Abstract
Reporter gene techniques have been applied toward studying the physiologic phenomena associated with tumor hypoxia, a negative prognostic indicator. The purpose of this study was to assess the potential adverse effects of hypoxic conditions on the effectiveness of four commonly used reporter genes: Renilla luciferase, monomeric red fluorescent protein, thymidine kinase, and lacZ. Tumor-forming A375 cells expressing a trifusion reporter consisting of Renilla luciferase, monomeric red fluorescent protein, and thymidine kinase were subjected to decreasing oxygen tensions and assayed for reporter expression and activity. A375 cells expressing β-galactosidase were similarly exposed to hypoxia, with activity of the reporter monitored by cleavage of the fluorescent substrate 7-hydroxy-9H-(1, 3-dichloro-9, 9-dimethylacridin-2-one)-β-galactoside (DDAOG). Generation of signal in in vivo tumor models expressing bioluminescent or β-galactosidase reporters were also examined over the course of hypoxic stresses, either by tumor clamping or the antivascular agent 5, 6-dimethylxanthenone-4-acetic acid (DMXAA). Our findings indicate that bioluminescent and fluorescent reporter activity are decreased under hypoxia despite minimal variations in protein production, whereas β-galactosidase reporter activity per unit protein was unchanged. These results demonstrate that combining β-galactosidase with the DDAOG optical probe may be a robust reporter system for the in vivo study of tumor hypoxia.
Reporter gene constructs that are expressed under the control of hypoxia-inducible factor (HIF)-1 were recently developed.3–6 The absence of oxygen (hypoxia) is a key prognostic factor in the management of cancer, with hypoxic tumors exhibiting increased aggressiveness, invasion, metastasis, and resistance to therapy.7–9 HIF-1 has emerged as a key component of the hypoxic cell phenotype. 10 A heterodimeric transcription factor whose a subunit rapidly undergoes proteosomal degradation under oxic conditions, HIF-1 facilitates oxygen-dependent gene expression by binding to promoters containing a hypoxia response element (HRE). 10 HRE-driven genes have been associated with energy metabolism, angiogenesis, extracellular matrix remodeling, and cellular growth and apoptosis. 11 A number of clinical and experimental studies have noted the overexpression of HIF-1 in cancer cells and have correlated its expression levels with the characteristics of the hypoxic tumor phenotype described above.12–16 The importance of HIF-1 as a transcriptional regulator and a mediator of tumor aggressiveness has encouraged further study of this factor as a potential diagnostic and therapeutic target in cancer.
Imaging of HIF-1-mediated transcriptional activation using reporter genes and their protein products assumes that the generation of imaging signals by these constructs is not adversely affected by the low oxygen concentrations that induce their expression. It has been shown that green fluorescent protein and its siblings require oxygen-dependent post-translational modification to become active fluorophores, whereas firefly and Renilla luciferase use molecular oxygen to generate photons following interaction with their substrates.17–19 This implies that under low oxygen tension, the absence of reporter signal may lead to an incorrect interpretation that HIF-1 is not expressed in the target tissue when, in fact, the reporter is being produced, but its activity is compromised by the lack of molecular oxygen. PET reporter approaches that rely on trapping of tritiated thymidine–labeled substrates in cells expressing a viral TK 20 have not been evaluated in this regard but may be sensitive to cellular energy levels at low oxygen tension as adenosine triphosphate (ATP) is required as a phosphate donor for the kinase reaction. These considerations may cause a deviation between signals obtained from HRE-based reporter genes and the actual level of HIF-1-mediated gene expression, as well as other reporter systems in which oxygen levels may be varying.
Our aim was to investigate the oxygen sensitivity of four commonly used reporter proteins: Renilla luciferase, red fluorescent protein, TK, and β-galactosidase. This was performed by observing reporter-expressing cells exposed to reduced levels of oxygen in vitro. The relationship between the level of reporter protein produced, reporter signal-generating activity, and oxygen levels was then assessed for each reporter gene. The applicability of the reporter gene behavior observed in cell culture to the in vivo situation was then determined using mouse models of tumoral hypoxia.
Materials and Methods
Cell Lines and Tumors
Human melanoma A375 engineered to stably express a triple-fusion gene construct composed of Renilla luciferase (hrl), monomeric red fluorescent protein (mrfp), and truncated herpes simplex virus type 1 sr39 thymidine kinase (ttk) driven by a cytomegalovirus (CMV) promoter (hrl-mrfp-ttk). 2 The human melanoma A375 cells were transfected with a CMV-lacZ construct (Promega, Madison, WI) using Lipofectamine reagent (Invitrogen, Carlsbad, CA), and a stable cell line was obtained by selection with hygromycin. All cell lines were cultured in Dulbecco's Modified Eagle's Medium supplemented with 10% fetal bovine serum. For in vivo imaging, 1 × 107 A375/hrl-mrfp-ttk cells or A375/lacZ cells were inoculated subcutaneously on the sacral region of male athymic nu/nu mice (Charles River Laboratories, Inc, Wilmington, MA). Tumors were imaged when they reached a size of 8 to 10 mm in largest diameter. All animal experiments were performed according to guidelines and directives set forth by the Stanford University Administrative Panel on Laboratory Animal Care.
