Abstract
Keywords
Infiltrating ductal carcinoma (IDC) and infiltrating lobular carcinoma (ILC) are the two most common histological subtypes of invasive breast cancer, with IDC occurring in more than three-quarters of patients [1,2]. Accordingly, treatment guidelines in breast cancer are mainly concluded from the outcome of patients with IDC, who markedly dominate the breast cancer population worldwide. Currently, both ILC and IDC subtypes receive the same treatment recommendations, depending mainly on stage and phenotype of the disease, without taking into account the potential impact of histological subtypes [3,4].
At presentation, ILC is rather difficult to detect, both clinically and radiologially. It is associated with an increased propensity for multicentric distribution and bilaterality [5–8]. Compared with IDC, ILCs are more commonly low grade, have positive estrogen receptor (ER) expression and poor response to neoadjuvant therapy [1,2,8–10]. These clinically relevant differences between ILC and IDC would certainly indicate that breast cancer histology has a true impact on tumor behavior, which is not fully explained at present.
Scarce data are available from the Middle East region regarding the incidence, clinicopathological features and treatment outcome of ILC patients. Hence, we conducted the current retrospective study to investigate the epidemiology, pathological features and outcome of ILC in Egyptian breast cancer patients, and discuss how these results compare with other parts of the world.
Patients & methods
We retrospectively identified all women with a diagnosis of non-metastatic invasive breast cancer, who were registered in the Cairo Oncology Center database between January 2000 and December 2008. Patients with IDC or ILC histology according to the criteria described by the WHO classification were included in the current study. Other histological subtypes including mixed ILC and IDC were excluded. All eligible patients had primary curative surgery for their disease. Patients referred for neoadjuvant treatment were excluded. ERs and progesterone receptors (PgR) were considered positive if greater than 1% of tumor cells showed expression by immunohistochemistry (IHC). HER2 was considered positive if scored 3+ by IHC or positive by FISH or silver in situ hybridization test. None of the patients in the current study had assessment of their proliferation marker, Ki-67, as the test was not routinely performed in Egypt at the time the study was performed. Hence, breast cancer luminal subtypes were defined using IHC surrogates based on the expression of ER, PgR and HER2 as follows:
Luminal A (favorable) group: ER+, PR+, HER2-
Luminal B (unfavorable) group: ER+, PR-, HER2-or ER+, any PgR, HER2+.
According to the treatment guidelines in our institution during the study period, adjuvant chemotherapy, endocrine or anti-HER2 therapy and radiotherapy were applied equally to patients with ILC and IDC.
Patient and tumor characteristics were analyzed separately for both IDC and ILC including age, menstrual status, bilaterality at presentation, pathological TNM staging (7th edition), disease phenotype, type of surgical treatment as well as time of first relapse and pattern of disease recurrence. Data on histological grade of ILC was not available in the vast majority of cases (88%) and was not analyzed in the present study.
Formal ethics committee approval was not required for conducting this retrospective analysis. However, handling of data and respecting patients confidentiality was followed per the principles outlined in the Declaration of Helsinki.
Statistical analysis
Descriptive statistical analysis was carried out to evaluate patients' demographics and clinical characteristics. The association between selected patient and tumor characteristics with the pathological subtype was assessed using the Chi-square test. We evaluated the cumulative incidence of breast cancer-related events, which included: locoregional disease recurrence, distant metastasis and contralateral breast cancer. The primary endpoint of this study was disease-free survival (DFS), which was defined as the time from the date of surgery to the date of any of the abovementioned breast cancer-related events or death of any cause or date of last follow-up. Survival plots were drawn using the Kaplan–Meier method [11]. The logrank test was used to assess the difference in DFS between strata. Multivariate Cox proportional hazards regression analysis was used to assess the independent prognostic significance of various clinical and histopathologic characteristics of the tumor on DFS [15]. All analyses were performed using SPSS statistical software (version 17.0).
Results
Patient characteristics
In the period between 2000 and 2008, there were 2070 women who were registered in our database as having a primary curative breast surgery with M0 disease, of whom 1758 were classified as IDC (85%), while 176 (8.5%) were classified as ILC. The remaining cases were diagnosed with other histological subtypes (including 52 cases with mixed ILC and IDC) and were excluded from further analysis (Table 1).
Histological subtypes.
