Abstract
Recruitment of CD4+ T cells into islets is a critical component of islet inflammation (insulitis) leading to type 1 diabetes; therefore, determining if conditions used to treat diabetes change their trafficking patterns is relevant to the outcome. Cotransfer of CD4+BDC2.5 (BDC) cells with non-CD4 splenocytes obtained from newly diabetic NOD mice, but not when they are transferred alone, induces accelerated diabetes. It is unclear whether these splenocytes affect diabetes development by altering the systemic and/or local trafficking and proliferation patterns of BDC cells in target and nontarget tissues. To address these questions, we developed an animal model to visualize BDC cell trafficking and proliferation using whole-body in vivo bioluminescence imaging and used the images to direct tissue sampling for further analyses of the cell distribution within tissues. The whole-body, or macroscopic, trafficking patterns were not dramatically altered in both groups of recipient mice. However, the local patterns of cell distribution were distinct, which led to invasive insulitis only in cotransferred mice with an increased number of islet-infiltrating CD11b+ and CD11c+ cells. Taken together, the non-CD4 splenocytes act locally by promoting invasive insulitis without altering the systemic trafficking patterns or proliferation of BDC cells and thus contributing to diabetes by altering the localization within the tissue.
Several noninvasive imaging techniques have been employed to image lymphocytes.8–11 For a longitudinal study of disease caused by chronic inflammation such as insulitis leading to type 1 diabetes, we have used in vivo bioluminescence imaging (BLI) in real time to noninvasively visualize autoantigen-specific diabetogenic T-cell trafficking. These studies were performed prior to and after the development of insulitis and overt diabetes. This approach offers the advantages of excellent temporal analyses, cell-labeling versatility, easily accessible instrumentation, and high sensitivity of signal detection.8,10,12–14 Use of luciferase (Luc) as an in vivo reporter provides excellent signal to noise ratios owing to a near absence of background luminescence from mammalian tissues. The photons emitted from Luc-expressing cells that are transmitted through tissues can be detected using charge-coupled device (CCD) cameras designed for low-light imaging. BLI allows for long-term longitudinal studies of cell-trafficking patterns because the reporter is encoded in the genome of the cells and the signal is, therefore, not diluted owing to cell division.15,16
The CD4+ BDC cells are islet antigen–specific and highly diabetogenic T cells.17,18 Activated BDC cells from BDC2.5 TCR transgenic NOD mice (BDC mice) induce an aggressive form of diabetes.19,20 To label T cells from these animals for trafficking studies, we crossed the BDC mice with a transgenic reporter mouse line 13 expressing luciferase from a strong, ubiquitous, constitutive promoter (luc-BDC mice). This synthetic hybrid promoter is composed of the chicken β-actin promoter and the immediate early enhancer from cytomegalovirus and drives expression of Luc in essentially all cells of the mice. 13 This results in stable expression of Luc in BDC cells, which allows for long-term longitudinal in vivo imaging studies of luc+BDC cells' trafficking to the pancreas and the islets, as well as other lymphoid and nonlymphoid tissues relative to disease state in animals.
Using an adoptive transfer model, we have imaged luc+CD4+BDC cells in real time and examined their trafficking patterns in NOD/scid recipients. It is known that transferring CD4+BDC cells alone into NOD/scid mice failed to induce diabetes; cotransfer of CD4 cell–depleted splenocytes isolated from diabetic NOD mice with CD4+BDC cells induced accelerated diabetes in recipient mice and that there can be a respectful peri-insulitis or invasive insulitis.20–22 In this study, we determined if the effect of the non-CD4 splenocytes is due to an altered systemic trafficking pattern of BDC cells or their effect on BDC cells occurs locally at tissue levels. We imaged BDC cells in vivo using BLI and compared their trafficking patterns in nondiabetic mice that received BDC cells alone and in prediabetic and newly diabetic recipients cotransferred with BDC cells plus non-CD4 splenocytes isolated from diabetic NOD mice. We used these data to guide us to the appropriate time and target tissues for further analyses using methods that would enable us to assess the effects of CD4 cell–depleted splenocytes on the local patterns of BDC cells at the tissue and cellular level. Our results showed that, at the times suggested by the BLI data, the non-CD4 splenocytes helped BDC cells induce diabetes by promoting invasive infiltration of BDC cells into the islets. The results also showed that there were increased CD11b+ and CD11C+ cells detected in the islet-infiltrating cells isolated from cotransferred mice. However, the systemic trafficking patterns of BDC cells in recipient mice and ex vivo signals in the pancreas were not altered by the cotransferred splenocytes. These data are expected to help direct the development of novel therapies by revealing that T-cell trafficking could be modulated locally within the pancreas and indicate that it may not be necessary to prevent diabetogenic T cells from reaching the pancreas.
Materials and Methods
Mice
The luc transgenic mice have been previously described. 13 The BDC 2.5 TCR transgenic NOD (BDC) mice 21 were obtained from Drs. Mathis and Benoist (Joslin Diabetes Center/Harvard Medical School, Boston, MA). Purified CD4+ T cells isolated from the mice in our BDC colony consistently do not transfer diabetes to NOD/scid recipient mice. All animals were kept in a specific pathogen-free animal facility. The F1 mice of luc transgenic mice and BDC mice crosses were backcrossed with BDC mice to generate luc-BDC mice. The luc-BDC mice (6–8 weeks old) obtained from at least N7 generation were used for the studies. These mice, like the BDC mice in our colony, do not develop diabetes by the age of 20 weeks.
Preparation of T Cells for Adoptive Transfer
Splenic CD4+ T cells were isolated from luc-BDC mice by negative selection using magnetic beads (Miltenyi Biotec, Auburn, CA). The purity of CD4+ T cells was confirmed by staining with anti-CD4 and the tetAg7/p79 tetramer that stains BDC cells.7,23 For cotransfer, CD4+ T cell–depleted splenocytes were isolated from newly diabetic NOD mice and were injected, intravenously, into 8- to 9-week-old female NOD/scid mice.
