Abstract
Women considering breast ablative therapy are confronted with a variety of decisions that include the type of mastectomy (partial, total, skin sparing, nipple areolar sparing), the type of reconstruction (prosthetic, autologous, oncoplastic) and the need for adjuvant therapy (chemotherapy, radiation). The parameters for each are multifactorial and require significant thought and counseling. Therapeutic options are essentially individualized and dependent upon a variety of factors such as tumor size and location, lymph node involvement, comorbidities, expectations and body characteristics. The role of reconstructive surgery is now well appreciated and an integral component of the multidisciplinary care of the patient and is influenced by the opinions and recommendations of surgical, medical and radiation oncologists. This manuscript will review the role of reconstructive surgery and the many factors to consider.
For many women, the breast represents a symbol of femininity, self-esteem and self-image. The diagnosis of breast cancer can be a significant threat to all of these factors and the possibility of losing a breast can result in depression, withdrawal and feelings related to ‘loss of control.’ The mission of reconstructive surgery is not only to restore a woman's breast but also to minimize the psychological impact of this dreadful diagnosis and to facilitate the transition from being a breast cancer patient to a breast cancer survivor. The journey is not easy as there are many life-altering decisions that need to be considered and made within a relatively short time. This manuscript will focus on factors that women with breast cancer or at high risk for breast cancer should be aware of. This will be reviewed from the perspective of a plastic surgeon with 20 years of experience and expertise in the arena of breast cancer and reconstruction.
Women diagnosed with breast cancer or found to carry the breast cancer gene will have many decisions to make regarding management. First and foremost is that the cancer be completely eradicated to ensure a cancer free survival. The topic of breast reconstruction following total or partial mastectomy is important and is usually reviewed at some point following the initial diagnosis. For some women, the decision to proceed with breast reconstruction is straightforward and without question; however, for others it is complicated and requires significant thought and research. There are several factors that can influence ones decision to proceed with reconstruction that include breast cancer stage, need for adjuvant therapies, as well as ones physical and mental status. Plastic surgeons and breast surgeons should provide information regarding the types of mastectomies performed (skin sparing versus nipple sparing), therapeutic versus prophylactic, reconstructive options (prosthetic devices versus autologous tissue versus oncoplastic), possibility of radiation therapy (RT), postoperative recovery and adverse events.
Surgical options
Women that have been diagnosed with breast cancer will typically opt for breast conservation that involves lumpectomy and radiation. Tumors that are small and not associated with a genetic mutation are most commonly treated with lumpectomy, sentinel lymph node biopsy and radiation. Reconstructive surgery is usually not necessary in this setting. Mastectomy may be necessary when there is tumor that is large in relationship to the breast size, or in multiple areas of the breast or maybe when chosen due to patient preference. Studies by Veronesse as well as Fisher have demonstrated equivalent survival between the two cohorts with 20-year follow-up [1,2].
Total mastectomy can be performed via skin sparing (SSM), nipple sparing (NSM) or areolar sparing approaches. All are safe and effective in properly selected patients [3]. Many women today are considering NSM especially in the setting of prophylactic mastectomy. Although this technique is considered safe and effective, patient selection is important. Ideal candidates include those with small-to-moderate volume breasts, small tumors <3 cm and tumors >3 cm from the nipple areolar complex (NAC). Women with larger tumors that are near the NAC, moderate to severe mammary hypertrophy, or previously radiated breasts may not be appropriate for NSM but may be candidates for SSM. It is important for all women considering total or partial mastectomy to discuss these options with their plastic as well as breast surgeons.
Another patient category is the high-risk patient or the gene carrier patient (BrCa). In these patient prophylactic therapies are considered and can include surveillance that sometimes consists of twice yearly ultrasound, mammography and yearly MRI. Other options include medical therapy consisting of tamoxifen or surgical therapy consisting of prophylactic mastectomy. These strategies can be 90% effective in reducing ones risk of developing breast cancer [4].
Breast reconstruction options
Current statistics in the USA demonstrate that nearly two-thirds of women that have a mastectomy will choose to proceed with reconstruction [5]. Breast reconstruction can be performed immediately following the mastectomy or on a delayed basis that can range from months to years. Some women choose never to have reconstruction for a variety of reasons that include reluctance for additional surgery, fear of tumor recurrence, cultural factors, lack of plastic surgical availability and the feeling that reconstruction is not necessary.
Reconstructive options can be divided into two types, prosthetic and autologous [6,7]. Prosthetic reconstruction is a simpler operative procedure and associated with a shorter postoperative recovery. The downside side is that prosthetic devices do not last forever and at some point in time may need to be removed or replaced. Autologous reconstruction on the other hand is a more complicated procedure with a longer recovery period. The advantage of autologous reconstruction is that when successful, will last forever. The decision as to which to choose will be based on patient factors, surgeon factors and oncologic factors.
Prosthetic reconstruction
Breast reconstruction using prosthetic devices is the most commonly utilized option in the USA and performed in nearly 80% [8]. There are two accepted methods by which this is performed that include the one-stage, also known as direct-to-implant option in which a permanent implant is inserted immediately following the mastectomy or the two-stage option in which a tissue expander is inserted to first stretch or preserve the post-mastectomy skin followed by a permanent implant several months later [9]. It should be remembered that one-stage does not imply 1-operation because revisions are sometimes necessary. Women who are good candidates for the one-stage procedures typically have a BMI <30, small-to-moderate breast volume, good quality mastectomy skin flaps and those having prophylactic mastectomy. Women that are good candidates for the two-stage procedure typically include those that have skin deficiency and will require expansion prior to permanent implant as well as patients with post mastectomy skin quality that is insufficient for a direct to implant reconstruction. The two-stage technique is preformed most often for the above reasons in addition to the fact that most plastic surgeons prefer it because it will provide another opportunity to optimize breast contour and position.
The devices themselves are an important factor for women considering prosthetic reconstruction [10]. Current devices include tissue expanders and implants that are all made with an outer shell composed of a silicone elastomer and an inner compartment that is filled with either saline or silicone gel. Tissue expanders are usually inserted immediately at the time of mastectomy but can also be used on a delayed basis. They are typically partially filled with saline in the operating room. Following the operation, the tissue expander is sequentially filled with saline through the integrated port until the desired volume is obtained. Following complete expansion, the tissue expanders are removed and exchanged for a permanent implant. These permanent implants will vary in terms of filler material (saline or silicone gel), surface (smooth or textured) and shape (round or anatomic). Most plastic surgeons will recommend and most women prefer silicone gel devices because they are soft and have a consistency that more closely resembles the natural breast. A few women however will express concern over the use of silicone gel devices based on the media reports from the early 1990s suggesting that they were not safe. Since then, hundreds of scientific studies have demonstrated that silicone gel implants are safe and effective and not associated with the development of connective tissue disorders, neuropathies, chronic fatigue, cancer or other clinical disorders. As a result, the Institute of Medicine and the US FDA have declared them safe and effective and permissible for use in all women [11]. It should be noted however that these devices will not last forever and that their life-span is typically 10–15 years. Over time, local complications will become more likely to occur and include capsular contracture and rupture that will likely require removal or replacement.
The surface and shape characteristics of the implants are also important factors to consider. Surface characteristics include smooth or textured and shape characteristics include round or anatomic [12]. Round devices can be textured or smooth whereas shaped devices are always textured. The texturing of shaped implants is important to promote adherence and prevent rotation. There are recent data to suggest that textured surface devices may result in less capsular contracture; however, some surgeons still prefer to use the smooth surface devices because they may be associated with less rippling and wrinkling [13]. Most patients that choose to have a shaped implant prefer silicone gel instead of saline. Physical characteristics of shaped implants include increased projection along the lower pole of the breast and improved contouring in the upper pole of the breast. Although these characteristics seem desirable, they are not necessary in all situations. It should be noted however, that shaped devices are composed of a more highly cohesive silicone gel making them firmer to palpation than the round silicone gel devices. This increased cohesivity with shaped devices is necessary for them to retain their shape when implanted.
Another factor to consider with prosthetic reconstruction is whether to have the devices placed partially under the pectoralis major muscle or completely under the muscle. It is estimated that approximately two-thirds of plastic surgeons in the USA perform the partial muscle coverage technique [14]. When the device is placed partially under the muscle, it is important to stabilize the inferior edge of muscle to prevent upward migration known as ‘window-shading.’ This can be achieved by stabilizing the cut inferior edge with sutures to the overlying skin or to use an acellular dermal matrix (ADM). The role of the ADM is to stabilize the position of the pectoralis major muscle, provide tissue support along the lower pole of the breast, provide an elastic environment and to compartmentalize the prosthetic device on the chest wall (Figure 1) [14]. These ADMs are composed of cadaveric materials such as human skin that have been processed to remove all cellular elements and to retain the collagen and elastic structure known as the extracellular matrix. They are inert biologic materials that do not cross-react with the host and may reduce the incidence of capsular contracture in nonradiated patients.
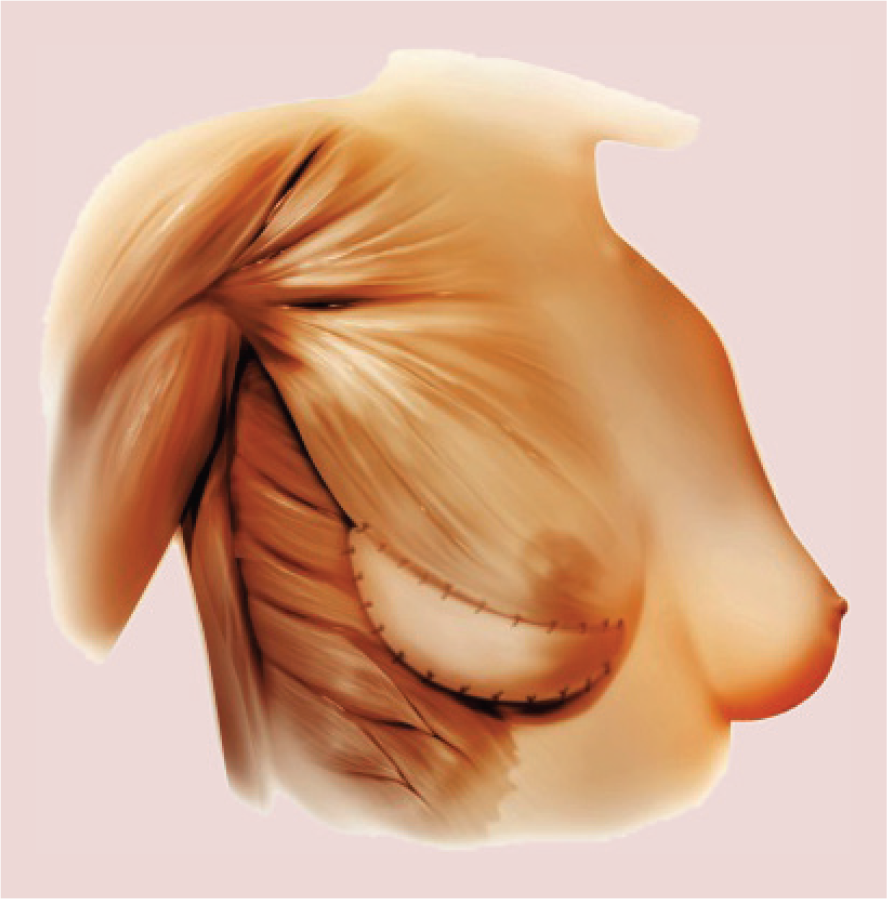
An acellular dermal matrix used along the lower pole of the breast for soft tissue support following mastectomy and reconstruction using a tissue expander or implant.
From a patients ‘perspective, the one-stage or direct to implant operation may seem more desirable than a two-stage operation; however there are factors that preclude this from being performed routinely. Many patients with breast cancer will have a mastectomy that is aggressive in terms the quantity of tissue removed. The skin flaps are often thin with a paucity of normal and desirable subcutaneous fat. A direct to implant reconstruction in this type of patient will more often than not result in the need for secondary procedures; thus, most plastic surgeons will recommend the two-stage procedure. This will provide more control of the reconstruction and ultimately lead to a good-to-excellent outcome. It is the goal of all surgical procedures to provide results that are predictable and reproducible. Figures 2–5 illustrate two scenarios that include two-stage prosthetic reconstruction in the setting of NSM (Figures 2 & 3) and SSM (Figures 4 & 5)