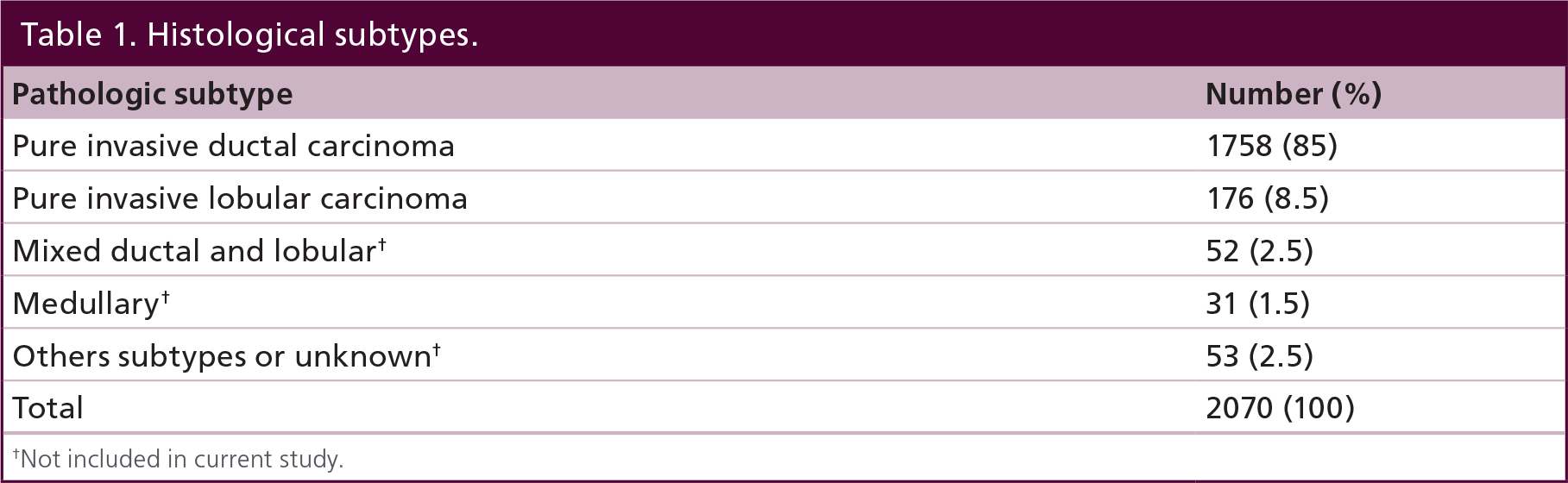
Not included in current study
The median age was the same in both groups (50 years), although a significantly lower proportion of patients with ILC were diagnosed below 35 years of age (3.4 vs 9.3% with IDC; p = 0.009). Bilaterality at presentation was encountered more frequently in patients with ILC compared with patients with IDC (4% and 1%, respectively; p = 0.001). Patients with ILC had a more advanced T stage (20.3 vs 13.4%; p = 0.027), and more positive nodal involvement (67.3 vs 60%; p = 0.004). Mastectomy was offered more frequently to ILC patients compared with those with IDC (79.4 vs 67.7%; p = 0.002). Compared with patients with IDC, ILC patients had more ER-positive disease (83.5 vs 70.3%; p < 0.001) and PgR-positive disease (75 vs 60%; p < 0.001), but less HER2 positivity (9.9 vs 25.9%; p < 0.0001). ILC was significantly associated with less unfavorable luminal phenotypes (8.1 vs 16.9%; p < 0.001) and less HER2-enriched disease (2.7 vs 11.5%; p < 0.001) (Table 2).
Clinicopathological characteristics according to histologic subtype.

BCS: Breast conservative surgery; ER: Estrogen receptor; IDC: Invasive ductal carcinoma; ILC: Invasive lobular carcinoma; MRM: Modified radical mastectomy PgR: Progesterone receptor.
Follow-up & survival analysis
At a median follow-up time of 64 months (range 28–122 months), 687 patients with IDC (39%) and 55 patients with ILC (31.2%) had relapsed (p = 0.019), while 76 patients with IDC (4.3%) and 6 patients with ILC (3.4%) had developed contralateral disease (Table 3).
Disease-free survival events according to histology (invasive ductile carcinoma versus invasive lobular carcinoma).

Disease-free survival events include all locoregional and systemic relapses of the primary disease(s)and the contralateral disease evolution.
% Does not add to 100% as many patients had more than one site of relapse.
Includes locoregional recurrence.
IDC: Invasive ductal carcinoma; ILC: Invasive lobular carcinoma; NA: Not applicable; NS: Not significant.