BLI of BDC Cells' Trafficking
We have determined the sensitivity of BLI for the detection of CD4+luc+BDC cells on isolated cells both in vitro and in vivo. A minimum of 3 3 105 isolated cells were required for in vitro detection using BLI at the same condition as that used for in vivo BLI, and the total flux (photon/s) signal emitted from the cells was proportional to the number of cells (data not shown). Transfer of at least 1 × 106 of CD4+luc+BDC cells to recipient mice was required for in vivo detection of bioluminescent signals (data not shown), and more optimal signals were obtained after transferring 5 to 10 × 106 cells to recipient animals. Therefore, in all BLI studies, we have transferred at least 7 × 106 cells per mouse for visualizing the trafficking of BDC cells.
Single-Cell Preparation from Various Tissues
Single-cell suspensions were prepared from dissected tissues in phosphate-buffered saline for fluorescence-activated cell sorter (FACS) analysis. The cells from the lung or the liver were further purified using Histopaque (Sigma, St. Louis, MO). The islet-infiltrating cells were prepared as previously described 23 and used for FACS analyses. Briefly, pancreatic lymph nodes were removed, and then the pancreas was distended with collagenase P to isolate islets using Histopaque gradient.
FACS Analyses
Stained cells were analyzed using FACSCalibure (Becton Dickinson, San Jose, CA), and the data were analyzed using FlowJo (Tree Star Software, San Carlos, CA).
Monitoring of Diabetes
Diabetes development was monitored by testing for urinary and blood glucose levels. Mice with glycosuria ⩾ 2% and blood glucose ⩾ 13.9 mmol (250 mg/dL) were considered diabetic.
Statistical Analysis
The two-tailed and two-sample unequal variance Student t-tests were used for data analysis, and p < .05 is considered significantly different.
Results
Noninvasive Real-Time In Vivo Bioluminescence Imaging of BDC Cell-Trafficking Patterns
We isolated CD4+luc+BDC cells from luc-BDC mice (Figure 1A) and adoptively transferred them into NOD/ scid mice for BLI analyses. Using this approach, we visualized just the BDC cells that express luciferase. To perform longitudinal studies on the trafficking behavior of CD4+BDC cells in recipient mice, the same living recipient mice were imaged at selected times over a period of 16 weeks following cell transfer (Figure 1B). Initial signals from the transferred BDC cells were apparent in the lung on the first day after transfer and then signals were detected in the pancreas and the liver by day 3. We detected the signals from luc+BDC cells in spleen and lymph nodes within 1 week after the cell transfer. These results demonstrated that the trafficking of BDC cells to the pancreas and other tissues in recipient mice can be visualized using BLI.
To quantify the bioluminescent signals detected from various tissues, the total flux (photons/s) was determined by drawing ROI and determining total photon output per ROI. ROI were defined visually on specific areas corresponding to selected organs of the body of recipient animals and control mice. The rates of increases in the in vivo bioluminescence signals detected in the ROI corresponding to the pancreas, spleen, and inguinal lymph nodes were closely correlated with each other (Figure 1C). These results further showed that the total flux from these tissues increased over time and reached a plateau, on day 56, that persisted in the pancreas and inguinal lymph nodes until the end of the study (day 113; see Figure 1C). The signals appeared to decline in the spleen after day 56. These results suggested that BDC cells eventually accumulated and proliferated in these tissues at the later time points, perhaps owing to homeostatic expansion. However, despite the fact that an increasingly larger number of BDC cells were present in the pancreas of recipient mice, none of these mice developed diabetes even at 20 weeks after the cell transfer (data not shown).
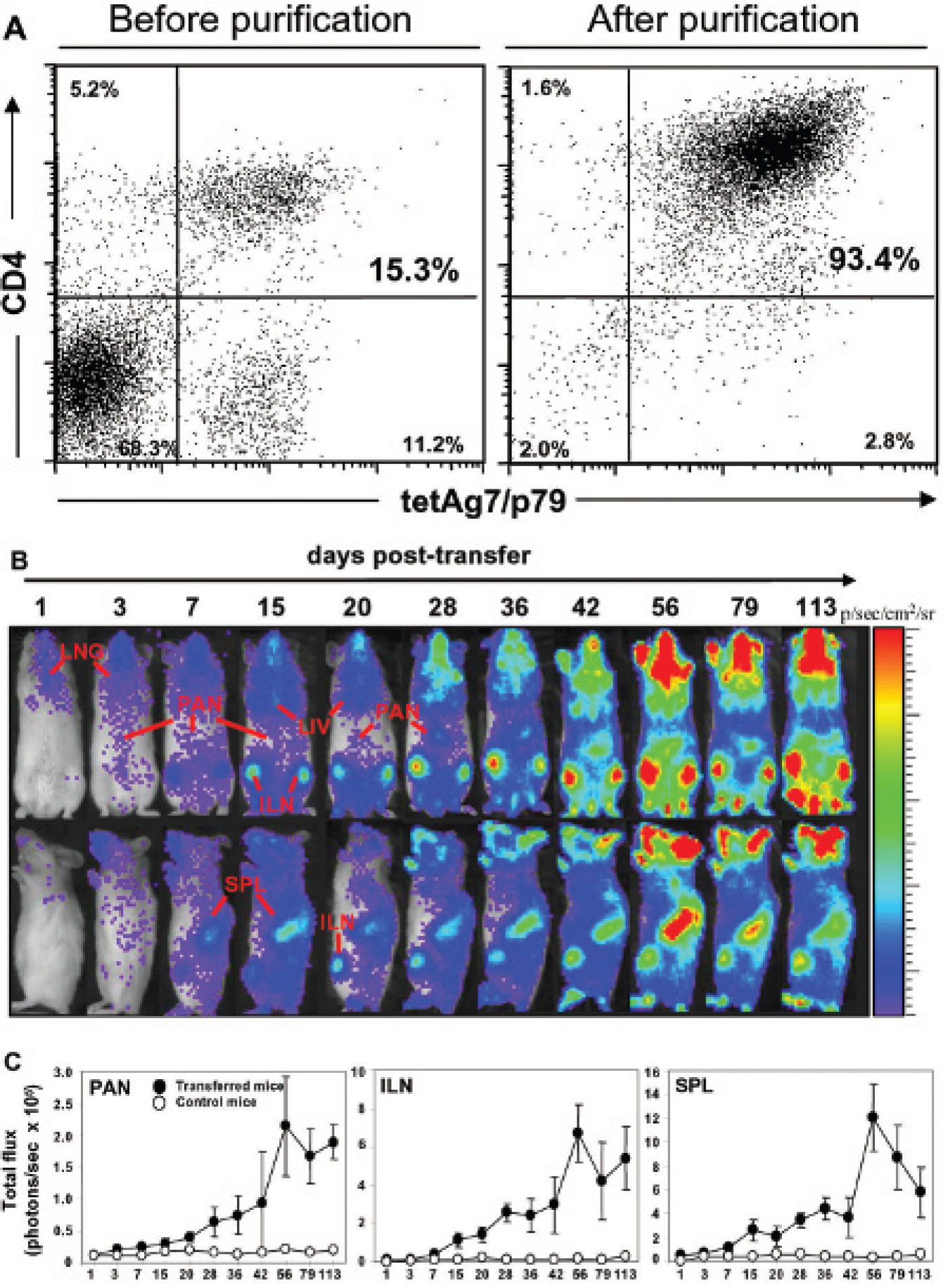
