Abstract
Ezetimibe (EZE), an inhibitor of cholesterol absorption, reduces atherosclerosis in apolipoprotein E–deficient (apo–/–) mice. The matrix protein ED-B fibronectin (ED-B) is upregulated in atherosclerotic lesions. Using a novel conjugate for near-infrared fluorescence (NIRF) imaging targeting ED-B, we studied the effect of EZE on plaque lesion formation in apoE–/– mice. ApoE–/– mice received EZE (5 μg/kg/d) or chow up to the age of 4, 6, and 8 months. NIRF imaging of aortic lesions was performed 24 hours after intravenous application ex vivo and in vivo. Plaque lesion formation was analyzed by histology and immunohistochemistry. Aortic lesion formation detected by Sudan staining and NIRF imaging was significantly reduced at 6 and 8 months (p < .001). Plaque areas determined by NIRF imaging significantly correlated with Sudan staining (p < .001). EZE treatment resulted in a significant reduction in plaque macrophage and ED-B immunoreactivity (both p < .05) in brachiocephalic lesions. There was a significant reduction in plaque size in brachiocephalic arteries in 8-month-old mice treated with EZE compared with mice during short-term treatment (p < .05), indicating EZE plaque regression. Targeted NIRF imaging showed a correlation to histologic lesion extension during therapeutical intervention in experimental atherosclerosis.
ATHEROSCLEROSIS is an inflammatory disease leading to plaque formation and expression of proinflammatory proteins, extracellular matrix proteins, and proteases. 1 In the recent decades, clinical imaging techniques have made substantial progress in identifying atherosclerotic disease; however, the structural information provided by computed tomography (CT), magnetic resonance imaging (MRI), optical coherence tomography (OCT), or ultrasonography does not reflect the biologic and pathophysiologic characters of plaque lesions and does not provide reliable information about the inflammation and vulnerability of plaque lesions.2–4
Therefore, imaging of plaque-associated proteins appears to be interesting, especially when their expression can be related to local inflammation or characteristics of plaque instability. 5 Recent reports suggest that this approach might be feasible for targeting thrombus formation or molecules selectively expressed during neovascularization6–9 or using substrates for protease activity.10,11 Matter and colleagues demonstrated that an antibody against ED-B fibronectin (ED-B) conjugate binds to atherosclerotic lesions in apolipoprotein E–deficient (apoE–/–) mice. 8 A recently developed conjugate with a single-chain antibody against ED-B for near-infrared fluorescence (NIRF) imaging could be successfully used for detection of atherosclerotic plaque lesions. 12 ED-B is a splice variant of fibronectin and can be expressed by fetal vascular smooth muscle cells (VSMCs) but less by adult VSMCs. 13 Balloon injury of rat arteries induced an increased expression and deposition of all fibronectins, including ED-B in the neointima and the media. 14 In atherosclerotic plaques from apoE–/– mice and humans, immunoreactivity for ED-B was closely associated with macrophage-rich lesions. 12
Ezetimibe (EZE), a potent cholesterol absorption inhibitor, has been shown to significantly lower serum cholesterol concentrations by selectively inhibiting cholesterol uptake at the brush border of the small intestine. The Niemann-Pick C1 Like 1 protein (NPC1L1) was identified as the protein essential for cholesterol absorption and target of EZE action.15,16 Treatment of apoE–/– mice with EZE significantly reduced athersclerotoic lesion formation. 17 Recently, the same authors found that deletion of the NPC1L1 gene resulted in a nearly complete prevention of lesion formation in apoE–/– mice. 18
In the present study, we investigated whether molecular imaging with an anti-ED-B NIRF conjugate is capable of detecting changes in plaque expansion occurring during pharmacologic intervention with EZE in a model of experimental atherosclerosis. Furthermore, we investigated the local expression of ED-B, the imaging target, in plaque lesions and its association with macrophage immunoreactivity and analyzed changes in plaque formation during short- and long-term therapy with EZE.
