, 20 We hypothesized that the high levels of intestinal radioactivity imaged in HSV1-tk reporter gene studies within 1 to 2 hours after injection could reflect not only the hepatobiliary excretion but also the translocation of the radionucleoside analogues from the bloodstream to the intestinal mucosa and consequent uptake of the radiopharmaceutical by normal intestinal flora. Although it is well known that radiolabeled nucleoside analogues are actively secreted into the small intestine through the hepatobiliary system, there is no evidence for direct secretion of nucleosides in the large intestine. The transport of radionucleoside analogues (such as 18F-FEAU, 124I-FIAU, 18F-FHBG) by the intestinal epithelium has never been investigated, although plasma membrane carriers are known to transport nucleosides and nucleobase analogues.21, 22 Nevertheless, the localization of radioactivity to the large bowel suggested that enteric bacteria could play a role.5, 14 To assess whether enteric bacteria contribute to high intestinal background in rodents, we studied the biodistribution of three radiolabeled nucleoside analogues in germ-free Swiss Webster mice and in the same strain of Swiss Webster mice exposed to a normal (nonsterile) vivarium environment.
To our knowledge, this is the first study to analyze the differences in intestinal background radioactivity among the three most commonly used radionucleoside analogues for imaging HSV1-tk or HSV1-sr39tk. The 18F-FEAU, 18F-FHBG, and 124I-FIAU biodistribution data are consistent in that the clearance of radioactivity from all organs, except for the intestine, is fairly rapid. At 2 hours, large intestine radioactivity levels are substantially higher than those in the small intestine, whereas with 124I-FIAU at 24 hours, both large and small intestine radioactivity levels are low. We demonstrated that the presence or absence of bacteria in the intestinal lumen does not affect intestinal background radioactivity. No statistically significant differences were observed in germ-free mice compared to control animals following injection of 18F-FEAU, 18F-FHBG, or 124I-FIAU. It is known that germ-free mice have distinctive characteristics other than the absence of bacteria, such as greater susceptibility to infection, reduced intestinal vascularity, digestive enzyme activity and muscle wall thickness, and fewer intraepithelial lymphocytes. 23 None of these factors appeared to have influenced our results. Our experimental design excluded bacteria as a major contributor to the high levels of intestinal background radioactivity, but it remains unknown whether a small, perhaps undetectable amount of radiotracer can be excreted by the intestinal epithelium and trapped by bacteria. The commensal flora is composed of aerobic, anaerobic, and facultative aerobic bacteria. It is known that the anaerobic proportion gradually increases from the proximal to distal regions of the intestine, reaching 99% of the inhabitants of the large intestine. 24 We demonstrated that differential accumulation of the radiotracer in the distal compared to the proximal part of the intestine is not related to a different bacterial population but rather to water reabsorption occurring in the distal sections of the intestine, thereby concentrating the radiotracer in the stool and reducing the intestinal progression and elimination of the radionucleoside.
Our biodistribution data for all of the radiotracers were comparable to those reported in the literature for 18F-FHBG4, 7, 25 and 18F-FIAU, 4 except for the intestine. We observed intestine radioactivity (%ID/g) values for 18F-FHBG that were almost 15-fold higher compared to those reported in nude mice by Allaudin and colleagues7, 25 and more than 20-fold higher than those reported in rats by Tjuvajev and colleagues. 4 Despite the differences in animal strain and species in these studies, the common denominator for this discrepancy is the technique used for harvesting and counting intestine samples, namely, intact intestine (containing feces) versus intestinal wall (cleansed of feces). It should be noted that a close correspondence was observed between the tissue sampling and microPET ROI radioactivity measurements (see Figure 4). This is also in accordance with our unpublished data, where the majority of intestinal radioactivity was found to reside in the stool, not in the intestinal wall. Lower levels of abdominal-intestinal background is one advantage for imaging HSV1-tk expression in target cells or tissue with 18F-FEAU and 124I-FIAU compared to 18F-FHBG.
