Abstract
The amount of the largest diameter of visible residual tumor after cytoreductive surgery remains one of the strongest prognostic factors In advanced ovarian cancer. The Implementation of a more aggressive surgical approach to Increase the proportion of patients without visible residual tumor Is, therefore, a rational concept. Thus, the surgical management of advanced ovarian, primary peritoneal and fallopian tube cancers now Incorporates more comprehensive surgical procedures. However, these more extensive surgical procedures are associated with an Increased risk of morbidity, which may have a negative Impact on the oncologic outcome. In addition, It Is unclear whether all patients benefit from a comprehensive surgical Intervention In the same way or If there are patients whose disease course will not be Influenced by this approach. The methodologic analysis of surgical effectiveness Is complex and controversial owing to a lack of prospective surgical trials. This review acknowledges controversies and alms to discuss novel developments In the field of cytoreductive surgery for patients with ovarian, primary peritoneal and fallopian tube cancers. The focus of the review Is to discuss the role of surgery at Initial diagnosis. The role of secondary and tertiary surgery In the recurrent setting Is beyond the scope of this review.
Keywords
Ovarian, primary peritoneal and fallopian tube cancers are the most lethal gynecologic malignancies, with 22,280 estimated new cases and 15,500 estimated deaths in 2012 in the USA [1]. The majority of patients (60–70%) present at an advanced disease stage at the time of initial diagnosis with overt peritoneal extrapelvic disease, lymph node involvement or distant metastatic disease beyond the peritoneal cavity and parenchymal liver metastasis (International Federation of Gynecology and Obstetrics [FIGO] stage III and IV, respectively) [1,2]. The natural history of advanced ovarian cancer is one of clinical remission after primary cytoreductive surgery (CRS) and postoperative chemotherapy; however, the majority of patients recur within 24 months and long-term cure rates languish between 20 and 30%.
Positive results from different improvement strategies have been reported over the past few years. Modification of the medical treatment after surgery demonstrated a benefit for patients treated with dose-dense intravenous (iv.) chemotherapy [3], for patients who underwent a combination of intraperitoneal (ip.) and iv. treatment [4], and for patients who were treated with a maintenance antiangiogenetic therapy after chemotherapy (monoclonal anti-VEGF antibody, bevacizumab, or oral angiogenesis inhibitor, pazopanib [5–7]). Risk-reducing strategies, for example, prophylactic salpingo-oophorectomy, reduce mortality in a high-risk population with germline mutations (
Role of surgery
In contrast to other solid malignancies of the upper abdomen (e.g., pancreas, gallbladder and stomach), ovarian, primary peritoneal and fallopian tube carcinomas are sensitive to platinum-based combination chemotherapy in up to 80% of cases. The distinct chemosensitivity justifies a more extensive surgical approach. Surgical cytoreduction of advanced stage ovarian cancer, also termed ‘tumor debulking’, is defined as an attempt to maximally resect all visible and palpable disease. Postoperative residual disease is a variable of great importance for estimating the prognosis and guiding further treatment of affected patients.
Radical surgical approach for advanced ovarian carcinoma
The term ‘radical surgery’ used for cancer surgery was introduced by William Halsted at the beginning of the 20th century and originates from the word radix, for example, removing the cancerous lesion including the root (radix) with tumor-free microscopic margins. In analogy, William Halsted surgically removed the entire breast, including the underlying muscle and the axillary lymph nodes to reduce the risk of local recurrence in patients with breast cancer. It was not until the late 1970s that Bernhard Fisher was able to prove equivalent outcomes for patients treated with less radical breast-conserving surgery followed by local radiation therapy in patients with breast cancer.