Material and Methods
Anti-ED-B NIRF Conjugates
Cloning and purification of the L19 antibody have been described. 19 For immunohistochemistry and imaging studies, a derivative of the single-chain antibody (scFv) L19 containing a C-terminal cysteine tag was generated. 12 This antibody was covalently linked to a tetrasulfonated carbocyanine-maleimide dye (absorption maximum ≈765 nm, emission maximum ≈790 nm) with cysteine-selective maleimide functionality theoretically yielding defined 1:1 conjugates for NIRF. Typically, measured dye to antibody molar ratios ranged between 0.9 and 0.7.
Animals
Animals used in this study were maintained in accordance with the Guide for the Care and Use of Laboratory Animals published by the National Institutes of Health. 20 All experiments were approved by the local animal committee. Eight-week-old apoE–/– mice (C57BL/6 background; Charles River Laboratories, Lyon, France) fed with a high-fat diet (21% fat; Altromin) mixed with and without EZE. Based on the daily food consumption, EZE was calculated at a daily dose of 5 mg/kg/d. Four-, 6-, and 8-month-old mice fed with and without EZE were subjected to an imaging protocol followed by sacrifice 24 hours after intravenous injection of NIRF antibody conjugate. Animals were injected intravenously into the tail vein with the antibody-dye conjugate dissolved in physiologic saline at a dose of 0.05 μmol/L/kg body weight. Typically, 400 μL was injected for a 40 g animal. Blood samples were drawn after sacrifice, the thoracic aorta was dissected, and the brachiocephalic artery was carefully removed, embedded in OTC, and frozen in liquid nitrogen. Aortas were prepared for Sudan staining and NIRF imaging studies.
NIRF Imaging
Aortas were carefully prepared immediately after sacrifice of the mice and maintained in phosphate buffered saline at 4°C. Images were obtained by using a cooled charge-coupled device (CCD) camera (Hamamatsu, Peltier, Model C4742-95 2ER, Herrsching, Germany) installed on a microscope (BH2, Olympus Optics) with a long–working distance objective lens (Olympus Optics). Samples were transilluminated with a near-infrared imager equipped with laser (742 nm, Model Ceralas PDT 742/1,5W, CeramOptec). Image analysis was performed using Compix Simple PCI 5.0 software, as published in detail recently. 12 NIRF-positive areas were quantified using SiS analysis software and expressed as a percentage of the total surface area.
In Vivo NIRF Imaging
In vivo imaging of animals with atherosclerotic lesions in the aorta was carried out using a specially built setup with transillumination geometry (Bayer Schering Pharma AG, Berlin, Germany). The animals were positioned between an illuminating glass fiber (OZ Optics LTD, QMMJ-55-IRVIS), which was connected to a Ti:Sa laser (Spectra Physics, Modell 3900s), and a CCD camera (Hamamatsu, C47842-98-26KAG2). Optical filters (Laser Components) were used to block the excitation light (748 nm) and to record NIRF images of the animals. Imaging was performed in anesthetized mice at different time points after intravenous administration of the antibody-dye conjugate dissolved in physiologic saline.
A significant accumulation of signal in the bladder and the liver was detected after 2 hours (data not shown). In all animals, ex vivo imaging was performed after euthanasia of the animals and preparation of the aortic arches.
Quantification of Aortic Atherosclerotic Lesions
The extent of atherosclerosis in the mouse aorta was also determined using the en face method after Sudan III staining. 21 Surface lesion areas were quantified using SiS analysis software and expressed as a percentage of the total surface area.
Immunohistochemical Analysis
Directly after sacrification, segments of the brachiocephalic arteries were obtained from every animal, were immersed in OTC compound, snap-frozen in liquid nitrogen–cooled methylbutane, and stored at −80°C. Serial aortic cross sections (5–6 μm) were cut on a cryostat and placed on tissue-section adhesive-coated slides. After air-drying, the slides were fixed in acetone at −20°C and incubated with primary antibodies overnight at 4°C. The labeled avidinbiotin method was used for immunohistochemical detection. Nonimmune rabbit, goat, or mouse immunoglobulin G was used as a negative control. The Mac3 antibody was from Santa Cruz (Heidelbeg, Germany). Slides were counterstained with hematoxylin. Color video images of stained sections were captured using the same image analysis system for quantifying the surface lesion area.