Chemicals
Coelenterazine was purchased from Biotium, Inc. (Hayward, CA). [8-3H]Penciclovir was obtained from Moravek Biochemicals (Brea, CA). 7-Hydroxy-9H-(1, 3-dichloro-9, 9-dimethylacridin-2-one)-β-galactoside (DDAOG) was purchased from Molecular Probes (Eugene, OR). 5, 6-Dimethylxanthenone-4-acetic acid (DMXAA) was obtained from Dr. Bruce Baguley, Auckland Cancer Society Research Centre, Auckland, New Zealand.
Hypoxia Treatment
Cells were plated on 6 cm Petri dishes and then transferred to variable hypoxia chambers containing either 2% or 0.5% oxygen (Biotrace Inc., Cincinnati, OH) or ⩽ 0.02% oxygen (Sheldon Corporation, Cornelius, OR) for 24 hours.
Western Blot
Cells were lysed in 9 M urea, 75 mM Tris-HCl, pH 7.5, and 0.15 M β-mercaptoethanol and sonicated briefly. Equal volumes of protein lysate were electrophoresed on 10% sodium dodecyl sulfate–polyacrylamide gel electrophoresis gels. The Renilla luciferase component of the triple-fusion construct was detected by monoclonal mouse anti–Renilla luciferase antibody (Clone 5B11.2) purchased from Chemicon International (Temecula, CA). This band was used as an estimate of levels of the intact trifusion reporter protein, as validated in previous studies. 2 β-Galactosidase was detected by monoclonal mouse anti-β-galactosidase purchased from AbCam (Cambridge, MA). For detecting loading controls, anti–glyceraldehyde-3-phosphate dehydrogenase (GAPDH) and anti-α-tubulin antibodies were obtained from Research Diagnostics, Inc. (Concord, MA). The CMIR_Image application developed at Massachusetts General Hospital was used to obtain quantitative estimates of protein levels by integrating the intensity of each band in a Western blot. Values obtained for the trifusion reporter and β-galactosidase were normalized relative to the corresponding loading control level and compared between hypoxic exposures.
In Vitro Bioluminescence, Fluorescence, TK, and β-Galactosidase Activity Measurements
For bioluminescence, cells were harvested and lysed using Passive Lysis Buffer (Promega, Madison, WI). After 15 minutes of exposure to lysing buffer, cell lysates were separated from debris at 12,000 rpm for 10 minutes in a bench-top centrifuge. This was performed in the hypoxia chambers, after which the tubes were sealed with parafilm to prevent reintroduction of oxygen on exposure to 21% oxygen in air. The lysates were mixed with the Renilla luciferase substrate coelenterazine, and photon release was measured immediately in a Monolight 2010 Luminometer (Analytical Luminescence Laboratory, San Diego, CA). Fluorescence intensity was assessed on live cells by flow cytometry. Cells were grown on 6 cm Petri dishes and following 24 hours of incubation at assigned oxygen settings were harvested and transferred to 5 mL Falcon tubes that were sealed with parafilm prior to removing the samples from the chambers. These samples were then studied using a FACSCalibur system employing CellQuest software (BD Biosciences, San Jose, CA). Background fluorescence levels were determined by measuring genetically matched cells that were not transfected with the reporter. The median fluorescence in positive cells was measured using histogram analysis of the FACS results. TK activity was assessed by the radiographic measurement of cellular accumulation of [3H] penciclovir, as described previously. 20 To assess β-galactosidase activity in vitro, A375/lacZ cells were plated on 6 cm Petri dishes and placed in either ⩽ 0.02, 0.5, 2, or 21% oxygen chambers when 60% confluent. The untransfected A375 cell line served as a negative control. Twenty-four hours later, the cells were washed with phosphate-buffered saline (PBS), suspended in Hanks' Buffered Salt Solution, and then transferred to the wells of a 96-well black flat-bottom plate. The optical probe DDAOG was added to a final concentration of 10 μM. β-Galactosidase activity was quantified by its ability to cleave its substrate DDAOG into the cleaved product, 7-hydroxy-9H-(1, 3-dichloro-9, 9-dimethylacridin-2-one) (DDAO). 21 Using an IVIS 200 optical imaging system (Xenogen Corporation, Alameda, CA), DDAOG was imaged using an excitation filter with a passband of 445 to 490 nm and an emission filter with a passband of 575 to 650 nm. The cleaved product DDAO was imaged with excitation and emission filters sensitive to 580 to 610 nm and 650 to 670 nm, respectively.