Unlike breast cancer, ovarian cancer involves the peritoneal surface with diffuse peritoneal carcinomatosis and the goal of CRS is to resect all macroscopic tumor lesions, not to remove the tumor with a tumor-free microscopic margin status. The goal of surgical cytoreduction of advanced stage ovarian cancer is to maximally resect all visible and palpable disease. There is a large body of evidence that demonstrates a survival benefit for patients who are left with minimal (‘optimally cytoreduced’) disease after surgery. For more than 30 years, it has been known that women with advanced ovarian cancer, who are left with an apparent small volume of residual disease following primary surgery and go on to receive systemic chemotherapy, have a superior outcome compared with patients who are left with a larger volume disease after initial surgery. In 1934, Meigs suggested removing as much tumor as possible to enhance the effectiveness of postoperative radiation therapy in patients with advanced ovarian cancer [9]. The radicality of this surgical approach is not related to the resection of predefined anatomic compartments but, rather, is based on multiple procedures individually targeting the organs involved with disease with the goal to achieve ‘optimal debulking’ status. These procedures include, but are not limited to, hysterectomy and salpingo-oophorectomy, peritonectomy with or without gastrointestinal surgery, lymph node dissection, omentectomy and upper abdominal surgery. In 1975, Griffiths hypothesized a threshold – a maximum tumor diameter – that may remain after CRS independent of the number of tumor lesions without compromising the benefit of the surgical procedure [10]. Hoskins
Prognostic impact of postoperative residual disease
Although, to date, the benefit of surgical cytoreduction has not been prospectively established through a well-defined prospective Phase III trial, CRS with the goal of achieving minimal residual disease in combination with platinum- and taxane-based chemotherapy is currently the accepted standard management. However, in the absence of prospective trials, we cannot say for sure whether the surgery itself is responsible for the superior outcome associated with small-volume residual disease or whether these patients are part of a biologically more favorable subgroup with excellent response to postoperative chemotherapy. The evidence strongly supporting the role of surgery has been derived from different types of analysis.
Single-center studies
A retrospective analysis of 465 patients at Memorial Sloan-Kettering Cancer Center (NY, USA) with ‘bulky’ FIGO stage IIIC disease (those stage IIIC patients with nodal involvement only were excluded) revealed three distinct groups with significantly different survival rates (p < 0.01): group one had no gross residual tumor, group two had gross <1 cm residual and group three had >1 cm [13]. Although the difference in survival in patients left with smaller volume <1 cm was significant when compared with those with suboptimal residual disease, there was a trend in patients with gross up to 0.5 cm residual compared with those with 0.6 cm up to 1 cm (p = 0.06), suggesting that each incremental decrease in residual tumor below 1 cm may be associated with an incremental improvement in OS. Aletti
Subanalyses from prospective multicenter trials
The prognostic impact of residual disease was evaluated in outcome data from several prospective trials evaluating the role of postoperative chemotherapy [11,14,15]. All studies enrolled advanced ovarian cancer patients and there was no significant difference in progression-free survival (PFS) or OS between the respective treatment arms. For each entire patient population, however, the postoperative residual tumor has been demonstrated as an independent prognostic factor. The strength of these studies is the large amount of prospectively acquired outcome data from multicenter patient populations. The weaknesses of these subanalyses include the fact that the aim of these prospective trials was the analysis of postoperative systemic treatment and not the surgical outcome. The patients included were randomized after postoperative recovery, not accounting for patients who did not meet study inclusion criteria, or patients who experienced complications from the surgical treatment that precluded them from participation in these trials. Since randomization in these trials was postoperative, patients included are highly selected. Therefore, results, with regard to surgical effect of these analyses, are hypothesis-generating, at best.
Meta-analyses
Bristow
Obtaining a higher rate of primary optimal cytoreduction
Paradigm shift towards more radical surgery: implementation of upper abdominal surgery
Advances have been made in successfully resecting pelvic disease, in addition to disease involving the intestinal tract; however, bulky tumor involving the upper abdomen has been noted as the main obstacle to attaining an optimal cytoreductive outcome without an increased risk of morbidity and mortality. A survey by the Society of Gynecologic Oncologists members demonstrated that tumors involving the diaphragm, bowel mesentery and portal triad consistently precluded optimal cytoreduction [19]. In general, over 50% of patients with advanced FIGO IIIC disease present with upper abdominal disease cephalad to the greater omentum [20]. Developments in surgical training, technique and perioperative care permit surgeons to maximize cytoreductive efforts in patients who have advanced stage IIIC and IV ovarian cancer; we are now able to increase the rate of optimal cytoreduction and target anatomical sites that were historically thought to be unresectable. This modified approach includes procedures, such as peritonectomy, diaphragm resection, splenectomy, distal pancreatectomy, partial liver resection, cholecystectomy and resection of tumor from the porta hepatis in cases where the primary surgeon deemed them necessary, to achieve optimal cytoreduction. A significant increase in optimal cytoreduction rates can be achieved after incorporation of comprehensive surgical efforts targeting the upper abdomen. For instance, following this programmatic change in surgical approach at the Memorial Sloan-Kettering Cancer Center, the rate of optimal cytoreduction for patients with stage IIIC ovarian cancer increased from 50% in the mid-1990s to greater than 75% starting in 2001 [18]. Although several groups now propose a new, more comprehensive approach to cytoreduction [16,21–24], others question the role of extensive surgery for this disease, and proposed that tumor biology is more important in determining the surgical resectability and survival outcome [25,26].
