Abstract
Worldwide ovarian cancer affects over 200,000 women per year. Overall survival rates are poor due to two predominate reasons. First, the majority of patients present with advanced disease creating significant difficulty with effecting disease eradication. Second, acquisition of chemotherapy resistance results in untreatable progressive disease. Advances in treatment of advanced ovarian cancer involve a spectrum of interventions including improvements in frontline debulking surgery and combination chemotherapy. Anti-angiogenic factors have been shown to have activity in frontline and recurrent disease while novel chemotherapeutic agents and targeted treatments are in development particularly for disease that is resistant to platinum-based chemotherapy. These developments aim to improve the progression-free and overall survival of women with advanced ovarian cancer
Keywords
Ovarian cancer is the third most common gynecological malignancy worldwide but it is the most lethal, responsible for 160,000 deaths per year [1]. Following surgical debulking, response rates to initial chemotherapy are high with up to 75% of patients having a complete clinical response [2]. However, the majority of patients will still go on to relapse. Once relapse has occurred treatment is not curative and unfortunately cure rates have changed little in the last 20 years. Ovarian cancer frequently responds to multiple lines of chemotherapy and new discoveries have increased the progression-free survival (PFS) and overall survival (OS) [3]. In this review, we discuss the recent discoveries and changes in the management of advanced ovarian cancer that have improved survival and we shed light on novel treatments and current areas of research.
Advances in surgery
Primary – maximal effort – cytoreductive surgery followed by chemotherapy is the mainstay of initial treatment for advanced ovarian cancer; Féderation Internationale de Gynécologie et d'Obstétrique (FIGO) stage IC-IV. Research in the 20th century in Europe and USA demonstrated that removal of as much visible (macroscopic) tumor as is technically possible prior to systemic chemotherapy was the most important factor in improving patient outcomes [4]. Initially, optimal debulking was defined as residual disease of less than 2 cm in the lesion of greatest diameter. More recently it has been shown that those patients who have no macroscopic disease at the end of surgery have greatly improved survival compared with those who were debulked with residual disease [5]. Data analysis has shown that patients who have no visible postoperative residual disease have a 5-year survival of 63%, but in those where the largest residual lesion is 1–10 mm this falls to 28.6%; demonstrating the poor prognosis for any residual disease however small [6]. Therefore complete macroscopic debulking surgery has become the target for primary surgery. Five-year survival falls further to 21.3% when residual disease is greater than 10 mm indicating that upfront surgery cytoreduction is of value even if total macroscopic tumor clearance is not feasible. This reduction in survival with increasing residual disease is demonstrated in Figure 1. The benefit of optimal surgical debulking within the peritoneal cavity is present even in patients with FIGO stage IV disease [7]. However, the reason for being classed as stage FIGO IV disease is important; stage IV due to positive pleura cytology alone (now stage IVA) is associated with a much higher OS than that due to intraparenchymal hepatic metastases (now stage IVB) and this finding is reflected in the 2014 update in FIGO staging [8].
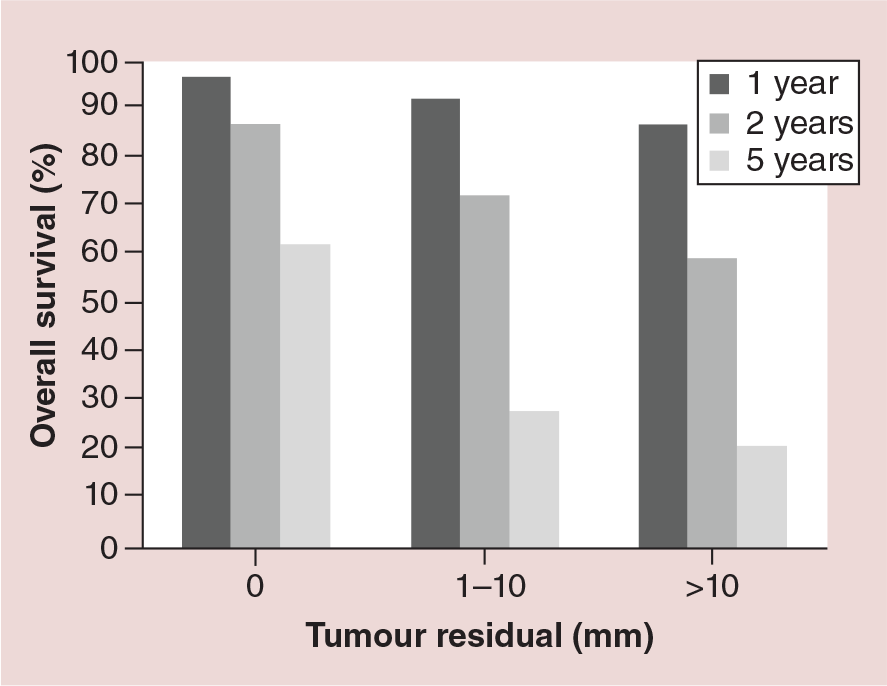
Overall survival at 1–5 years is dependent upon the size of largest residual tumor deposit following debulking surgery.
