Abstract
Protein kinases play pivotal roles in almost all cellular signaling pathways, and modulation of their activity is desirable in both disease and studies of function. Information on the activity of kinases in vivo is scarce owing to a lack of appropriate methods. To obtain such information, we produced mice in which protein kinase A (PKA) activity can be monitored noninvasively in vivo. The model uses luciferase, which has been mutated to contain a target sequence of PKA, thus making luminescence from the enzyme dependent on its state of phosphorylation. The PKA-sensitive luciferase, termed luciferasePKA, was incorporated into the mouse genome, and transgenic animals exhibited a rapid β-adrenergic response, that is, reduced luminescence, in various organs, including the pancreas, muscle, liver, and fat, after isoproterenol injection. This study shows that luciferase can be used for in vivo measurements of kinase activity, suggesting that different kinase target sequences in luciferase can monitor kinase activity modulation.
PROTEIN KINASE A (PKA) mediates signals in response to the major second-messenger cyclic adenosine monophosphate (cAMP), conveying signals from a number of cell surface receptors, such as G protein-linked membrane-spanning receptors. PKA and cAMP mediate physiologic signals in neurotransmitter signaling and play an essential role in regulation of metabolic processes in tissues. Particularly, shifts in energy metabolism and stress signals are mediated by stimulation of adenylate cyclase activity followed by increased intracellular concentrations of cAMP. PKA consists of four subunits, two regulatory (R) and two catalytic (C) subunits, which are expressed in all mammalian tissues tested. There are several splice variants of both R and C subunits with significant redundancy. C subunits require cAMP-dependent release from the R subunits to become active kinases phosphorylating a large number of substrates. Overexpression, knockout, and knockdown studies have to some extent shown the importance of PKA in different tissues in vivo.1,2 However, little information is available on the dynamic changes in PKA activity in vivo under different normal and disease conditions owing to a lack of appropriate methods. In vivo measurements of kinase activity can thus possibly be monitored by changes in an externally detectable substrate activity containing the target sequence. One substrate that is detectable in live cells is luciferase.3–5 We now present evidence that PKA-sensitive luciferase (luciferasePKA) driven by the constitutive cytomegalovirus (CMV) promoter is expressed in many tissues and organs in transgenic mice. Posttranslational phosphorylation of an engineered serine in position 220 in luciferase containing a target sequence for PKA reduces the ability of the enzyme to use luciferin to yield photons by oxidation, as described by Sala-Newby and Campbell. 6 Thus, direct stimulation of PKA activity reduces luminescence, and inhibition of basal or stimulated PKA activity increases luminescence (unpublished observation, 2005). 7 The present study demonstrates that luciferasePKA can be used for in vivo, noninvasive, real-time monitoring of PKA activity in mice by bioluminescence imaging.
Materials and Methods
LuciferasePKA Expressed from CMV Promoter (pCMVβ-LuciferasePKA)
pGL3-luciferasePKA was constructed by polymerase chain reaction (PCR) amplification of pGL3-control (Promega, Madison, WI) with forward primer GGGGCATGCGAGAATCTCCTGCAGGCAGTTCTATG and reverse primer CGAAACAAAACAAACTA to introduce the PKA target sequence. The PCR product was then digested with SphI and BglII and ligated into pTAL-luc (Clontech, Palo Alto, CA) digested with the same enzymes. The resulting plasmid was digested with NcoI and XbaI and ligated into pGL3-control digested with the same enzymes to produce pGL3-luciferasePKA. pGL3-luciferasePKA and pCDNA3 (Invitrogen, Carlsbad, CA) were digested with HindIII and XbaI. The 1,655 bp fragment from pGL3-luciferasePKA was ligated into pCDNA3 using T4 deoxyribonucleic acid (DNA) ligase (Promega) to obtain pCDNA3-luciferasePKA. In vivo expression of foreign genes is enhanced when the transgene contains an intron between the promoter and the exon. Therefore, pGL3-luciferasePKA was digested with XbaI and HindIII, and the resulting fragment was ligated into the NotI site of pCMVβ (Clontech, GenBank U02451) after treatment with T4 DNA polymerase for blunt end ligation, to obtain pCMVβ-luciferasePKA.
Transgenic Animals
Transgenic mouse lines were created by pronuclear injection of purified pCMVβ- luciferasePKA DNA into fertilized eggs (B6CBA F2), according to standard protocols. 8 Animal care was in accordance with national legislation and institutional guidelines. All animal experiments were performed with the F1 generation produced by breeding of transgenic founders with nontransgenic C57 BL/6S.
