Abstract
Symptoms of vulvar and vaginal atrophy (VVA), including dyspareunia and vaginal dryness, have a distinct negative impact on a woman's quality of life. The REVIVE survey highlighted the lack of awareness of VVA symptoms among postmenopausal women with vaginal symptoms, with many women reluctant to initiate discussions with their healthcare professionals despite the presence of vaginal symptoms. The REVIVE survey also provided insights into women's views of VVA treatments. Women reported displeasure with the vaginal administration route, lack of symptom relief with over-the-counter products, and concerns about the safety of estrogen therapies. With the high prevalence of VVA, obstetricians/gynecologists should become vigilant in identifying women with VVA by implementing screening and discussion of symptoms during routine office visits - providing patients with information about appropriate therapies based on the severity and impact of symptoms, keeping in mind individual preferences and perceptions.
Keywords
The peri- and post-menopausal years are associated with a decline in estrogen levels and, ultimately, a hypoestrogenic state. This leads to physiological changes that affect multiple organ systems, particularly the endocrine and genitourinary systems [1]. In the urogenital tract, these physiological changes typically lead to vulvar and vaginal atrophy (VVA), a condition that is characterized by changes in the maturation index of the vaginal epithelial cells (decreased superficial cells and increased parabasal cells), an elevation in vaginal pH (loss of acidity), and increased susceptibility to infection. There is also a reduction in vaginal moisture and loss of tissue elasticity [1–3]. Additionally, there is a decline in secretion of dehydroepiandrosterone and circulating dehydroepiandrostenedione sulfate, which may also impact normal vaginal physiology [4,5]. These involutional changes frequently lead to bothersome symptoms that can negatively impact a woman's quality of life [2,6,7].
The vast majority of postmenopausal women will develop VVA at some point in their lives [8–10], and up to 50% will experience physical symptoms of vaginal atrophy, particularly dyspareunia (pain with intercourse), vaginal dryness, and vaginal irritation [1,7,11,12]. Furthermore, the prevalence of VVA in postmenopausal women is likely to increase, secondary to factors such as the aging population, increased longevity, and a decline in the use of systemic hormone therapy [13–15]. Despite the increased prevalence of VVA, many women report that their healthcare professional (HCP) did not inquire about specific conditions such as dyspareunia during routine examinations [16]. In a recent survey of women aged 45–65 years in the USA, only 36% of HCPs acknowledged that they ‘often’ discussed the potential of pain associated with intercourse and only 10% of women indicated that their HCP had initiated a conversation about dyspareunia [17].
Until recently, therapies for symptomatic VVA have been limited to vaginal products (i.e., over-the-counter [OTC] moisturizers or lubricants and/or estrogen products prescribed as creams, rings, or vaginal tablets). Current recommendations suggest that systemic estrogen therapy be used primarily as an option for women who require treatment for other postmenopausal symptoms (e.g., hot flushes) [18]. In February 2013, the US FDA-approved ospemifene, a new oral selective estrogen receptor modulator, as a nonestrogen option for the treatment of moderate-to-severe dyspareunia due to menopause. Ospemifene is a tissue-specific estrogen agonist/antagonist that, based on Phase III clinical trials, is effective, generally well tolerated, and approved for the treatment of moderate-to-severe dyspareunia associated with postmenopausal VVA [19–21].
Several surveys of postmenopausal women in the USA and abroad have documented a negative impact of VVA symptoms on quality of life of women [12,17,22–24]. Despite differences in study populations, design, and overall objectives, all surveys reported that VVA had a substantially negative impact on overall sexual health and identified numerous barriers to treatment. The most critical and frequently identified barrier was the reluctance of postmenopausal women to broach the topic of any type of vaginal discomfiture with their HCP [13]. Many of the women surveyed were unaware that VVA symptoms were a direct result of menopause and hypoestrogenism and that their symptoms, unlike vasomotor symptoms, would not resolve over time. Additionally, many HCPs surveyed thought that VVA and the associated symptoms were too personal or racy, and they hesitated to initiate conversation about sexual health.