In Vivo Optical Imaging
Male athymic nu/nu mice bearing A375/hrl-mrfp-ttk tumors were anesthetized with 2 to 2.5% isoflurane, after which each mouse was placed in a light-tight imaging chamber of the IVIS 200 optical imaging system (Xenogen Corporation). Five microliters of a 3 μg/mL PBS stock solution of coelenterazine was administered directly into the tumor with a 27-gauge needle, and a series of images were acquired immediately. After measuring the bioluminescence signal for approximately 10 minutes following substrate injection, blood flow and oxygen delivery were occluded by lifting the skin encasing the subcutaneous tumor mass and applying a clamp. Bioluminescence images were acquired before, during, and after clamping. With the clamp in place, a second intratumoral injection of coelenterazine was administered to both clamped and unclamped control tumors. The mice remained anesthetized throughout the study. Total photon emissions from the control and clamped tumors over the course of the experiment were quantified using the Living Image software (Xenogen Corporation).
An identical experimental course was used to assess reporter mRFP fluorescence before, during, and after clamping. In this case, the IVIS 200 was operated in fluorescence mode using excitation and emission filters specific to mRFP.
The effect of decreasing tumor oxygenation on bioluminescence was similarly assessed in the tumors following intraperitoneal administration of the antivascular agent DMXAA (24 mg/kg). 22 The antivascular effects of DMXAA have been described in a number of reports.22,23 DMXAA has been shown to cause inhibition of blood flow in murine tumors24,25 and increases pimonidazole-assessed tumor hypoxia. Coelenterazine was administered intratumorally at 20-minute intervals and images were acquired using an IVIS 200 optical imaging system. As above, this experimental procedure was repeated substituting fluorescence imaging for bioluminescence imaging to evaluate mRFP under these conditions.
To image β-galactosidase activity in vivo, mice bearing subcutaneous A375/lacZ tumors were anesthetized with isoflurane, as described above. The A375/lacZ tumors were clamped or unclamped as above and then directly injected with ≈5 μL volume of a 10 μM solution of DDAOG. Images were acquired for 10 seconds at 5-minute intervals. On an IVIS 200 optical imaging system (Xenogen Corporation), DDAOG was imaged using an excitation filter with a passband of 445 to 490 nm and an emission filter with a passband of 575 to 650 nm. The cleaved product DDAO was imaged with excitation and emission filters sensitive to 580 to 610 nm and 650 to 670 nm, respectively. The obtained images were analyzed using CMIR_Image. Calibration factors relating the images measured using the filter sets specific to DDAOG and DDAO were calculated based on in vitro measurements of DDAOG exposed to purified β-galactosidase protein. These factors were used to normalize the DDAOG and DDAO images and calculate the ratio DDAO/(DDAOG + DDAO) on a pixel-by-pixel basis. This value gives an indication of the percentage of injected probe converted to DDAO by β-galactosidase.
Results
Protein Production in Low Oxygen Environments
A375/hrl-mrfp-ttk cells and A375/lacZ cells were incubated for 24 hours in either ⩽ 0.02%, 0.5%, or 2% hypoxia prior to analysis. There was little variation in the level of protein production at ⩽ 0.02%, 0.5%, 2%, and 21% oxygen (Figure 1) in either transfected cell line.
Oxygen Sensitivity In Vitro
The activity of the luciferase enzyme, demonstrated by photon emission, significantly dropped at ⩽ 2% oxygen compared with 21% oxygen. Although the amount of reporter protein produced did not differ significantly at low oxygen concentrations compared with 21% oxygen, the activity of protein present at the time of analysis was significantly decreased under low oxygen conditions (Figure 2A). There was a significant correlation between the drop in oxygen concentration and the decrease in bioluminescence (r = .998, p = .0022, t-test). Fluorescence intensity was reduced with a drop in oxygen to ⩽ 2% (Figure 2B). TK activity also decreased as the reporter cells were exposed to decreasing oxygen concentrations (Figure 2C). β-Galactosidase activity in lacZ-expressing cells remained constant, with no significant changes across the varying levels of oxygen: ⩽ 0.02%, 0.5%, or 2% oxygen compared with 21% oxygen (Figure 2D).
