Abstract
To examine inflammatory tissue, an initial and common symptom of various types of pathogenesis, we designed inflammation-targeted T1 contrast agents prepared by bioconjugation of gadolinium diethylenetriaminepentaacetic acid (Gd-DTPA) with anti-intercellular adhesion molecule 1 (ICAM-1) antibody. The anti-ICAM-1 antibody was coupled with DTPA and was then conjugated with Gd. The specific binding of the Gd-DTPA-anti-ICAM-1 antibody complex to the ICAM-1-expressing cells was examined in the cultured endothelial cells where ICAM-1 expression was stimulated. Inflammation-specific T1 imaging was then assessed using a mouse abscess model with the 1.5-Tesla module. The Gd-DTPA-anti-ICAM-1 antibody displayed increased r1, which was two times higher than that of Gd-DTPA and showed predominant binding to cultured endothelial cells, which expressed a high level of ICAM-1. Moreover, the inflammation-specific T1 enhancement was imaged with the Gd-DTPA-anti-ICAM-1 antibody in the mouse acute inflammation model. The Gd-DTPA-anti-ICAM-1 antibody showed significantly increased vascular circulation time, which thereby offered a greater chance for its binding to the target cells. The Gd-DTPA-anti-ICAM-1 antibody displays a potential targeted T1 contrast agent specific to the inflammatory tissue that expresses ICAM-1.
CONTRAST AGENTS FOR MAGNETIC RESONANCE IMAGING (MRI) are mainly chelates of paramagnetic or superparamagnetic cations. As a member of the lanthanide group, stable chelates of gadolinium are widely used as magnetic resonance contrast agents because of their physicochemical properties, seven unpaired electrons, eight or nine coordination sites, and favorable electronic relaxation time.1–8
Targeted molecular MRI was recently developed to monitor the cells for both diagnostic and therapeutic purposes. Imaging of cancers expressing the Her2/neu receptor was attempted by conjugation of avidin-Gd and biotinylated anti-Her2/neu receptor antibody for potential application in the diagnosis of breast cancer. 9 The gadolinium (Gd)-conjugated human β-amyloid peptide derivative has been suggested as enabling in vivo MRI for the early detection of individual amyloid plaques in the brains of possible Alzheimer's disease patients. 10 The blood coagulation-specific T1 contrast agent has also been produced by conjugation of a macrocyclic chelator, 1,4,7,10-tetraazacyclododecane-N,N′,N″,N‴-tetraacetic acid (DOTA), and Gd to the substrate of tissue factor XIII (TF XIII), 11 as well as the Gd-conjugated peptide probe EP-1873, 12 to produce a thrombosis-specific diagnostic paramagnetic contrast agent.
To develop inflammation-specific T1 imaging, we introduced molecular imaging technology involving bioconjugation of Gd-diethylenetriaminepentaacetic acid (DTPA) with an inflammation-specific probe. Otherwise, in MRI of inflammatory tissues with the conventional Gd chelates, such as Gd-DTPA, it is difficult to discriminate inflammatory liver tissues from necrotic liver tumors or liver abscesses.
In vivo visualization of inflammatory blood vessels and activated immune cells involves an enormous physiopathologic importance, including monitoring the onset of pathogenesis and immune activity in tissue, such as infection, neoplasm, artherosclerosis, and ischemia, thereby resulting in the early diagnosis of these diseases even before anatomic changes become evident.13–17
Endothelial cells are closely involved in the inflammation processes as these cells have a central role in leukocyte extravasation, which is a key feature of inflammation. The inflammation process is regulated by various adhesion molecules, such as integrins, selectins, and intercellular adhesion molecule (ICAM), etc., which trigger a series of inflammation reactions on endothelial cells.18–22 Leukocyte-endothelial cell adhesion is regulated by the interactions of receptor-ligand ICAM pairs, such as integrin Mac-1 and ICAM-1. ICAM-1 is also highly expressed in the activated leukocytes and infiltrated macrophages, which interact with the leukocyte function antigen 1 of T lymphocytes. Accordingly, neutrophil trafficking is impaired in ICAM-1-deficient mice and is accompanied by decreased functional responses to both inflammatory and immune stimuli. 23
In this study, to visualize inflammatory tissues on T1 imaging, anti-ICAM-1 antibody was linked to DTPA using a bioconjugation reaction. The DTPA monoclonal antibody was then chelated with GdCl3 to form the inflammation-specific T1 contrast agent Gd-DTPA-anti-ICAM-1 antibody. The Gd-DTPA-anti-ICAM-1 antibody bioconjugate was then tested for its T1 relaxivity, cytotoxicity, and molecular specificity against ICAM-1 expression both in vitro and in vivo. The ICAM-1-expressing endothelial cell-specific targeting by the Gd-DTPA-anti-ICAM-1 antibody was assessed in the mouse endothelial cells stimulated with 2 nM tumor necrosis factor α (TNF-α), a potent inducer of ICAM-1 expression. In the acute inflammatory mouse model generated by injection of Staphylococcus aureus into the mouse thigh muscle, enhancement of the T1 intensity of the inflammatory tissue by the Gd-DTPA-anti-ICAM-1 antibody was compared with that of the control antibody conjugate, Gd-DTPA-immunoglobulin G (IgG).