Preoperative image of a woman scheduled for bilateral nipple sparing mastectomy.

Postoperative image following bilateral nipple sparing mastectomy and two-stage device reconstruction.

Preoperative image of a women scheduled for bilateral skin sparing mastectomy.

Postoperative image following bilateral two-stage breast reconstruction and nipple areolar reconstruction.
Monitoring and surveillance of women with breast implants following mastectomy is another important factor to consider [15]. Because the breast parenchyma has been removed, mammography is not usually necessary. MRI or ultrasound is typically recommended when there are oncologic concerns or to assess the integrity of the implants. For women with silicone gel breast implants, the FDA has recommended that imaging be performed at 3-year intervals to assess for device rupture based on the prevalence of silent rupture and the complexities associated with its detection. Because these devices are made with a highly cohesive gel, rupture is not usually associated with gel migration to other regions of the body. Instead, the silicone gel usually remains within the fibrous capsule of the implant; however, migration of silicone gel outside the capsule can sometimes occur in implants places prior to 1990. In the event of a rupture, device exchange or removal is usually recommended.
Autologous tissue
The other principle method of breast reconstruction following mastectomy is to use a woman's own tissues commonly referred to as flap or autologous reconstruction [16]. Through ingenuity and innovation, plastic surgeons have designed a variety of flaps that are currently in use in order to minimize donor site morbidities and to maintain high esthetic quality. The composition of flaps can include skin, fat and muscle. Flaps that include skin and fat only are usually referred to as perforator flaps, for example, deep inferior epigastric perforator (DIEP), superior gluteal artery perforator (SGAP) (Figure 6); whereas flaps that include skin, fat and muscle are referred to as musculocutaneous flaps (e.g., TRAM, LD). Musculocutaneous and perforator flaps are derived from virtually all territories of the body that include the abdomen, posterior and lateral thorax, gluteal region, as well as the medial and posterior thigh. Some of these flaps are robust and can provide large volumes for total breast reconstruction and others are less voluminous and ideally suited for partial breast defects. Given the diversity in flap characteristics and the various donor sites, the ability to properly evaluate and select patients for autologous reconstruction is important. This entails evaluation of a woman's general body habitus, breast volume and donor site considerations.