The recently published REVIVE survey [25], conducted in a large cohort of postmenopausal women in the USA, confirmed that women were relatively unaware of VVA as a specific medical condition, despite the negative impact of dyspareunia and/or vaginal dryness. Additionally, the REVIVE survey found that many women do not communicate well with their HCPs and that most symptomatic postmenopausal women remain untreated. The REVIVE survey also provided unique insights into women's views regarding treatment options for VVA [25]. This review addresses the implications of the REVIVE survey and discusses the availability of a new oral treatment option for dyspareunia associated with VVA.
The REVIVE survey: survey design
The REVIVE survey was conducted from 31 May through 14 June 2012, among women in the USA who reported having VVA and/or symptoms consistent with VVA. Postmenopausal women with VVA symptoms were identified through KnowledgePanel®, a 56,000-member panel demographically representative of the USA population developed and maintained by GfK Knowledge Networks. Of the 15,576 women in KnowledgePanel who were contacted, a total of 10,486 women responded with 8081 (77%) self-identifying themselves as postmenopausal. A total of 3046 (38%) postmenopausal women reported they had experienced symptoms of VVA, with 44% reporting that they had experienced dyspareunia/pain with sex (Figure 1). Each participant then completed a structured, online quantitative and qualitative questionnaire that collected basic demographic data and information on a range of areas. This 45-min online questionnaire, developed by menopause experts/clinicians, explored a range of topics including women's knowledge about VVA, the impact of vaginal symptoms on their lives, interactions with HCPs about VVA, and their experiences with and attitudes toward treatment options for VVA [25].

Survey participants from KnowledgePanel®.
REVIVE survey findings
Although all participants who participated in the REVIVE survey had reported at least one symptom consistent with VVA, only 38% of women were aware of the condition and only 24 and 12% attributed VVA symptoms to menopause or hormonal changes, respectively. Approximately half (56%) of the women had discussed their symptoms with an HCP, but only 13% reported that the HCP had initiated the discussion. Only 19% of women who had seen an HCP for any reason in the previous year indicated that they were asked about their sexual health during routine examinations; however, 40% reported they expected their HCP to initiate dialog about menopausal symptoms. The two most common reasons that women selected for not reporting VVA symptoms to their HCP were the perception that their symptoms were: a natural part of aging (50%); and/or not bothersome enough to mention (40%) [25].
In contrast to the low levels of awareness and communication regarding VVA, 59% of women reported that VVA symptoms interfered with their enjoyment of sex. Negative effects on intimacy and several activities of daily life were also reported. Postmenopausal women rated sexual satisfaction as important (55% of women with an intimate partner and 32% of women without an intimate partner). Women with an intimate partner indicated that the impact of VVA on loss of intimacy was ‘more on their mind’ (47%) than loss of youth (12%). Relative to women with a partner, fewer women without an intimate partner indicated loss of intimacy (20%) and more selected loss of youth (19%) as more on their mind. Of note, approximately one in ten women without a partner (12%) reported that VVA symptoms were the reason for them not seeking a new partner [25].
REVIVE dyspareunia cohort
Of the 3046 postmenopausal women with VVA who completed the REVIVE survey, 44% reported dyspareunia as one of their vaginal symptoms, with 467 (15%) women reporting dyspareunia as their only vaginal symptom. The baseline demographics and other characteristics of this dyspareunia cohort were very similar to the total REVIVE survey population, with the only minor difference being in marital status (78% in dyspareunia cohort versus 71% in the total population) (Table 1). Over 80% of women experiencing only dyspareunia in the REVIVE survey reported experiencing painful symptoms due to sexual intercourse in the past year.
Baseline characteristics of the dyspareunia cohort in the REVIVE survey.