Luminometry and Protein Measurements
Excised organs were homogenized for 15 to 20 seconds in Reporter Lysis buffer (Promega) with Ultra Turax homogenizer before undissolved material was removed by centrifugation at 12,000g for 15 minutes at 4°C. Protein concentration in supernatants was determined by Bio-Rad Protein Assay (Bio-Rad Laboratories, Hercules, CA) and used for normalization of luminescence measurements. Luminescence was measured as recommended by the manufacturer of the Luciferase Assay System (Promega) with a TD 20/20 luminometer (Turner Design, Sunnyvale, CA).
In Vivo Imaging and Luminescence Measurements
Mice were anesthetized with isoflurane, injected with ≈130 mg/kg luciferin (Biosynth, Basel, Switzerland) intraperitoneally, and subjected to imaging using the IVIS 100 Imaging System from Xenogen (Alameda, CA), before or after injection of isoproterenol (Rikshospitalets Apotek, Oslo, Norway). Live animals were imaged intact or with their internal organs exposed. Luminescence was integrated over a period of 2 seconds to 1 minute depending on the intensity of the signal after an initial period of 7 minutes, during which luciferin entered systemic circulation. Images obtained were analyzed by counting of photons in a specific area including most of the abdominal region (when not indicated otherwise) after removal of the fur by shaving. Photon counting was performed using the software Living Image version 2.20 (Xenogen).
Results
Expression of LuciferasePKA In Vivo
To prove the concept of noninvasive in vivo protein kinase measurements, we first made a construct with luciferase containing the PKA target sequence R217R218F219S220 (Figure 1, A and B), driven by a CMV-derived promoter. Pronuclear injection of this construct gave rise to six founders, which displayed bioluminescence from various regions on the abdominal side of the animal, suggesting different expression of the construct and/or differences in basal PKA activity in various organs (Figure 1C). The transgene did not produce any obvious changes in the behavior or general health of the animals. Disregarding the paws and nose region, most positive animals displayed the highest reporter activity in the abdominal region, but there were considerable differences in the luminescence pattern in this region. However, there seems to be a recurrent pattern, in that all six animals have one or two bright spots in the abdominal region. The difference in luminescence among founders may be explained by insertion of the transgene in the genome at loci with different chromatin structure and transcriptional activity. Two animals have strong luminescence also from the thoracic region (PK11 and PK12). There was apparently no correlation between luminescence patterns and sex (data not shown). The CMV promoter is regarded as a strong constitutive promoter and yields high expression in many cells transfected with CMV-containing expression vectors. 10 We have also found that the CMV promoter drives expression of luciferasePKA in several cell lines, including transfected primary mouse cells (unpublished observation, 2005).

Schematic illustration and bioluminescence from protein kinase A-sensitive luciferase (luciferasePKA). (A) Elements of the construct and (B) position of mutations in model of luciferase structure 9 used for producing transgenic animals are illustrated. C, Offspring from six different mice made transgenic with luciferasePKA were anesthetized with isoflurane, and luciferin was injected intraperitoneally. Animals were then imaged after 7 minutes with an IVIS 100 Imaging System, lying on their dorsal side. Two exposures were made, one regular photograph and one in complete darkness. The two images were then superimposed on each other; luminescence is represented by pseudocoloring. The correlation between photon counts and color is illustrated by the color bar. D, PK8 offspring (littermates, n = 5) were sacrificed by cervical dislocation before indicated tissues, and organs were dissected out within 10 minutes. Tissues were homogenized, and samples were analyzed by luminometry; data are presented as an average of luminescence units, ± SD, on a log scale y-axis. ATP = adenosine triphosphate; CMV = cytomegalovirus; PK = protein kinase; WT = wild type.
The differences in bioluminescence from various organs were substantiated by bioluminescence measurement in tissue homogenates from offspring from one of the founders, PK8 (Figure 1D). The largest difference was between liver and muscle (approximately 200-fold). The individual response varied significantly, particularly in the liver and in the stomach. A possible explanation is that individual mice were in metabolically different status regarding stress and/or feeding. Glycogen metabolism is closely associated with food intake and stress, factors that may induce hormonally regulated glycogen degradation. Glycogen degradation is signaled through receptors linked to adenylate cyclase, cAMP synthesis, and PKA activation, which, in turn, modulate the activity of glycogen phosphorylase. It is conceivable that differences in animal handling prior to PKA assessment may account for some of the observed differences.
