Abstract
Hereditary angioedema (HAE) is a rare disorder resulting from decreased functional levels of C1 -inhibitor (C1-INH), which manifests as periodic episodes of localized edema which can be extremely painful, debilitating and even fatal if the swelling affects the larynx. HAE can complicate many aspects of obstetric/gynecologic care, and an awareness of the disease is critical for clinicians involved in the care of women because of potential HAE-related complications pertaining to pregnancy, labor and delivery, and other women's health issues. This article provides a review of published literature specific to HAE and its management in female patients, including important concerns regarding obstetric/gynecologic care. A growing body of relevant experience is presented to help guide the care of women with HAE.
Hereditary angioedema (HAE) is a rare, autosomal dominant disorder that is characterized by attacks of swelling, usually of the extremities or abdomen but sometimes of the genitalia, trunk, face or larynx [1,2]. While obstetricians/gynecologists would not typically be involved in the direct management of HAE itself, an awareness of the disease is critical because of potential complications related to contraception, pregnancy, labor and delivery, and other women's health issues. Furthermore, some HAE treatments are contraindicated in pregnancy. Following a brief overview of HAE and its treatment in general, this paper will review currently available literature specific to HAE and its management in women, including important concerns regarding obstetric/gynecologic care.
Pathophysiology of HAE
Most cases of HAE are caused by any one of numerous genetic defects that lead to decreased functional levels of C1-INH [3]. In the contact system, activated factor XII (factor XIIa) converts prekallikrein to kallikrein, which in turn cleaves off the small peptide bradykinin from high molecular weight kininogen; bradykinin is a potent inducer of vasodilation and vascular permeability (Figure 1) [4]. C1-INH inhibits factors XII and XIIa and kallikrein, thus regulating the production of bradykinin [3]. Lack of functional C1-INH results in excessive production of bradykinin, which is primarily responsible for the angioedema symptoms of HAE [3].

There are two subtypes of HAE that result from low functional C1-INH activity (C1-INH-HAE). Type I (85% of C1-INH-HAE cases) results from mutations that reduce or prevent secretion of C1-INH from the mutated allele [1]. Type II (15% of C1-INH-HAE cases) results from mutations that cause production of a nonfunctional form of C1-INH from the mutated allele [1]. These two types of HAE are clinically indistinguishable [1].
HAE also occurs in individuals with normal C1-INH (HAE-nC1-INH) activity [5,6], a phenomenon first described in the 1980s [7]. This type (formerly called ‘type III’ HAE) is less common than C1-INH-HAE and affects women predominantly, but not exclusively [8,9]. In contrast, C1-INH-HAE generally affects men and women in similar proportions. Within the subtype HAE-nC1-INH, some cases are associated with mutations in the gene that codifies factor XII (HAE-FXII), although the pathophysiologic importance of this mutation is not well understood [9–11]. In other cases of HAE-nC1-INH, the underlying genetic defect cannot be identified, leading to the designation of HAE of unknown origin (U-HAE). Exacerbation from estrogen exposure is particularly common in HAE-nC1-INH [7]. In most, but not all, patients with HAE-nC1-INH, disease severity is exacerbated by increased exposure to estrogens, such as during pregnancy or while taking exogenous estrogen-containing therapies [9,12–14].
Female sex hormones appear to play an important role in the onset of HAE and the frequency of attacks:
Women have more frequent attacks of HAE than men [2]; Women often have more severe HAE symptoms than men and require hospitalization for attacks more often [15]; In a survey of 150 women with HAE, 62% reported that the disease worsened during puberty and 80% that it worsened while they were taking estrogen-containing oral contraceptives [16]. Menstruation and ovulation have also been found to trigger HAE attacks, and use of progestin-only OCs has been reported to reduce the frequency of attacks [16]; Initiation of estrogen replacement during menopause has been reported to exacerbate HAE [17]; A high proportion of women who have HAE-nC1-INH report that their symptoms began when they initiated estrogen-containing OCs or became pregnant for the first time, or that their symptoms are exacerbated by OC use or pregnancy [12]. In 29% of patients, symptoms occur only during periods of increased exposure to estrogen [12]; Women with C1-INH-HAE may have a higher risk of polycystic ovary syndrome [15].