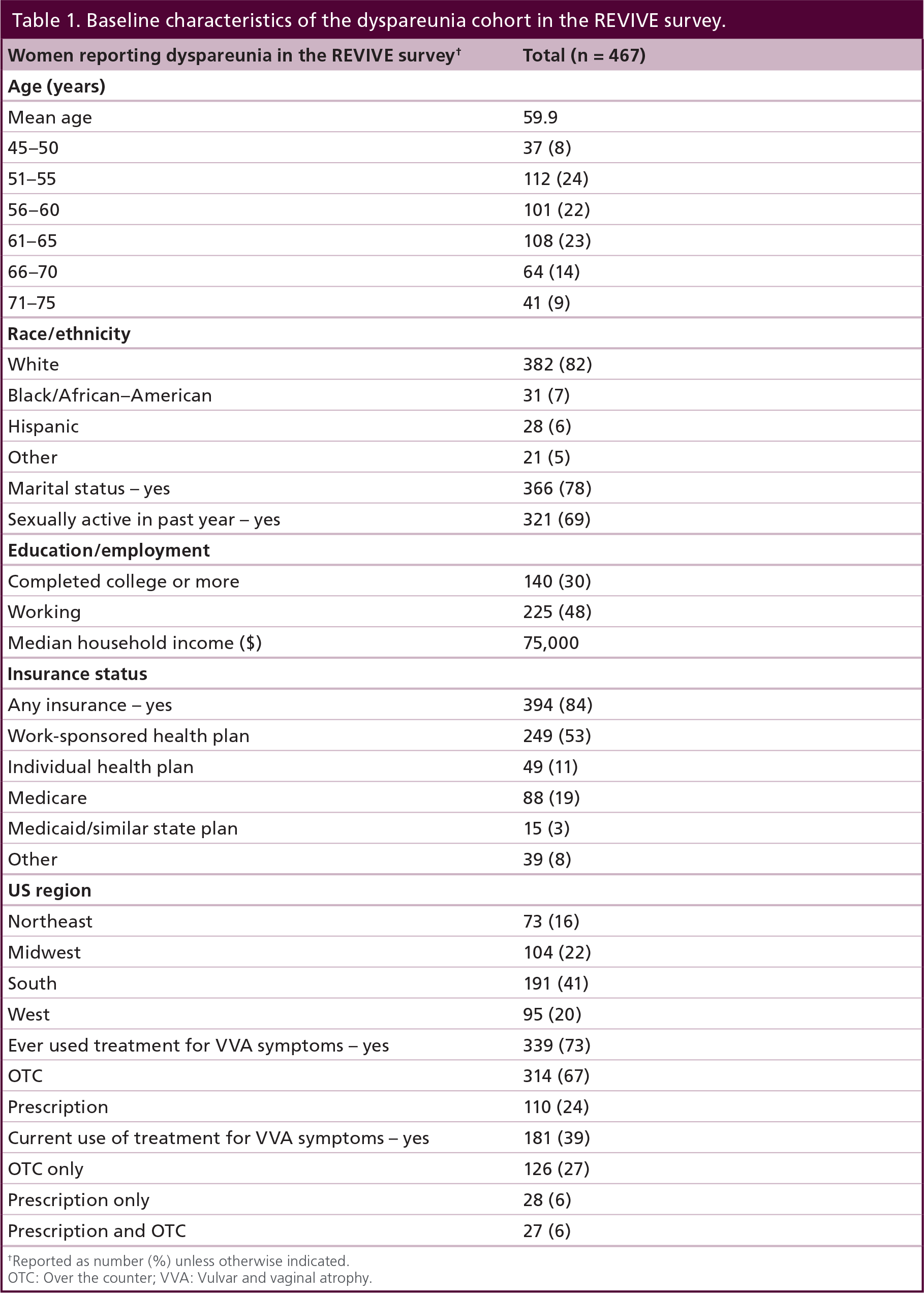
Reported as number (%) unless otherwise indicated.
OTC: Over the counter; VVA: Vulvar and vaginal atrophy.
Approximately half of women with dyspareunia recalled discussing their VVA symptoms with their HCP. Of the women who initiated the first conversation regarding their VVA symptoms, 79% did so during their regularly scheduled office visit. Of the 38% who had never discussed their VVA symptoms with an HCP, 41% thought VVA was a natural part of aging and 23% were too embarrassed to talk about their symptoms. Only a small proportion (14%) of symptomatic women in the dyspareunia cohort attributed their dyspareunia to menopause, with 29% simply attributing their dyspareunia directly to vaginal or vulvar dryness (Table 2). Similar to the overall REVIVE survey population, 67% of women with dyspareunia had no knowledge of the terms ‘VVA’ or ‘vaginal atrophy’ and only 28% felt that their HCP was comfortable talking with them about their symptoms.
Perceived cause of dyspareunia for postmenopausal women with vulvar and vaginal atrophy in the REVIVE survey.