A deep inferior epigastric perforator perforator flap demonstrating separation of the artery and vein from the underlying muscle.
The indications for autologous reconstruction are variable and include women that do not want prosthetic reconstruction, have a sufficient quantity of donor site tissue, have had a prior failed prosthetic reconstruction or have had prior radiation to the breast or chest wall. This section will review some of the factors associated with flaps based on the anatomic area of origin.
Abdominal flaps
The lower abdominal region has become the preferred donor site for the majority of autologous breast reconstruction procedures and is the source for the pedicle transverse rectus abdominis musculocutaneous (TRAM), free TRAM, DIEP and superficial inferior epigastric artery (SIEA) flaps [17]. The blood supply to these flaps is based on either the inferior or superior epigastric artery or vein.
In order to understand the nature of these flaps, it is important to understand the differences in flap nomenclature. A pedicle flap is defined as one that is transferred to the breast without disrupting the vascular supply, in other words, it is transposed from an adjacent region of the body to the breast, for example, pedicle TRAM flap. A free flap or a tissue transfer is defined as the transfer of tissue from a remote donor site by dividing the artery and vein supplying the flap and then attaching the donor site artery and vein to the recipient artery and vein, in other words, internal mammary or thoracodorsal vessels. Free flaps can be perforator or musculocutaneous and include the DIEP, SIEA and free TRAM. Free flaps all require microsurgery to reattach the small donor vessels to the recipient vessels using an operative microscope, microsurgical instruments and thin sutures. Patients that are considering an abdominal flap should note that the abdomen can be used once; however, it can be used for either a unilateral or bilateral reconstruction. It is not usually possible to stage a reconstruction such that one half of the abdomen is used for one breast at one operation and then to use the other half for the other breast at a later date.
The DIEP flap has become the most commonly utilized option for autologous breast reconstruction [17,18]. It utilizes the skin and fat from the lower abdomen to reconstruct the breast. The advantage of the DIEP flap is that abdominal strength is usually not compromised because the rectus abdominis muscle is not removed and the nerves to the muscle are preserved. However, the muscle is split longitudinally via a myotomy to dissect out the artery and vein. The DIEP flap can be used for women with a low or high BMI as long as the volume requirements of the breast and abdomen are similar (Table 1). In women that have had prior abdominal operations, the ability to perform an abdominal flap may be compromised because of disruption of the blood supply to the flap. In these women as well as women in whom there is some uncertainty about the vascularity to the flap, preoperative vascular imaging may be recommended. There are two commonly utilized imaging modalities that include computerized tomographic angiography and magnetic resonance angiography. The purpose of imaging is to identify the location and caliber of the source vessels as well as the specific perforating vessels to facilitate performing the operation [19]. Figures 7 & 8 illustrate a woman who had a unilateral DIEP flap.
Algorithm for using a deep inferior epigastric perforator, Free transverse rectus abdominis musculocutaneous or superficial inferior epigastric artery flap†

+ = consider; ++ = preferred.
DIEP: Deep inferior epigastric perforator; SIEA: Superficial inferior epigastric artery; TRAM: Transverse rectus abdominis musculocutaneous.


Postoperative image following left breast reconstruction with a deep inferior epigastric perforator flap and staged nipple areolar reconstruction.
The free TRAM is also commonly performed to reconstruct a breast following mastectomy [18]. The free TRAM is distinguished from the DIEP flap in that it incorporates a small segment of the rectus abdominis muscle (Figure 9). The free TRAM is further classified as a muscle sparing based on the amount and location of the rectus abdominis muscle that is removed (Table 2). It is classified as a nonmuscle sparing if the full width but limited length of the muscle is harvested. The question that is commonly asked is whether there is a functional consequence by removing a small segment of the rectus abdominis muscle with the muscle sparing free TRAM compared with the DIEP flap in which no muscle is removed. Comparative studies have demonstrated that there is no difference in abdominal strength between the muscle sparing free TRAM and DIEP flap when used for unilateral breast reconstruction procedures; however then may be a slight reduction in the ability to perform sit-ups with the muscle sparing free TRAM following a bilateral reconstruction [18]. The decision to perform a muscle sparing free TRAM is made preoperatively in women that are obese or have had certain abdominal procedures and intraoperatively in women that have an insufficient system of perforators. The reason that a free TRAM is preferred in these cases is that a network of 3–5 perforators is typically included with the small segment of muscle rather than just one or two perforators typically dissected with the DIEP flap. Figures 10–12 illustrate a woman that had a bilateral free TRAM flap breast reconstruction.
Classification of muscle sparing.

DIEP: Deep inferior epigastric perforator; MS: Muscle sparing.

A muscle sparing free transverse rectus abdominis musculocutaneous flap demonstrating a small segment of the rectus abdominis muscle.

Preoperative image of a woman scheduled for bilateral mastectomy.