Real-time in vivo bioluminescence imaging of CD4+ BDC cells. A, Flow cytometric analyses of CD4+ BDC cells isolated from splenocytes of luc-BDC mice. Noncultured CD4+ T cells were isolated from the spleen of luc-BDC mice (N7) using magnetic beads by negative selection. The cells were stained with an anti-CD4 antibody and tetAg7/p79 tetramer before and after purification. The purity of the isolated CD4+, tetAg7/p79+ T cells is usually ≈92 to 95%. Our previous studies have shown that the tetAg7/p79 tetramer effectively stains BDC cells.7,23 B, Isolated naive CD4+ luc+BDC cells (7 × 106/mouse) were injected (intravenously) into 8- to 9-week-old female NOD/scid mice. Bioluminescence images of the transferred BDC cells were taken with a 2-minute acquisition time using a charge-coupled device camera at 15 minutes after intraperitoneal injection of
These noninvasive in vivo imaging studies revealed that although BDC cells migrate to the pancreas rapidly after cell transfer and the signals of BDC cells present in the pancreas increased significantly with time, the recipient mice did not develop diabetes. Therefore, these results revealed in real time the rapid appearance and the presence of a large number of BDC cells in the pancreas without development of diabetes, and this is consistent with the idea that islet-antigen-specific diabetogenic T cells traffic to the pancreas regardless of whether they induce diabetes.
Ex Vivo Imaging Confirms the Presence of BDC Cells in Different Tissues
To confirm the location and intensity of the in vivo signals, we performed luciferase assays on excised tissues as a means of confirming the presence and numbers of BDC cells in various tissues of recipient mice. The ability to detect optical signals from inside living animals is determined by absorption and scattering of the signals; thus, surface intensity is influenced by the depth of the source in the body and is surface weighted. 24 Therefore, evaluation of signals in excised tissues should help locate tissue sites that harbor fewer BDC cells than those identified in Figure 1 but where the bioluminescent signal could not be detected by in vivo imaging. These studies confirmed the presence of BDC cells in the pancreas, spleen, inguinal lymph nodes, and lung (Figure 2A). In addition, bioluminescent signals were detected in excised pancreatic lymph nodes and the pancreas, which are anatomically in close proximity and not readily distinguished in intact mice but easily detected in excised tissue (see Figure 2A). We were also able to demonstrate the presence of BDC cells in mesenteric lymph nodes and not in the surrounding gut tissues. In comparison, no bioluminescent signal was detected from tissues of the control mice (see Figure 2A).

Ex vivo bioluminescence imaging of various tissues in mice receiving naive CD4+ BDC cells. For ex vivo visualization of the transferred BDC cells present in various tissues of recipient mice, the mice were dissected on day 113 after cell transfer. Images were obtained at 15 minutes after intraperitoneal injection of
We then quantified the bioluminescent signals by selecting ROI corresponding to selected tissues (Figure 2B). The total photon flux was greatest in the spleen and lung, with the pancreas and mesenteric lymph nodes being significantly higher than the other tissues analyzed. The BLI signals from control mice were negligible. Quantification of luciferase activity in the dissected tissues is subject to oxygen availability and levels of substrate and adenosine triphosphate and may, therefore, not constitute a quantitative measurement. Additionally, although these BLI signals may not be directly compared among different tissues without normalizing the tissue volume, the ex vivo BLI results are confirmed by using the FACS analyses described below. These BLI data are able to reveal sites of BDC cells' homing that can be further confirmed using other quantitative assays, such as flow cytometry analyses of tissue-infiltrating cells. Therefore, the use of whole-body in vivo imaging serves as an excellent guide for the selection of tissues and times for more labor-intensive assays at necropsy. Moreover, the patterns of signal localization are similar among animals that are analyzed similarly, indicating consistency in the biology, which can also be confirmed using other methods, such as flow cytometry.