As surgery is operator dependent, improving surgical skill and technique may improve outcomes if all patients are operated on with the aim of achieving no macroscopic residual disease. A large metanalysis of 3126 patients [6] showed that postoperative residual disease overrides the FIGO stage in terms of predicting prognosis, with advanced stage optimally debulked patients showing higher survival rates than suboptimally debulked patients of a lower stage. However, there is a subset of patients in whom complete macroscopic debulking is not technically possible due to distribution of disease and therefore these patients have poorer outcomes. It is not clear whether these patients are disadvantaged because their disease is unresectable and therefore improvement in surgical technique may improve their outlook or whether such patients have disease in which inability to carry out resection is a surrogate marker of unfavorable disease biology.
A review of the data of SCOTROC by Crawford et al. [7] shed light on this issue of favorable versus unfavorable disease. It first showed that patients in the UK generally had a lower proportion of patients that were optimally debulked compared with similar patients in Australia and the USA, which is an ongoing area of controversy. It also showed that optimal debulking was associated with improved PFS mainly in patients with less extensive disease at diagnosis, rather than across all patients as might be expected if degree of debulking is a universally critical issue in ovarian cancer. This suggests that more extensive disease may represent a more aggressive subtype of disease and that the poor outcomes from incomplete debulking in these patients may be due to differences in tumor biology rather than lack of technical skill. This may also suggest why complete removal is associated with better PFS; because it is a less aggressive subtype of disease. Definitive evidence for this theory is still sought.
Neoadjuvant chemotherapy
Although primary cytoreductive surgery is considered the standard of care in many centers there has been considerable interest in the use of neoadjuvant chemotherapy followed by interval debulking surgery, due to the morbidity associated with maximal effort cytoreductive surgery. In their 2010 paper, Vergote et al. [9] presented a randomized trial in which patients with stage IIIC and IV ovarian cancer were randomized to primary cytoreductive surgery or neoadjuvant chemotherapy followed by interval debulking. The results showed no difference in survival between the two groups suggesting that neoadjuvant chemotherapy followed by interval debulking was not inferior. Since primary cytoreductive surgery carries greater morbidity and mortality risk due to the extent of surgery, it may be interpreted that neoadjuvant chemotherapy is preferable. However, in this trial surgical quality was strongly inhomogeneous with total macroscopic tumor clearance rates ranging from 3.9 to 63%. In addition OS was just 29 months in the upfront surgery group, compared with 49 months in a comparable group in the GOG172 [10] study carried out in the USA. These differences in survival are believed by many practitioners to be primarily due to a lower rate of low-volume debulking in the Vergote et al. study which contributed to the view held by the majority of US practitioners that primary debulking surgery is still the best practice for most women with ovarian cancer. This leaves unanswered the question of adequate timing of surgery with standardized total macroscopic debulking. The differences between US and European philosophies mean that neoadjuvant chemotherapy remains controversial and surgical management of ovarian cancer is typically much less aggressive in Europe than in the USA.
In Vergote et al. 80.6% of the neoadjuvant group of patients were resected to less than 1 cm with no concurrent increase in OS, as compared with expectations from upfront surgery. This suggests that the survival advantage of being optimally debulked is lost if neoadjuvant chemotherapy has been given and therefore that the definition of postoperative residual disease after chemotherapy is not the same as the definition of residual disease in chemo-naive patients. This poses the question of oncologic safety and validity of surgery at a delayed time point and the need to redefine the meaning of optimal surgery in the neoadjuvant setting.
This view is supported by the Gynecologic Oncology Group (GOG) 152 trial in which patients with advanced ovarian cancer and residual tumor exceeding 1 cm after primary surgery despite maximal surgical effort in the hands of experienced surgeons, were randomized after three cycles of adjuvant chemotherapy to interval cytoreductive surgery plus three further cycles of chemotherapy or to chemotherapy alone [11]. In this study patients had higher optimal debulking rates after three cycles of chemotherapy compared with their initial surgery, but there was no survival advantage of reoperation after suboptimal initial debulking. This study emphasizes the point that the impact of tumor residual disease depends on the timing of the cytoreductive procedure. In order to address the issue of survival benefit of upfront debulking counterbalanced with higher surgical morbidity, a further trial is currently being developed with strictly defined qualification criteria for the participating centers to ensure surgical quality. This will answer the question of the optimal timing of chemotherapy. The study is planned as a collaboration between the GOG, AGO, GINECO, NOGGO, MITO and MANGO organizations and is expected to include more than 700 primary patients with advanced stage III and IV disease
Secondary debulking surgery
Despite the strong evidence for primary cytoreductive surgery in ovarian cancer, the evidence for surgery after disease relapse is less well defined. Surgery at relapse has two aims; cytoreduction or palliation of otherwise untreatable symptoms. Even in cases where cytoreduction is the aim, surgery is not curative.
In many centers relapses are surgically excised based on local practice before further chemotherapy is administered in the belief that this improves survival. Retrospective analysis of the DESKTOP dataset of patients who have undergone selective secondary debulking surgery has shown significant improvement in survival if recurrent disease was completely resected; median survival in complete resection 45.2 versus 19.7 months if incomplete (hazard ratio [HR]: 3.71; 95% CI: 2.27–6.05; p < 0.001) [12]. A study prospectively randomizing resectable patients to secondary debulking surgery or chemotherapy, DESKTOP III [13], has been designed to directly compare these strategies. In this trial patients with presumed resectable disease are randomized between second-line chemotherapy or secondary debulking followed by chemotherapy. This trial, together with the equivalent American trial GOG 213 [14] will define the prospective value of surgical cytoreduction in platinum-sensitive relapse.