A strategy to increase intestinal motility to facilitate the progression of stool and hepatobiliary excreted radionucleoside through the intestine was investigated. For this study, we selected a single agent, 18F-FEAU, because it has the best HSV1-tk imaging characteristics and it is likely that 18F-FEAU will be used to image HSV1-tk reporter gene expression more extensively in the future. It is also likely that these bowel-cleansing strategies will be more effective in experiments involving radionucleoside probes with higher intestinal background activity, such as 18F-FHBG. A laxative agent was successfully used in mice by Pickhardt and colleagues to cleanse the bowel prior to virtual CT colonoscopy. 26 We attempted a similar approach by increasing bowel motility with Nulytely and food deprivation. Despite the strict monitoring of the animals for signs and symptoms of dehydration, according to the institutional guidelines, the prolonged laxative treatment resulted in dehydration (14% weight loss) and systemic retention of radioactivity owing to decreased urine output. The effects of dehydration were reflected in higher blood and tissue radioactivity levels and a higher background visualized on microPET imaging compared to that in control animals. Given that the intestine has a highly branched and developed vascular system, we suggest that the majority of radioactivity observed in the large and small intestine samples of the Nulytely-treated animals is attributed to the blood (compare panels A and B in Figure 3). This does not appear to be the case for the control and Peptamen-treated animals. Therefore, normalization of tissue radioactivity to blood values is a better reflection of biodistribution differences between animal groups. Moreover, the higher levels of radioactivity obtained in the PET ROI measurements compared to the tissue sampling measurements (%ID/g) may reflect the analysis of the whole intestinal compartment without precise discrimination between intestine and abdominal vessels (blood pool). Food deprivation and feces loss induced by the PEG-based laxative also contributed to the weight loss. Nulytely was most effective in cleansing the intestine of radioactive stool compared to the other treatment groups, as measured by the normalized tissue to blood ratio data (see Figure 3). At necropsy, macroscopic viewing of the large and small intestine revealed the near-total absence of stool.
Peptamen has been used successfully by Cottart and colleagues as an alternative to laxative agents in cystic fibrosis transgenic mice to prevent intestinal obstruction. 27 A therapeutic benefit with prolonged survival was described for animals fed with Peptamen for more than 60 days. In our experiments, animals tolerated the Peptamen feeding well for 14 days, without any significant weight loss compared to control animals. Surprisingly, although the liquid diet was thought to transit the gastrointestinal tract more rapidly, the intestinal/abdominal level of radioactivity in the Peptamen group was not statistically different compared to control animals. Despite the higher lipid content of Peptamen compared to the standard pelleted diet A03 (Safe, Augy, France) (lipids g/100kcal: 3.7 vs 1.6), 27 it has been reported that an amino acid–containing diet may not adequately stimulate cholecystokinin (CCK) release. 28 The known effect of CCK in stimulating the contraction of the gallbladder, the lower esophageal sphincter, and the sphincter of Oddi, as well as its role in increasing the motility of the stomach and intestine, was reduced during Peptamen treatment. This probably explains the slightly higher values (although not statistically significant) observed in the Peptamen group compared to the control group. Moreover, the high variability in the gallbladder data and the small number of animals per group (n = 5) preclude resolution of this issue.
The disadvantages of two of the strategies were reduced by combining them (Peptamen plus Nulytely). The combination resulted in only a modest weight loss (6%) compared to control animals, which was most probably due to the reduction of intestinal contents and fecal mass. Importantly, the continuous feeding with Peptamen during Nulytely administration allowed us to partially overcome the dehydration issue. We did not observe signs of dehydration; therefore, supplemental fluid administration was not needed. The combined treatment gave a 62% and 69% decrease in large intestine (with feces) radioactivity, respectively, for tissue sampling and microPET measurements compared to controls.
Our final goal was to optimize the visualization of HSV1-tk reporter expression in tumor-bearing mice. This was assessed by comparing radioactivity measurements in HSV1-tk-expressing (transduced) xenografts to background activity measured in wild-type (nontransduced) xenografts and in the abdomen/large intestine. The microPET ROI analysis of the images showed an improvement of 2.2-fold (p < .001) in the HSV1-tk-bearing tumor to abdomen ratio for the Peptamen plus Nulytely group compared to the control group. In contrast, there was no change in the transduced to wild-type RG2 tumor ratio between the treatment groups. A more complete evaluation of the utility of the combination Nulytely-Peptamen treatment should be performed using different HSV1-tk transduced models, such as monitoring lymphocyte trafficking or metastatic bone disease, where abdominal/intestinal background radioactivity could be a greater problem and obscure visualization of HSV1-tk expression in abdominal structures.
Footnotes
Acknowledgments
We thank Ms. Valerie A. Longo (Small Animal Imaging Core), Aziz Toma, and Yuri Igarashi (Research Animal Resource Center) for technical assistance and Mr. Bradley Beattie and Dr. Vladimir Ponomarev for helpful discussion.
Financial disclosure of authors: This work was supported by an R25T Molecular Imaging Grant from the National Cancer Institute (CA096945) and by the MSKCC Center for Molecular Imaging in Cancer (P50-CA086438). Technical services were provided by the MSKCC Small-Animal Imaging Core Facility, supported in part by an NIH Small-Animal Imaging Research Program grant (R24 CA83084) and an NIH Center grant (P30 CA08748).
Financial disclosure of reviewers: None reported.