The results of a paradigm shift in surgical management were published by Chi
Results of retrospective studies evaluating the implementation of extensive cytoreductive surgery.
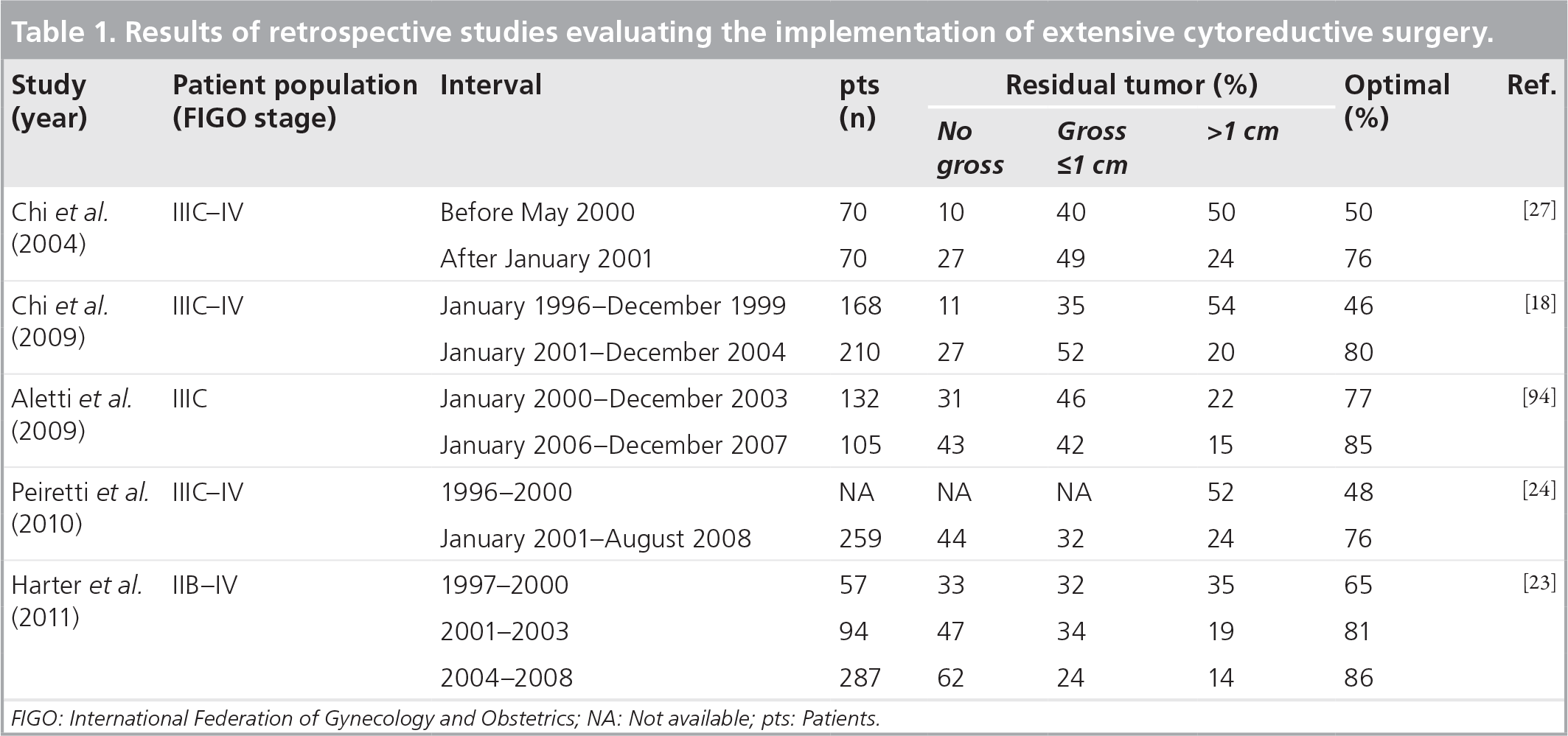
FIGO: International Federation of Gynecology and Obstetrics; NA: Not available; pts: Patients
The role of neoadjuvant chemotherapy
Predicting optimal cytoreduction represents a critical point in the management of advanced ovarian cancer patients. However, until today no single preoperative predictor has demonstrated sufficient accuracy to predict surgical outcome. It is well established that patients who undergo suboptimal cytoreduction sustain the morbidity of surgery without the associated survival benefit [31–34]. Avoiding suboptimal (unnecessary) surgery is an important goal. One strategy to increase optimal cytoreduction rates by decreasing preoperative tumor burden is neoadjuvant chemotherapy. Two recently published randomized trials compared upfront CRS followed by six cycles of platinum-based chemotherapy with a neoadjuvant treatment approach, including three cycles of chemotherapy before CRS followed by three cycles after interval debulking surgery [35,36]. Both trials showed no significant differences in median PFS or OS.