Plaque Morphology and Morphometry
All morphometric analyses were made on elastin-stained sections, and three vessel cross sections were quantified per mouse. Morphometry was performed with SiS analysis. The lengths of the internal and external elasticae were recorded. These were used to derive the media area by assuming them to be the circumferences of perfect circles. The plaque area was measured directly and was subtracted from the area enclosed by the internal elastic lamina to derive the true lumen area.
Determination of Plasma Lipid Concentration
Total cholesterol was determined enzymatically in heparinized plasma.
Statistics
Correlations between plaque areas and antibody-conjugated near-infrared fluorochrome uptake as assessed by NIRF were performed by using linear regression analysis (SPSS version 12.01, SPSS Inc., Chicago, IL). Bland-Altman plots were constructed to determine the level of agreement between optical NIRF and Sudan III measurements. 22 Comparisons of areas were made using the Mann-Whitney U test. Statistical significance was attributed to p values < .05. Values are given as mean or mean ± SEM.
Results
EZE Reduced Cholesterol Levels
A significant reduction in total cholesterol was observed in the presence of EZE treatment at the ages of 6 and 8 months. No significant difference in cholesterol was observed at the age of 4 months, which reflects 2 months of therapy with EZE. Total body weight demonstrated an age-dependent increase without significant differences between the groups (Table 1).
Serum cholesterol levels and body weights of all investigated groups. Results are expressed as mean+/2SEM.
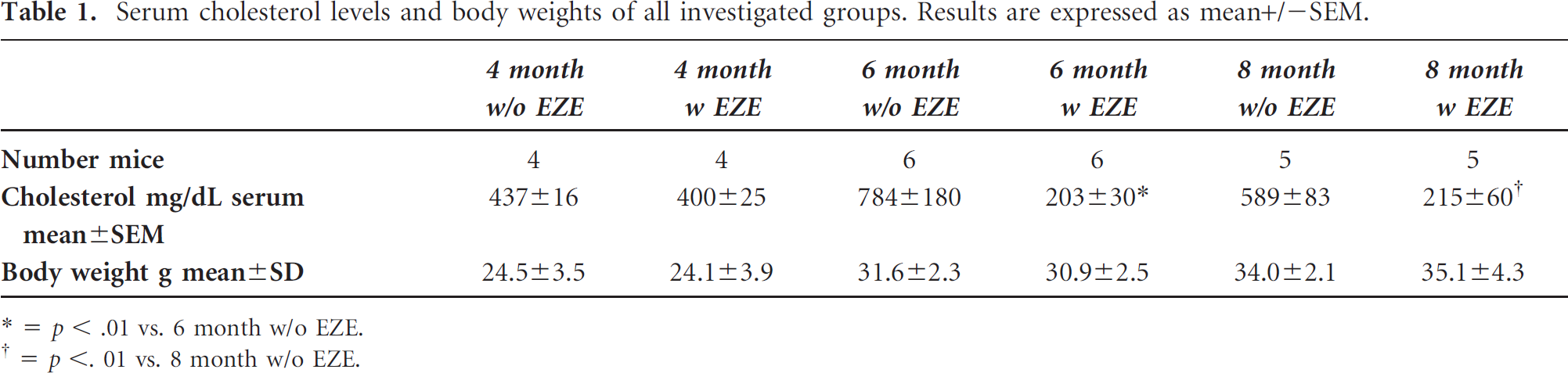
* = p < .01 vs. 6 month w/o EZE.
= p < .01 vs. 8 month w/o EZE.