Materials and Methods
The rat hybridoma for the anti-ICAM-1 antibody was purchased from the American Type Culture Collection (ATCC) and was cultured in Dulbecco's Modified Eagle's Medium with 4 mM
Antibody Purification
The ICAM-1 antibody was purified from the culture supernatant of rat hybridoma using protein A/G-coupled affinity chromatography. The purified antibodies were identified by the bicinchoninic acid (BCA) test, sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE), and Western blot analysis.
Bioconjugation Reaction
Diethylenetriaminepentaacetic acid bisanhydride (DTPABA) was dissolved in dimethylformamide and then added to the antibody solution. The anti-ICAM-1 antibody was conjugated to DTPABA in phosphate-buffered saline (PBS) (pH 8.5) for 1 hour at room temperature. The reaction solution was purified using the PD-10 column (Sephadex G-25M, Amersham Biosciences, Buckinghamshire, UK). The content of the antibodies was measured using the protein assay method.
Gd chloride was dissolved in deionized water and added to the DTPA-antibody conjugate solution. One part of the DTPA-antibody complex was then reacted with 40 parts of Gd chloride in 0.5 M sodium acetate (pH 5.5). The solution was loaded into the PD-10 column and eluted with 0.15 M NaCl (pH 5.5). Gd-DTPA antibody conjugates were analyzed using Matrix-assisted laser desorption ionization-time of flight (MALDI-TOF) (Voyager-DE STR 4349) spectrometry. The Gd content was determined by inductively coupled plasma mass spectrometry (ICP-MS) (Agilent 7500A, Agilent Technologies, Palo Alto, CA), and the final concentration of the conjugated antibody was measured using BCA analysis. The molar ratio of gadolinium and antibody was then calculated.
Cytotoxicity Test
Subconfluent cells were incubated with various concentrations of Gd-conjugated IgG or with anti-ICAM-1 antibody at 37°C for 12 hours. The cells were stained with Coomassie Brilliant Blue R-250 0.5% (w/v) in 45% (v/v) methanol and 10% (v/v) acetic acid for 5 minutes after washing three times with PBS buffer. The stained cells were measured by A450 in a plate reader, Spectra Max M2 (Molecular Devices, Sunnyvale, CA).
Determination of Plasma Half-Life
After the Gd-DTPA antibody conjugates (9 nmol of Gd per gram of body weight) were intravenously injected to four BALB/c mice, approximately 50 μL of blood through bleeding from the tip of the tail was time-dependently collected with heparinated tubes for 24 hours. The blood samples were then analyzed with ICP-MS (Agilent 7500A).
Immunoblot Analysis
Cells and inflammatory tissues were lysed and homogenized, respectively, in radioimmunoprecipitation assay (RIPA) buffer containing 1 mM phenylmethylsulfonylfluoride (PMSF), 5 μg/mL leupeptin, and 5 μg/mL aprotinin. The cell lysates and homogenates (25 μg) reconstituted in reducing the Laemmli buffer were subjected to 12% SDS-PAGE and were then transferred to a nitrocellulose membrane. Following blocking with a 5% skim milk solution in tris buffered saline (TBS) at room temperature for 30 minutes, the membrane was incubated with 0.25 ng/mL of an antimouse ICAM-1 antibody (BD Transduction Laboratories, San Jose, CA) or with 1 ng/mL of an anti-β-actin antibody (Santa Cruz Biotechnology, Santa Cruz, CA). These monoclonal antibodies were detected using 0.2 ng/mL of horseradish peroxidase (HRP)-conjugated second antibody and an enhanced chemiluminescence (ECL) kit (Amersham Bioscience) following the manufacturer's instructions.