The rate of patients with no gross residual tumor was significantly higher after neoadjuvant chemotherapy when compared with patients who underwent primary CRS (52.1 vs 20.3%). However, in a subgroup analysis in patients who had a complete gross tumor resection after neoadjuvant chemotherapy, OS was lower compared with patients with no gross residual disease after primary CRS (median OS of 38 months [90% CI: 33–44] vs 45 months [90% CI: 34–54]) [36]. Thus, the increase in complete gross resection rates after neoadjuvant chemotherapy does not appear to translate into improved outcomes observed in patients who undergo primary CRS.
Although the multicenter, randomized Phase III trials evaluating the role of neoadjuvant chemotherapy showed no difference in outcomes, the optimal resection rates achieved in patients undergoing primary CRS were comparably low, and the studies are the subject of ongoing critique. Many gynecologic oncologists raise some concerns regarding whether surgical skills and efforts in centers participating in these trials were equivalent to earlier mentioned single center experiences [12,13]. The rate of optimal surgical cytoreduction of patients with stage III and IV disease who were treated at the participating centers, but not included in these trials, is unknown. A potential selection bias is, therefore, incalculable, and a comparison to outcomes from other studies or pooled data is, therefore, problematic and debatable.
The comparison of resection rates: main indicator for quality?
Published data suggest that a survival advantage can be achieved if CRS is performed by surgeons who are trained in gynecologic oncology, compared with general surgeons or generalist gynecologists. Despite this knowledge, there is still a wide variance in reported rates of optimal cytoreduction – from 15 to greater than 85%. The rate of optimal cytoreduction in centers treating advanced ovarian cancer patients may be an indicator for the quality of surgical care. However, whenever comparing cytoreduction rates of institutions or study groups, it is important to account for patients included in these analyses. There are different optimal cytoreduction rates and complete cytoreduction rates observed when analyzing patients with FIGO stage II–IV disease versus patients with bulky stage IIIC and IV disease. Kommos
In addition, there is a wide variance of patients treated with neaoadjuvant chemotherapy versus primary surgery among centers. In a recently published prospective, multicenter analysis of five European gynecologic cancer centers of 275 consecutive patients with FIGO stage II–IV epithelial ovarian cancer (EOC), 173 patients (63%) underwent primary CRS and optimal cytoreduction (<1 cm residual tumor) was achieved in 68% [39]. A French multicenter study evaluated 527 patients with advanced ovarian cancer. Of those, 190 underwent primary CRS and the complete gross resection rate was 65% [40]. The remainder were treated with neoadjuvant chemotherapy. Thus, patient selection plays an important role when comparing resection rates of different centers and groups; this should be considered when determining quality of surgical care.
Morbidity & mortality
Surgical cytoreduction is associated with a risk of morbidity and mortality. Single center data quantified the mortality rate after CRS in advanced ovarian cancer between 1.4 and 2.5%, which compares favorably to population-based studies with a mean mortality rate of 3.7% [17,41,42]. Risk factors for higher mortality in these studies are age, performance status and comorbidities, as well as emergency treatment compared with patients who were treated electively [43–45].
A recently published study has shown that patients undergoing extended surgical procedures have a higher rate of complications and initiate chemotherapy with a significant delay. Among 3991 patients with stage III–IV ovarian cancer who were aged 65 years or older, 479 (12%) did not receive postoperative chemotherapy. Perioperative complications significantly delayed initiation, but did not increase the chance of omission of chemotherapy [46]. In a randomized trial (EORTC 55971) comparing primary CRS followed by adjuvant chemotherapy with neoadjuvant chemotherapy followed by interval debulking surgery, 18 out of 315 patients (5.7%) in the primary CRS arm failed to receive adjuvant chemotherapy, and 21 out of 326 patients (9.5%) in the neoadjuvant treatment arm did not undergo interval debulking surgery [36].