Of note, in all retrospective series so far, cytoreductive surgery at relapse has been shown to be associated with prolongation of OS and PFS only if a total macroscopic tumor clearance could be obtained. However, it is difficult to predict this preoperatively. Based on the DESKTOP dataset the German Arbeitsgemeinschaft Gynäkologische Onkologie (AGO) defined a clinical ‘AGO score’ which could predict total macroscopic tumor clearance in more than two of three patients if all of the following factors were met; ascites less than 500 ml, tumor-free surgery at initial presentation of disease and good performance status at relapse. The number and localization of the relapsed lesions were of no significant value if total macroscopic tumor clearance could be obtained. Interestingly, in the DESKTOP series the patients with disseminated peritoneal carcinomatosis at relapse had the same survival as those without peritoneal carcinomatosis if they had no macroscopic disease postoperatively [15].
There is also evidence that the value of cytoreductive surgery holds beyond first disease recurrence, such that wherever complete surgical resection of disease is possible and the disease is responsive to platinum chemotherapy it may be recommended, since surgical effort appears to be associated with significantly prolonged OS and PFS [16].
Intraoperative mapping
Given the importance of complete macroscopic cytoreductive therapy for prolonged survival in ovarian cancer, advances that can improve the visualization of miniscule tumor deposits during surgery would be beneficial. Ovarian cancer cells are known to over express folate receptors and intra-operative visualization of cancer cells by the development of a folate receptor-α-targeted fluorescent agent has shown experimental potential in these patients. By helping to guide surgeons in their debulking efforts these experiments aim to increase the degree of tumor clearance and therefore survival [17]. Other agents such as integrin fluorescent imaging are in development and may come to play an important role in fully resecting advanced ovarian cancer [18]. However, all of the agents described in this section are in the early stages of research and development.
Adjuvant chemotherapy
Adjuvant chemotherapy results in a long-term cure in 10–15% of advanced ovarian cancer patients and improves OS in the remainder [3]. For more than a decade platinum analog-based adjuvant chemotherapy regimens have been the standard of care following primary cytoreductive surgery. Carboplatin which has equivalent efficacy and comparatively less toxicity compared with cisplatin has become the most widely used platinum-based agent [3]. Many different platinum containing combinations have been investigated and carboplatin plus paclitaxel given on a 3 weekly basis has become the consensus frontline treatment [19]. Despite this consensus there is evidence that the benefit of adding paclitaxel to a carboplatin regimen is small and that in patients who are unable to tolerate paclitaxel, carboplatin alone is a reasonable first-line option [20]. The most important dose limiting toxicity of paclitaxel is neuropathy, the risk of which can be reduced with the substitution of docetaxel but at the cost of increased myelosuppression [21].
Dose dense regimes
A study by Katsumata et al. [22] in Japan has suggested that dose dense paclitaxel may provide a significant survival advantage over standard treatment. In this trial women were randomized to a conventional regimen of 3 weekly carboplatin and paclitaxel or a dose dense regimen of weekly paclitaxel plus 3 weekly carboplatin. Median OS was higher in the dose dense regimen group (100.5 months) than in the conventional treatment group (62.2 months; HR: 0.79; CI: 0.63–0.99; p = 0.039) [23]. To confirm these findings in a wider ethnic group the International Collaboration on Ovarian Neoplasm (ICON)8 study [24] is currently trialing conventional 3 weekly carboplatin and paclitaxel, to dose dense paclitaxel, with conventional and dose dense carboplatin. Two trials have already published further data on dose-dense paclitaxel. The completed Multicenter Italian Trials in Ovarian Cancer (MITO-7) study showed no difference between its trial arms of three weekly carboplatin (AUC 6) plus paclitaxel (175 mg/m2) versus weekly carboplatin (AUC 2) plus paclitaxel (60 mg/m2) [25]. However, the lower total dose of weekly paclitaxel used in this study may have been responsible for the negative study findings. GOG262 has published preliminary results [26]. This study compared dose-dense weekly paclitaxel (80 mg/m2) and carboplatin (AUC 6) with a conventional three weekly carboplatin and paclitaxel. Preliminary results show no difference between the two arms. However, crucially both arms provided the option of additional bevacizumab. This addition makes results difficult to interpret as it appears that bevacizumab may reduce the advantage of dose dense paclitaxel.
Angiogenesis inhibitors
Despite the improvement in chemotherapy regimens the overall improvement in PFS since the discovery of the platinum agents has been disappointing. Therefore, recent research has been focused on understanding the biology of ovarian cancer in order to discover new methods of targeting the disease.