In Vivo Imaging of PKA Activity in Response to β-Adrenergic Stimulation
We then tested whether the animals and tissues responded to stimuli mediated by changes in PKA activity, such as the β-adrenergic agonist isoproterenol. When isoproterenol was injected intravenously, bioluminescence from the abdominal region was reduced to 20 to 60% of the control (before injection) in four of six different founders within 5 minutes (Figure 2A). Thereafter, there was little change in luminescence up to 30 minutes after isoproterenol injection. Luminescence during this period is most likely a product of ongoing luciferase phosphorylation/dephosphorylation, luciferase translation, and a gradual decrease in available luciferin. Two founders responded poorly (PK11 and PK17), possibly owing to expression of the luciferase in β-adrenergic nonresponsive tissue. The large variation in luminescence from animals injected with phosphate-buffered saline may reflect responses to handling, metabolic shifts during anesthesia, or other physiologic processes in which PKA is involved. Imaging of abdominal regions at time points between 0 and 6 minutes after isoproterenol injection shows that a rapid initial response (1 minutes) is partly reversed over the next 5 minutes compared with the control, suggesting that phosphorylation of luciferasePKA is reversible (Figure 2B). This reversal may be mediated by phosphatase P1 (PP1) and reflect a physiologically relevant response to a strong β-adrenergic stimulus in several organs, although translation of unphosphorylated and enzymatically active luciferase cannot be ruled out. β-Adrenergic responses are particularly interesting in brain and muscles as PKA mediate aminotropic and metabolic responses in the two tissues, respectively.11,12 We therefore performed in vivo imaging of these tissues in live animals after injection of isoproterenol and found a substantial decrease in bioluminescence (ie, increased PKA activity; Figure 2, C and D). Interestingly, bioluminescence observed from the outside of the brain was not evenly distributed; a bright spot was observed at the position where fissure longitudinalis cerebri ends against the cerebellum. This could possibly reflect variability in PKA activity in different brain structures, although differences in expression cannot be ruled out and need further testing. PKA plays a central role in several brain functions, 13 including long-term potentiation and memory. 14 Our results demonstrating in vivo measurements of brain PKA activity suggest that this model may be very useful for studies of PKA in the brain as a drug target.

In vivo imaging of β-adrenergic modulation of protein kinase A-sensitive luciferase (luciferasePKA) activity. A, Offspring from six transgenic founders (▴ = PK1, ▪ = PK11, • = PK2, × = PK12, + = PK17, – = PK8) were anesthetized and injected with luciferin before imaging and photon counting with the Living Image version 2.20 software after 7 minutes. Then isoproterenol (60 μg) was injected intravenously, and the animals were imaged and photons counted in the entire abdominal area after the indicated time periods. Luminescence is presented as a percentage of control (before isoproterenol) in a typical experiment. B, Offspring from PK2 and PK8 were injected with luciferin and imaged after 7 minutes. Then isoproterenol (60 μg, PK2 [▪], n = 3, and PK8 [▴], n = 3) or phosphate-buffered saline (300 μL, PK8 [Δ], n = 4) was injected, and animals were imaged at the indicated time points. Luminescence is expressed as a mean percentage of control (before isoproterenol injection) ± SD. C, Offspring from PK8 were anesthetized and had the fur over the skull removed before injection of luciferin intraperitoneally. The dorsal side of the head (the shaved area) was then imaged after 7 minutes before injection of 60 μg of isoproterenol intravenously and subsequent imaging after the indicated time points. Data are presented as percent luminescence of control (before isoproterenol injection), average of three animals, ± SD. The lower panel shows images from a typical experiment. D, PK8 offspring were anesthetized with isoflurane, and luciferin was injected intraperitoneally. After 7 minutes, the hind limb muscles (gastrocnemius with fur removed) were imaged (basal activity) before 10 μg of isoproterenol was administered intramuscularly (gastrocnemius) at time 0. The animals were then imaged 90 or 210 seconds after isoproterenol injection. Luminescence from the shaved area of the muscles was quantitated and presented as mean percent luminescence (compared with basal activity) in both hind limbs, n = 4, ± SD. The lower panel shows images from a typical experiment.