The purified rat antimouse ICAM-1 antibody fractions from the culture supernatant of the hybridoma were electrophoretically separated and were transferred to a nitrocellulose membrane. The membrane was incubated with 0.2 ng/mL of HRP-conjugated goat antirat IgG, and the antigen-antibody-HRP complexes were also visualized using the ECL.
In Vitro Magnetic Resonance Analysis
The relaxivity of the Gd conjugates from three different preparations in test tubes was determined using a mixed inversion recovery/spin echo (IR/SE) sequence in a 1.5-Tesla clinical magnetic resonance scanner (Gyroscan Integra, Philips Medical System, Eindhoven, The Netherlands) assisted with the knee coil (repetition time [TR]; spin echo (SE)/inversion recovery (IR)/IR del = 920/2300/500, echo time [TE] [multi] = 30, flip angle (FA) = 90, number of signal acquisitions (NSA) = 2, field of view [FOV] = 50 mm). The endothelial cell-specific contrast effect was assessed by MRI of its differential contrast effects on the endothelial cells, MS-1, and the nonendothelial cells, HT1080. Cells (5 × 106) were cultured and incubated with the Gd-DTPA antibody complexes (390 nmol of Gd) in the presence or absence of anti-ICAM-1 antibody (40 nmol) at 4°C for 4 hours. The cells were then washed three times with PBS and were harvested by scraping. The cells were then pelleted by centrifugation. An MRI of the cell-containing tubes placed in a water-filled chamber was obtained using a spin echo sequence with the imaging parameter (TR = 500, TE 15, FOV = 60 mm, FA = 90, slice thickness 2 mm) in the 1.5-Tesla module. The T1 intensity of the cell pellets was normalized against that of the surrounding water. Each experiment was triplicated, and the intensity was shown with mean ± standard error.
In Vivo Magnetic Resonance Analysis
Bacterial inflammation was made in the left thigh of 15 BALB/c mice by injection of 0.1 mL of S. aureus (106 cells) using a 30-gauge needle. After 24 hours, MRI was performed with injection of Gd-DTPA (100 nmol of Gd/g of body weight) and Gd-DTPA antibody (9 nmol of Gd/g of body weight) on five mice with each type of injection. Dynamic T1 imaging was obtained after intravenous injection of the contrast agents. MRI was conducted using a 1.5-Tesla scanner with a 47 mm surface coil. T1-weighted spin echo images (TR/TE = 500/15 milliseconds) were then acquired. The FOV, the image acquisition matrix, and the slice thickness were 60 mm, 224 × 224, and 1.5 mm, respectively.
Immunohistochemical Analysis
After the mice were analyzed on MRI, the inflammatory tissue was collected and the expression of ICAM-1 was then examined using immunohistochemical analysis. The tissue section was fixed with 4% paraformaldehyde and reacted with the rabbit antimouse ICAM-1 antibody or rat antimouse macrophage-specific antibody, rat anti-mouse F4/80 antibody for 2 hours at 37°C. The primary antibody bound to the ICAM-1-expressing cells was then visualized with the peroxidase-conjugated second antibody and the substrate dye. To detect the injected Gd-DTPA-anti-ICAM-1 antibody, the adjacent section was incubated with only the second antibody (goat antirat IgG antibody) for the Gd-conjugated antibody (rat antimouse ICAM-1 antibody) and was detected with the substrate dye.
Analysis of Data
The data are presented as mean ± standard error of the means, and the significance of the data was assessed by paired t-test.