Quantification of BDC Cells in Tissues by Flow Cytometry
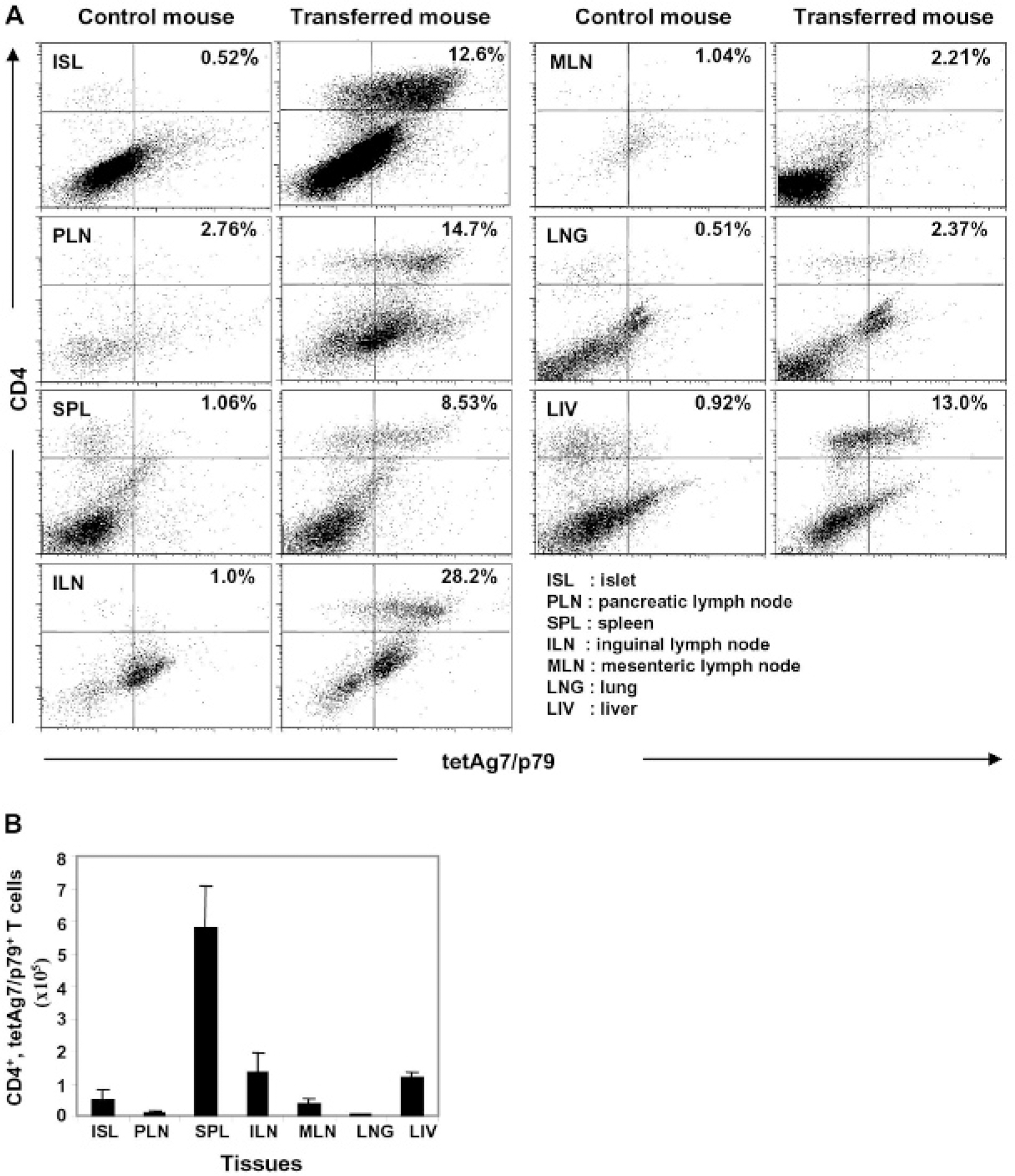
Although bioluminescent signals obtained both in vivo and on excised tissues showed the presence of BDC cells in selected tissues, we performed additional studies to confirm and further quantify the number of BDC cells present in various tissues. We prepared single-cell suspensions from cells infiltrating each tissue and stained the cells with anti-CD4 plus tetAg7/p79 for analysis by flow cytometry (Figure 3A). The results showed that BDC cells were detected in all of the analyzed tissues, including pancreatic lymph nodes and the islets at the relative frequencies predicted by the bioluminescent signal intensities. The fraction of cells in islets that were BDC cells was 16%, with greater than 50% of the cells in pancreatic lymph nodes being BDC cells. The fractions in the lung and liver were 8 and 20%, respectively.
We also have calculated the total number of BDC cells that were present in each tissue 113 days post-transfer (Figure 3B). As expected from BLI results, a large number of BDC cells were present in the spleen, whereas a smaller number of cells were present in other tissues, including the pancreas, pancreatic lymph nodes, and inguinal and mesenteric lymph nodes. 25 The dissected pancreas preparation used for flow cytometric analyses was processed to enrich for islets; therefore, it contained T cells that have trafficked to and proliferated in the islets. 26 These results also show that, although a strong bioluminescence signal was initially detected in the lung and liver, the number of BDC cells measured using flow cytometry in these two tissues was lower than those of other tissues in which the bioluminescence signals at day 113 were relatively high.
Altogether, these results demonstrated that the macroscopic whole-body in vivo BLI analysis can be used to reliably visualize T-cell trafficking in animals and reveal the systemic trafficking patterns prior to and after the development of severe insulitis. This noninvasive imaging approach also provides a measure of the relative number of T cells present within tissues such as the pancreas and lymphoid tissues. Bioluminescence analyses of the excised tissues can be used to further resolve signals from tissues that are closely associated in vivo, including pancreatic lymph nodes with the pancreas, and, together with macroscopic whole-body in vivo imaging, these approaches provide a picture of the general cell-trafficking patterns.