The role of female sex hormones in HAE is not fully understood, although various physiologic associations have been observed, particularly with regard to the kallikrein–kinin system. Women treated with estrogens exhibit increased plasma levels of coagulation factor XII, bradykinin and kallikrein [18–22], likely due to increased transcription [23–25]. Estrogen has also been shown in animal studies to regulate bradykinin B2-receptor gene expression and function [26].
Clinical presentation of HAE
The symptoms of HAE result from extravasation of plasma into the deep layers of skin or mucous membranes [27]. The majority of attacks occur in the skin, particularly the skin of the extremities or in the GI tract [2]. Attacks affecting the face (Figure 2) or genitals are less common, but at least two-thirds of patients have experienced them at some point in their lives [2]. The skin swelling of HAE is nonpitting and occurs without urticaria or pruritus but may be accompanied by a characteristic rash called erythema marginatum. Because many physicians are unfamiliar with HAE, years may pass between a patient's first HAE symptom and a correct diagnosis of the disease [28].

Hereditary angioedema patient (A) experiencing a facial attack and (B) under normal circumstances.
Most individuals with C1-INH-HAE experience frequent abdominal attacks characterized by severe crampy or colicky pain, sometimes accompanied by distention, nausea, vomiting or diarrhea [27,30]. If untreated, attacks can last for 2–5 days and hospitalization occasionally is necessary [30]. In as many as 4.4% of abdominal attacks, fluid shifts result in circulatory collapse and shock, sometimes leading to loss of consciousness [30]. Misdiagnosis of abdominal attacks of C1-INH-HAE has led to unnecessary surgery in as many as 20% of patients with C1-INH-HAE [28]. Laryngeal edema, which can be fatal, occurs in fewer than 1% of C1-INH-HAE attacks but about half of all patients experience a laryngeal attack at some time [2,31].
Patients who have HAE-nC1-INH have a later mean age of symptom onset (30 vs 14 years), more frequent swelling in the larynx, lips, face and tongue, and less frequent swelling in the abdomen compared with C1-INH-HAE patients [32,33].
In addition to exposure to estrogen, triggers of HAE attacks include trauma, such as injury, dental work or medical procedures; repetitive work, such as typing or yard work; sexual activity; emotional stress; infections; and the use of medications such as angiotensin-converting enzyme inhibitors that decreased degradation of bradykinin [34]. However, most attacks occur without an identifiable trigger [1].
Treatment options
Three treatment strategies are commonly used for HAE: on-demand (acute) therapy given at the onset of swelling to terminate attacks; long-term prophylaxis for preventive care; and, in some cases, short-term prophylaxis before medical or dental procedures or other known triggers [35]. It is important to recognize that traditional interventions for histamine-mediated (allergic) angioedema such as corticosteroids, antihistamines and epinephrine are not effective in HAE.
Several HAE-specific agents are currently available, including attenuated androgens, plasma-derived C1-INH concentrate (pdC1-INH; Berinert®/CSL Behring GmBH; Cinryze®/Shire ViroPharma), ecallantide (Kalbitor®/Dyax Corp.), icatibant (Firazyr®/Shire) and recombinant C1-INH (rhC1-INH/Ruconest®; Pharming Group NV). Regional availability, regulatory approvals and/or cost barriers may prevent access to some of these medications, in which case other less proven therapies may have a role, including antifibrinolytics, and fresh frozen plasma (Table 1). Attenuated androgens such as danazol and stanozolol have been used for decades for HAE prophylaxis; their benefits include oral administration, good efficacy in most patients and low cost. However, androgens are not effective for alleviating attacks and have potentially undesirable side effects in female patients, including weight gain, virilization, acne and menstrual irregularities [36]. With the exception of attenuated androgens, no gender differences in the safety/tolerability or efficacy of agents used for the treatment of HAE have been reported.
Drugs/products used in the treatment of hereditary angioedema.