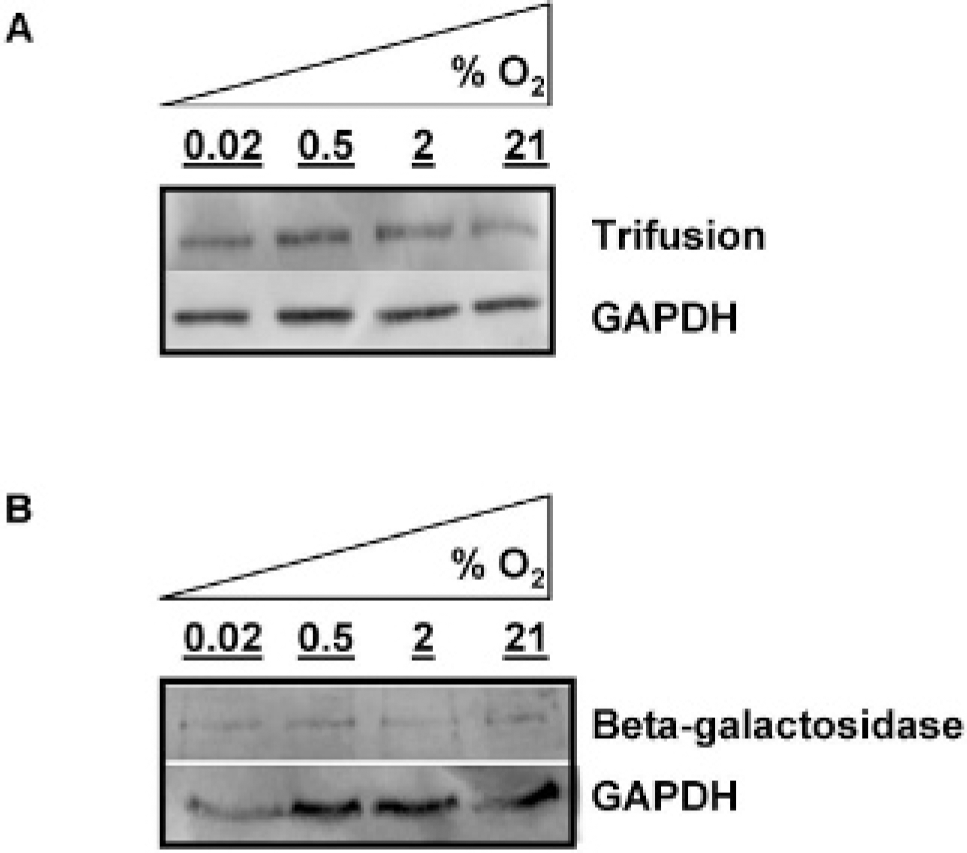
Under decreasing levels of oxygen, production of the trifusion protein and β-galactosidase remained constant in A375 cytomegalovirus-hrl-mrfp-ttk cells and A375/lacZ cells. Western blot of (A) trifusion protein production in A375/hrl-mrfp-ttk cells and (B) β-galactosidase in A375/lacZ cells. Cells were harvested after 24 hours of incubation at ⩽ 0.02, 0.5, 2, or 21% oxygen, as indicated. GAPDH = glyceraldehyde-3-phosphate dehydrogenase.
Oxygen Sensitivity of Renilla Luciferase and mRFP In Vivo
To test Renilla luciferase protein activity in vivo, male athymic nu/nu mice bearing subcutaneous tumor-forming melanoma A375/hrl-mrfp-ttk tumors were anesthetized by inhalation with isoflurane and positioned side by side in the light-tight chamber of the IVIS 200 imaging system (Figure 3A). Photon release is indicated in the panel on the left following the administration of coelenterazine directly into tumors A and B. Three minutes following the administration of substrate, the tumor on mouse B was clamped to occlude blood flow and tumor oxygen delivery, significantly abrogating photon release in the clamped tumor compared with the unclamped tumor. When coelenterazine was reintroduced to both tumors, photon release increased as expected in the unclamped tumor on mouse A but only marginally in clamped tumor B. Photon production by the luciferase-coelenterazine reaction in the clamped tumor B was recovered following reoxygenation when the clamp was removed. These trends were quantified by region of interest (ROI) analysis and are presented graphically (Figure 3B). Bioluminescence is dramatically decreased in the clamped tumor compared with the unclamped tumor control. The substrate coelenterazine is administered at times 0 and minute 14 following the beginning of the imaging session, as indicated by the downward-facing arrows (see Figure 3B). The clamp was added at 3 minutes following the start of the imaging session. A similar experiment measuring mRFP fluorescence under these conditions demonstrated no change in signal levels before, during, and after clamping (data not shown). Photon production of the coelenterazine-luciferase reaction was abrogated when delivery of oxygen to the tumor was decreased by the administration of the antivascular agent DMXAA (Figure 3C). At the beginning of the experiment, coelenterazine was injected and photon release was monitored over time. After 20 minutes, bioluminescence decreased by 80%, reflective of the rapid metabolism of this substrate by Renilla luciferase. 26 Coelenterazine was again injected into the tumor, whereas DMXAA was administered via an intraperitoneal injection. Coelenterazine was injected two more times at 20-minute intervals following DMXAA administration to the animals to demonstrate the persistent decrease in bioluminescence signal following DMXAA treatment. The peak in bioluminescence signal obtained after each substrate injection decreased approximately 65% within 30 minutes of DMXAA treatment. As with clamping, no change in mRFP fluorescence was observed before and after administration of DMXAA to reporter-expressing tumors (data not shown).