BLI of CD4+luc+BDC Cells during the Development of Insulitis Leading to Diabetes
Although many luc+CD4+BDC cells are present in pancreatic lymph nodes and islets, the recipient mice failed to develop diabetes. This is not due to the introduction of luciferase transgene into BDC mice because purified CD4+BDC cells obtained from BDC mice of the same age in our mouse colony that have not been crossed with luc transgenic mice also did not induce diabetes in recipient mice (data not shown). One possibility explaining these results is that the study was terminated prior to diabetes development, although control mice receiving whole BDC mouse splenocytes develop diabetes within 6 weeks after cell transfer. 19 This seems unlikely because our additional studies have shown that recipient mice still did not develop diabetes at 23 weeks following the transfer of BDC cells alone (data not shown). Alternatively, BDC cells may require the presence of other types of cells for them to destroy the islets and induce diabetes. It has been shown that transfer of T cells alone did not induce diabetes and the presence of non-CD4+ cells such as CD8+ T cells are necessary.27–29 Previous studies also have shown that cotransfer of the clonal CD4+BDC2.5 T cells with CD4+ cell–depleted splenocytes from newly diabetic NOD mice induced accelerated diabetes in the recipient animals. 22 To better understand the trafficking behavior of BDC cells during islet inflammation that led to diabetes, we cotransferred purified CD4+luc+BDC cells and CD4+ cell–depleted luc− splenocytes from newly diabetic NOD mice into NOD/scid mice (Figure 4A). Although the recipient mice receiving these non-CD4+ splenocytes alone did not develop diabetes, the mice that received both populations of cells developed diabetes as early as on day 9 following cell transfer, and all recipient mice developed diabetes within 3 weeks (Figure 4B).

FACS analyses of BDC cells present in various tissues of the recipient mice. A, FACS analyses of BDC cells. To confirm the presence of CD4+ BDC cells in the various tissues of recipient mice transferred with CD4+ BDC cells, the mice were dissected on day 113 after cell transfer. These mice were not diabetic at this time point. Single-cell suspensions from tissues were prepared for staining with an anti-CD4 antibody and tetAg7/p79 and FACS analyses. The data shown are representative of three different experiments. B, Total number of CD4+, tetAg7/p79+ BDC cells present in various tissues. The numbers of CD4+, tetAg7/p79+ cells in these tissues were calculated by multiplying the total number of cells obtained from these tissues by the percentage of CD4+, tetAg7/p79+ cells obtained from the FACS analyses shown above in A.
Our BLI studies showed that the kinetics of luc+BDC cells' trafficking in mice cotransferred with both CD4+luc+BDC cells and CD4+ cell–depleted luc− splenocytes from newly diabetic NOD mice was similar, at the level of the whole body and organs, to that in mice that received BDC cells alone (Figure 5). Regardless of whether BDC cells were transferred alone or in combination with the luc− non-CD4 splenocytes, they migrated first to the lung and then could be detected in the pancreas, inguinal lymph nodes, and spleen within 1 week. The presence of BDC cells in various tissues of the diabetic recipient mice with both cell types was confirmed using flow cytometry (Figure 6).
To further quantify and compare the changes in bioluminescent signals in various tissues of the two different groups of recipient mice at the same time points prior to and after the cotransferred mice became diabetic, we imaged BDC cells and analyzed ROI drawn on the pancreas, inguinal lymph nodes, and spleen. Analyses of ROIs from these two groups of mice imaged at the same time showed that the total flux gradually increased over time (Figure 7). A similar trend of total flux increase was detected in the ROI over the pancreas in both groups of mice, although there appeared to be continued proliferation of cells in the mice that received both cell types in the lymph nodes and spleen. Comparable levels of total flux over time were also detected in the inguinal lymph nodes and spleen in both groups of mice up to day 15 after cell transfer, a time when all mice receiving both types of cells became diabetic after cotransfer of the cells. However, it appeared that the total flux in lymph nodes and spleen increased at a faster rate in mice that received both cell types during the postdiabetic period, between days 15 and 20, than that observed in nondiabetic mice that only received BDC cells. Therefore, cotransfer of CD4+ cell–depleted splenocytes resulted in diabetes but did not otherwise change the overall trafficking patterns of BDC cells to these tissues during the prediabetic or early diabetic stage. These results suggested that the cotransferred splenocytes, capable of helping BDC cells to induce accelerated diabetes, do not do so by modulating BDC cells' trafficking to the pancreas and the total number of BDC cells in the pancreas, but they may serve to increase the numbers of BDC cells in the lymph nodes and spleen, suggesting increased proliferation of these cells outside the pancreas.

Isolation of CD4+ and CD4+ cell–depleted splenocytes and diabetes development in recipient mice. A, Noncultured CD4+ BDC cells were isolated from the splenocytes of luc-BDC mice (N7) using magnetic beads by negative selection. The purity of the cells was determined by staining the cells with an anti-CD4 antibody and tetAg7/p79. The CD4+ cell–depleted splenocytes were isolated from newly diabetic NOD mice (within 3 days after blood glucose level is ⩾ 250 mg/dL) using magnetic beads by negative selection and their purity was analyzed by FACS. B, Diabetes development in recipient mice. The recipient NOD/scid (8–9 weeks old) mice were adoptively cotransferred with noncultured CD4+luc+BDC cells (7 × 106/mouse) plus CD4+ cell-depleted luc− splenocytes (7 × 106/mouse) on day 0. Recipient mice were monitored for blood glucose. Mice were considered diabetic when their blood glucose was ⩾ 250 mg/dL for 2 consecutive days. Data represent cumulative diabetes incidence from three different experiments (n = 5). Circles = CD4+BDC cells plus CD4+ cell–depleted splenocytes; triangles = CD4+ cell–depleted splenocytes only.