Angiogenesis is a complex process regulated by a number of endogenous pro- and anti-angiogenic factors and plays a key role in the growth and metastases of solid tumors including ovarian cancer. VEGF is a key mediator of angiogenesis and expression of intratumoral VEGF and its receptor are associated with poor prognosis in ovarian cancer [27]. Bevacizumab is a monoclonal antibody that binds to the isoform VEGF-A and has proven efficacy in a range of cancers. The ICON7 [28] and the GOG 218 study [29] were two landmark trials that defined the role of bevacizumab in front-line treatment of advanced ovarian cancer. Patients were randomized to three weekly carboplatin and paclitaxel with or without bevacizumab. Median PFS was improved by 1.5 months in ICON7 (HR: 0.81; 95% CI: 0.70–0.94; p = 0.004) and 3.8 months in GOG218 (HR: 0.72; 95% CI: 0.62–0.82; p < 0.001) and was restricted to those patients who received maintenance bevacizumab for a year or more. Although PFS was improved with bevacizumab in GOG218 and ICON 7, a benefit in OS has not been shown. The PFS advantage then appeared to wane once bevacizumab was discontinued and the optimal period of time to continue the drug remains unclear. Bevacizumab is usually well tolerated but common toxicities include hypertension and proteinuria. An increase in the frequency of bowel perforation has also been observed. Bevacizumab is the first anti-angiogenesis drug to show an improvement in PFS and on the basis of these trials the EMA has approved bevacizumab for front line use. In contrast the National Institute for Clinical Excellence has not provided funding on the basis of lack of cost–effectiveness [30], as bevacizumab costs GB£128,000–£161,000 per quality-adjusted life year (QALY) gained. The appropriate role for bevacizumab in ovarian cancer in the up-front adjuvant setting is not yet determined.
A number of VEGF pathway inhibitors have been trialed besides bevacizumab. Pazopanib is an oral tyrosine kinase inhibitor that inhibits several kinase receptors including VEGF, PDGF and FGF. The PDGF and FGF pathways have been shown to be involved in angiogenesis and may be involved in the resistance mechanisms to VEGF receptor inhibitors [27]. AGO-OVAR-16 [31] is a large Phase III trial comparing 24 months of maintenance pazopanib vs placebo in 940 women with advanced ovarian cancer. Patients in the pazopanib arm had a PFS of 17.9 months versus 12.3 months in the placebo arm (HR: 0.76; 95% CI: 0.64–0.91; p = 0.002). The most common toxicity was hypertension (31% grade 3/4), with other toxicities including diarrhoea, hepatic impairment and neutropenia. The result of this trial suggests that maintenance pazopanib is a potential oral alternative to bevacizumab, although head to head comparison trials have not been undertaken. However, as the use of VEGF antagonists in the up-front setting remains controversial, they have not been incorporated into clinical trials as the ‘standard’ arm in the treatment of advanced disease.
Alternative angiogenic pathways are also being targeted as potential treatments for ovarian cancer. One of these pathways involves angiopoietins; which are cytokines involved in the regulation of angiogenesis and inflammation. Novel peptibodies which selectively neutralize specific angiopoietins have shown promise in Phase II clinical trials in recurrent ovarian cancer [27] and in the future may provide new adjuvant treatments.
Intraperitoneal chemotherapy
As the development of new targeted therapies for ovarian cancer has been limited, research has also focused on delivering existing chemotherapeutic agents in novel ways. Since ovarian cancer frequently has an intraperitoneal distribution at diagnosis, intraperitoneal instillation of chemotherapy has been investigated as a way of exposing tumor tissue to extremely high levels of chemotherapy. Studies have shown that intraperitoneal concentrations of intraperitoneal cisplatin may reach 12–21-fold the concentration of the plasma [32]. Therefore the intraperitoneal route may be expected to have improved disease control in addition to reduced systemic adverse effects. The first randomized trial of intraperitoneal chemotherapy, published in 1996 GOG 104 [33], supported both of these aspects. In this trial patients were given the same dose of cisplatin (100 mg/m2) intraperitoneal or intravenous (iv.) along with cyclophosphamide iv. for six cycles. Median survival in the intraperitoneal arm was significantly longer at 49 months compared with 41 months in the iv. arm (HR: 0.76; CI: 0.61–0.96; p = 0.02). Grade III/IV neutropenia, clinical hearing loss, and neuromuscular toxic effects were all reduced in the intraperitoneal group. Two subsequent trials; GOG114 [34] and GOG172 [10] confirmed the finding of increased survival. However, they showed increased grade III/IV adverse effects, particularly fatigue, gastrointestinal and hematological effects. As a result of these adverse effects only 18% and 42% of patients completed the assigned intraperitoneal chemotherapy in the respective trials leading to serious concerns about intraperitoneal toxicity. An additional problem of GOG172 and GOG114 trials lay in their design. While GOG104 was a simple substitution of iv. for intraperitoneal chemotherapy, both of the subsequent trials had unequal control and treatment arms (Figure 2) such that it is difficult to be certain that the positive results of these trials were due to superiority of the intraperitoneal route. Consistent evidence of significantly increased survival led the National Cancer Institute (NCI) in the USA to recommend the use of frontline intraperitoneal chemotherapy in 2006 [35]. Improvements in port placement and management may significantly reduce toxicities and there are a number of centers worldwide that use and develop this promising form of therapy. However, lack of experience with this route of chemotherapy and uncertainty over its risk of complications has meant that it has yet to be widely adopted.

Design of GOG114and GOG172.
Areas for further research include substituting cisplatin with carboplatin to look for reduced intraperitoneal toxicity and a definitive trial with balanced chemotherapy doses to confirm that intraperitoneal is a superior route of administration.