Anti-ED-B NIRF Imaging Detects Aortic Plaque Areas
At the ages of 4, 6, and 8 months, thoracic aortas were analyzed for Sudan-positive areas and NIRF imaging 24 hours after intravenous injection of the ED-B NIRF conjugate. At the age of 4 months, discrete lesion formation was visualized by Sudan III stain and consecutive quantitative analysis in untreated and treated mice (Figure 1A). There was a significant increase in aortic lesion formation at 6 and 8 months compared with each time point (p < .01). At 8 months, up to 40% of the total aortic area was Sudan III positive in untreated apoE–/– mice on a high-fat diet. In contrast, only minimal Sudan III stain and NIRF signal intensity were detectable under EZE treatment at 6 or 8 months (see Figure 1A). Quantitative analysis demonstrated that EZE decreased the Sudan-stained areas and NIRF imaging at 6 and 8 months (Figure 1, B and C). The signal to noise ratios (SNRs) for NIRF imaging were not significantly different by 4 months but increased significantly (sixfold) at 6 and 8 months

Specific binding of near-infrared fluorescence (NIRF) fluorochrome conjugated with anti-ED-B fibronectin antibody to atherosclerotic lesions in apolipoprotein E–deficient (apoE–/–) mice. A, Fat staining of longitudinally opened aortas 24 hours after injection in 4-, 6-, and 8-month-old apoE–/– mice with (with EZE) or without ezetimibe (w/o EZE) from one representative mouse and NIRF images from three representative animals/group. NIRF signals were detected in small lesions. EZE treatment resulted in a significantly reduced lesion formation determined by quantitative analysis of Sudan-stained areas (B), NIRF imaging–positive areas (C), and a significant reduction of signal to noise ratio (D) in mice fed with EZE for 6 and 8 months.
(p < .05). EZE significantly reduced SNRs at 6 and 8 months (Figure 1D). Comparison of both methods for the detection of plaque areas demonstrated a significant correlation between Sudan stain and NIRF imaging and excellent correlation using Bland-Altmann analysis (Figure 2).

A, Correlation between Sudan fat staining–positive areas and near-infrared fluorescence (NIRF) signal–positive areas 24 hours after intravenous injection of the NIRF conjugate (n = 28). Corresponding areas were compared using linear regression (r = .881, p < .001). B, Blant-Altmann plot correlates of the mean of NIRF-positive areas and Sudan-stained areas of the total vessel area {expressed as a percentage of the total aortic area} and the difference between Sudan- and NIRF-positive areas. Reference lines mark the double level of significance and the mean of differences.
In vivo measurements were performed in a cohort of apoE–/– mice treated or untreated with EZE for 8 months using an imaging system with transillumination geometry. As presented in Figure 3, we observed a strong NIRF signal in the thoracic field in mice fed a high-fat diet without EZE and a high plaque load, whereas EZE treatment reduced the signal intensity, corresponding to the minimal plaque load in the aortas of these mice. The signal was observed after 8 hours and was detectable for 48 hours. The SNR at 24 hours was 278 ± 35 without EZE and 108 ± 34 in EZE-treated animals (n = 3, p < .05). A significant thoracic signal was detected only in mice with significant lesion formation (without EZE treatment), which was confirmed in the ex vivo imaging study and by Sudan staining (see Figure 3).

In vivo measurement was performed in 8-month-old apolipoprotein E–deficient mice with (with EZE) or without ezetimibe (w/o EZE). Specific binding of near-infrared fluorescence (NIRF) fluorochrome conjugated with anti-ED-B fibronectin antibody was determined 24 hours after intravenous injection into the tail vein. Mice were sacrificed after 48 hours to perform ex vivo imaging and Sudan stain of the thoracic aortas. Representative images are shown here: in vivo NIRF image (upper row) and the corresponding ex vivo NIRF image and Sudan stain of the aorta from the same animal (lower row). There was significant in vivo NIRF activity detectable in untreated mice in the thoracic field between the right foreleg (RF) and the left foreleg (LF), whereas very low activity was detected in mice with EZE treatment.