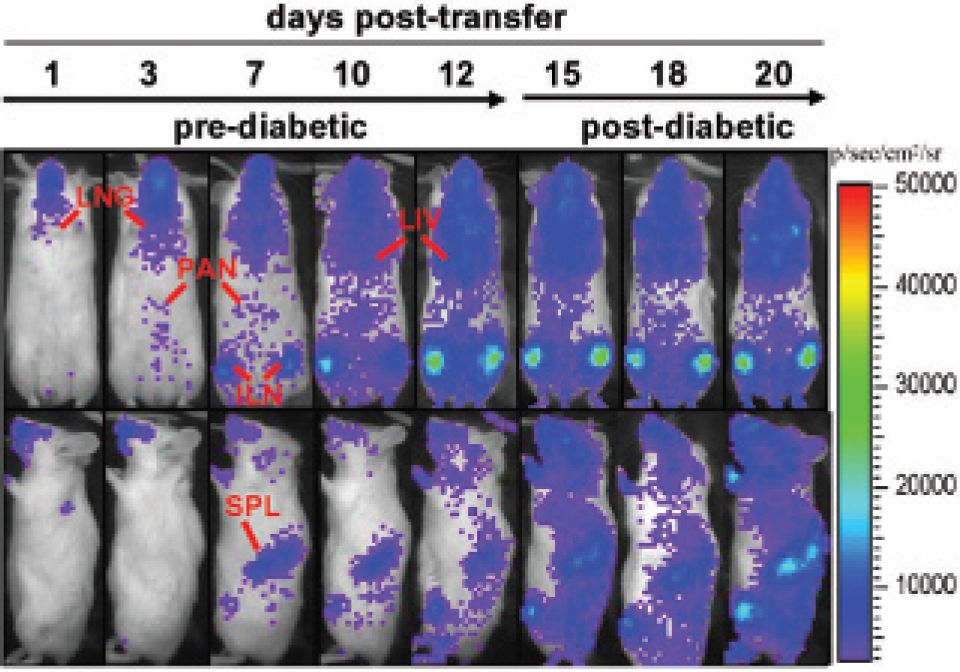
Bioluminescence imaging analyses of CD4+luc+BDC cells cotransferred with luc− CD4+ T cell–depleted NOD mouse splenocytes. Real-time in vivo bioluminescence imaging analyses of CD4+ BDC cells adoptively cotransferred with CD4+ T cell–depleted NOD mouse splenocytes into NOD/scid mice. Naive CD4+ BDC cells (7 × 106/ mouse) isolated from splenocytes of luc-BDC mice (N7) were intravenously injected together with CD4+ T cell–depleted diabetic NOD splenocytes (7 × 106/mouse) into female NOD/scid mice of 8 to 9 weeks of age. Bioluminescence images of BDC cells were taken with a 2-minute acquisition time using a charge-coupled device camera at 15 minutes after intraperitoneal injection of
Cotransferred CD4+ Cell–Depleted Splenocytes Help BDC Cells Induce Severe Invasive Insulitis and Destroy Islets
BDC cells induce insulitis in both groups of recipients, but only the recipients receiving BDC cells and CD4+ cell–depleted splenocytes developed diabetes. These results would suggest that it is not the total cell numbers present in the pancreas that correlates with the development of diabetes but rather the cellular function within the target tissue and the microscopic localization of the cells within the tissue. Different degrees and types of insulitis may occur in the recipient animals after transfer of different cell populations. These include peri-insulitis that does not lead to islet destruction, whereas invasive insulitis destroys islets and leads to diabetes.20,21 To further evaluate why BDC cells plus the cotransferred CD4+ cell–depleted splenocytes, but not BDC cells alone, induced accelerated diabetes, we examined whether these splenocytes may increase infiltration of BDC cells into their target organ in the pancreas (ie, the islets) and induce invasive insulitis, whereas BDC cells alone induce peri-insulitis in recipients. To make these distinctions, it was necessary to perform microscopic imaging of the target tissue since no macroscopic imaging method could distinguish cell distribution differences at this resolution.
The results of our initial ex vivo BLI analyses of excised pancreas showed that the bioluminescent signals in the pancreas of both groups of recipients were not significantly different from each other (Figure 8, A and B). These results further support the contention that BDC cells can traffic to the pancreas with or without being cotransferred with other splenocytes. We then performed histologic analyses of the pancreatic tissues to determine whether mice cotransferred with both cell types may develop more severe invasive insulitis than in mice that received BDC cells alone. Indeed, the islets in the pancreatic sections obtained from cotransferred mice on day 20 following cell transfer showed invasive insulitis that were heavily infiltrated with leukocytes and appeared severely damaged, whereas the islets from mice transferred with BDC cells only showed signs of peri-insulitis and their islets remained intact (Figure 8C). These results suggest that the cotransferred splenocytes may enhance the local infiltration of BDC cells into the islets.
To better understand which cell populations may contribute to the invasive insulitis observed in the cotransferred mice, we performed additional FACS analyses of islet-infiltrating cells isolated from both groups of recipient mice. We found that the percentage of both CD11b+ cells and CD11c+ cell populations were increased only in the islets of cotransferred recipient mice (Figure 9A). It is unknown whether these two populations of cells may be directly involved in the enhanced invasiveness of BDC cells into the islets. One possibility is that these cells may promote BDC cell activation, and, thus, more BDC cells may be activated in the islets of cotransferred mice. We examined the activation status of BDC cells present in the islets by measuring the expression of CD69 and CD62L on BDC cells. Our results show that there is no difference in CD69 and CD62L expression on BDC cells isolated from the islets of both groups of recipient mice (Figure 9B).
Overall, the results are consistent with the concept that although BDC cells are able to traffic efficiently to the pancreas in the absence of cotransferred splenocytes, the presence of non-CD4 splenocytes helps them locally to infiltrate and destroy the islets without altering their systemic or global trafficking patterns in animals. These observations could not have been made without imaging over a range of scales from macroscopic imaging, or whole-body BLI, to microscopic imaging via light microscopy of tissue sections.
FACS analyses of BDC cells present in various tissues of cotransferred recipient mice. A, FACS analyses of BDC cells. Various tissues of diabetic recipient mice that were cotransferred with noncultured CD4+luc+BDC cells and CD4+ T cell–depleted luc− splenocytes from newly diabetic NOD mouse were dissected on day 13 after cell transfer. The results shown were obtained from a mouse that became diabetic on day 11 after cell transfer. Single-cell suspensions from these tissues were prepared for staining using an anti-CD4 antibody and tetAg7/p79. The data shown were obtained from a representative mouse of five mice from three different experiments. B, Total number of CD4+, tetAg7/p79+ BDC cells present in various tissues of cotransferred recipient mice (n = 3). The numbers of CD4+, tetAg7/p79+ cells in these tissues were calculated by multiplying the total number of cells obtained from these tissues by the percentage of CD4+, tetAg7/p79+ cells obtained from the FACS analyses shown above in A.