Sexual satisfaction was rated as ‘important’ or ‘very important’ by over 67% of women in the REVIVE survey experiencing dyspareunia, with 73% of women with dyspareunia reporting that their VVA symptoms directly interfered with enjoyment of sex (Figure 2). Dyspareunia had a major emotional (23%) and physical (54%) impact on the overall health and well-being of postmenopausal women with VVA in the survey. Dyspareunia also negatively impacted intimacy (56%), sexual spontaneity (55%), and the relationship with their partner (48%). In addition, women with dyspareunia felt a sense of aging (45%), an attitude of ‘it is what it is’ (41%), and an overall displeasure with their diagnosis of VVA (37%). Most women (81%) reported that their dyspareunia resulted in decreased libido. There was considerable dissatisfaction with current treatment options among women with dyspareunia in that only 32% of women indicated they were satisfied with their current treatment and only 8% were satisfied with their current vaginal estrogen therapy. However, despite the impact of dyspareunia and dissatisfaction with available therapies, only 54% of women had ever discussed dyspareunia with their HCP.

Interference with sexual and daily activities reported by REVIVE survey participants suffering from dyspareunia/pain with sex (n = 1352).
Over 75% of the women in the REVIVE survey with dyspareunia expressed concerns regarding long-term use of their current vaginal estrogen therapy. These concerns often led to discontinuation of their current treatment, with 55% of all women indicating they had not refilled their vaginal estrogen therapy: 50% discontinued because of efficacy, 41% because of concerns about safety or side effects, 19% due to convenience, and 20% due to cost. The most common safety-related concerns reported by survey respondents were associated with long-term use (33%), hormone exposure (21%), breast cancer (19%), and other unwanted side effects (18%). Treatment goals for women with dyspareunia were focused on enjoying sex more (56%) and restoring the vagina to a more natural state (42%), as well as the ability to ‘feel like my old self again’ (22%).
Many women have never used any medication for their VVA symptoms. Over 25% of postmenopausal women with dyspareunia in the REVIVE survey had never used vaginal estrogen or an OTC lubricant/moisturizer. The main reasons for never having initiated therapy were questions regarding efficacy (63%), safety (25%), and convenience (17%). Many women with dyspareunia felt that their symptoms were not bothersome enough to initiate treatment (36%), that the symptoms would simply abate over time (30%), or they had concerns about side effects/unwanted effects (29%). Eleven percent of women with dyspareunia in the REVIVE survey declined therapy of any kind because of safety concerns and/or fear of side effects.
Implications for clinical practice
Obstetricians/gynecologists (OB/GYNs) need to be aware of the prevalence of VVA and the associated physical symptoms. More likely than not, patients will not directly raise issues such as dyspareunia and vaginal dryness because of embarrassment or the belief that nothing can be done medically to treat the condition [17,25]. Initiating discussion can be accomplished readily by raising awareness of potential menopausal changes and physical symptoms, as well as using simple screening questions about VVA during routine visits for women approaching menopause or beyond (Box 1). The need for awareness has become more significant given that the new guidelines for Papanicolaou smear frequency [26] may result in fewer annual visits to HCPs in the future.
For a woman who identifies vaginal symptoms such as dyspareunia or vaginal irritation, VVA can be confirmed by visual examination of the vagina (common signs include vaginal dryness, petechiae, pallor, friability, and redness of the mucosa). If such clinical signs are observed in women who do not report experiencing VVA symptoms, the findings on clinical examination should prompt a discussion regarding VVA. Women should be informed that the decline in estrogen and androgen levels may lead to physical changes in the vagina (thinning and reduced moisture) and may cause symptoms such as pain and discomfort during sex as well as vaginal dryness. In particular, women can be educated that VVA is common among post-menopausal women, yet unlike hot flushes that resolve over time, VVA is a chronic and progressive condition. A woman may not report her VVA symptoms for a myriad of reasons, so HCPs must be proactive in their discussions in order to avoid delays in treatment. Patients should be reassured that, although VVA is chronic and progressive, it should not be accepted as inevitable.