Long-Term EZE Treatment Reduced the Atherosclerotic Lesion Area
The plaque areas from brachiocephalic arteries were significantly increased after 6 and 8 months compared with 4 months, but without a difference between 6 and 8 months (Figure 4). There was no significant effect of EZE on plaque size at 4 months (7.2% to 6.9% with EZE). At 6 and 8 months, a clear reduction in plaque size (p < .001) was observed (8 months: 32.0% to 2.1%, p < .001). There was a significant reduction in plaque size in the EZE-treated mice between 4 and 8 months (from 6.9% to 2.1%, p < .05), which suggests that the EZE not only prevented plaque formation but also mediated regression of plaque lesions in the brachiocephalic artery (see Figure 4).

Mean values of plaque areas in brachiocephalic arteries in mice after 4, 6, and 8 months of being fed a high-fat diet without and with ezetimibe (EZE) demonstrated a significant reduction in plaque areas under EZE treatment. Furthermore, longer treatment with EZE resulted in a significant reduction in plaque area after 6 and 8 months compared with the 4-month group (mean ± SEM, n = 6–10).
Immunohistochemical analysis of the brachiocephalic arteries demonstrated changes in the expression of ED-B and the macrophage marker Mac3 in the atherosclerotic lesions. The ED-B-positive and Mac3-positive areas were increased at 6 and 8 months. EZE treatment reduced the immunoreactivity of ED-B- and Mac-3-positive areas significantly after 6 and 8 months of EZE treatment (Figure 5). The immunoreactivity for both ED-B and Mac3 were lowest in lesions after 8 months of treatment with EZE.

Representative micrographs of immunohistochemical staining of atherosclerotic lesions of apolipoprotein E–deficient (apoE–/–) mice at the ages of 4, 6, and 8 months after being fed a high-fat diet without and with ezetimibe (EZE) using (A) anti-ED-B fibronectin antibody staining and (B) Mac3 antibody staining for detection of macrophages. C, Quantitative analysis demonstrated a significant reduction in ED-B fibronectin–positive areas (percentage of the total plaque area) after 6 and 8 months compared with the apoE–/– control mice without EZE (p < .05). D, A similar effect was observed for Mac3-immunoreactive areas (p < .05; mean ± SEM, n = 6–10).
Discussion
ED-B is a potential target for molecular imaging. Its expression highly correlates with macrophage accumulation within the plaque.8,12 In the present study, we evaluated whether ED-B NIRF imaging can be used for monitoring pharmacologic intervention using the cholesterol absorption blocker EZE, which is efficient in reducing atherosclerotic lesion formation in this model. 17 We compared data obtained by targeted NIRF imaging of atherosclerotic aortas from apoE–/– mice with data obtained from Sudan III staining, a well-established method for quantization of plaque expansion in the mouse aorta. 21 The results obtained in this study demonstrate an excellent correlation between Sudan III stain and NIRF imaging and confirm that ED-B NIRF imaging is an excellent tool for monitoring plaque progression and prevention in this model.
We recently demonstrated that plaque macrophages and plaque ED-B expression are closely associated, although VSMCs are the major source of ED-B production. 12 In the present study, we also investigated the local expression of the ED-B and macrophages in brachiocephalic arteries. ED-B and macrophage immunoreactivity was significantly reduced under EZE therapy and was maximally increased in 6-month-old mice. Again, there was a close association between the expression of both macrophages and ED-B during the investigated time course from 4 to 8 months. Reduced immunoreactivity under pharmacologic intervention was also seen for both ED-B and macrophages. The present finding demonstrates that ED-B NIRF imaging represents a reliable tool for the monitoring of interventional therapy in experimental atherosclerosis.
EZE nearly completely reduced plaque formation and NIRF imaging in 6- and 8-month-old apoE–/– mice, whereas aortic lesion formation was constantly increasing in untreated mice from months 4 to 8. This effect is in accordance with previous studies in EZE-treated apoE–/– mice 17 and recently in NPC1L1-deficient apoE–/– mice, which lack the target protein inhibited by EZE. 18 In contrast to the previous reports by Davis and colleagues, which presented only end point data after 6 months of therapy,17,18 we also monitored plaque formation under EZE at different time points. The effect of EZE was not detectable at 4 months, which corresponds to 2 months of therapy. Clearly detectable effects were seen at 6 and 8 months on aortic plaque expansion determined by Sudan III and on plaque lesion size in brachiocephalic arteries. The content of macrophages and ED-B was also significantly reduced in lesions from these arteries at these time points under EZE therapy. Monitoring the effect of EZE, we observed a significant reduction in brachiocephalic lesion size after 8 months compared with 4-month-old apoE–/– treated mice, which was accompanied by a reduction in Mac3 immunoreactivity. This suggests that EZE not only prevents but also regresses plaque lesion formation, which probably had already started in young mice.