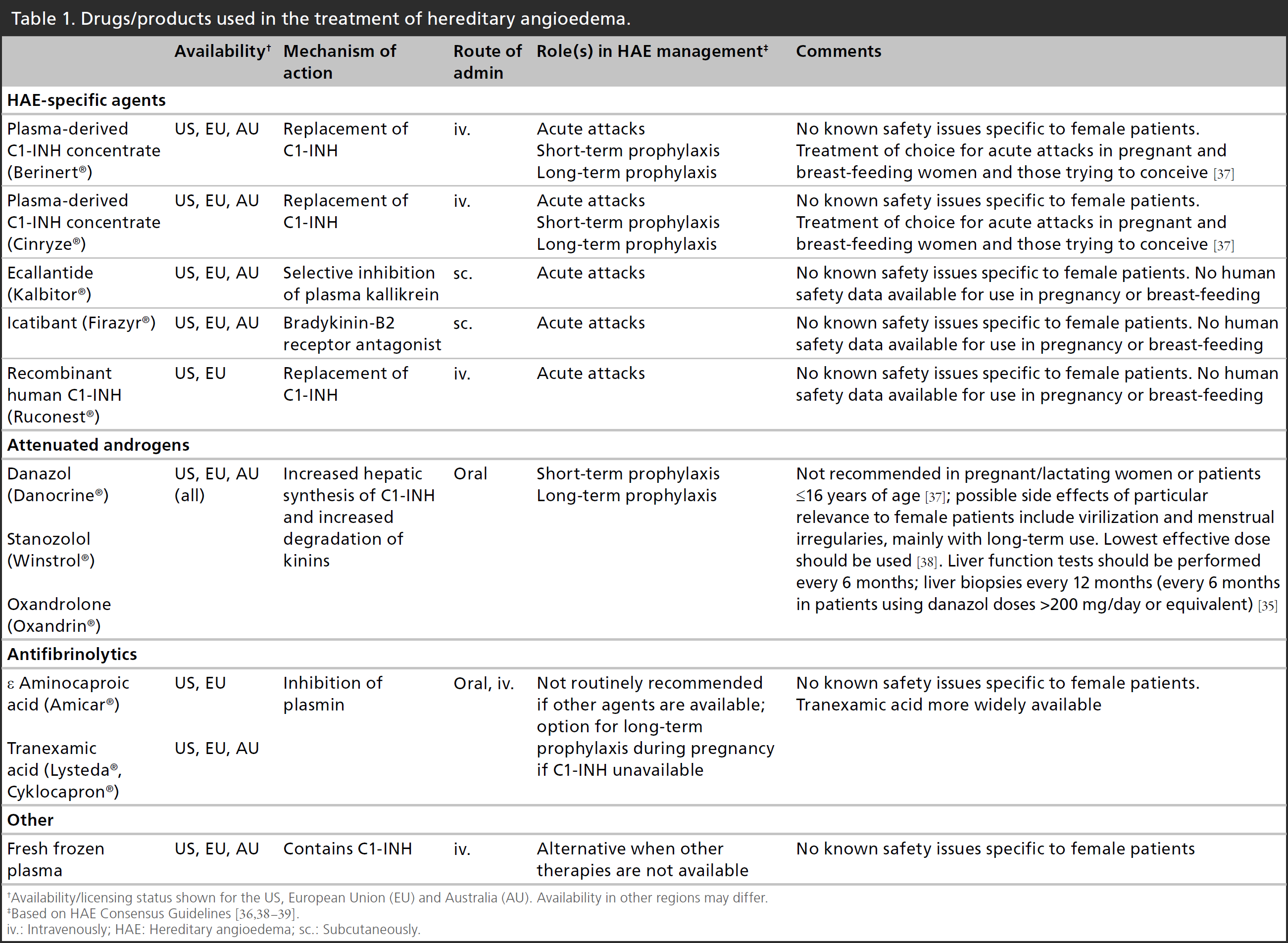
Availability/licensing status shown for the US, European Union (EU) and Australia (AU). Availability in other regions may differ.
Based on HAE Consensus Guidelines [36,38–39].
iv.: Intravenously; HAE: Hereditary angioedema; sc.: Subcutaneously.
The newer HAE-specific agents are given intravenously (pdC1-INH, rhC1-INH) or subcutaneously (ecallantide, icatibant). Self-administration of pdC1-INH is recommended, feasible and widely implemented [40] [R
Data sources & study selection
The English-language literature was searched for relevant articles using PubMed (1990-) and EMBASE (1993-) (start dates reflect database file origin points). The following search terms were employed: hereditary angioedema; bradykinin; female patient; women; pregnancy; labor; delivery; contraception; termination of pregnancy; lactation; genetic counseling; menstruation; menopause; infertility; and gynecologic surgery. Randomized clinical trials, retrospective studies, prospective observational studies, clinical guidelines and case reports considered to have clinical merit with regard to the manifestation or management of HAE in female patients, specifically, were included for review. Letters or comment-oriented citations were not included, nor were articles focused on complex diagnostic, genetic or nonclinically relevant issues. Furthermore, use of the search terms ‘female’ and ‘women’ resulted in a large number of irrelevant hits with abstracts including the terms, but only as descriptors for study population characteristics. The search process yielded 150 unique citations, of which 30 were deemed relevant for inclusion. Bibliographies of reviews and other papers were also scanned for additional source material pertinent to the topic. Additional references, not specific to care of the female patient, have been cited to support general discussions of HAE. While the purpose of this paper is to review published data on the topic, a recent consensus document [38] reflecting the recommendations of HAE experts regarding management of female patients is also cited throughout as appropriate and the reader is referred to that guideline for additional reading.