Preoperative markings for a skin sparing mastectomy and muscle sparing free transverse rectus abdominis musculocutaneous flap reconstruction.
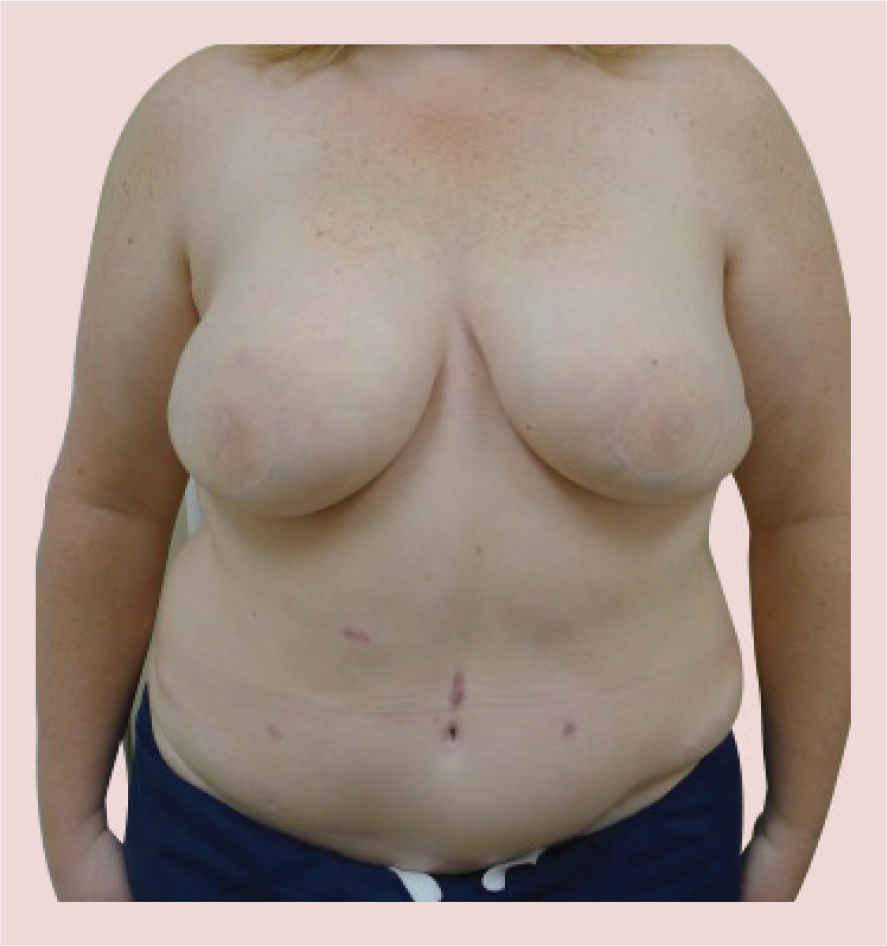
Postoperative image following bilateral breast reconstruction with muscle sparing free transverse rectus abdominis musculocutaneous flaps.
The pedicle TRAM flap was the first abdominal flap described and was introduced in the early 1980s. It includes the same skin and fat territories as the free TRAM and DIEP flap but requires removal of the entire length and sometimes width of rectus abdominis muscle (Figure 13). The reason to remove the muscle with the pedicle TRAM is because the superior epigastric artery and vein courses through it; thus, the muscle essentially serves as a carrier for the blood supply. The flap is typically tunneled under a skin bridge between the abdomen and breast into the breast pocket. Because the blood supply is not disrupted, a vascular anastomosis is not necessary. The disadvantage of the pedicle TRAM compared with the muscle sparing free TRAM or DIEP flap procedures is that there is usually a functional consequence because the muscle is removed. Abdominal weakness and postoperative contour abnormalities may be more likely. In order to minimize the incidence of abdominal contour abnormalities associated with the pedicle TRAM, the use of a mesh material is sometimes necessary. These materials are occasionally needed with the free TRAM and rarely needed with the DIEP flap. In some women that may have a larger abdominal girth or be overweight or obese, a delay procedure may be recommended. With a surgical delay procedure, the deep inferior epigastric artery and vein are ligated 2 weeks prior to the definitive operation in order to optimize flap perfusion via the superior epigastric artery and vein. This maneuver has been demonstrated to reduce the incidence of fat necrosis within the flap. Figures 14 & 15 illustrate a patient following a unilateral pedicle TRAM flap breast reconstruction.

A muscle-sparing pedicle transverse rectus abdominis musculocutaneous flap.

Preoperative image of a woman scheduled for right mastectomy.
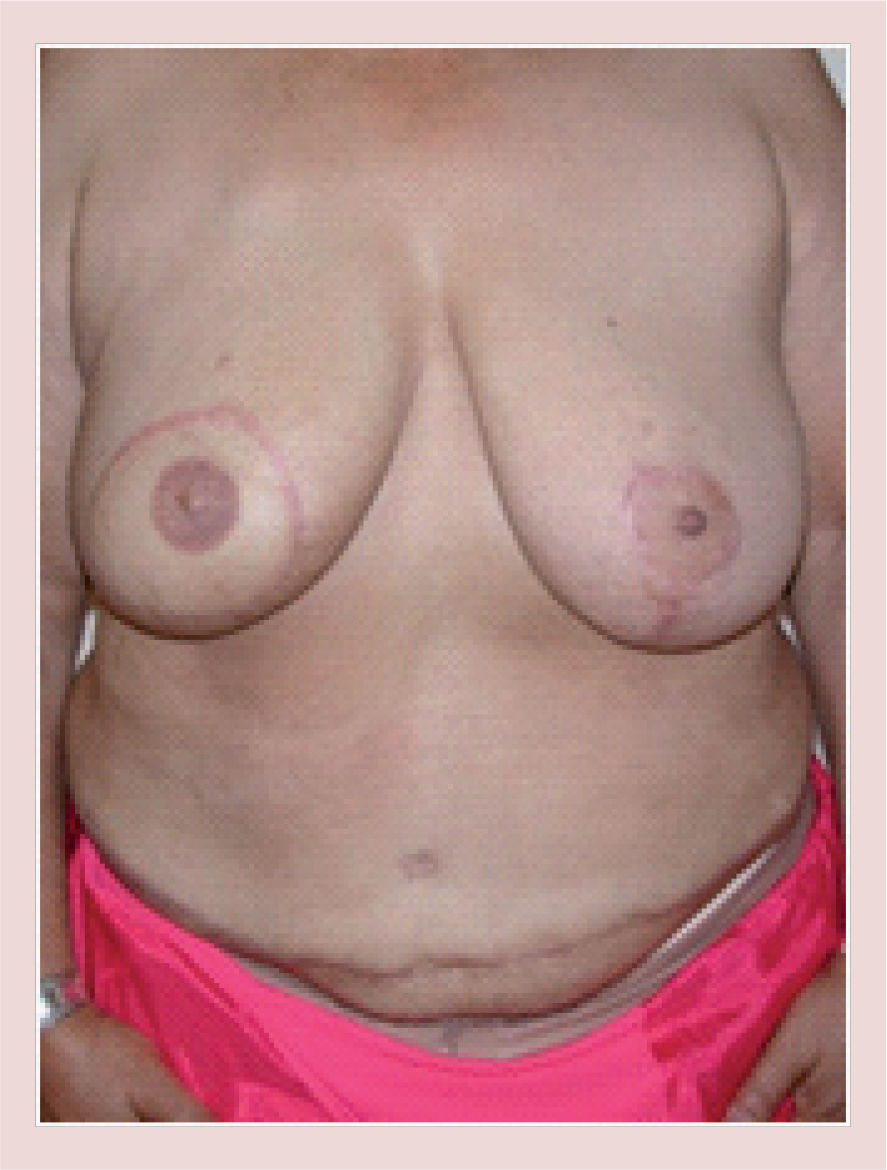
Postoperative image of a woman following right breast reconstruction with a pedicle transverse rectus abdominis musculocutaneous flap.
The SIEA, unlike the pedicle TRAM, free TRAM and DIEP flap does not violate the supportive structures of the anterior abdominal wall. It does not require an incision in the anterior rectus sheath or the rectus abdominis muscle. Thus, the SIEA flap represents a true abdominoplasty flap. Unlike TRAM or DIEP flap that derive their blood supply from the superior and deep inferior epigastric vessels, respectively, the SIEA flap derives its blood supply from an entirely different source, the superficial inferior epigastric vessels. The principal disadvantage of this flap is that the blood supply is unpredictable and is usually associated with a smaller caliber vessel. Thus, most surgeons agree that it can be performed only 50% of the time at best. Because the vessels are smaller, it is also associated with a higher reoperation rate due to vascular complications that can result in flap failure.
Latissimus flaps
The principal flap derived from the back is the latissimus dorsi (LD) musculocutaneous flap [20]. This flap has been performed since the mid 1970s and is considered to be one of the most reliable and predictable. The LD flap requires the removal of part or all of the LD muscle. This muscle is principally used for extension and adduction of the arm. Removal of the muscle typically does not interfere with arm movement but will result in some limitation of forceful arm movements (pull downs, rowing motions). Many surgeons will recommend and use this flap for total as well as for partial breast reconstruction. Because the flap is typically associated with less volume that its abdominal counterpart, it is usually combined with an implant. Figures 16–18 illustrate a woman following bilateral breast reconstruction using an LD flap.