ED-B NIRF imaging in this study was not limited to the ex vivo studies, although the low penetration depth of the laser system used is still a significant problem. In vivo data obtained from a small cohort of apoE–/– mice demonstrated a significant NIRF signal in the thoracic field that corresponded to the aortic arch. This signal was significantly reduced when animals received EZE. So we have promising data from these preliminary in vivo studies that a target-specific accumulation of ED-B-targeted NIRF dye can be visualized in living mice. This observation still has to be validated in a larger group of animals.
An attractive alternative to the method used here is OCT, which partially overcomes this limitation, resulting in improved resolution, penetration depths up to 10 cm, and better quantification performance, 23 as has been demonstrated by studies using activatable probes detecting matrix metalloproteinase (MMP) activity.10,11 Still, combination with another imaging modality such as MRI or CT is needed for exact anatomic colocalization of optical signals. MRI, which already provides high accessibility, robust imaging quality, and high spatial resolution in clinical settings, is an especially promising technique.6,7 Another alternative could be positron emission tomography (PET)-CT. Matter demonstrated that PET-CT using 18F-fluorocholine detects plaque lesions in apoE–/– and correlated with local macrophage accumulation. 24 Recently, a more specific substrate, high-resolution imaging of diseased carotid arteries, was used to investigate and analyze plaque content to identify lesions at risk.25–27
Different probes to target atherosclerotic lesions have been developed, including high-density lipoprotein-like nanoparticles, 28 protein ligands for adhesion molecules, 9 antibodies against macrophage scavenger receptors 29 or αvβ3 integrin, 7 and enzyme substrates for cathepsins or MMPs.10,11 However, most imaging experiences have been gained with nonspecific magnetic nanoparticles, which accumulate in vascular macrophages.30–32 Most of the ligand-based probes target vascular macrophages or lipid depots. Owing to the different kinds of models and imaging modalities, a comparison between different probes is difficult. The important goal of atherosclerosis imaging is to detect the disease early and provide information about inflammatory activity, which is probably the most important surrogate for plaque vulnerability. 5 ED-B is solely expressed in tumors and atherosclerotic lesions. It is associated with local macrophage accumulation in vascular plaque lesions. 12 So the antibody is an indicator for inflammation but also an indicator for vascular disease. In animal models, ED-B expression has already been observed in low-grade lesions, indicating early onset of ED-B expression at an early stage of atherosclerotic disease.
The developed anti-ED-B antibody also has a very high affinity and specificity to ED-B in solid tumors. 19 Consequently, a derivative of this single-chain antibody (used as a backbone of a fusionprotein) 33 has been developed for targeted antitumor therapy in patients with solid tumors using immunocytokines as effector molecules, which is currently in clinical evaluation.
At the moment, we do not know how much such surrogates for local inflammation will help to achieve an estimate of the biologic activity of plaque lesions. In the present study, we can demonstrate that molecular imaging targeting ED-B provides information about increasing inflammatory plaque formation and about the effect of pharmacologic intervention reducing plaque macrophage content. A combination of noninvasive imaging techniques with targeted imaging probes such as anti-ED-B might provide insights into the biologic activity of disease and enable us to monitor the effects of (successful) therapeutic interventions.
In summary, EZE-induced prevention of plaque formation can be successfully monitored using a novel NIRF imaging probe targeting ED-B. In addition, the changes in plaque formation at different time points of EZE therapy indicate that EZE not only prevents but also regresses already existing plaque lesions.
Footnotes
Acknowledgments
We thank Stefan Wisniewski and Astrid Knop for their excellent technical contributions.