Results
Pregnancy
Pregnancy has variable effects on the frequency of HAE attacks: angioedema episodes may become more frequent, less frequent or remain unchanged [16,38,42]. An abdominal HAE attack may be difficult to distinguish from other complications of pregnancy; detection of free peritoneal fluid and edema of the intestinal wall by abdominal ultrasound helps confirm the diagnosis [43]. Some reports suggest that attack frequency is highest in the first trimester and declines in the second and third, whereas others found that symptoms worsened in the third trimester [42–44]. These patterns likely reflect the most active changes in estrogen levels especially in relation to progesterone levels, which occur during the first and third trimesters [38]. Carrying a fetus with the HAE mutation is associated with a higher frequency of third-trimester attacks [43]. The frequency of attacks during one pregnancy does not predict the frequency in subsequent pregnancies [38,44]. Research in rats suggests that bradykinin, the major mediator in HAE attacks, has a procontractile effect on uterine smooth muscle [45,46]. One case series [47] found higher rates of spontaneous abortion or premature labor in women with HAE compared with non-HAE relatives, while other reports have described similar rates of prematurity, spontaneous abortion and cesarean delivery in women with and without HAE [16,43].
Numerous case reports in the literature describe successful management of HAE during pregnancy with administration of prophylactic or on-demand pdC1-INH [48–54]. Additionally, expert guidelines support the use of pdC1-INH as needed during pregnancy [38,55–56]. Long-term prophylaxis should be considered for women with histories of miscarriage, high-risk pregnancies and those with frequent, severe attacks [38]. Medication dosage is suggested to be the same as in nonpregnant patients [38].
When available, pdC1-INH is generally recommended over other treatment options during pregnancy [38]. A retrospective review of 16 HAE patients treated with pdC1-INH as on-demand or prophylactic therapy during pregnancy found that attack frequency was reduced and attacks were managed successfully without significant adverse effects on mother or child [57]. A prospective observational study of 35 pregnancies in 22 women found that on-demand and prophylactic therapy with pdC1-INH was safe and effective [42]. Retrospective analysis of 118 pregnancies in 41 women in Hungary found pdC1-INH to also be safe and effective when used as long-term prophylaxis, short-term prophylaxis or on-demand treatment of attacks [44].
Other agents may not be optimal for treatment of HAE during pregnancy. Attenuated androgens are contraindicated during pregnancy because they cross the placenta and may affect fetal development [15,58]. Although no harmful effects of antifibrinolytics on the fetus have been reported, specific studies have not been performed in humans, therefore they can be considered for prophylaxis of HAE attacks during pregnancy only if pdC1-INH is not available, and after careful consideration of their potential benefit and harm [38,48,59–60]. One case report describes the successful use of fresh frozen plasma to manage HAE attacks during pregnancy [61]. No published data were identified regarding the safety of icatibant or ecallantide during pregnancy.
Labor & delivery
Vaginal delivery is preferred for patients with HAE because surgery or general anesthesia with endotracheal intubation may provoke an attack [62]. Somewhat surprisingly, HAE attacks are uncommon during vaginal delivery, occurring at a rate of 6–8%, therefore routine prophylaxis against an HAE attack is not recommended [16,43]. In case an attack does occur, acute treatment (preferably pdC1-INH) should be available and a physician familiar with the management of HAE should be consulted [38]. If the patient has a history of severe attacks or genital attacks secondary to trauma or her attacks have been frequent during the third trimester, short-term prophylaxis with pdC1-INH before vaginal delivery is advisable [38,48,50,52–53,58,60–61,63]. Administration of pdC1-INH is also recommended before forceps delivery or vacuum extraction [38].