Since CRS is often associated with multiorgan resection, in addition to general surgical complications such as bleeding, cardiac events, thromboembolic events and surgical site infections, organ-specific surgical complications are also frequently observed. Bowel-related morbidity and complications, including anastomotic leak, range from 2 to 30.5% [47,48]. With the preferred use of stapled (double-stapling technique) rather than hand-sewn techniques, and the performance of tension-free and adequately perfused anastomoses, low anastomotic leak rates of less than 2% are achievable in patients with advanced ovarian cancer undergoing complex cytoreductive procedures. The rectosigmoid is the bowel segment most frequently resected during CRS for ovarian cancer. Peiretti
The role of diaphragm surgery was evaluated in several studies [30,54–60]. In a study on the incidence and management of pleural effusions after diaphragm peritonectomy or diaphragm resection in 59 patients with advanced ovarian cancer, Eisenhauer
Complications involving the debulking of the left upper quadrant have also been studied. Magtibay
Surgery in patients with stage IV disease
The role of residual tumor and radical surgery in stage IV disease is more controversial [67,68]. Patients with stage IV disease, including extraperitoneal or parenchymal liver spread, have worse prognosis than those with stage III disease. To improve the accuracy of the FIGO staging, patients with pleural effusions may benefit from a video-assisted thoracoscopic surgery for diagnosis and intrathoracic cytoreduction [69–71]. In 50% of patients with moderate-to-large pleural effusions, solid pleural-based tumors that were not observed in previous imaging were identified. The results of intrathoracic cytoreduction may influence the management of abdominal surgery: if the residual intrathoracic tumor is >1 cm, radical surgery in the abdominal cavity is debatable.
Conclusion
The natural history of advanced ovarian cancer is one of clinical remission after primary CRS and postoperative chemotherapy; however, the majority of patients recur within 24 months, and long-term cure rates languish between 20 and 30%. Since the amount of the largest diameter of visible residual tumor after CRS remains one of the strongest prognostic factors, an aggressive surgical approach is taken to increase the proportion of patients without visible residual tumor. However, more extensive surgical procedures are associated with an increased risk of morbidity, which may have a negative impact on the oncologic outcome. One promising new treatment strategy is to combine surgical cytoreduction with hyperthermic intraoperative ip. chemotherapy (HIPEC). Further study of HIPEC, and development of additional treatment strategies, are needed.
Future perspective: the role of HIPEC
Since EOC remains confined to the peritoneal cavity for most of its natural history, numerous theoretical studies evaluated the use of ip. drug delivery as a management strategy [72–74]. The studies predicted a pharmacologic advantage for ip. therapy with improved tumor cell access, longer half-life in the peritoneal compartment, increased dose intensity and slow peritoneal clearance, while still reaching sufficient levels of systemic exposure for longer periods of time. Further preclinical data for platinum agents revealed that resistence to the drugs can be overcome by exposing the tumor cells to higher ip. concentrations. Animal studies confirmed that the peritoneal cavity can be exposed to substantially greater concentrations of cytotoxic agents [75–77]. Owing to the production of severe side effects, these concentrations are not realistically attainable within the systemic compartment. Based on these data, several Phase III clinical trials have explored the clinical utility of postoperative ip. therapy of small-volume residual disease following maximal CRS of advanced EOC [4,78,79]. In these trials, after postoperative recovery cisplatin-based ip. therapy was delivered through an ip. access device and compared with standard iv. chemotherapy. In the most recent of these trials (GOG-172), there was a statistically significant increase in the median PFS and OS in the cisplatin/paclitaxel ip. treatment compared with the iv. group, with a benefit of 6 and 16 months, respectively. These findings have prompted the National Cancer Institute (MD, USA) to make a clinical announcement in 2006; since then, postoperative ip. treatment in patients with minimal residual disease after surgical cytoreduction is increasingly used in the USA and continues to be the subject of ongoing randomized trials with new regimens and new/additional drugs [80]. However, a substantial percentage of the patients (60%) were unable to complete six cycles of cisplatin/paclitaxel ip. chemotherapy, most commonly secondary to abdominal discomfort due to postoperative adhesion barriers and ip. catheter-related complications. Therefore, the European Organization for Research and Treatment of Cancer (Brussels, Belgium) and other international organizations are still unconvinced by available data and still consider this treatment experimental [81]. However, despite only 48% of the patients receiving three or fewer cycles of ip. therapy, the ip. group had significant improvement in median survival compared with the iv. group.