Inspection of whole-body images revealed several bright spots in the abdominal region that displayed reduced bioluminescence after isoproterenol injections, whereas skin in the extremities and nose region responded less clearly to the β-adrenergic stimulus. To quantify this variation and identify some of these tissues and organs, we injected luciferin into PK8 mice before tissues and organs were exposed while the animal was still alive. Isoproterenol was then injected intravenously, and images were taken 150 seconds after injection (Figure 3A). Photons from selected identifiable organs and tissues were counted and related to luminescence before isoproterenol injection (Figure 3B). The liver responded to the largest extent, and luminescence was reduced to 40% of control, whereas fat responded poorly (75% of control).
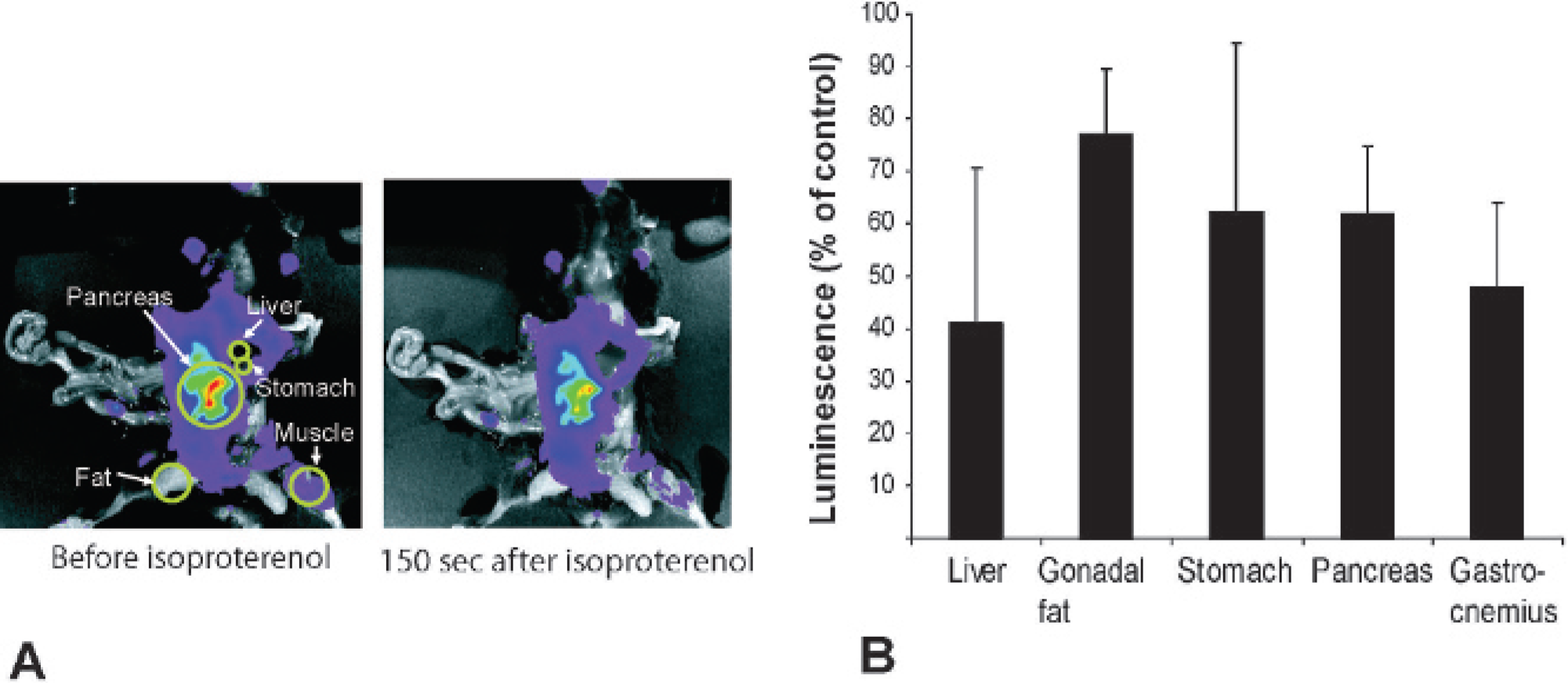
In vivo imaging of protein kinase A-sensitive luciferase (luciferasePKA) in exposed tissues in response to isoproterenol. A, PK8 offspring were anesthetized with isoflurane and luciferin was injected intraperitoneally. After 7 minutes, the abdomen was opened and several organs and tissues were exposed; the animals were imaged (basal activity) before 60 μg of isoproterenol was administered intravenously at time 0. The animal was then imaged 150 seconds after isoproterenol injection (right panel). B, For quantification of response, PK8 offspring were treated as above, and luminescence in the regions marked with circles in the left panel in A corresponding to the indicated tissues was quantitated and presented as average percent luminescence of control (before isoproterenol), n = 5, ± SD.