Results
Preparation of Gd-DTPA-Anti-ICAM-1 Antibody and Assessment of Its Cytotoxicity and Plasma Half-Life
Bioconjugation of the anti-ICAM-1 antibody and non-immunized IgG with DTPABA and GdCl3 was characterized using analytic chemistry (Figure 1). The purified antibody fraction was identified by Coomassie blue staining of the electrophoretically separated gel (see Figure 1A). The reduced IgG was separated into heavy (50 kDa) and light (25 kDa) chains. These rat antibody fragments were also identified using Western blot analysis (see Figure 1B). The intermediate product, the DTPA antibody, and the final product, the Gd-DTPA antibody, were quantified by BCA assay for their antibody content and were characterized using MALDI-TOF spectrometry analysis for the conjugated product (see Figure 1C). An increase in the molecular weight represented conjugation of the antibody with DTPA and Gd. Quantitative analysis of the Gd contents in the Gd-DTPA antibody was performed using ICP-MS. The molar ratio of Gd per IgG molecule was 20 ± 3. From the routine conjugation procedure, the DTPA antibody complex was obtained with more than 95% yields and the Gd-DTPA antibody with 80% yields, based on the antibody contents. The Gd-conjugated antibodies did not show any cytotoxic effect on cultured endothelial cells (see Figure 1D). The half-life of the Gd-DTPA antibody conjugate in the body circulation was also measured with ICP-MS from the blood samples collected after injection of the contrast agent. The calculated plasma half-life was 3.6 ± 0.16 hours, which is more than 10 times longer than that of Magnevist (Berlex, Germany) (≈20 minutes) (see Figure 1E).
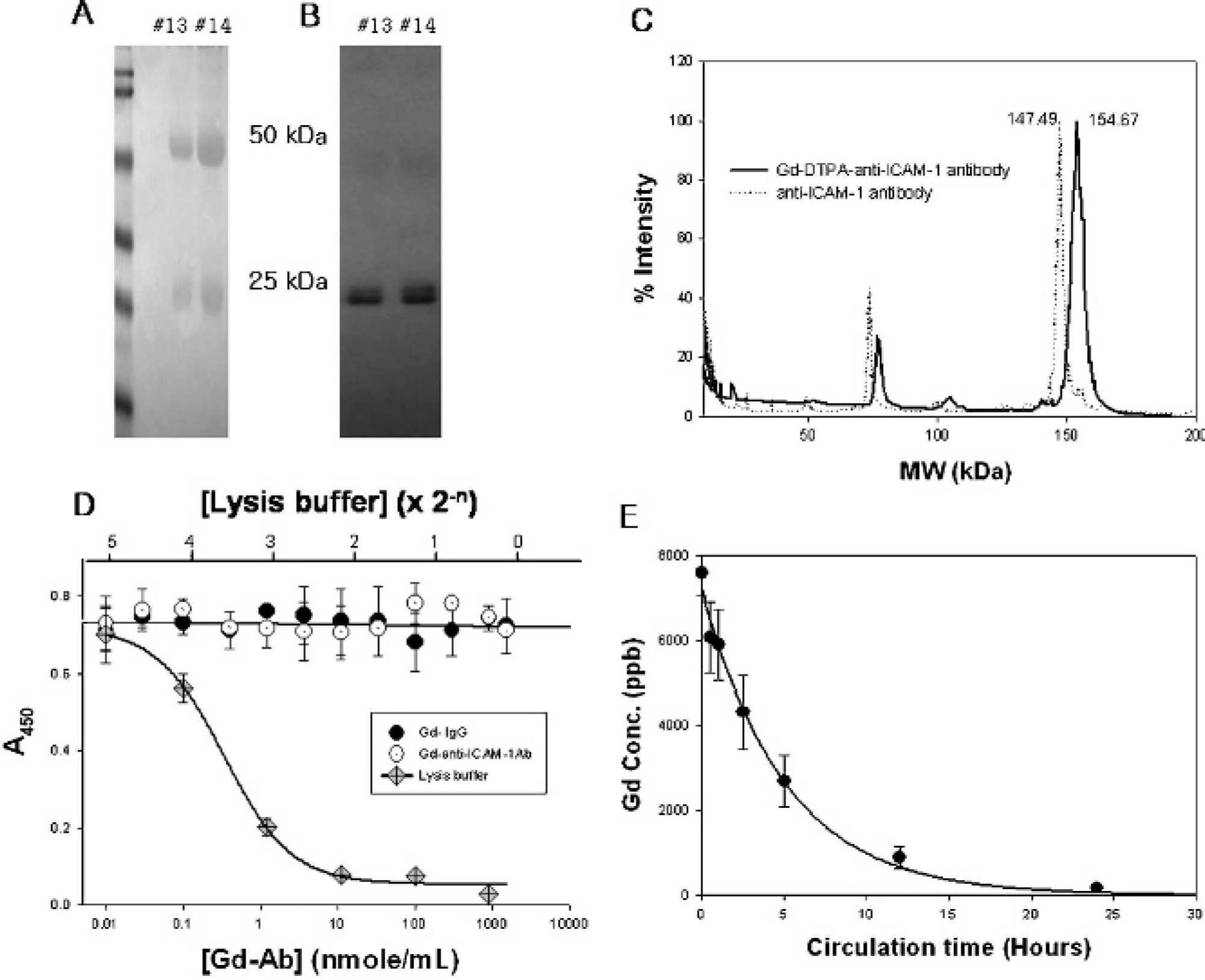
Preparation of gadolinium diethylenetriaminepentaacetic acid (Gd-DTPA)–anti-intercellular adhesion molecule 1 (ICAM-1) antibody and assessment of cytotoxic effects and plasma duration time. A, The purified protein fraction was electrophoretically separated visualizing the heavy (50 kDa) and light (25 kDa) chains of the immunoglobulin G (IgG). B, The antibody was identified by Western blot analysis, which