Preoperative image of a patient following partial bilateral nipple sparing mastectomy without reconstruction.

Preoperative markings in preparation of a bilateral latissimus dorsi flap.

Postoperative image following bilateral latissimus dorsi flap breast reconstruction.
There are several methods by which this flap can be harvested. The most common technique involves excising an ellipse of skin and fat from the right or left back and then removing the LD muscle with it. A muscle sparing LD flap can also be performed by only removing the portion of the muscle that is in continuity with the fat. The LD flap can also be harvested endoscopically through the anterior mastectomy incision. With this technique, no skin is removed from the back and only the muscle and some overlying fat are harvested. This is typically performed in the setting of a skin sparing or nipple-sparing mastectomy. Finally, there is a perforator version of this flap called a thoracodorsal artery perforator flap (TDAP). This flap is typically used for partial breast reconstruction and used for inferior or laterally based breast defects.
Gluteal flaps
Gluteal flaps are usually considered when there is insufficient skin and fat in the abdomen. There are two general categories of gluteal flaps that include musculocutaneous and perforator based. Most surgeons that perform gluteal flaps are using the perforator versions that include the SGAP and the inferior gluteal artery perforator (IGAP) flaps [21]. These flaps are derived from the upper and lower buttock region respectively. Both flaps are generally used to reconstruct breasts of mild to moderate volume (300–600 g). The SGAP flap is considered by most microsurgeons to be one of the more difficult flaps to harvest because the artery and vein are located deep in the gluteal region and are usually thin in caliber and short in length. This flap will tend to lift the buttock and will cause some degree of asymmetry and distortion when performed unilaterally. The IGAP flap is typically harvested in the crease of the lower buttock. A criticism of the IGAM is that it may result in more thigh and leg pain because of its proximity to the sciatic nerve. One advantage of gluteal flaps regardless of the type is that they can be performed unilaterally or bilaterally and either simultaneously or staged because the flaps are not in continuity with each other. Abdominal flaps on the other hand can only be performed once for a given patient because the right and left halves are in continuity. Figures 19–21 illustrate a woman following staged bilateral breast reconstruction with SGAP flaps.

Preoperative image of a woman prior to mastectomy and staged superior gluteal artery perforator flaps.

Postoperative image of the gluteal scars following bilateral superior gluteal artery perforator flaps.
Thigh flaps
The medial and posterior thigh donor sites have also demonstrated success for breast reconstruction. These flaps are again considered alternatives to the abdomen in the event that the abdomen is not suitable. There are several flaps based around the thigh region that have been described and include the transverse upper gracilis, diagonal upper gracillis and transverse musculocutaneous gracilis and the profunda artery perforator flaps [22]. These flaps are all capable of providing 200–500 g of tissue and are useful for mild-to-moderate volume breast reconstruction. Potential adverse events associated with the transverse upper gracilis flaps include postoperative lymphedema, complex scarring and thigh distortion. The advantage of the diagonal upper gracillis is to minimize disruption of the superficial lymphatic vessels and to conceal the incision when viewed from the frontal and lateral sides. The advantages of the profunda artery perforator flap over gluteal flaps and medial thigh flaps are that lymphedema risk is minimal, pedicle length is increased and gluteal contour is not affected.
Oncoplastic surgery
Oncoplastic surgery represents the most recent option in the reconstructive armamentarium of plastic and breast surgeons. This option is frequently considered in women who require a large resection of breast tissue in order to complete their breast cancer resection rather than total mastectomy. Breast conservation therapy (BCT) is the most common form of cancer ablation and accounts for approximately 2/3 of women. The primary limitation of BCT is that 20–40% of women will have a contour abnormality especially following the RT. The principle behind oncoplastic surgery is that following the partial mastectomy, the adjacent tissues are rearranged to prevent or minimize any contour abnormality [23]. This may be in the form of a mastopexy, breast reduction or by using a flap such as the TDAP or LD. This can occur either immediately following the partial mastectomy or on a staged basis. The staged approach is to ensure that the pathologic margins of the specimen are free of tumor and is usually performed 1–2 weeks later and before the RT. The best candidates for oncoplastic surgery are usually women with larger breasts in whom the lumpectomy defect can be easily corrected using reduction mammaplasty techniques. In women with smaller breasts, adjacent breast tissue rearrangement may not be possible and therefore distant tissues such as the LD flap are considered. Figures 22 & 23 illustrate a woman following oncoplastic reduction mammaplasty.

Preoperative image of a woman with left breast cancer scheduled for partial mastectomy and oncoplastic reconstruction.