Discussion
PKA plays a pivotal role in many physiologic processes and is modulated by many external and internal stimuli. It should be kept in mind that the isoproterenol response observed here is done at a physiologic state influenced by isoflurane anesthesia, which was chosen for practical reasons, allowing rapid awakening after imaging. Another anesthetic, halothane, has been shown to inhibit β-adrenergic stimulation of lipolysis in adipocytes 15 ; thus, the effects of anesthetic drugs must be considered when these results are interpreted. Signaling by neurotransmitters such as dopamine and acetylcholine is mediated in part by PKA. Acetylcholine reduces cAMP levels, whereas dopamine, serotonin, norepinephrine, and adenosine may reduce or increase cAMP levels depending on the subtype of neurotransmitter receptors involved. 16 Anesthetics working through γ aminobutyric acidB receptors, which are heterodimeric G protein-coupled seven-membrane-spanning receptors, may reduce PKA activity. 17 Isoflurane also has been shown to increase PKA-mediated protein phosphorylation in platelets, 18 and isoflurane attenuates isoproterenol-mediated vasodilatation of rat arteries. 19 It has also been reported that isoflurane and ketamine influence β-adrenergic responses in the heart differently. 20 Ketamine antagonizes glutamate receptors and has been shown to inhibit PKA 21 ; consistent with this, in the present study, there was a two- to threefold increase in luminescence from the abdomen (data not shown). On the other hand, ketamine has been shown to induce twofold increases in cAMP in the brain. 22 For the above reasons, isoflurane was used throughout this study.
Isoproterenol is known to activate KATP channels, 23 and this activation may influence intracellular Mg++ concentrations, of which luciferase is dependent. The net effect of this is, however, difficult to predict. In kidney cells 24 and platelets, 25 isoproterenol has been shown to promote uptake of Mg++, whereas in other cell types, activation of KATP channels reduces intracellular Mg++. It can thus not be ruled out that the effect of isoproterenol in our system is partly influenced by the availability of intracellular Mg++. If so, it may still be an effect of PKA stimulation, although indirectly via activation of KATP channels.
Luciferase-positive tissues, such as the pancreas, muscle, liver, stomach, and fat, responded to isoproterenol by a reduction in luminescence, as expected. Interestingly, the organ with the highest luciferasePKA activity was the pancreas. This can be interpreted as low PKA activity or constitutively high expression of the reporter in this organ compared with other tissues or a combination of the two. A high pancreatic expression of the reporter is corroborated by the work of Collaco and Guesz, who also found high CMV-driven wild-type luciferase activity in the pancreas. 26 The high signal in the pancreas makes our model useful for studies on PKA responses related to the neuroendocrine responses in the pancreas and food intake and the action of insulin, glucose, and glycogen on pancreas cells.
Expression of PKA-sensitive luciferase in our system is driven by the strong CMV promoter. The CMV promoter has four cAMP responsive elements (CRE) recognition sites. CRE sites are bound by CRE-binding protein (CREB), and CREB is activated by PKA27,28; thus, high PKA activity could yield more efficient transcription of luciferasePKA and subsequent strong luminescence. On these grounds, one could speculate that the luminescence seen in various tissues in the animals could be lower or higher than it actually is owing to high or low PKA-mediated transcription of the reporter, depending on the intracellular cAMP concentration. In long-term experiments with PKA enhancers or inhibitors, this could give rise to an underestimation of the effects of PKA modulation. It should be noted, however, that, to our knowledge, PKA-modulated transcription from the CMV promoter in vivo has yet to be demonstrated.
Fluorescence resonance energy transfer (FRET) was used by Zhang and colleagues to monitor PKA activity in cells.29,30 This system relies on phosphorylation of a linker sequence between yellow and cyan fluorescent protein to bring them sufficiently close to achieve energy transfer. This system could possibly also be used in vivo; however, discrimination between a PKA-specific FRET signal and a strong autofluorescence background is technically challenging. The advantage over luminescence would be that this probe gives better resolution.
To our knowledge, transgenic luciferasePKA mice are the first model in which intracellular protein kinase activity can be measured in intact living animals and facilitates studies of PKA-mediated hormone actions. The model is a proof of concept for using luciferase as a reporter for kinase activity in vivo and opens up the possibility of engineering other target sequences in luciferase for screening of kinase modulators in drug development under highly relevant physiologic conditions.