In an exploratory retrospective subanalysis of GOG172 by Lesnock et al. the tumors were analyzed for expression of the Breast Cancer 1 (BRCA1) protein [36]. This study showed that patients with tumors that had reduced BRCA1 expression had longer OS in response to intraperitoneal chemotherapy (84 months) than iv. (47 months; HR: 0.67; 0.67 CI: 0.47–0.97; p = 0.03). However, the benefit of intraperitoneal vs. iv. chemotherapy was lost in tumors with normal BRCA1 expression. It would therefore appear that the benefit of intraperitoneal chemotherapy may be restricted to those with reduced BRCA1 expression. Prospective trials are needed to confirm this finding and with a 36 month survival advantage it would appear prudent to design trials involving intraperitoneal treatment in those with aberrant BRCA1 expression, particularly those with inherited BRCA1 mutations.
BRCA1- & BRCA2-related disease
BRCA1 and BRCA2 are dominantly inherited cancer susceptibility genes that were first discovered in families with high rates of breast cancer. Mutations in these genes have been found to be common in women with high-grade serous cancer (HGSC), which is the most common histological subtype of ovarian cancer. BRCA germline mutations are seen in 13–16% of HGSC and somatic mutations are detected in another 6% of cases. The lifetime risk of HGSC is up to 60% in BRCA1 mutation carriers, and up to 27% in those with BRCA2 [37]. Women who are known carriers are offered pre-emptive salipingo-oophrectomy after completion of their families to substantially reduce their risk of developing ovarian cancer.
Patients with HGSC that carry BRCA mutations have been shown to be particularly sensitive to platinum-based chemotherapy, and frequently have sustained response to repeated treatments with these agents [38]. This increased sensitivity to platinum agents may explain the prolonged survival in the intraperitoneal trials mentioned previously, since BRCA patients may have particular sensitivity to the higher intratumoral concentrations of platinum agents.
PARP inhibitors
The BRCA genes encode proteins that sense DNA damage and are involved in double standed (ds) DNA repair via homologous recombination. Cell lines carrying these mutations are therefore deficient in dsDNA repair and rely upon single stranded (ss) DNA repair mechanisms to resolve DNA damage. Poly(ADP-ribose) polymerase (PARP) enzymes are a family of proteins shown to be involved in ssDNA repair. PARP inhibitors are a new class of drug that prevent PARP enzyme activity leading to persistence of spontaneous ssDNA breaks that progress to dsDNA breaks on replication [39]. BRCA mutated cells cannot repair these breaks leading to cell death, while normal cells are spared. This targeting of BRCA mutated cells by exploitation of their specific sensitivity to PARP inhibitors is called ‘synthetic lethality’ and has been the focus of a great deal of research in recent years. Olaparib is one of a number of PARP inhibitors that have been investigated. A Phase III study of maintenance olaparib versus placebo has already been completed in women with recurrent HGSC [40]. Women receiving olaparib had an improvement in PFS which was much more pronounced in those who harboured a BRCA mutation (11.5 vs 5.6 months; p < 0.00001) [41]. This evidence, although involving retrospective BRCA evaluation, suggests that testing for BRCA status in women with HGSC has clinical significance. A Phase III trial called SOLO (Study of OLaparib in Ovarian cancer) has been designed to determine the benefit of olaparib as a maintenance monotherapy specifically in BRCA mutated ovarian cancer patients. This strategy will be evaluated in both the first-line (SOLO1) [42] and second-line (SOLO2) [43] setting.
Predicting relapse
Unfortunately the majority of patients with advanced ovarian cancer will go on to have relapse of their disease after frontline surgery and chemotherapy. There are many options for surveillance to detect relapse including serial measurement of the tumor marker cancer antigen 125 (CA125), physical exam and a number of imaging modalities. Significant controversy exists about the relative effectiveness of each modality and their effect on survival and whilst detection of recurrences by physical exam alone is rare (4% of relapses), the combination of examination, history and elevated CA125 will detect up to 90% of recurrences [2].
CA125 is a glycoprotein which is elevated in up to 88% of advanced ovarian cancers and has a role in both monitoring disease response and in follow-up. Rising CA125 may precede development of symptoms by a number of months and represents a significant management dilemma; whether to treat when CA125 is first elevated or on the development of symptoms? This was addressed in a study by Rustin et al. [44] in which women who were in complete remission were randomized to early treatment of relapse on asymptomatic CA125 rise or treatment only on symptomatic recurrence. There was no significant difference between the two treatment arms (median survival: 25.7 vs 27.1 months) but women in the early treatment arm received more chemotherapy overall and had an earlier deterioration in quality of life. Therefore measuring CA125 alone is not sufficient to dictate timing of further treatment but must be used in conjunction with other investigations. Continuing developments in chemotherapy treatment and increasing use of secondary debulking constantly change the options for disease treatment and mean that early detection may in the future allow improved outcomes
Although there are many imaging modalities available for ovarian cancer, there has been no OS benefit shown for serial radiological studies [2]. The most commonly used modality to guide treatment is computer tomography (CT). However, as an elevated CA125 may precede CT findings by many months alternative imaging has been investigated to decide who requires immediate treatment. 18Fluorine deoxyglucose PET is a method of imaging based on the increased glucose metabolism of malignant tumors. It has been shown to detect ovarian relapses at an earlier stage during follow-up with a sensitivity of up to 96% [45]. This suggests that fluorine deoxyglucose PET could play a role in early detection of relapses but it has yet to been proven to be useful in clinical trials.