also detected the heavy and light chains. The purified anti-ICAM-1 antibody and its bioconjugates, anti-ICAM-1 antibody-DTPA and anti-ICAM-1 antibody-Gd-DTPA, were analyzed with MALDI-TOF spectrometry to monitor the molecular weight shift of the bioconjugates. C, The molecular weight shift from anti-ICAM-1 to anti-ICAM-1 antibody-Gd-DTPA in MALDI-TOF spectrometry analysis represents conjugation of the antibody with Gd-DTPA. D, The cytotoxic effects of the Gd conjugates were examined in the cultured endothelial cells. E, Plasma circulation time of the Gd-DTPA-anti-ICAM-1 antibody was determined with ICP-MS analysis of blood samples collected from the tail.
Relaxivity of the Gd Conjugates
The relaxivity (mmol−1 sec−1) of the Gd conjugates was measured on an 1.5-Tesla magnetic resonance scanner. The differential r1 of the reactant, intermediates, and final conjugate was the Gd-DTPA antibody > GdCl3 > Gd-DTPA, indicating the potential significance of the Gd-DTPA-anti-ICAM-1 antibody as a T1 contrast agent (Table 1).
Relaxivity of the Gadolinium Conjugates

DTPA = diethylenetriaminepentaacetic acid; Gd = gadolinium; ICAM-1 = intercellular adhesion molecule 1; IgG = immunoglobulin G.
The data were obtained from the three different preparations of the contrast agents and are presented as mean ± SD.
ICAM-1-Specific MRI with Gd-DTPA-Anti-ICAM-1 Antibody in the Cultured Endothelial Cells
To examine whether the Gd-DTPA-anti-ICAM-1 antibody conjugate can bind cells in an ICAM-1-specific manner, the contrast agent was incubated with the MS-1 cells (Figure 2), which were stimulated with 2 nM TNF-α, a potent inducer of ICAM-1 on endothelial cells in the presence or absence of a competitive binding inhibitor, the anti-ICAM-1 antibody. The competitive antibody was added with two times higher concentration than the anti-ICAM-1 antibody content of the Gd-DTPA-anti-ICAM-1 antibody complex. As the contrast agent was treated to the cells at 4°C to avoid nonspecific phagocytosis of the contrast agent, the results represent pure detection of the cell-surface ICAM-1 by the contrast agent.