Postoperative image following bilateral oncoplastic reduction mammaplasty and localized left breast radiation therapy.
Radiation therapy
RT is sometimes recommended for women with breast cancer treated with mastectomy to destroy microscopic cancer cells that may reside following surgical removal of the breast and primary tumor. The indications for RT following mastectomy are expanding and based on the number of positive lymph nodes, the size of the primary tumor and the margin status following excision. If four or more lymph nodes are positive for cancer, there is good consensus among physicians and RT is recommended. However, with larger tumors (2–5 cm) with 1–3 positive lymph nodes, a consensus regarding RT is lacking. In this cohort, there are no randomized controlled trials demonstrating efficacy; however, there are subset analyses from the Danish Breast Cancer Trial and the British Columbia randomized clinical trials that have demonstrated improvement in survival associated with post mastectomy radiation for patients with 1–3 positive nodes [24,25]. Smaller tumors (<2 cm) as well as specimens with close margins may also be considered appropriate for RT. RT is usually targeted to include the chest wall and breast as well as the axillary, internal mammary and sometimes the infraclavicular lymph node basins.
Although RT is effective at destroying cancer cells, it can also adversely affect normal cells and affect the vascularity, elasticity and texture of the remaining skin and underlying tissues. These changes can make breast reconstruction more challenging. The early appearance of the radiated breast includes erythema, desquamation and inflammation. Controversy exists as to whether or not immediate or delayed reconstruction is beneficial in cases where RT is recommended. When RT is administered to a breast that has been reconstructed with a prosthetic device or autologous tissue, there can be negative consequences [26–29]. In the event of prosthetic reconstruction; capsular contracture, displacement and discomfort can occur. With autologous reconstruction; flap shrinkage, distortion and indurations have been described. As a result, there are several strategies that have been popularized to minimize the likelihood of adverse events associated with RT and breast reconstruction.
Many plastic surgeons and radiation oncologists agree that it is preferable to radiate a prosthetic device (tissue expander, implant) rather than a flap although this remains controversial. The reason for this is because devices can be exchanged or modified, whereas it may be more difficult to revise a radiated flap. There are several strategies to minimize the untoward effects of RT on the reconstructed breast. Some women are not interested or may not be candidates for flap reconstruction. In these situations, several strategies can be considered. Some surgeons will place a permanent implant at the time of the mastectomy and proceed with radiation. Other surgeons may place a tissue expander at the time of the mastectomy and exchange this for a permanent implant either before or following radiation.
In women that ultimately desire a flap, a commonly utilized strategy is the delayed-immediate approach. In women in whom the likelihood of radiation is high (>50%), a tissue expander is placed following the mastectomy. Following the expansion and completion of the RT, the tissue expander is removed and replaced either with a flap or a permanent implant. In the event that the tissue expander is replaced with a permanent implant, the timing of the operation is variable. Some surgeons prefer to wait 6–12 weeks following the RT as this interval corresponds with the subacute phase of radiation injury and will permit some degree of soft tissue manipulation to positively impact the quality of the final reconstruction. Other surgeons prefer to wait 6–12 months to ensure that post radiation inflammatory response has subsided. In the event that the tissue expander is replaced with a flap, the operation usually occurs 6–12 months following the RT. The rationale for this is to allow the inflammation surrounding the source vessels of the flap as well as the recipient site to subside. Premature flap reconstruction in the setting of previous RT may result in more complications including flap failure.
Another strategy is to delay the reconstruction until the RT is completed. In this situation, reconstruction with prosthetic devices is usually not advised because the tissues have become more fibrotic and less elastic. Tissue expansion in these situations is fraught with more complications including capsular contracture, pain, breast distortion and chest wall/rib distortion. It is therefore preferred by the majority of plastic surgeons to use autologous tissues. This can be performed with either free or pedicle flaps from either the abdominal, thoracic, gluteal or thigh donor sites. Some surgeons are recommending fat grafting to rebuild the breast that will be described in the following section.
Fat grafting
Fat grafting has added another treatment modality in the armamentarium of the plastic surgeon [30]. It has been used successfully to correct contour abnormalities of the reconstructed breast, improve the quality of radiated or damaged skin and enhance the volume of flaps. The technique involves the aspiration of fat from a remote area of the body such as the abdomen or thigh region, processing of the fat to remove excess fluids and oils, then injection of the fat into the specific area in question. It is more common than not to require several treatment sessions for fat grafting especially in previously radiated areas. Because the fat is transferred without a blood supply, it has to be acquired from the recipient site. Some of the fat will acquire a blood supply and survive; the body will resorb the fat that does not. Hence, subsequent fat grafting procedures are usually needed in order to achieve the desired result. Figures 24 & 25 illustrate a woman following fat grafting in the setting of autologous reconstruction and radiation.