Platinum-sensitive relapse
At relapse the period of time that has elapsed since last treatment with platinum-based compounds is prognostic [3]. Patients who have relapsed more than 1 year after their last platinum-based treatment are classified as platinum sensitive and have approximately a 60% chance of response to further platinum containing chemotherapy. If relapse occurs following a platinum-free interval (PFI) of 6–12 months patients are classified as partially platinum sensitive and have a reduced response rate of 25–40% to platinum agents. Patients who relapse with a PFI of less than 6 months are classified as platinum resistant and are not rechallenged with platinum-based regimes due to unacceptably low response rates.
In platinum sensitive relapse, second-line treatment will most commonly involve carboplatin. Research into combination therapy has shown that the addition of paclitaxel to a platinum regime gives superior survival at relapse [46]. However, this combination is accompanied by significant toxicity including peripheral neuropathy, neutropenia and alopecia. Carboplatin plus gemcitabine is one alternative regime which has also shown superiority to carboplatin alone [47]. The CALYPSO [48] study compared the combination of pegylated liposomal doxorubicin (PLD) and carboplatin with standard carboplatin and paclitaxel at first relapse. The PLD arm had superior PFS (HR: 0.82; 95% CI: 0.72–0.94; p = 0.005), although in subsequent analysis there was no difference in OS, potentially due to unequal post study cross-over [49]. Overall more patients discontinued treatment in the standard arm due to severe nonhematologic toxicity, with the PLD arm having significantly less neuropathy and alopecia, but more nausea, mucositis and hand-foot syndrome. Therefore in the relapse setting carboplatin plus PLD is less toxic and likely more effective than carboplatin plus paclitaxel.
Angiogenesis inhibitors
Anti-angiogenic agents are known to have activity in the setting of platinum sensitive recurrence. The largest randomized Phase III trial is the OCEANS trial [50] in which carboplatin and gemcitabine was trialled with and without the addition of concurrent bevacizumab, which was continued until disease progression. PFS was superior in the bevacizumab arm (HR: 0.48; 95% CI: 0.39–0.60; p < 0.0001) and has therefore been approved for use by the EMA and the cancer drugs fund (CDF) for first-line platinum-sensitive relapse in conjunction with carboplatin and gemcitabine only. There is currently no published data on the effectiveness of bevacizumab for relapse after having received the drug in the first-line setting.
Cediranib is an oral tyrosine kinase inhibitor that also has inhibitory effects on VEGF/PDGF/FGF receptors and is similar in action to pazopanib [27]. The ICON6 study enrolled patients following their first relapse with ovarian cancer. The control arm was standard chemotherapy, with two experimental arms of concurrent cediranib plus placebo maintenance or concurrent plus 18 months maintenance cediranib. The trial showed the combination of cediranib and chemotherapy provided a 2.7 month advantage in OS (17.6 vs 20.3 months; HR: 0.7; p =0.04) and also extended PFS by 3.1 months (9.4 vs 12.5 months; HR: 0.68; p = 0.0022) thus showing a significant survival benefit to cediranib use [51].
The OCEANS trial which showed improvement in PFS but not OS and the ICON 6 trial which showed modest OS and PFS improvement strengthen the evidence for a benefit from targeting the angiogenesis pathway in recurrent ovarian cancer, in addition to the evidence for targeting it in the frontline setting. On publication of the final results of ICON6 cediranib will likely be an oral alternative to bevacizumab in recurrent ovarian cancer.
Partially platinum-sensitive relapse
The trials thus far described in this section primarily recruited patients with platinum-sensitive recurrence. For patients that relapse with a PFI of 6–12 months there is less consensus regarding rechallenge with platinum-based regimes due to decreased response rates. In addition, platinum-based toxicities may be greater with a short PFI and therefore there is need for alternative nonplatinum agents. Trabectedin is a chemotherapy agent originally derived from the sea squirt Ecteinascidia turbinate, that has proven efficacy in relapsed ovarian cancer [52]. In the OVA-301 trial trabectedin plus PLD was shown to have superior activity to PLD alone in relapsed ovarian cancer [53]. A total of 214 patients in this study had partially platinum sensitive disease and a subanalysis of these patients showed a median PFS of 7.4 months in the combination arm versus 5.5 months in the PLD arm (HR: 0.65; 95% CI: 0.45–0.92; p = 0.015) [54]. This was the largest difference for all PFI subsets analyzed therefore showing evidence for a specific benefit of trabectedin/PLD in partially platinum-sensitive disease. In addition, clinical data suggest that the artificial expansion of PFI with an intervening nonplatinum therapy may be beneficial [55], possibly by reversing platinum resistance. Within OVA-301 partially platinum sensitive patients in the trabectedin/PLD arm survived significantly longer after subsequent platinum treatment (HR: 0.63; p = 0.0357; median: 13.3 vs 9.8 months) than those on PLD alone, thus supporting the hypothesis that artificially increasing PFI is important. The additional benefit for trabectedin/PLD therapy in terms of PFS and survival after further platinum therapy provides a new option for partially platinum-sensitive patients. However, because platinum containing regimes remain a reasonable treatment option in this group and the evidence is based on subset analysis only, a trial is planned comparing carboplatin/PLD using the regimen evaluated in the CALYPSO trial with trabectedin/PLD [54].