The activity of bioluminescent, fluorescent, and thymidine kinase reporters decreased in vitro despite relatively constant levels of reporter protein production in low oxygen environments, whereas β-galactosidase activity remained constant across all oxygen concentrations, corresponding with a constant level of protein production. A demonstrates densitometric analysis of protein content versus bioluminescence at the concentrations of oxygen indicated on the x-axis. B shows changes in median fluorescence intensity in cells in comparison with protein production. C indicates a decrease in thymidine kinase activity compared with protein expression. D, β-Galactosidase activity is assessed by its ability to cleave its substrate, 7-hydroxy-9H-(1, 3-dichloro-9, 9-dimethylacridin-2-one)-β-galactoside (DDAOG). 7-Hydroxy-9H-(1, 3-dichloro-9, 9-dimethylacridin-2-one (light grey) was quantified at 35 minutes following the exposure of A375/lacZ cells to DDAOG. *p ⩽ .005 compared with reporter activity at 21% oxygen. Error bars represent standard deviation of three independent measures. Units were normalized relative to the 21% O2 measurement within each data set. Dark grey: protein expression; light grey: protein activity.
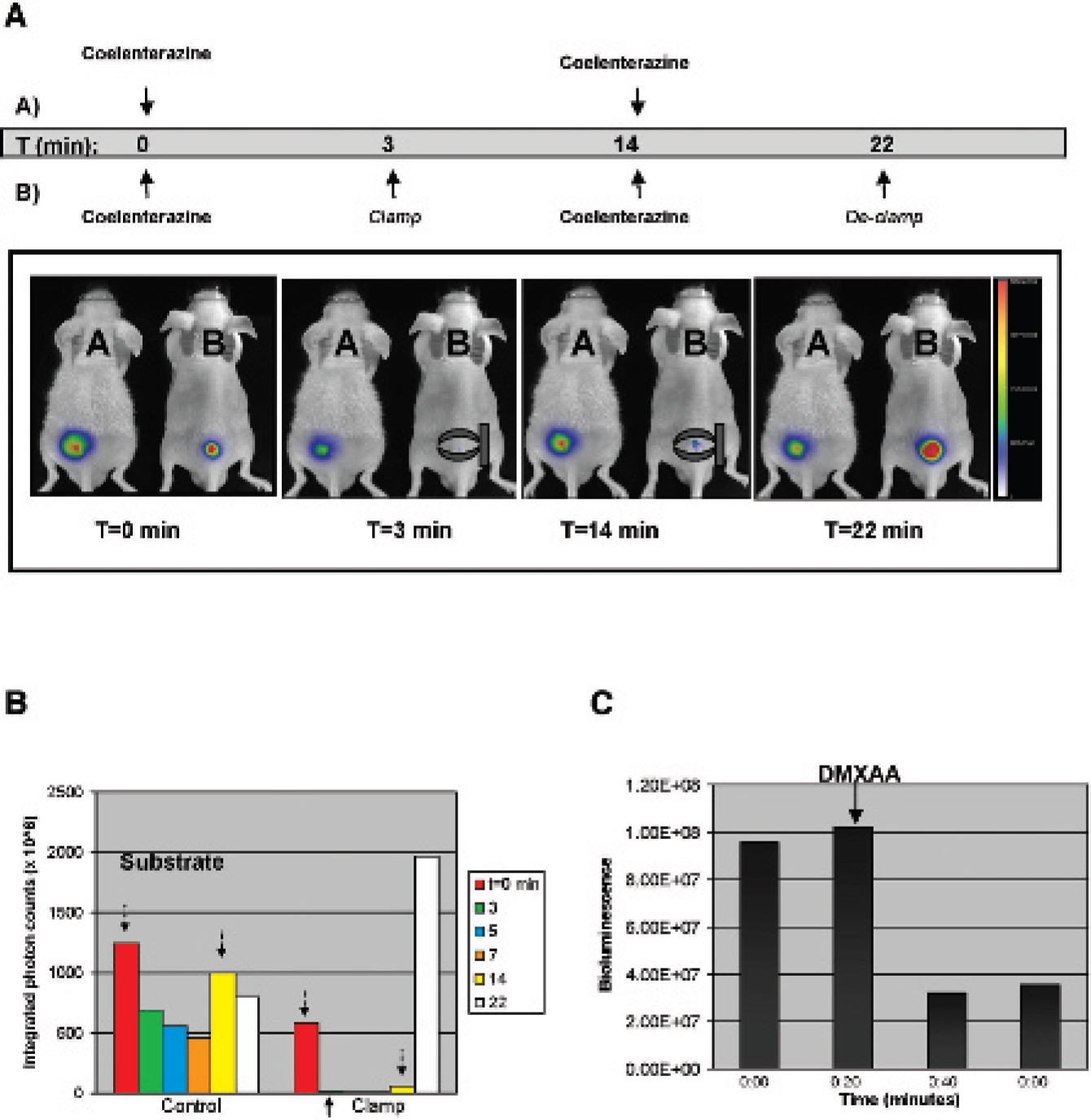
Bioluminescence was abrogated in vivo in tumors that were clamped or treated with the antivascular agent 5, 6-dimethylxanthenone-4-acetic acid (DMXAA). Inhibiting blood and oxygen delivery to tumors significantly abrogated bioluminescence activity. A, From left to right, the four panels show A375 cytomegalovirus-hrl-mrfp-ttk tumors imaged before, during, during with reintroduction of coelenterazine, and postclamping in mouse B, respectively, whereas the tumor on mouse A remained unclamped. The tumor clamp is represented schematically. Three minutes postadministration of coelenterazine, the signal in tumor B was abrogated significantly. Once coelenterazine was reintroduced to the tumor on both mice, at 14 minutes after the initial injection, photon release from the unclamped tumor on mouse A increased as expected, whereas the signal in the clamped tumor of mouse B did not. Following 1-minute acquisition, the clamp was removed with a significant increase in photon release in the tumor on mouse B. The bar on the right of each figure indicates photon/second/cm2, with a range of 0 to 2.25 × 108. B, This bar graph summarizes a quantitative region of interest analysis of signal detected from the tumors. Intratumoral administration of coelenterazine substrate is indicated by the downward-facing arrows. Temporarily occluding tumor blood flow by clamping the skin surrounding a subcutaneous tumor decreases photon production by the hrl-coelenterazine reaction. C, Photon production from the hrl-coelenterazine reaction was inhibited following the administration of the antivascular agent DMXAA. Even with repeated coelenterazine injections at intervals of 20 minutes, photon release was abrogated.