Before cesarean delivery, pdC1-INH should be administered to reduce the risk of an HAE attack [14,38,51,54]. Epidural anesthesia is preferred because intubation for general anesthesia might trigger a laryngeal attack [14,38]. Emergency procedures should not be delayed if pdC1-INH is not immediately available [38].
Patients may experience an increased frequency of HAE attacks during the postpartum period [43]; patients with perineal swelling after delivery are at particularly high risk [18]. Close follow-up for 72 h after delivery by staff familiar with the management of HAE is recommended, and the patient should be informed of the treatment plan in the event of an HAE attack [38].
Termination of pregnancy
Short-term prophylaxis before surgical abortion is recommended; alternatively, the procedure may be carried out without prophylaxis, provided that acute treatment is immediately available. A case report described successful medical abortion in a woman with HAE using tranexamic acid for short-term prophylaxis; pdC1-INH was available but not needed [64].
Genetic counseling
Genetic counseling should be offered to all patients with HAE, with establishment of the pedigree and education regarding inheritance [38]. As an autosomal dominant disorder, HAE in a parent confers a 50% chance of inheritance on each child; however, disease expression is highly variable, and the severity of the parent's disease does not predict that of the child's [27]. Few patients request prenatal diagnosis in an established pregnancy; it can be performed only if the parent's genetic mutation is known [38]. Genetic testing for the specific mutation is performed with cells from a chorion villus sample taken after the 10th week of gestation or from an amniotic fluid sample taken after the 15th week [38]. For patients undergoing IVF, preimplantation genetic testing for HAE may be performed if requested [65]. Diagnostic testing for HAE in infants has low reliability, therefore if testing is performed during that period it should be repeated after 1 year of age for confirmation [66].
Lactation
Lactation may be associated with an increased frequency of HAE attacks, possibly associated with increased serum prolactin levels and discontinuing lactation might reduce the frequency of attacks [43,44]. Tranexamic acid is excreted into breast milk and should not be used in patients who are breastfeeding. It is not known whether anabolic androgens are excreted in breast milk, but because of their potential adverse effects in children they should be avoided during lactation. Consensus guidelines recommend pdC1-INH for on-demand or prophylactic therapy of HAE in lactating women [38].
Contraception
In as many as 80% of women, use of estrogen-containing oral contraceptives precipitates the onset of HAE attacks in a previously healthy patient or exacerbates previously diagnosed HAE, and discontinuing use usually relieves or reduces symptoms [16,62,67–68]. Parenteral estrogen-progestin contraceptives (patch or vaginal ring) would be expected to have effects similar to those of estrogen-containing OCs [38].
A retrospective analysis found that use of progestin-only OCs reduced the frequency of attacks in 45 (81.8%) of 55 women with HAE [69]. In 11 patients, improvement while on progestin-only OC enabled them to discontinue long-term prophylactic therapy. Antigonadotropic agents (lynestrenol, nomegestrol and chlormadinone acetate) were more effective than low-dose progestin-only pills (L-norgestrel, norgestrienone and desogestrel).
IUDs are generally well tolerated by women with HAE [16]. Short-term prophylaxis before insertion is not considered necessary, but treatment for an attack should be available. For emergency contraception, a progestin-only agent should be used, and estrogen-containing agents should be avoided. Barrier methods of contraception have not been found to have any effect on occurrence of HAE symptoms [38].
Menstruation
In women with HAE, attacks are reported to be triggered by menses in 35% and by ovulation in 14% [16]. Diagnosing an abdominal HAE attack during menses is difficult; the primary differential is pelvic endometriosis; severe abdominal pain, ascites or intestinal edema, and improvement after administration of an HAE treatment suggests HAE [38].
Menopause
In a survey of women with HAE, among those who had experienced menopause, 32% reported symptom worsening, 13% reported improvement and 55% reported no change [16]. Estrogen replacement therapy (including phytoestrogens) is contraindicated in women with HAE. Progesterone and progestins are alternatives for treatment of hot flashes. Nonhormonal treatments for the symptoms of menopause are not known to trigger HAE attacks [38].
Infertility
Fertility is unaffected by HAE, however no data are available regarding the effect of HAE medications on fertility [16]. Short-term prophylaxis is recommended before salpingography. If intrauterine insemination or IVF is attempted, estrogen induced by injectable gonadotropins may trigger an HAE attack. Therefore if possible IVF should be performed during spontaneous cycles when estradiol levels are lower and less likely to cause hyperstimulation. Short-term prophylaxis with pdC1-INH should be provided before procedures that might trigger an attack, and should be used in the event of an attack [38].