Intercellular adhesion molecule 1 (ICAM-1)-positive endothelial cell-specific binding of the gadolinium diethylenetriaminepentaacetic acid (Gd-DTPA)–anti-ICAM-1 antibody (Ab). A, The induction of ICAM-1 in the endothelial cell line (MS-1) and nonendothelial fibrosarcoma cell line (HT1080) by 2 nM of tumor necrosis factor α (TNF-α) was identified by Western blot analysis. B, Both TNF-α-stimulated MS-1 and HT1080 were incubated with Gd-DTPA-ICAM-1 antibody, and the cell-bound Gd-chelate was T1 imaged after washing and centrifugation. The relative contrast to noise ratio (CNR) of the T1 intensity was calculated against the intensity of surrounding water. C, Induction of ICAM-1 by TNF-α resulted in high T1 intensity by the Gd-DTPA-anti-ICAM-1 antibody. *p < .05; the binding was significantly inhibited by anti-ICAM-1 antibody (n = 3).
The treatment of TNF-α, which did not affect the cell population, stimulated expression of the ICAM-1 level in the MS-1 cells. Immunoblot analysis showed induction of the ICAM-1 level in MS-1 cells but not in HT1080 (see Figure 2A). The relative contrast to noise ratio (CNR), calculated as {(region of interest [ROI] cell )-(ROI water )}/(ROI water ), shows a significant increase in the T1 signal intensity in the TNF-α-treated MS-1 but neither the untreated MS-1, the HT1080, nor the TNF-α-treated HT1080 showed a meaningful contrast-enhanced T1 intensity (see Figure 2, B and C). Moreover, binding of the Gd-DTPA-anti-ICAM-1 antibody complex to the ICAM-1-expressing cells was significantly inhibited by the anti-ICAM-1 antibody. The T1 enhancement in the ICAM-1-expressing MS-1 cells by Gd-DTPA-anti-ICAM-1 antibody in vitro implies the potential significance of the contrast agent as an inflammation-targeted T1 contrast agent in vivo.
Inflammation-Specific MRI with Gd-DTPA-Anti-ICAM-1 Antibody in a Mouse Model
The inflammation-targeted MRI with Gd-DTPA-anti-ICAM-1 antibody was examined in the inflammation-induced mouse model (Figure 3). The quick T1 enhancement by Gd-DTPA appeared within 1 minute after injection and completely disappeared 1 hour later (see Figure 3A). However, the Gd-DTPA-anti-ICAM-1 antibody displayed significant signal enhancement and accumulation in the inflammatory tissues until 24 hours after the contrast agent injection. The Gd-DTPA-IgG, the Gd-conjugated control antibody with the same amount of Gd as the Gd-DTPA-anti-ICAM-1 antibody, showed no significant enhancement in T1 until 24 hours after injection (see Figure 3B). Interestingly, even though the Gd content of the Gd-DTPA-anti-ICAM-1 antibody was almost 10 times lower than that of Gd-DTPA, a significant amount of T1 enhancement by the Gd-DTPA-anti-ICAM-1 antibody was observed (see Figure 3C). The CNR of dynamic T1 by Gd-DTPA and Gd-DTPA-anti-ICAM-1 antibodies is summarized in Figure 3D and shows the completely different pattern of signal enhancement in these two contrast agents. The T1 enhancement pattern of the contrast agents in individual mice was similar, although the size of the inflammatory area varied, as shown in Figure 3D. We also monitored long-term changes of the inflammatory T1 enhancement by the Gd-DTPA-anti-ICAM-1 antibody until 11 days after injection. The CNR of the inflammatory tissue was slowly decreased with a sigmoidal decay manner (Figure 3D, inset).

Inflammation-specific T1 imaging by gadolinium diethylenetriaminepentaacetic acid (Gd-DTPA)–anti-intercellular adhesion molecule 1 (ICAM-1) antibody. Dynamic T1 imaging of the inflammatory tissue was monitored before and after intravenous Gd-DTPA injection (2.5 μmol of Gd per mouse), Gd-DTPA-immunoglobulin G (IgG) (225 nmol of Gd per mouse), and Gd-DTPA-anti-ICAM-1 antibody (225 nmoles of Gd per mouse). A, T1 intensity in the inflammatory tissue was quickly increased by Gd-DTPA but disappeared within 1 hour. B, No significant T1 enhancement was observed after Gd-DTPA-IgG administration. C, The T1 signal intensity in the inflammatory area gradually increased and accumulated by the Gd-DTPA-anti-ICAM-1 antibody for 24 hours, resulting in significant T1 targeting of the inflammatory tissue. D, The dynamic enhancement by the Gd-DTPA-anti-ICAM-1 antibody (triangles) was compared with enhancement of Gd-DTPA (closed circles) and Gd-DTPA-IgG (open circles) according to the pattern of the contrast to noise ratio (CNR) changes. Inset, CNR changes in the inflammatory tissue of mice injected with Gd-DTPA-anti-ICAM-1 antibody were monitored until 11 days after injection. *p < .01, significantly different T1 enhancement (n = 5).