Oxygen Sensitivity of β-Galactosidase Activity In Vivo
The filter sets used to detect uncleaved DDAOG and its cleaved product DDAO are outlined schematically in Figure 4A. Decreasing blood and oxygen delivery to the A375/lacZ tumor using a tumor clamp did not affect the ability of β-galactosidase to cleave the optical imaging probe (Figure 4B). ROI analysis using the formula [DDAO]/[DDAOG + DDAO] performed using CMIR_Image revealed no difference in the fraction of DDAOG cleaved to DDAO between clamped and unclamped tumors.
Discussion
In this report, we assessed the activity of four reporter proteins relative to their production in cells incubated in low oxygen atmospheres to determine which protein may be most useful for detecting tumor hypoxic regions using molecular imaging techniques. It was imperative to compare the activity of reporter constructs containing genes driven by the same promoter as the transcriptional control of promoters may be differentially affected in low oxygen environments. For example, hypoxia leads to the activation of proteins such as nuclear factor κB (NF-κB), potentially leading to the increased transcription of reporter genes driven by the CMV promoter containing three NF-κB consensus binding sites.27,28 Although this may lead to an indirect increase in the level of protein produced in these cells, our studies were not focused on the transcriptional control under hypoxia but instead on how well the activity of the reporter protein is maintained in low oxygen environments.
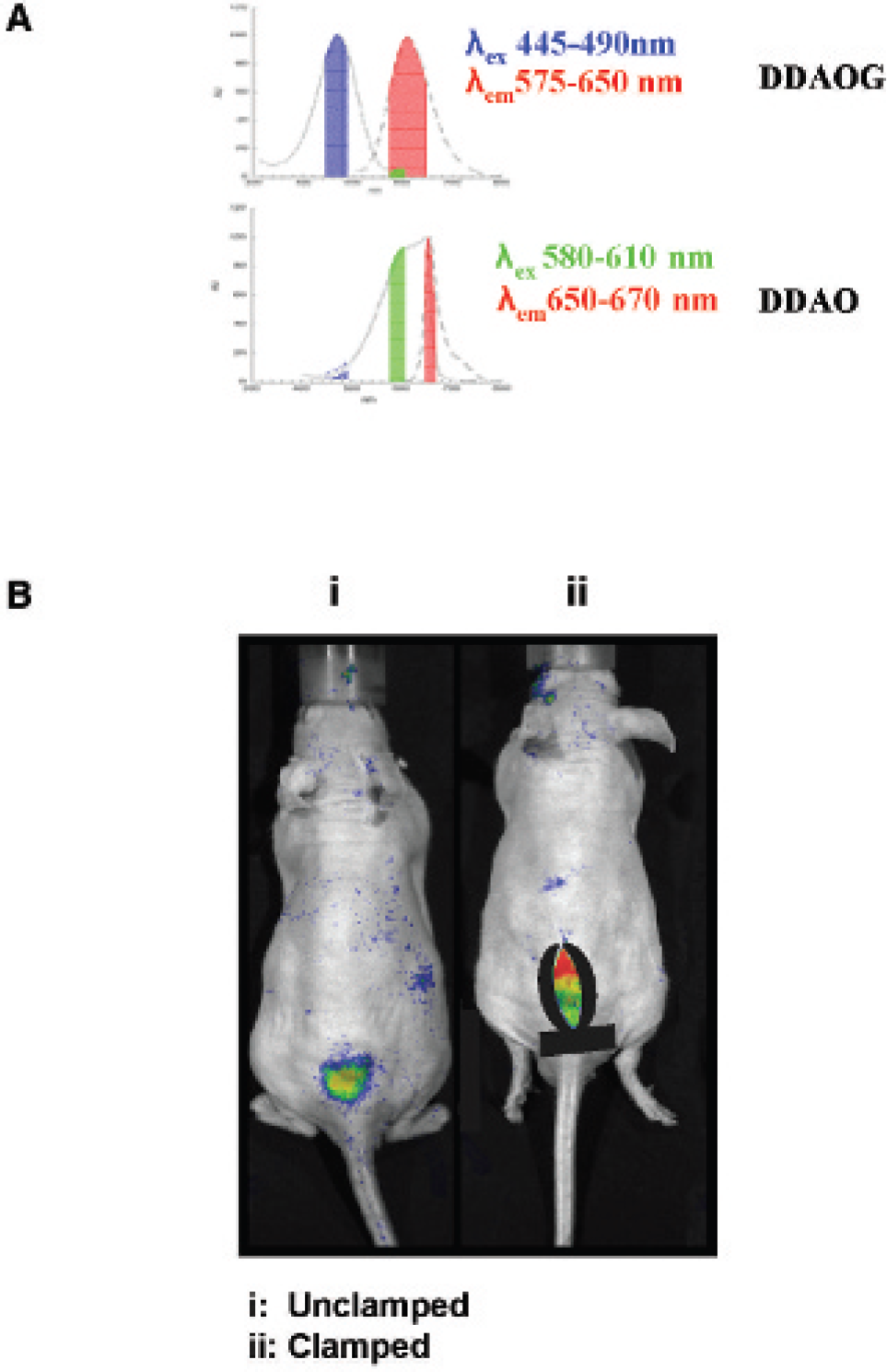
The cleavage of 7-hydroxy-9H-(1, 3-dichloro-9, 9-dimethylacridin-2-one)-β-galactoside (DDAOG) to 7-hydroxy-9H-(1, 3-dichloro-9, 9-dimethylacridin-2-one) (DDAO) by β-galactosidase is preserved after clamping of A375/lacZ tumors. A, A description of the excitation and emission wavelength filter sets used on the Xenogen IVIS 200 to detect DDAOG and its cleaved product, DDAO. In B, mouse i was left unclamped, whereas the tumor on mouse ii was clamped; DDAOG was then administered directly into both tumors, and the images shown were acquired at 5-minute intervals following the DDAOG injections. The amount of cleaved DDAOG relative to the total probe was assessed on a per-pixel basis with CMIR_Image analysis using the formula DDAO/(DDAOG + DDAO).