A comparison of the kinetics of in vivo bioluminescent signals detected from tissues of single-transferred versus cotransferred recipient mice. Noncultured luc+CD4+ BDC cells (7 × 106/mouse) isolated from splenocytes of luc-BDC mice were intravenously injected alone (A, BDC alone) or together (B, BDC + splenocytes) with CD4+ T cell–depleted diabetic NOD splenocytes (7 × 106/mouse) into female NOD/ scid mice of 8 to 9 weeks of age. Bioluminescence images of BDC cells were taken with a 2-minute acquisition time using a charge-coupled device camera at 15 minutes after intraperitoneal injection of D-luciferin (150 mg/kg). The total flux was determined for the region of interest of the body corresponding to the three selected tissues (inguinal lymph node [ILN], spleen [SPL], and pancreas [PAN]) and normalized with signals from control mice. The experiments were terminated on day 20 after cell transfer, when all of the cotransferred mice developed diabetes. At least four mice were used in each of the studies from four different experiments.
Discussion
Trafficking of diabetogenic CD4+ T cells such as BDC cells to the pancreas is necessary for them to induce insulitis and cause diabetes. Diabetogenic CD4+ T cells can also facilitate the homing of CD8+ T cells to the islets during diabetes development. 30 However, the longitudinal trafficking behavior of diabetogenic CD4+ T cells to the pancreas during islet inflammation and diabetes development has not been determined. Although it is possible to trace T cells such as BDC cells using antigen-specific tetramers, 23 these methods are invasive, requiring tissue removal; hence, only terminal data can be obtained. To better understand the trafficking of diabetogenic T cells in animals and evaluate the effects of various diabetes treatment regimens on the trafficking of these T cells, it is important to noninvasively monitor cell trafficking using whole-body in vivo imaging and use these data to guide tissue sampling for the invasive assays. Whole-body in vivo imaging will help us perform longitudinal studies that examine the in vivo behavior of diabetogenic T cells and assess whether treatments to prevent diabetes or islet graft rejection can be mediated through modulating the systemic trafficking T cells in living animals in addition to other cellular functions. For this purpose, we have developed a novel animal model that uses BLI to visualize, in real time, the trafficking behavior of CD4+luc+BDC cells in living animals under conditions that either lead to diabetes or not. These novel studies have revealed the kinetics, tissue localization, and relative cell numbers in various tissues in living mice during the development of islet inflammation and diabetes. We also used this method to examine whether the conditions that induce diabetes in an adoptive transfer model may do so by causing altered global and/or local trafficking patterns of T cells.

Comparable ex vivo BLI signals were detected in the pancreas of both groups of recipients, although only cotransferred mice developed severe invasive insulitis. A and B, Ex vivo bioluminescent imaging of CD4+BDC cells. Recipient mice were single-transferred with 5 × 106 of CD4+BDC cells (BDC alone) or cotransferred with an equal number (5 × 106) of BDC cells and CD4-depleted splenocytes from newly diabetic NOD mice (BDC + splenocytes). A, The region of interest analyses of ex vivo bioluminescent signals detected from pancreas excised from recipients on day 10 to day 11. During these time points, cotransferred mice were 3 to 4 days after diabetes onset, whereas none of the single-transferred mice were diabetic. The total flux was normalized using control mice that received no cells. At least four mice per group were used in three independent experiments. The total flux between both groups were not significantly different from each other (p > .9). B, Ex vivo bioluminescent signals of the pancreas from representative mice from both groups of recipient mice. The pancreas was excised from recipient mice of both groups on day 11 after cell transfer for BLI analyses. C, Mice were single-transferred with CD4+BDC cells (BDC alone, 5 × 106/mouse) or cotransferred with an equal number of CD4+BDC cells plus CD4 cell–depleted splenocytes (BDC+ splenocytes, 5 × 106/mouse) isolated from newly diabetic NOD mice. Pancreas obtained from single-transferred or cotransferred recipient mice were analyzed on day 20 after cell transfer using hematoxylin and eosin histologic staining. At this time point, the representative cotransferred mouse has been diabetic for 4 days, whereas the single-transferred mouse remained diabetes free. Scale bars = 50 μm.
In our experiments in which noncultured CD4+luc+BDC cells were transferred alone and the recipients did not develop diabetes, we were able to image the trafficking of BDC cells to various tissues and monitor them in mice for at least 20 weeks. Although the BDC cells were isolated from NOD mouse background, the percentage of luc+CD4+ cells that express non-BDC TCRs is low (less than 5% of CD4+ BDC cells); thus, their BLI signals do not affect the BLI signals generated by BDC cells. In addition, the location of tissues may affect the measured photons 31 ; thus, superficial tissues, such as lymph nodes, may generate stronger BLI signals than deeper tissues, such as the pancreas. However, we have confirmed and validated the results obtained using BLI with analyses of excised tissues and by flow cytometry. These results showed that the BLI signals detected from each tissue indeed represented the presence and relative numbers of BDC cells in these tissues, and the relative intensity of BLI signals and the total flux of each tissue's ROI are proportional to the number of BDC cells present in these tissues. Although in vivo BLI may not clearly distinguish between the signals from tissues in close proximity to each other, such as the pancreas and pancreatic lymph nodes, the presence and location of BDC cells in these tissues were determined and confirmed using other methods. Together, the results demonstrated that whole-body in vivo BLI is a useful and reliable tool for examining cell-trafficking patterns in the entire organism during the onset of islet inflammation and diabetes. This method is also useful for monitoring the effect of disease treatments on T cells through visualizing cell trafficking in long-term longitudinal studies of chronic diseases such as islet inflammation and diabetes.