Expression of ICAM-1 in the Bacteria-Induced Inflammatory Tissues
The inflammatory tissue induced by the administration of bacteria was also examined using immunoblot and immunohistochemical analysis for ICAM-1 expression to assess whether the inflammation-specific imaging by Gd-DTPA-anti-ICAM-1 antibody corresponds to the ICAM-1 expression in this tissue (Figure 4). The expression of ICAM-1 was extremely high in the inflammatory area, where ICAM-1-positive monocytes had largely infiltrated (see Figure 4, A and B). Moreover, the injected Gd-DTPA-anti-ICAM-1 antibody was also detected in the endothelial cells and in the infiltrated macrophages in the inflammatory tissues. Anti-F4/80 antibody was used to identify the macrophages in the inflammatory tissue. The immunostaining showed that infiltrated macrophages were primary in the inflammatory area (see Figure 4D). The ICAM-1 expression was detected by Western analysis in the tissue homogenates 12 to 24 hours after S. aureus was inoculated, suggesting that the ICAM-1-dependent T1 enhancement of the Gd-DTPA-anti-ICAM-1 antibody, which was injected 24 hours after the bacterial administration, corresponds with the time-dependent expression of ICAM-1 (see Figure 4E).

Histologic examination of intercellular adheison molecule 1 (ICAM-1) expression in the inflammatory tissue. The ICAM-1 expression level in the inflammatory tissue was assessed by immunohistochemical analysis. A, Histologic examination of the inflammatory area (I) of the muscle tissue (M) by hematoxylin-eosin staining shows that inflammation was generated in the muscle tissue and that a number of monocytes had infiltrated there. B, The immunohistochemical staining of the adjacent tissue section with antimouse ICAM-1 antibody reveals that ICAM-1 is highly expressed in the inflammatory tissue. C, The injected gadolinium diethylenetriaminepentaacetic acid (Gd-DTPA)–anti-ICAM-1 antibody was also identified with its second antibody-coupled dye. D, The infiltrated macrophages in the inflammatory area were stained with anti-F4/80 antibody (X 100 original magnification). Staining with a control nonspecific second antibody did not show any significant staining (data not shown). E, The tissue homogenates at an indicated time after the bacterial abscess were analyzed with immunoblot for time-dependent ICAM-1 expression.
Discussion
Inflammatory tissue-specific targeting by the magnetic resonance contrast agents has been constantly demanded owing to its versatility to efficiently examine the initial progress of disease. The essential technical element needed to develop cell- or tissue-specific targeted magnetic resonance contrast agents is the bioconjugation of the probe molecule with paramagnetic or superparamagnetic cations.7,24 Targeted contrast agents for both T1 and T2 imaging must be developed owing to their clinically potential significance in providing as much information as possible for patient diagnosis.
In terms of the sensitivity of magnetic resonance contrast agents, it is important to increase their concentration on the target sites to achieve significant signal enhancement. A variety of conjugation strategies for the high conjugation ratio of moles of Gd/mol of the probe molecule have been investigated to generate a sufficient T1 signal.25–29 Our results show that Gd(III) is conjugated with a high content using a simple chelating agent, DTPA, in the high molecular weight probe, the IgG molecule, which contains more than 100 amine side chains. Interestingly, bioconjugation of the Gd-DTPA with the high molecular weight IgG molecule (150 kDa) accomplishes reasonable size control of the contrast agent to achieve a long life span of plasma circulation, thereby resulting in successful delivery of the contrast agent to the target tissues. Therefore, Gd(III), which is small in size, is considered to be a useful lanthanide cation for the preparation of targeted molecular T1 contrast agents, especially for the high molecular weight immunoglobulin molecules, because the superparamagnetic iron oxide (SPIO)- or ultrasmall SPIO-based magnetic resonance contrast agents may be too large to be conjugated with the antibody, thereby resulting in an unstable complex and a low conjugation ratio of the antibody to the contrast agent.