Findings from the REVIVE survey regarding the negative impact of VVA and dyspareunia underscore the importance of initiating appropriate treatment. VVA and its consequences, especially dyspareunia, are important quality-of-life issues that remain under-reported in current clinical practice and deserve far more attention.
Undertreatment & limitations of current treatment options for VVA
An aspect of the REVIVE survey that differentiates it from previous patient surveys is the close examination of women's attitudes about available treatment options for VVA. Despite the impact of VVA reported by survey participants, less than 40% of postmenopausal women in REVIVE with dyspareunia symptoms were currently using a VVA-specific treatment (OTC vaginal moisturizer or lubricant [27%]; prescription vaginal estrogen therapy [6%]; combination OTC/vaginal estrogen treatment [6%]), and less than 30% of all women surveyed had ever used local vaginal estrogen. Women who had discussed VVA symptoms with an HCP were twice as likely to utilize therapy; however, despite conversations with an HCP, 62% were using OTC products alone, 23% were using vaginal estrogen, and 15% were using both. This is of particular concern because women using OTC products reported continued symptoms at higher rates than those using prescription therapies [25].
Women identified a range of limitations with available treatment options (Table 3). The most common limitations/issues reported for OTC products included messiness and inadequate efficacy, whereas concerns about safety were more commonly reported for estrogen therapies. Inconvenience and problems with administration were mentioned as important limitations of all these vaginal products, with many women discontinuing treatment because of issues related to safety, convenience, and lack of efficacy. Similar to the overall REVIVE study cohort, reasons for treatment discontinuation in the dyspareunia cohort included ‘didn't restore my vagina to a natural state/didn't reverse the changes that have occurred’, ‘not enough relief of symptoms’, and ‘no relief of symptoms’ with their vaginal prescription treatment.
Views of postmenopausal women with dyspareunia currently using treatment for vulvar and vaginal atrophy1
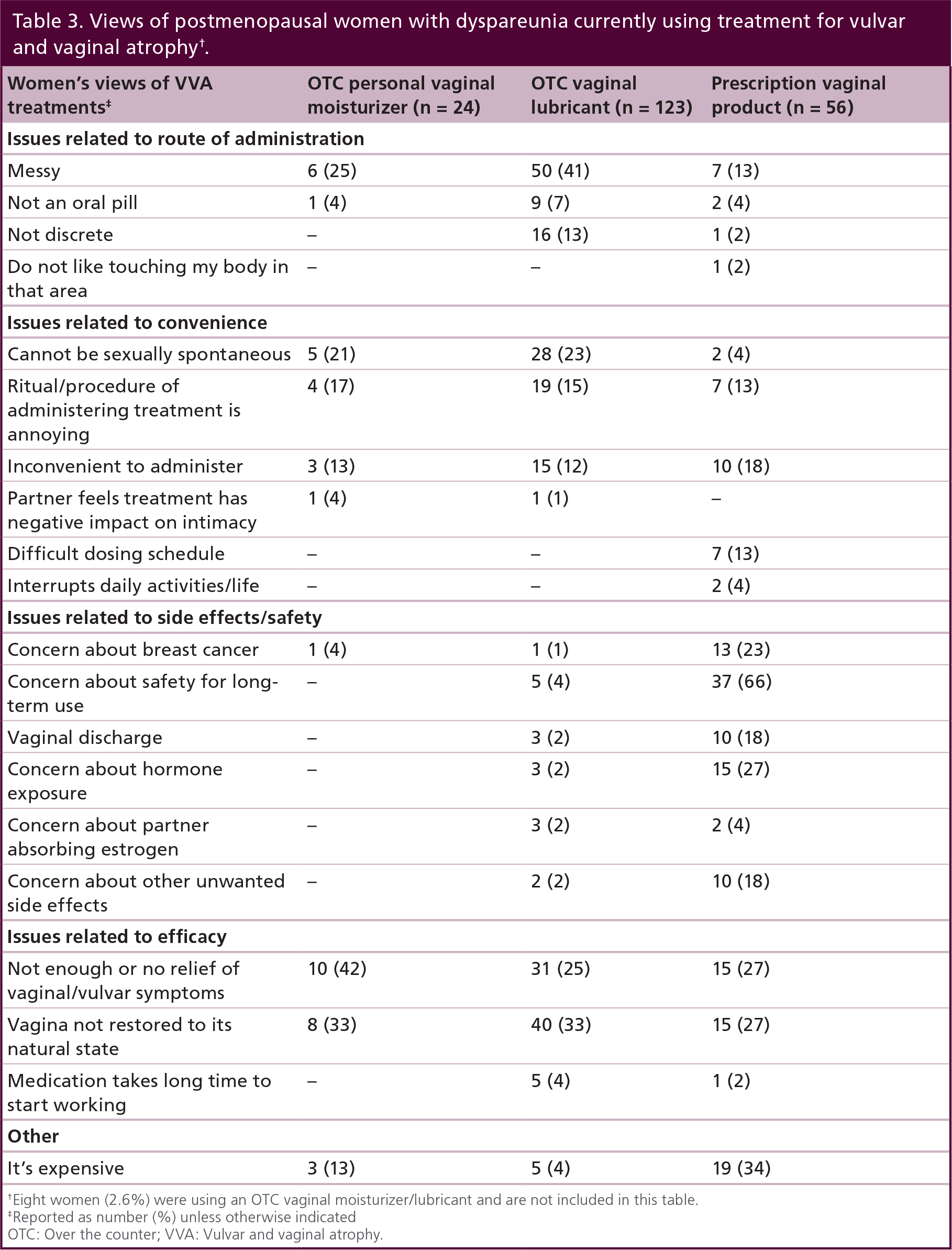
Eight women (2.6%) were using an OTC vaginal moisturizer/lubricant and are not included in this table.
Reported as number (%) unless otherwise indicated
OTC: Over the counter; VVA: Vulvar and vaginal atrophy.
Implementation for clinical practice