In cells stably expressing the trifusion reporter construct hrl-mrfp-ttk, a decrease in bioluminescence was observed with decreasing oxygen concentrations, although the amount of protein produced did not change. The Renilla luciferase used in this study catalyzes the oxidation of coelenterazine to coelenteramide, in the process generating carbon dioxide and a photon. Although it is functionally similar to firefly luciferase, the Renilla luciferase–coelenterazine reaction does not require ATP for the generation of photons. Therefore, the lack of ATP at low oxygen concentrations was dismissed as the cause of our observation. The oxygen dependence of this reaction is not an obstacle in assays in which a sample expressing hrl as a reporter gene can be removed from its native environment and allowed to reoxygenate. However, this is not the case for reporter genes intended for use in noninvasive live animal imaging. Therefore, great care was taken to minimize exposure of the samples to normal air prior to analysis. In situations in which tissue oxygen concentrations may be varying, particularly in tumors with spatially and temporally heterogeneous vascular distributions, relating signal obtained from a luciferase reporter gene to gene expression may be difficult. This is evidenced by the lack of correlation between reporter protein levels and bioluminescent activity when the oxygenation of the cell samples was varied (see Figure 2).
The data presented in this report suggest that the fluorescence of mRFP is compromised in chronic low oxygen environments. Although the fluorescence in the cells used in this report does not completely diminish under nearanoxic conditions, it is dimmed compared with that observed in air. Unlike luciferase activity in which, owing to the chemistry of the luciferase-coelenterazine reaction, one can expect to observe a drop in photon production with a decrease in oxygen, fluorescence depends on oxygen-dependent post-translational maturation of a fluorophore. A fluorescent protein requires just one molecule of oxygen to become an active fluorophore, after which it may generate many fluorescent photons with appropriate excitation. The oxygen requirement for fluorescent proteins is therefore considerably less stringent than that of bioluminescent enzymes, where one molecule of oxygen is needed for each photon generated. This difference in oxygen requirements is supported by the observed dependence of reporter fluorescence and bioluminescence on oxygen. It will be of critical importance to assess the significance of this loss in an in vivo tumor model when this construct is driven by a hypoxia-responsive promoter.