Among relapsing patients, lung metastases occurred more frequently in patients with IDC compared with patients with ILC (30.7 vs 21.8%; p = 0.036). On the other hand, patients with ILC had a higher incidence of bone metastases (69.1 vs 47.5%; p = 0.044) and also a higher incidence of peritoneal/ovarian metastases (9.1 vs 0.7%, respectively; p = 0.001). There was no difference in the incidence of contralateral disease evolution between the two groups during the follow-up period (4.3% in IDC and 3.4% in ILC). The estimated 5-year DFS for ILC and IDC was 61.5% and 45.8%, respectively (hazard ratio [HR]: 0.76; 95% CI: 0.58–50.99; p = 0.011) (Figure 1). In a multivariate model adjusted for age, tumor size, nodal status, histological grade and IHC (ER, PgR and HER2, both individually and in groups), ILC histology was independently associated with better DFS (HR: 0.58; 95% CI: 0.36–0.93; p = 0.023) (Table 4).
Multivariate analysis for disease-free survival.

HR: Hazard ratio; IDC: Invasive ductal carcinoma; IHC: Immunohistochemistry
ILC: Invasive lobular carcinoma; LA: Luminal A subtype; N: Pathological nodal status
N0: Negative axillary lymph node involvment; N+: Positive axillary lymph node involvment
T: Pathological tumor size.

Luminal A subtype.
Discussion
In this relatively large study, ILC constituted 8.5% of Egyptian women with breast cancer. In the Middle East region, studies from Turkey and Israel have reported ILC at an incidence of 12% and 10.9%, respectively [12,13]. A much lower incidence has been observed in the majority of the Far East countries (~2–4 % in Japan and Korea) [14,15]. In Western countries, studies including patients diagnosed before the 1990s have reported a relatively low incidence of ILC (~6%) [2]. However, more recent data has consistently reported a higher incidence of ILC (10–15%), which was attributed to the increased use of hormone-replacement therapy during the last two decades, which appears to increase the risk of ILC more than other histologic subtypes [16–18]. Therefore, while no specific racial/ethnic predisposition for ILC has been suggested, it seems that lifestyle parameters including hormone-replacement therapy and delayed pregnancy are among the most relevant risk factors implicated in the etiology of this histologic subtype [18].
In our series, ILC was associated with a more advanced T stage compared with IDC, which has been consistently reported by others [1,2,7,8]. A delay in the diagnosis of ILC is common, since ILC is hard to detect clinically and radiologically owing to its peculiar diffuse pattern of growth within the breast parenchyma [6–8].
Nearly 70% of patients in our series were subjected to mastectomy, which is significantly higher than figures from Western data sets. This reflects the higher prevalence of advanced tumors at diagnosis in this part of the world, which is largely due to lack of early detection screening programs during the study period. Our finding that ILC was treated more often with mastectomy than IDC is in line with almost all published data [19–21]. This is attributed – at least in part – to the more advanced T stage encountered in patients with ILC. The inherent difficulty to define tumor margins may obviously increase the rate of mastectomy in these patients compared with IDC [20,21]. Whether BCS is equivalent to mastectomy in patients with ILC was a subject of debate for a long time, however, accumulating evidence has strongly suggested that if clear surgical margins are obtained, BCS is associated with similar local and distant relapse rates compared with mastectomy in ILC [21,22]. In our study, we did not observe an increase in the rate of local relapse in the limited number of ILC cases who were treated with BCS compared with those treated with mastectomy.
We observed more disease bilaterality in ILC patients, which was also reported in other studies [23]. However, no further increase of contralateral disease was seen in the ILC group This is in line with a recent large IBCSG study including 767 cases with ILC and 8607 cases with IDC; the 20-year incidence of contralateral breast cancer was 8.1% and 5.7% for the two subtypes, respectively [2]. Our study – in addition to others – may argue against the known dogma that ILC is essentially associated with a much higher rate of contralateral disease compared with IDC. Longer follow-up.
In agreement with other studies [2,8,23], we found that lobular histology had a significantly lower incidence of lung metastasis, and a higher incidence of bone and peritoneal relapses (Table 3). Our study was also in general agreement with the notion that ILC has a significantly higher frequency of favorable luminal breast cancer phenotype [2,8,23,24]. Using gene expression profiling, most ILCs fall into luminal-A molecular subtype (~75%) followed by luminal-B (~20%), although some ILCs clustered with either HER2 or apocrine subtypes [25,26]. Furthermore, ILC demonstrated significantly lower p53 and Ki-67 expression, and rare c-myc amplifications compared with luminal IDC, denoting its potentially indolent nature even when compared against the luminal-A subtype of IDC [27]. All of these characteristics may suggest that ILC likely originates from more differentiated luminal cells that may behave differently in terms of relapse pattern and response to therapy [28].