HCPs should be encouraged to initiate a discussion about preferences and perceptions regarding available VVA treatment options and to assist patients in their selection of therapy. However, since only 32% of HCPs indicated that they are extremely or very knowledgeable about symptoms of VVA such as dyspareunia [27], significant patient education is also needed to ensure they are aware of the relationship between the physical symptoms of VVA (i.e., dyspareunia and vaginal dryness) and menopause. Additionally, patients should be informed that OTC lubricants and moisturizers may provide temporary relief but that they do not prevent or reverse the unrelenting, progressive atrophic physical changes responsible for their vaginal symptoms. In line with a recently published position statement from The North American Menopause Society regarding the management of symptomatic VVA, a patient who presents with mild symptoms may choose to try an OTC product initially, but the HCP should consider a parallel treatment approach of providing a prescription that can be filled if the OTC product is not effective within a few weeks [2].
Suggestions for routine vulvar and vaginal atrophy screening.
Preface screening question(s) with general statement to place patient at ease: ‘Vaginal changes are very common for women who are going through menopause/postmenopausal.’†
If prior to vaginal examination: ‘Have you experienced any vaginal symptoms such as pain during sexual activity or vaginal dryness?’
If clinical signs of atrophy are present on vaginal examination‡: ‘I see some vaginal changes that can occur during menopause/postmenopause and may cause symptoms like pain, discomfort and/or dryness. Have you experienced any vaginal symptoms?’
Tailor language to patient's status (i.e., regarding undergoing menopausal transition or postmenopausal)
Common clinical signs of vulvar and vaginal atrophy include vaginal dryness, petechiae, pallor, friability, and redness of the mucosa
For a patient with moderate-to-severe VVA, therapy with proven efficacy should be considered first line. Vaginal estrogen therapies are effective, but their utility may be limited in women with safety concerns or in breast cancer survivors [2,28]. The recent approval of ospemifene provides postmenopausal women with moderate-to-severe dyspareunia due to menopause with an effective alternative treatment option. The increase in superficial cells and reduction in parabasal cells should improve vaginal moisture, while the reduction in vaginal pH should restore the normal ecosystem in the urogenital tract and reduce the risk of infection [1]. Regardless of the treatment selected, women should be advised as to when to expect symptom relief and to understand the possibility of long-term treatment. Such pre-emptive discussion will hopefully increase the likelihood of patient adherence.
Executive summary
The decline in estrogen levels associated with menopause can lead to vulvar and vaginal atrophy (VVA), a chronic and progressive medical condition characterized by bothersome physical symptoms such as dyspareunia, vaginal dryness, irritation, and itching. These symptoms can negatively impact sexual function and quality of life.