Gynecologic surgery
Before intubation or major procedures of any type, short-term prophylaxis should be considered and can include androgens for 5 days prior to the procedure or pdC1-INH. If pdC1-INH is used, it should be administered as close to the procedure as possible, preferably within 1 h and definitely within 6 h [55]. No optimal dose has been established, but expert guidelines recommend 10–20 units/kg, with a second dose of equal amount available during the procedure [55].
Gynecologic cancer
Androgens have potential adverse effects in patients with breast cancer, therefore they should be avoided if possible in patients with HAE and breast cancer [70–72]. Antiestrogens may have some agonistic effects on estrogen receptors; tamoxifen has been reported to exacerbate HAE symptoms and should be used with caution; an aromatase inhibitor may be preferable [73]. Androgens are not contraindicated in endometrial or cervical cancer [38].
Conclusion
The care of women during pregnancy, labor and delivery, lactation, contraception, menopause and surgical procedures may be complicated by the presence of HAE. Close cooperation between the obstetrician/gynecologist and the physician treating HAE is recommended to ensure optimal patient care without exacerbation of HAE symptoms.
Executive summary

Hereditary angioedema (HAE) is a rare, chronic disorder characterized by sporadic, painful and debilitating edematous attacks which are usually spontaneous but can be triggered by physical trauma, emotional stress or hormonal fluctuation. HAE symptoms appear more prevalent among females than males, and HAE can complicate, or be aggravated by, a number of obstetric/gynecologic aspects of women's health. Treatment strategies include acute treatment of individual attacks, long-term prophylaxis, short-term prophylaxis prior to a potential triggering event or some combination of these.
Pregnancy has the potential to change the frequency of HAE attacks. The use of plasma-derived C1-inhibitor (pdC1-INH) during pregnancy is recommended by recent expert guidelines when treatment is necessary. Attenuated androgens are contraindicated during pregnancy.
Vaginal birth is preferable to cesarean delivery in women with HAE. HAE prophylaxis during delivery is not recommended as a routine practice, but might be considered in certain cases depending on personal risk factors. Epidural anesthesia is preferable to intubation to reduce the risk of a potentially fatal laryngeal attack. Lactation can increase the frequency of HAE attacks; pdC1-INH is recommended for women who desire to breastfeed.
Estrogen-containing birth control pills often exacerbate HAE and should be avoided, while progestin-only contraceptives may lessen attack frequency. lUDs are generally well tolerated by women with HAE.
Recent years have witnessed a marked increase in the number of treatment options available for HAE and additional therapies are expected to emerge over coming years. The underlying pathophysiology of HAE, including various subtypes, continues to be characterized more fully, including the role of estrogen in some variants. Ongoing discoveries in this area may improve treatment strategies in women with HAE. Optimal management of female patients with HAE will continue to require close collaboration between HAE-treating physicians and women's health professionals.
Future perspective
Over recent years, significant advances have been made with regard to HAE management and the potential is greater than ever before for affected patients to move closer to living a life that is not overshadowed by disease morbidity. A range of effective therapies have become available, and treatment can now be managed at home by most patients, even with injectable agents. A growing understanding of the underlying pathophysiology of HAE has facilitated the development of a number of targeted therapies, and it is likely that ongoing progress in this area will continue to improve prophylactic options for HAE as well as refine how the disease is managed, particularly in recently identified subtypes with normal C1 inhibitor. Additional laboratory and clinical research may identify biomarkers useful in predicting clinical phenotypes and/or response to various therapeutic agents.
Footnotes
This review and manuscript development was funded by CSL Behring, King of Prussia, PA, USA. A Banerji has received research funding from Dyax, Shire, CSL Behring and ViroPharma, and has served as a scientific consultant for Dyax, Shire, CSL Behring and Santarus. M Riedl has received research funding from CSL Behring, BioCryst, Dyax, ViroPharma, Shire and Pharming; he has served as a scientific consultant for CSL Behring, BioCryst, Dyax, ViroPharma, Shire, Santarus and Isis, and as a speaker for CSL Behring, Dyax, Shire and ViroPharma. The authors have no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed.
Writing assistance was provided by Greg Otis and Sandra Westra, of Churchill Communications, Maplewood, NJ, also funded by CSL Behring. The authors retained full control over manuscript content.