In our study, we found that patients with ILC had better 5-year DFS compared with those with IDC. In general, studies reporting on 5-year survival data – as in our study – have reported a superior outcome in patients with ILC [9], whereas studies with longer follow-up periods tend to show a similar or even worse outcome for ILC than IDC [2,29]. In the IBCSG studies, Pestalozzi et al. have found an early significant 5-year DFS and overall survival advantages for the ILC patients, which was reversed with longer term follow-up in favor of IDC patients [2]. The observation of having high incidence of late relapse has been described as a feature of endocrine-sensitive tumors, especially Luminal-A disease [30,31], which is the most frequent phenotype in ILC patients. Nevertheless, late relapses in ILC may also occur independent of ER status, denoting that the diagnosis of ILC carries distinct biologic implications. Interestingly, disseminated tumor cells were identified more commonly in ILC patients compared with IDC patients, raising the question of whether the higher frequency of disseminated tumor cells in patients with ILC might be mechanistically responsible for the phenomenon of late recurrence in this histologic subtype [32].
Putting these data into a therapeutic context, it seems that chemotherapy modulation is less likely to improve treatment outcome in ILC as the latter is known mainly to reduce events on the short-term. Further evidence from a recent meta-analysis clearly showed that IDCs have a three-times higher chance of achieving pCR to neoadjuvant chemotherapy, underscoring that ILCs are less chemoresponsive [10]. We believe that improvement in endocrine treatment strategies for these patients could perhaps be the best way to go. A recent analysis from the BIG 1–98 trial has shown that ILC patients benefit significantly more from letrozole than tamoxifen [33]. Furthermore, and acknowledging the relatively high rate of late events in ILC, it would be more interesting to explore the potential use of extended adjuvant endocrine therapy in these patients. Results from the MA17 [34], ATLAS [35] and aTToM [36] trials have unequivocally proved that extended endocrine therapy beyond 5 years could improve long-term survival. The benefit of this approach has been mentioned to be most significant in patients with ER, PR-positive, HER2-negative disease [34], which is the dominant phenotype among patients with ILC, as demonstrated in many studies including the current one.
We realize that our study has many limitations that should be considered. It is a retrospective study and hence we had missing information for some of the evaluated parameters, particularly for HER2 expresWsion, which was not routinely tested in our center prior to the year 2005. The median follow-up period (~5 years) is relatively short to generate solid conclusions regarding the natural history of breast cancer, especially in patients with ILC. Nevertheless, it is still one of the largest series evaluating the behavior of ILC in a non-Western population, which may add important information and insights to literature in this field. On the other hand, and irrespective of the histologic subtype, our study has shown that breast cancer women in Egypt are diagnosed at a younger age, with more advanced stages of the disease compared with Western women, which confirms some of the known facts about breast cancer in the Middle East region [37,38].
In conclusion, our study indicated that classifying invasive breast cancers into distinct histological subtypes is clinically relevant. We showed that lobular histology is associated with biological features of good prognosis. Yet, and independent of that, lobular histology per se is associated with better DFS at 5 years and a unique pattern of relapse, at least in the short term. These differences between ILC and IDC may potentially provide useful therapeutic tools for a more individualized treatment decisions in patients with early breast cancer.
Future perspective
Future studies should be looking into biological explanations of the differences between ILC and IDC. In the era of genomic profiling and tumor sequencing, elucidating the molecular heterogeneity between both subtypes remains an open question that needs to be addressed.
Financial & competing interest disclosures
The authors have no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
No writing assistance was utilized in the production of this manuscript.
Ethical conduct of research
For studies involving data relating to human or animal experimental investigations, appropriate institutional review board approval has been obtained and is described within the article (for those investigators who do not have formal ethics review committees, the principles outlined in the Declaration of Helsinki have been followed).
Executive summary
Lobular carcinoma constitutes approximately 8-10% of breast cancer in the Middle East.
Lobular carcinomas are more commonly diagnosed in slowly proliferating endocrine-sensitive tumors.
At 5 years, outcome is better than invasive ductal carcinoma.
More bone and peritoneal metastasis are observed in invasive lobular carcinoma but less lung metastasis.
Data from the Middle East are rather in line with published series from Western countries.
Lobular histology is associated with biological features of good prognosis. Yet, and independent of that, lobular histology per se is associated with better disease-free survival at 5 years and a unique pattern of relapse, at least in the short term.