Platinum-resistant relapse
Where the PFI is less than 6 months platinum agents have low response rates and the median survival is less than 12 months [56]. The mainstay of treatment is single agent therapies which have varying levels of activity. Weekly paclitaxel appears to have response rates of up to 25% [57]. Other commonly used therapies such as topotecan and PLD have response rates of 10–15% [56]. Improvements on these poor response rates have been sought by adding anti-angiogenic factors. The AURELIA study investigated the addition of bevacizumab to single agent therapy from any of the previous mentioned single agents. Choice of therapy was investigator directed depending on previous patient treatment tolerances. The addition of bevacizumab showed a PFS advantage with a median PFS of 6.7 months with chemotherapy plus bevacizumab versus 3.4 months with chemotherapy alone (HR: 0.48; 95% CI: 0.38–0.60; p < 0.001) [58]. The improvement in PFS is not dissimilar to the 1.5–3.8-month improvement in PFS seen in the adjuvant use of bevacizumab [28,29]. However, in the platinum-resistant setting there is considerably less exposure to bevacizumab to gain the same improvement in PFS. This shows that anti-angiogenic treatment is active in all presentations of ovarian cancer resulting in an approximate 3 month improvement in PFS independent of PFI.
Novel chemotherapeutic agents
In the search for novel nonplatinum-based chemotherapeutic agents various lines have been investigated. Over 80% of ovarian cancers overexpress the membrane bound folate receptor (FR), whereas it is poorly expressed or absent in most normal cells making it an excellent candidate for targeted therapy. Farletuzumab, an FR targeted antibody, has shown disappointing results in Phase III trials [18]. However, vintafolide, a folate conjugated with the vinca alkaloid desacetylvinblastine monohydrazine, is specifically designed to target the FR delivering potent chemotherapy directly to cells overexpressing FR. PRECEDENT [59] is a Phase II study which assessed vintafolide with PLD versus PLD alone in recurrent ovarian cancer. This showed an improvement in PFS with a median survival of 5.0 months for the combination arm versus 2.7 months for the PLD alone arm (HR: 0.63; 95% CI: 0.41–0.96; p = 0.031). This trial was run alongside evaluation of etarfolatide, which is a folate-technetium conjugate imaging agent that selectively binds FR-expressing cells analogous to the action of vintafolide. This allowed imaging of FR-expressing tumors via single photon emission computed tomography. Patients who had 100% of lesions positive for FR, had an improved PFS median of 5.5 months in the experimental arm compared with 1.5 months for PLD alone. PROCEED, a randomized placebo controlled trial of vintafolide alongside etarfolatide imaging has been undertaken [60], but enrolment has been suspended due to a disappointing interim analysis. It is hoped that future developments of FR targeted drugs may be more successful.
Another area of interest has been in inhibition of the Phosphoinositide 3-kinase (PI3K)/AKT pathway. This has been shown to be overactive in ovarian cancer and may be involved in the resistance of ovarian cancer to platinum agents [61]. Inhibition of AKT is postulated to reverse chemotherapeutic resistance and therefore a Phase I/II study is underway to investigate a novel AKT inhibitor GSK2110183 when given alongside carboplatin and paclitaxel [62].
Immunotherapy
There is evidence that the human immune system generates tumor reactive immune cells that have anti-tumor potential. Therefore modulation of this response represents a rational therapeutic approach to the treatment of ovarian cancer [18]. The most well developed area of research in ovarian cancer immunotherapy is dendritic cell (DC)-based vaccines. DCs capture, process and present antigens including tumor-associated antigens thereby inducing the generation of effector and memory T cells [63]. The activity of these cells is believed to be responsible for the observed antitumor effects. The design of DC therapy in ovarian cancer is in the early stages but there have been encouraging findings in the treatment of prostate cancer [64]. DC vaccines are usually prepared ex vivo. Autologous immune cells collected from patients are treated with a range of peptides, mRNA, proteins, tumor lysates or other agents before reintroduction to the patient [63]. The best immunizing antigens are currently unknown but the use of a cell surface associated mucin 1 dendritic vaccination is the most extensively studied and CA125 responses have been seen in response to their use [65]. A Phase II trial (CAN-003) randomizing 56 patients in remission after first or second-line therapy to DC vaccine or standard observational care has demonstrated safe use of a mucin 1 DC vaccine and a significant increase in PFS in patients after second-line therapy (median PFS for observation 4.94 months vs ‘not reached’ but greater than 12.91 months for DC vaccine [p = 0.04]) [66]. A larger randomized Phase II trial called CANVAS has been initiated to further investigate this vaccine's effectiveness in first- and second-line remission [67].
Programed cell death 1 (PD-1) is an inhibitory surface receptor expressed by a number of immune cells including B and T cells that binds to the programed cell death ligand 1 (PD-L1). The expression of PD-L1 in tumors is inversely correlated with survival of patients, as tumor cells are believed to activate the PD-1 inhibitory pathway to evade the immune response. In animal models antibody mediated PD-1 blockade enhanced tumor immunity [68], suggesting that targeted interventions against this pathway may increase antitumor immunity in ovarian cancer and may compliment developments in DC vaccination.