Preoperative image of a woman following right breast reconstruction and radiation therapy demonstrating a upper pole fat deficiency.
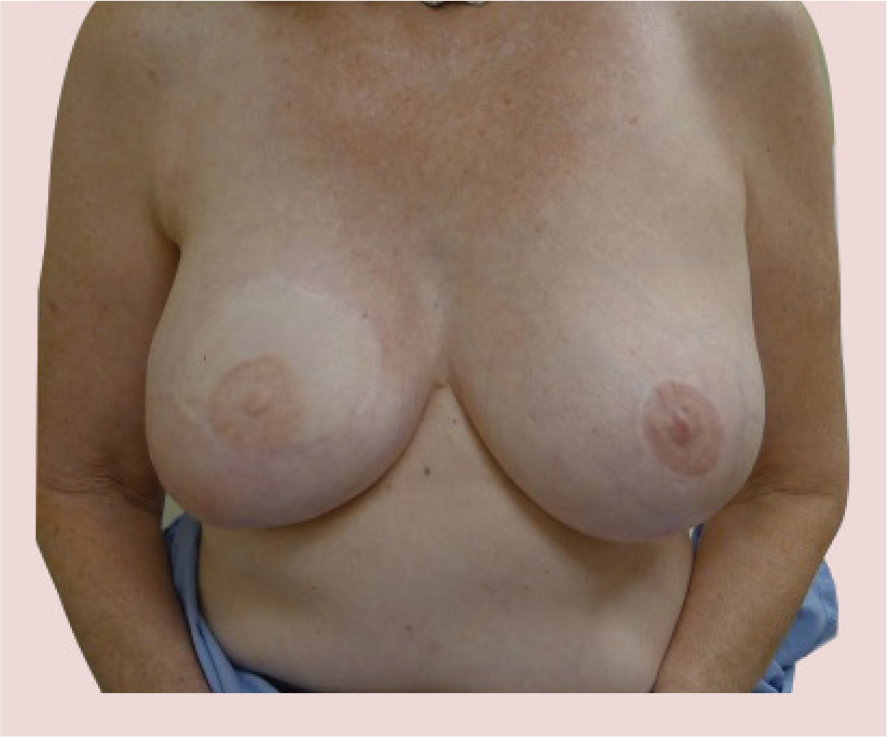
Postoperative image following the third session of fat grafting demonstrating an improvement in contour.
Some plastic surgeons have used these fat grafting techniques for total breast reconstruction. In these situations, the use of an external expansion system known as breast augmentation with vacuum assistance (BRAVA) is used and applied over the mastectomy site. The device is utilized for 10 weeks for 10 h/day. The proliferation of connective tissue and vascularity creates an environment in which the grafted fat may more likely acquire a blood supply and survive. There are other components to this technique that are beyond the scope of the article that can be accessed in the literature. Currently, there are only a few surgeons that use this technique as it is still being evaluated for safety and efficacy.
Postoperative recovery
The postoperative recovery following breast reconstruction will depend upon the nature of the reconstruction, prosthetic or autologous. Autologous reconstruction is considered the more complex procedure and therefore associated with the more lengthy recovery. Most women that have flap reconstruction will remain in the hospital for 2–5 days depending upon the specific flap. Within the hospital, the flap will be monitored for intervals that range from 1–4 h to assess circulation and viability. This is usually performed with a hand-held Doppler to listen to the arterial and venous signals within the flap or by using an implantable or external monitoring device. Pain management typically is achieved using a patient controlled anesthesia in which the patient will administer a predetermined narcotic dose at specified intervals. Patients are encouraged to get out of bed and begin ambulating on postoperative day (POD) 1. Women usually resume their normal diets the morning following surgery. Physical and occupational therapy consultations are typically obtained. Bandages are usually removed on POD 2.
With prosthetic reconstruction, women usually remain in the hospital for <24 h. Pain managements, antibiotics, diet and therapy instructions are similar to the flaps. Medication to relieve spasm of the pectoralis major muscle is also usually prescribed. It should be remembered that tissue expanders are partially or totally under the muscle and partially filled with saline in the operating room so the postoperative breast volume may be less than expected. These expanders are filled to completion during the postoperative visits. Some women may have direct to implant reconstruction. In these cases the breast volume is typically restored. In situations where a nipple-sparing mastectomy has been performed (prosthetic or autologous), the viability and circulation of the nipple is assessed on POD 1. If at all compromised, appropriate intervention is performed and may include suture removal, application of Silvadiene (topical antibiotic) or reoperation.
Upon discharge, most women are advised to restrict exertional physical activity for 4–6 weeks to allow for ample time for the incisions to heal. Most women will have drains placed in the operative sites that typically remain in place for 1–2 weeks. All patients are typically discharged to their home with medication to relieve pain and some women will be prescribed oral antibiotics to lessen the risk of infection. The first postoperative visit is usually within 1 week. At that time, drain output is assessed and sometimes removed. Drain removal is usually based on duration and output. They are typically removed when the output is <30 cc/24 h for 2 consecutive days. Women are advised to return to work somewhere between 2 and 8 weeks following breast reconstruction depending on the nature of the reconstruction and the speed of their recovery.
Complications
The topic of complications or adverse events is another important factor for all women to consider prior to breast reconstruction. There is no surgical procedure that is without risk. Fortunately, complications are relatively uncommon; however, of those that do occur the ones that are common to all include bleeding, infection and scar. The nature of the scar will depend on patient age, skin tone and location. Normal scars will manifest as a fine line but abnormal scars can be raised, discolored, wide, hypertrophic or keloid. Bleeding is also uncommon but the likelihood is increased in women with bleeding tendencies or disorders such as factor 5 deficiency, platelet abnormalities, hemophilia and von Willebrand disease. Drains are sometimes inserted into the operative site to help identify abnormal postoperative bleeding and to reduce fluid accumulations. Infection typically occurs in less than 5% of cases and is often caused by the patients' own bacteria. Infection can manifest as a cellulitis (skin infection) or a deep space infection (abscess). Abscess will often require surgical drainage whereas a cellulitis may respond to antibiotics alone. Almost all patients will receive preoperative antibiotics just prior to surgery and many patients will receive postoperative antibiotics upon completion of the operative procedure. With nipple sparing mastectomy, it is important to remember that the nerves supplying the NAC are divided, therefore, the NAC will be numb. Some sensation may return over the ensuing months to years, but it rarely returns to normal.
With prosthetic reconstruction, it is important to recognize that implanted devices do not last forever. They are generally good for 10–15 years before requiring removal or replacement. Adverse events that warrant replacement or removal may include capsular contracture, rupture, distortion and pain. Capsular contracture often manifests as a progressive hardening of the implant that may result in distortion, displacement and pain. The treatment is usually surgical and includes capsulectomy or capsulotomy. Rupture of implants is also uncommon but the likelihood will increase with the age of the implant. Rupture becomes more of a concern with devices that are over 10 years of age. With saline devices, the breast will simply deflate as the saline is absorbed. With silicone gel devices, the rupture may be more difficult to detect and is commonly referred to as ‘silent rupture.’ For this reason, the FDA has recommended MRI of silicone gel implants every 3 years. It is important to remember that these devices are not harmful and have not been associated with connective tissue disorders, neuropathy, cancer or chronic fatigue based on scientific studies.
With autologous reconstruction, the most dreaded complication is flap failure. In this situation, the transferred skin, fat and muscle (when included) dies usually because of abnormal blood circulation. This may be due to occlusion of the artery or vein. In the event of a free tissue transfer or a pedicle flap, an expeditious return to the operating room is required in order to attempt flap salvage. If the flap is deemed nonsalvageable, then it is removed and another form of breast reconstruction is considered. Fortunately, flap failure occurs less than 2% of the time in the hands of experienced plastic surgeons. Other flap related complications include fat necrosis, partial flap necrosis and delayed healing all of which are related to abnormal perfusion that is localized rather than diffuse. The donor sites may also be susceptible to adverse events. With the abdomen, there are risks of abnormal contour that may manifest as a bulge or hernia. There can also be abdominal weakness depending on the amount of rectus abdominis that is sacrificed or damaged. These occurrences range from one to 10%. The primary complication with the LD flap is a seroma or watery fluid accumulation. This typically can occur in 20–30% of cases and for this reason, two drains are typically used and left in place for 2 weeks or so. With the gluteal and thigh flaps, the primary risks include flap failure, contour irregularity, lymphedema and complex scars.
Secondary procedures