An increased population of CD11b+ or CD11c+ islet-infiltrating cells is present in cotransferred recipient mice. Islet-infiltrating cells were prepared from recipient mice that were single-transferred with BDC cells alone (BDC alone) or were cotransferred with BDC cells plus CD4-depleted diabetic NOD mouse splenocytes (BDC + splenocytes) on day 11 after cell transfer. Some mice were also transferred with CD4-depleted diabetic NOD mouse splenocytes (splenocytes alone) as controls. A, The cells were stained with antibodies against CD11b or CD11c. The islet-infiltrating cells of cotransferred and not single-transferred recipient mice contained a significantly increased population of CD11b+ and CD11c+ cells (p < .02). The results shown are representative of three independent experiments. Dashed lines = control mice transferred with no cells; solid lines = mice transferred with cells. p < .02. B, The cells were also stained with anti-CD69 and anti-CD62L antibodies. The stained cells were analyzed by FACS analysis. The cotransferred mice (BDC + splenocytes) were 2 days post–diabetes onset, whereas the single-transferred (BDC alone or splenocytes alone) mice were not diabetic.
Our study also demonstrated that BDC cells, with or without cotransfer of CD4+ cell–depleted splenocytes from newly diabetic NOD mice, were detectable in the pancreas shortly after cell transfer and remained in this tissue for prolonged periods of time. Although the results of BLI and confirmatory assays revealed comparable numbers of BDC cells in the pancreas of recipient mice during the first 2 to 3 weeks following cell transfer, only the mice with cotransplanted cells developed diabetes during the study period. Further accumulation of BDC cells in lymphoid tissues and the islets at late time points, which may be due to potential homeostatic expansion, did not lead to diabetes in mice receiving CD4+BDC cells alone. These results show that the mere presence of BDC cells in the pancreas does not necessarily lead to diabetes, even after prolonged periods of time (eg, 23 weeks after cell transfer), consistent with previous results.20–22 Other types of splenocytes and signals provided by these other cell types appear to be required for islet destruction and diabetes but perhaps not for the general trafficking patterns. The cotransferred splenocytes did not alter the overall kinetics of appearance of BDC cells in the pancreas of recipient mice, although their presence may have increased cell numbers infiltrating locally into the islets.
Our additional studies to understand the cellular mechanisms showed that the numbers of CD11b+ and CD11c+ cells present in the islets were increased. However, the CD62L and CD69 expression on BDC cells is not altered between both recipient mouse groups, which suggests that these splenocytes did not change the activation status of BDC cells. Further studies would be necessary to determine whether these splenocytes may directly help or enhance the infiltration of BDC cells into the islets and cause severe invasive insulitis and islet destruction. In comparison, mice that received BDC cells alone only developed peri-insulitis and their islets remained intact. Altogether, these results indicate that the localization and function of BDC cells at the tissue level differ between the two groups of recipient mice. In addition, these results are consistent with the findings based on ROI imaging analyses that BDC cells are present in the pancreas with no significant difference in total cell numbers, but these cells do not destroy islets and cause diabetes when administered alone. The trafficking of BDC cells to pancreatic lymph nodes precedes their infiltration into islets. 32 Therefore, altogether, these results suggest that the cotransferred splenocytes helped BDC cells induce accelerated diabetes not by promoting their global trafficking but by promoting their local trafficking from pancreatic lymph nodes to the islets.
Several nonexclusive possibilities may explain these observations. First, it is likely that antigen-presenting cells such as the CD11b+ and CD11c+ splenocytes can help BDC cells to induce diabetes. Our results suggest that they may not do so by changing the activation status of BDC cells. It remains possible that these cells may promote the trafficking of more BDC cells and/or other types of splenocytes to the islets. In addition, a recent report suggests that macrophages may be the final effector cells killing islet β-cells in BDC cell–induced diabetes. 33 Thus, another reason that the mice receiving CD4+BDC cells alone did not develop diabetes may also be due to insufficient recruitment or inefficient function of macrophages in the pancreatic lymph nodes or the islets. Second, it has been shown that both CD4+ and CD8+ T cells are required for the initiation of beta-cell destruction and for adoptive transfer of diabetes to NOD mice.27–29,32,34–36 CD8+ T cells may be required during the early stage of disease induction and help CD4+ T cells induce diabetes.22,28 Nevertheless, in our model system, CD8+ cells are not likely to be involved in the invasive insulitis found in cotransferred recipient mice because we did not detect a significant number of these cells in the islet-infiltrating cells of both groups of recipients (data not shown). The CD4-depleted splenocytes contained an enriched population of CD8+ T cells. It is currently unclear whether the CD8+ T cells may have an effect on the trafficking, number, and localization of CD4+ BDC cells in the islets and other tissues. Third, the presence of CD4+CD25+ T regulatory cells in noncultured CD4+BDC cells may be responsible for the suppression of BDC cells, when they are transferred alone, to induce diabetes in recipient mice. The suppression of BDC cells by T regulatory cells may be blocked by other cells present in CD4+ cell–depleted NOD splenocytes.
Altogether, these results demonstrate that the whole-body in vivo BLI analyses can help us evaluate the trafficking behavior of autoreactive T cells that cause autoimmune disease such as type 1 diabetes and guide us to the times and tissues where the unique biology is taking place, and will be a useful method for monitoring the effect of various treatments that may regulate cell trafficking and inhibit diabetes. The complementary methods of whole-body imaging and the microscopic analyses have revealed that localization of cells within the tissues and potential cell–cell communication at the site of tissue inflammation and destruction may also offer another target for disease intervention. Therefore, the approaches that span the macroscopic to microscopic range of scales in cell imaging are complementary and will be helpful for evaluating approaches to regulating T-cell trafficking that lead to inflammation and autoimmune diseases such as type 1 diabetes.
Footnotes
Acknowledgments
We would like to thank Drs. Mathis and Benoit for providing the BDC2.5 TCR transgenic mice. These studies were supported in part by the H.L. Snyder Medical Foundation, American Heart Association, Juvenile Diabetes Research Foundation, and National Institute of Health.