The avidin/biotin-mediated conjugation was widely introduced to crosslink Gd and contrast agents 30 because it is easy to conjugate the reporter and probe molecules and because the direct labeling of immunoglobulins with Gd-DTPA has been shown to be insufficient to achieve a T1 contrast effect. 31 However, in terms of the clinical application of contrast agents, biotin and avidin are expected to cause potentially unfavorable effects, such as difficulties in humanization and tissue clearance of the avidin from the patient's body. Therefore, we determined the reaction conditions for direct conjugation between Gd-DTPA and antibody featuring size regulation for the long circulation and regulation of the Gd-DTPA conjugation rate for the T1 signal and the probe activity. Our results suggest that direct conjugation of the antibody and Gd-DTPA display enough contrast effect to produce target-specific T1 imaging when the conjugation reaction was controlled to obtain a reasonable conjugation ratio of the Gd to the antibody. Direct incorporation of the antibody and DTPA can reduce the cost to produce contrast agents without using expensive materials, such as avidin or streptavidin, which, if used, require even more antibodies for the additional conjugation reaction (antibody/avidin or antibody/biotin). It has been reported that the functionally effective conjugation ratio of Gd/IgG is less than 37 (mol/mol), without compromising the affinity of the antibody to the antigen, although the Gd concentration of the antibody conjugates is important for the sensitivity of magnetic resonance. 32
In addition, another important factor in increasing the sensitivity of molecular contrast agents is enhancement of their relaxivity to visualize the target tissue from the surrounding water-containing environment. Conjugation of the paramagnetic Gd(III) with the heavy molecular weight complexes increases the r1 of the local water protons and therefore provides enhanced T1 contrast.2,6,33 We also found that the higher the conjugation ratio (Gd/antibody) achieved, the greater the relaxivity (R1) of the contrast agents (data not shown).
However, there are still several substantial problems to be resolved with the antibody-conjugated contrast agents. First, immunoglobulin can cause an unfavorable immune response from the injected animals or patients. To reduce the host immune reaction to the contrast agents, we must modify the immunoglobulin by converting it to F(ab'), although we also observed inflammation-targeted imaging with the Gd-DTPA-anti-ICAM-1 antibody in both immunodeficient mice and BALB/c mice (data not shown).
Our results show that the Gd-DTPA-anti-ICAM-1 antibody complex appears to specifically bind the ICAM-1-expressing endothelial cells, which are the typical characteristics of inflammation, thereby suggesting that the Gd-DTPA-anti-ICAM-1 antibody is a useful molecular T1 contrast agent for inflammatory tissue. In addition, the high molecular weight protein, immunoglobulin conjugated with Gd-DTPA, enables the contrast agent to increase its plasma half-life, which is 10 times longer than that of Gd-DTPA (≈20 min), 34 thereby increasing its chance to bind the target tissue-expressing antigen. Moreover, the high relaxivity of the Gd-DTPA-anti-ICAM-1 antibody exhibits significant potential as a T1 contrast agent. It was also confirmed that unlike Gd-DTPA, the Gd-DTPA-anti-ICAM-1 antibody displays a significant T1 enhancement, even with 10-fold less Gd content than that of Gd-DTPA, in an acute inflammatory mouse model. The low dose of the contrast agent may constitute a distinct clinical advantage for patients. We suggest that this ICAM-1-specific T1 imaging would be used in various inflammatory disease models, such as brain abscess models or rheumatoid arthritis models in mice.
In conclusion, we produced the inflammatory cell-targeted T1 contrast agent with anti-ICAM-1 antibody, which was bioconjugated with Gd. This molecular contrast agent provided ICAM-1-positive endothelial cell-specific T1 imaging in vitro. Moreover, Gd-DTPA-anti-ICAM-1 antibody complexes notably displayed inflammation-specific MRI in the mouse abscess model.
Footnotes
Acknowledgment
We would like to thank Bonnie Hami, MA, for her comments and editing.