It should be noted that following any breast reconstructive procedure, secondary operations might be necessary [31,32]. This may be because of a complication such as infection, flap failure or device failure or it may be to improve the symmetry between the two breasts. Fortunately, insurance carriers following the Women's Health and Cancer Rights Act of 1998 usually cover these procedures. Secondary procedures may be necessary for the reconstructed breast or the nonreconstructed breast. These procedures may include revision of a reconstruction such as contouring, fat grafting, implant exchange or capsule modification. In the case of a unilateral reconstruction, the contralateral breast may also need an operative procedure for symmetry that can include a mastopexy or breast reduction.
Nipple reconstruction is also a secondary procedure that essentially completes the reconstructive process [33,34]. Creation of a nipple is relatively simple and completed by various techniques that include small incisions and creation of local flaps to create a nipple on the breast or it may be via a nipple sharing procedure from the opposite breast in a women with a prominent nipple. Skin grafting from the thigh can also be performed but is less common that the other methods described. Areolar color is usually achieved using tattoo (Figures 5, 8, 15, 20 & 25). Women with prosthetic reconstruction that have been radiated are usually discouraged from nipple reconstruction because of the increased risk of delayed healing and infection. In these patients, areolar tattooing is recommended as a sole procedure. In some centers, 3D nipple areolar tattooing is performed. The principle limitation of most nipple reconstruction technique is flattening due to scar contracture. Fortunately, nipple reconstruction can be repeated using various filler materials.
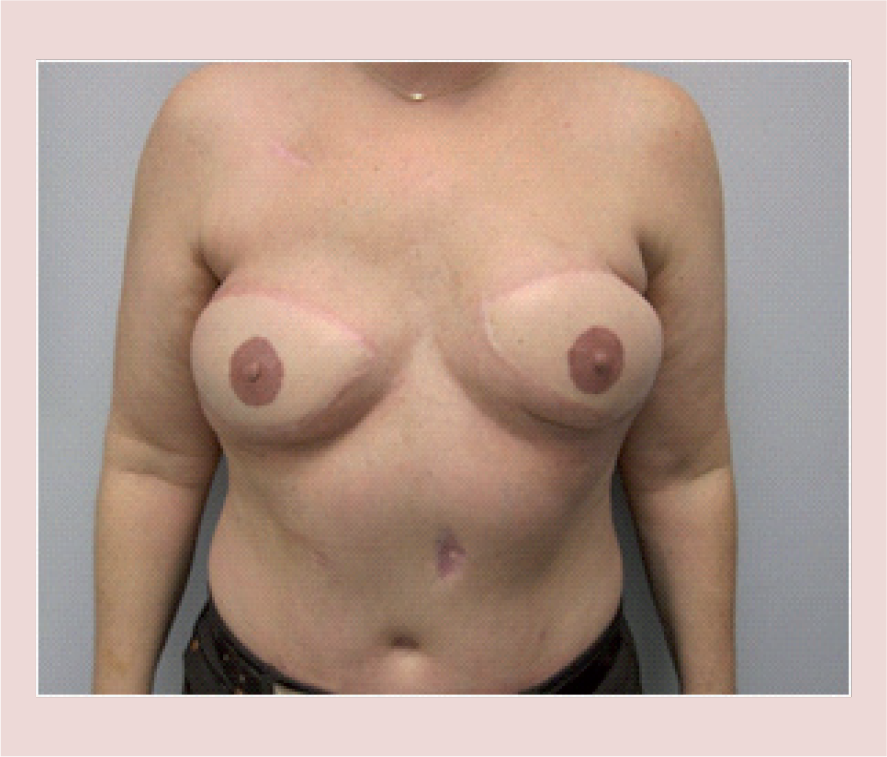
Postoperative image following staged bilateral breast reconstruction with superior gluteal artery perforator flaps.
Conclusion
In summary, advances in breast reconstruction over the past few years have generated numerous options. Women can choose prosthetic or autologous reconstruction and achieve excellent esthetic outcomes. With the prevalence of nipple sparing mastectomy and direct to implant reconstruction on the rise, the number of subsequent operations can be reduced. It is important for all women diagnosed with breast cancer to consult with a plastic surgeon to review the options for reconstruction and to determine which of these reconstructive options would be appropriate. These operations are safe and effective and have been demonstrated to improve the quality of life for many patients that have gone through this process.
The journey from becoming a breast cancer patient to a breast cancer survivor can be frightening and difficult. The process is long and arduous. The mission of the comprehensive breast cancer teams across the country is to make this journey a smooth and strengthening experience. Our goal is to evaluate each woman, discuss all options in a simple fashion with photographs and provide the best reconstruction that will satisfy their emotional, psychological and physical requirements.
Future perspective
The nature of Plastic and Reconstructive Surgery is to innovate and to strive for perfection. This is certainly true when one considers the advancements over the past 20 years that include perforator flaps, acellular dermal matrices and oncoplastic surgery. The next 10 years should be just as exciting. Regenerative medicine is still in its early stages of evolution. The role of fat grafting and stem cell therapy may one day allow surgeons to reconstruct a breast with minimally invasive techniques. Using external expansion to stretch and vascularize the chest wall or breast tissue, fat grafting for total breast reconstruction may soon become commonplace. Acellular dermal matrices may one day be created as 3D constructs and used for partial mastectomy defects. The integration of these matrices with growth factors such as VEGF, TGF and EGF as well as stem cells may permit breast surgeons to prevent contour abnormalities following breast conservation. Another fascinating concept is the ability to decellularize fat and to be able to use it as a construct for fat/breast regeneration.
Executive summary
Prosthetic reconstruction accounts for approximately 80% of all breast reconstruction procedures in the USA.
Two-stage reconstruction with tissue expanders followed by permanent implants is considered to be more predictable and reproducible.
Acellular dermal matrices provide tissue support, implant compartmentalization, an elastic space and may minimize the incidence of capsular contracture.
Women with prosthetic devices are encouraged to have surveillance every few years to assess for silent rupture.
The abdominal donor site is most common and includes the deep inferior epigastric perforator, transverse rectus abdominis musculocutaneous and superficial inferior epigastric artery flaps. Each can create a natural shape but differ in their donor site morbidity profile.
The latissimus is a reliable flap that does not usually require microsurgery. It can create a natural breast but often requires use of an implant.
Gluteal and thigh flaps are alternative donor sites that require microsurgical techniques.
Oncoplastic reconstruction involves partial breast excision with glandular remodelling prior to radiotherapy.
Oncoplastic techniques include tissue rearrangement, mastopexy, reduction mammaplasty and local flap reconstruction
Radiation therapy is always recommended for tumors >5 cm and when greater than four lymph nodes are positive.
Radiation therapy can have adverse consequences on breast reconstruction that include capsular contracture with devices and flap shrinkage with flaps.
Fat grafting has been demonstrated to correct contour abnormalities, improve the quality of radiated skin and to correct volume discrepancies.
Its benefit is primarily based on the presence of stem cells obtained within the lipoaspirate.
All reconstructive techniques can result in complications. The most common include infection, bleeding and scar.
Nipple areolar sparing mastectomy may result in loss of sensation, asymmetry, necrosis and removal.
Prosthetic reconstruction can result in capsular contracture, displacement, rupture and premature removal.
Autologous reconstruction can result in total or partial flap failure as well as donor site morbidities such as a bulge, hernia and muscle weakness.
Secondary procedures are sometimes necessary following breast reconstruction to achieve symmetry and to correct complications.
Prosthetic devices continue to improve and may one day be customized to ideally match the natural breast. Device failure may be minimized such that they last forever without worrying about rupture or capsular contracture. The various implant shapes, surface textures and filler characteristics continue to improve. Air filled tissue expanders are on the horizon that can be used instead of the traditional saline-filled tissue expander. The expansion is automated so the patient does not need to make weekly visits to the plastic surgeon for expansion. Technique modifications are also evolving. As tissue perfusion techniques continue to evolve, we can better assess vascularity following mastectomy allowing surgeons to perhaps place devices in the subcutaneous space rather than the submuscular space that may reduce animation deformities.
The number of autologous options has expanded significantly over the past 10 years and will certainly continue. Surgeons can now remove tissues from virtually anywhere on the body to recreate a breast. Fabricated flaps are possible and can be constructed based on desired location. Microvascular success continues to improve and will soon reach 100% given advancements with vascular couplers and monitoring techniques.
With continued research and the support of industry, the specialty of plastic and reconstructive surgery will continue to provide patients with options that will optimally fulfill expectations and outcomes.
Financial & competing interests disclosure
The author has no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
No writing assistance was utilized in the production of this manuscript.