Future perspective
As we begin to understand how different ovarian cancers respond to standard chemotherapy, novel agents and targeted agents, the tailoring of treatment to a cancer's histological and genetic makeup will become increasingly important. Whole-exome DNA sequencing of 316 ovarian cancer samples has shown that TP53 mutations occur in virtually all (96%) patients and that a number of genes are mutated in a subset of high-grade serous ovarian cancers. BRCA1 and BRCA2 had germline mutations in 9 and 8% of cases, respectively, and there were somatic mutations in a further 3% of cases. Six other genes were also shown to be statistically recurrently mutated: RB1, NF1, FAT3, CSMD3, GABRA6 and CDK12 [69]. Although the clinical significance of these mutations has yet to be fully elucidated, it lends credence to the theory that there may be other genetic mutations besides BRCA that could be targeted to improve outcomes. Spatial heterogeneity of cancer mutations within the same person suggests that single biopsies should not be used as the sole specimen when deciding on personalized medicine [70].
The TP53 tumor suppressor gene is a key regulator of cell cycle arrest or cell death upon oncogenic stress. Most TP53 mutations are missense mutations that result in dysfunctional p53 protein allowing cell survival and division. Restoration of wild type p53 function can therefore induce cell death and tumor regression. APR-246 is a compound that can restore wild type conformation to mutant p53. It has been tested in a Phase I/II clinical trial involving 22 patients with hematological malignancies or hormone-refractory prostate cancer. Drug safety has been confirmed and clinical effects have been observed [71]. Due to the frequency of TP53 mutations in ovarian cancer, restoring p53 activity with APR-246 is an area of current research. PiSARRO, a Phase Ib/II study assessing carboplatin combination chemotherapy with or without APR-246 in relapsed platinum-sensitive disease has been designed [72] specifically to investigate the effectiveness of modifying dysfunctional p53 to attain disease control.
A potential barrier to targeted therapy is that the mutations may change over time, such as when the tumor becomes resistant to chemotherapy treatment. Therefore, a method of detecting these changes is important and in early development are techniques such as circulating tumor DNA analysis [73]. The ALFApump® system provides an alternative way to obtain whole cell DNA. It has been developed to continuously drain ascites into the bladder [74] and is currently being trialed in advanced ovarian cancer. An exciting possibility is to continuously monitor the genetic mutations of cancer cells present within urine due to the presence of the pump. As the urine may include cells from multiple tumor sites within the peritoneal cavity it may provide a method to monitor both spatial and temporal genetic change allowing a more comprehensive assessment for personalized medical care.
Executive summary
Primary cytoreductive surgery to no macroscopic disease shows the greatest improvements in 5-year survival.
The benefit of optimal surgical debulking within the peritoneal cavity is present even in patients with stage IV disease.
Neoadjuvant chemotherapy reduces morbidity due to surgery but there are concerns that surgery at a delayed time point may compromise effective surgical disease clearance.
Debulking surgery at relapse may be indicated if complete macroscopic tumor clearance can be achieved. Ongoing trial DESKTOP III.
Carboplatin and paclitaxel three weekly is the standard of care. The intraperitoneal route may be considered in selected patients.
Dose dense paclitaxel given with carboplatin may have a survival advantage. Ongoing research is with ICON8.
Bevacizumab and pazopanib in addition to standard chemotherapy improve progression-free survival.
Intraperitoneal chemotherapy has superior efficacy at the potential cost of increased adverse effects.
BRCA-related disease is more sensitive to platinum-based chemotherapy.
Intraperitoneal platinum chemotherapy may be more effective in BRCA mutation carriers.
PARP inhibitors show an improvement in progression-free survival on initial trials. Further research will be undertaken in the SOLO 1 & SOLO 2 trials.
The majority of patients with advanced ovarian cancer will go on to have relapse of their disease.
There is evidence that CA125 monitoring is not necessary during follow-up.
An asymptomatic CA125 rise with no lesion on imaging represents a prognostic dilemma and current information indicates they do not need treatment until symptomatic.
Rechallenge with a platinum-containing regime is mandatory if tolerated.
The combination of pegylated liposomal doxorubicin and carboplatin is less toxic and likely more effective than carboplatin paclitaxel.
Use of bevacizumab or cediranib show a progression-free survival advantage when given with standard chemotherapy.
Trabectedin is a novel chemotherapy agent with appears most effective in partially platinum-sensitive disease.
Where the platinum-free interval is less than 6 months platinum agents have low response rates.
Chemo-monotherapy is the mainstay of treatment.
The addition of bevacizumab to chemo-monotherapy in the AURELIA study showed a progression-free survival advantage similar to that seen in the adjuvant setting.
Folate-receptor targeted chemotherapy agents are an area of current interest.
Immunotherapy using dendritic cell vaccines is a promising area of future research.
The use of targeted therapies based on the detected mutations of the tumors will be of increasing importance in tailoring anticancer therapy.
TP53 is almost ubiquitously mutated in ovarian cancer and drugs that can modify dysfunctional p53 are an important area of research. Ongoing trial PiSARRO.
Spatial and temporal variation in tumor characteristics and mutations is a significant barrier to effective personalized medicine.
In summary the treatment of ovarian cancer draws on a wide knowledge of expertise. Advances in its treatment are diverse and include new surgical treatments, use of an increasing number of imagining modalities, new uses of existing therapies and the development of novel chemotherapeutic agents and targeted therapies. It is likely that each of these will have an impact on the disease such that there will be a continued improvement in 5-year survival and a gradual increase in the percentage of patients who achieve a long-term cure. The future is promising, and with each step forward the lives of patients will be inexorably improved.
Financial & competing interests disclosure
S Blagden has received an honorarium from Roche and research funding from Merck. The authors have no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed.
No writing assistance was utilized in the production of this manuscript.