An alternative to the postoperative local drug delivery is the intraoperative chemotherapy. For several reasons, there has been increasing interest in the surgical oncology community for combining surgical cytoreduction and hyperthermic ip. drug delivery (HIPEC) in the management of solid tumors whose natural history remains principally localized to the peritoneal cavity. The advantages are as follows:
By giving the chemotherapy intraoperatively, drug exposure is optimal secondary to direct contact with the cancer cells without the barriers of postoperative adhesions; Intraoperatively, the chemotherapy can be delivered under highly standardized procedures and the surgeon can guarantee optimal distribution of the chemotherapy, and dwell times can be controlled; Hyperthermia has been shown to increase the cytotoxic effect of many chemotherapeutic agents by increasing DNA-crosslinking and increasing the tumor penetration [82–84].
A comprehensive review of HIPEC is beyond the scope of this review, but such publications are available [85–92]. This approach has been explored in the treatment of patients with peritoneal malignancies, including pseudomyxoma peritonei, gastric cancer and colorectal malignancies, suggesting a survival benefit using this approach. Unfortunately, there is essentially no evidence from prospective trials to objectively confirm any claimed benefits associated with this approach for patients with advanced EOC. The existing studies on HIPEC are mostly retrospective in nature, heterogeneous with regards to combined inclusion of primary and recurrent disease, and lack robust dose-finding and pharmacokinetic data. Until evidence-based trials yield further data, it is reasonable to conclude that surgical cytoreduction with HIPEC offers a rational, but still investigative, approach in the management of EOC, the use of which should be employed under the umbrella of Institutional Review Board-approved clinical trials.
Executive summary
Ovarian, primary peritoneal and fallopian tube cancers are the most lethal gynecologic malignancies.
Retrospective studies and meta-analyses have demonstrated and confirmed that the amount of residual disease after cytoreductive surgery (CRS) inversely correlates with progression-free and overall survival.
Increasing optimal cytoreduction rates with more extensive and complex surgical efforts is associated with improved outcomes.
Advances in surgical training, technique and perioperative care have allowed surgeons to maximize cytoreductive efforts, enabling them to increase the rate of optimal cytoreduction.
The rate of patients with no gross residual tumor was significantly higher after neoadjuvant chemotherapy when compared with patients who underwent primary CRS. However, the increase in complete gross resection rates after neoadjuvant chemotherapy does not translate into improved outcomes observed in patients who undergo primary CRS.
Surgical cytoreduction is associated with a risk of morbidity and mortality. Single center data quantified the mortality rate after CRS in advanced ovarian cancer to be between 1.4 and 2.5%.
Risk factors for higher mortality in these studies are age, performance status and comorbidities, as well as emergency treatment compared with patients who were treated electively.
Patients with pleural effusions may benefit from a video-assisted thoracoscopic surgery for diagnosis and intrathoracic cytoreduction.
Combining surgical cytoreduction and hyperthermic intraperitoneal drug delivery is a rational, but still investigative, approach in the management of epithelial ovarian cancer.
One randomized Phase II trial has been initiated recently at the Memorial Sloan-Kettering Cancer Center [93]. This is a randomized trial designed to compare the efficacy and safety of carboplatin at a dose of 800 mg/m2 administered as HIPEC after secondary CRS for recurrent platinum-sensitive EOC. Patients with minimal residual disease of 5 mm or less will be randomized 1:1 to CRS with HIPEC (arm A; n = 49) or CRS only (arm B; n = 49). Both arms will receive standard postoperative iv. chemotherapy. The primary objective is to determine the proportion of patients who are without evidence of disease progression at 24 months after CRS with or without HIPEC.
Current ongoing clinical trials are listed in Table 2.
Ongoing studies evaluating the role of hyperthermic intraperitoneal chemotherapy for patients with ovarian cancer.

FIGO: International Federation of Gynecology and Obstetrics.
Financial & competing interests disclosure