To this point, the behavior of TK in low oxygen environments has not yet been assessed. The data presented in this report suggest that TK activity is compromised when cells expressing a TK reporter gene are grown under hypoxic conditions. This may be dismissed as a bystander effect of low ATP levels at nearanoxic conditions since TK activity depends on the presence of ATP. However, ATP is incorporated in the TK activity assay, a fact that perhaps rejects this as a reason for our observation. Further study will be required to assess the mechanism and significance of the loss of TK reporter activity in hypoxic environments.
Our live animal optical imaging study demonstrates the dependence of Renilla luciferase activity to oxygen for photon production. Comparing mice bearing the subcutaneous A375/hrl-mrfp-ttk tumor, we demonstrate that acute abrogation of blood flow and tumor oxygen delivery by clamping tumor-feeding vessels significantly hampers photon release, whereas high activity remains in the undisturbed control tumor. Tumor clamping is an established method of inducing acute hypoxia in tumors. 29 To dismiss the possibility that our observation was due to an acute halt in the vascular delivery of coelenterazine to the tumor once the clamp was in place, substrate was introduced directly into both tumors rather than the usual intravenous route of administration. Despite the direct delivery of coelenterazine to the clamped tumor, this procedure did not lead to a significant rise in photon emission, presumably because of the lack of oxygen. On releasing the clamp, a large increase in photon emission is observed in the previously hypoxic tumor. This supports the interpretation that during clamping, a pool of coelenterazine had accumulated that was unable to be metabolized owing to a lack of oxygen. On the reintroduction of oxygen with removal of the clamp, the luciferase reporter has access to an excess of substrate and achieves corresponding high levels of photon release. Measurements of mRFP fluorescence over a similar course of clamp application and release demonstrated no significant change in signal during or after clamping, suggesting that mRFP signals are unaffected by such an acute hypoxic challenge. Because of the difficulties of performing PET examinations in a clamp model, we did not evaluate the sensitivity of the TK reporter to changes in oxygenation in vivo. Further, more sophisticated experiments will be required to determine whether the changes in TK activity with oxygen observed in vitro affect its ability to act as a reporter gene for hypoxia-related processes in vivo.
Under normal circumstances, a tumor will contain a heterogeneous distribution of hypoxic regions. For this reason, one may predict that constitutively expressed reporter genes will respond along a gradient of oxygen concentrations, causing some hypoxic regions to exhibit less reporter signal than others. Because the in vivo molecular imaging techniques used to image these reporters have spatial resolutions on the order of 1 mm, the imaging signals detected from reporters will, in general, represent an average over these hypoxic gradients. High-resolution microscopic techniques such as intravital microscopy may be used to examine microscopic variations in oxygenation using reporter approaches. The rapid kinetics of bioluminescent signal changes in response to changes in oxygenation discussed above suggest that constitutively expressed reporter constructs could be used to image regions of acute hypoxia by looking for signal decreases (negative contrast). When using macroscopic imaging methods with constitutively expressed reporters, signal averaging will tend to reduce the impact of misquantitation because of hypoxia-mediated loss of reporter signal. However, hypoxia-induced reporter genes will be especially sensitive to oxygen-mediated loss of reporter activity because of their overexpression in regions of poor oxygenation. This situation encourages rigorous assessment of the quantitative accuracy of hypoxia-driven reporter gene techniques.
The bacterial lacZ gene product β-galactosidase has long been applied as a reporter protein to study gene expression and regulation. Typically, its expression is assessed in reporter assays or detected histologically by staining with 5-bromo-4-chloro-3-indolyl-β-
Recently, several reports have demonstrated the use of reporter genes expressed under hypoxic conditions via promoters containing HREs responsive to HIF-1 to visualize tumor hypoxia by noninvasive imaging.8–11 These studies have used either luciferase alone, fluorescence alone, or bifusion constructs combining fluorescence and PET reporters. It is important to note that the reporting ability of a gene construct under hypoxic conditions is a balance between the amount of protein expression an HRE promoter can achieve and the amount of protein activity that is lost by the reporter product under hypoxia. The development of tumor models expressing the HIF-specific, HRE-driven trifusion reporter construct still remains a key interest as a tool for identifying hypoxia and hypoxia-related physiology in tumors. However, it will be critical to compare signal intensity with other noninvasive forms of assessing hypoxia, such as hypoxia-specific PET tracers, such as, 18F-fluoromisonidazole, 64Cu-diacetyl-bis (N(4)-methylthiosemicarbazone), to rigorously establish the accurate quantitative range of reporter gene measurements.
The data presented highlight the varying dependence on oxygen among the reporter proteins studied. Our findings suggest that β-galactosidase is a more robust reporter under hypoxic conditions compared with Renilla luciferase, red fluorescent protein, and TK in vitro, exhibiting a closer correlation between protein expression and the signal produced. These findings were confirmed in in vivo models of tumor hypoxia for Renilla luciferase, red fluorescent protein, and β-galactosidase. Among the panel of reporters, the oxygen-independent activity of β-galactosidase encourages its application in the development of quantitative measures of tumor hypoxia, in addition to other applications in oxygen-deprived environments such as stroke and myocardial infarction.