Therapies for symptomatic VVA include over-the-counter moisturizers or lubricants and/or estrogen products prescribed as creams, rings, or vaginal tablets. Ospemifene, an oral selective estrogen receptor modulator, is a nonestrogen option for the treatment of moderate-to-severe dyspareunia due to menopause.
The REVIVE survey assessed women's knowledge about VVA, the impact of vaginal symptoms on their lives, interactions with healthcare professionals (HCPs) about VVA, and their experiences with and attitudes toward treatment options for VVA.
The REVIVE survey included 3046 postmenopausal women in the USA who reported having VVA and/or symptoms consistent with VVA.
Each participant completed a structured, online questionnaire that collected basic demographic data and information on a range of areas related to their experience with VVA.
The REVIVE survey identified unmet needs for postmenopausal women with VVA symptoms, such as poor awareness and understanding of VVA symptoms and low rates of treatment despite the presence of symptoms.
Women reported displeasure with the vaginal administration of VVA treatments, inadequate symptom relief with over-the-counter products, and concerns about the safety of estrogen therapies.
Of women with VVA who completed the REVIVE survey, 44% reported dyspareunia as one of their symptoms.
Two-thirds of women in the REVIVE dyspareunia cohort rate sexual satisfaction as important or very important and the majority report that their VVA symptoms directly interfere with enjoyment of sex.
Similar to the overall REVIVE survey population, women with dyspareunia expressed considerable dissatisfaction with current treatment options.
Despite the impact of dyspareunia and dissatisfaction with available therapies, only slightly more than half of women had ever discussed dyspareunia with their HCP.
HCPs should be aware of the prevalence of VVA and they should be proactive in educating their patients on the causes of VVA and the associated physical symptoms such as dyspareunia and vaginal dryness. HCPs should assist patients in selecting a therapy based on the severity of symptoms, risk-benefit profile of treatment options, and patient preferences.
The REVIVE survey confirms the low awareness of VVA among postmenopausal women and shows that women living with VVA are reluctant to initiate discussion of vaginal symptoms with HCPs, despite the negative impact on their lives.
Participants in the REVIVE survey reported various issues with VVA treatments, including problems with the vaginal route of administration, lack of symptom relief with over-the-counter products, and concerns regarding the safety of estrogen therapies.
HCPs can educate patients about this medical condition and help patients find an effective treatment.
Conclusion
The REVIVE survey confirms the low awareness of VVA among postmenopausal women and shows that women experiencing VVA are reluctant to initiate discussion of vaginal symptoms with HCPs, despite the negative impact on their lives. These findings should raise HCPs' awareness of women at risk and make VVA discussion a part of routine visits for women who are either menopausal or approaching menopause.
The majority of women in the REVIVE survey reported that they were using OTC products. Patients should be advised that, although such products may alleviate symptoms initially, the atrophic process and involution continue. Patients should be advised to seek prescription therapy if and when they do not experience symptom relief with OTC products.
Participants in the REVIVE survey reported various issues with VVA treatments, including problems with the vaginal route of administration, lack of symptom relief with OTC products, and concerns regarding the safety of estrogen therapies. A new selective estrogen receptor modulator, ospemifene, has been approved for the treatment of moderate-to-severe dyspareunia in postmenopausal women with VVA and represents an effective and generally well-tolerated oral treatment alternative to vaginal estrogen therapies. By considering factors such as the severity of VVA symptoms, the impact on quality of life, and the individual patient's preferences and perceptions regarding treatment, HCPs will be better prepared to select appropriate therapy for their patients.
By initiating the discussion of VVA and dyspareunia, HCPs can educate patients about the importance of this medical condition that heretofore has remained largely unaddressed. VVA is a chronic medical condition that can have a major impact on sexual health and quality of life in postmenopausal women, and there are a number of effective therapies that are readily available. VVA and dyspareunia remain an awareness issue… so just ask!
Future perspective
With the population of postmenopausal women expected to increase significantly over the next 10 years, it is expected that OB/GYNs and other HCPs will experience an increased diagnostic frequency of VVA. The REVIVE survey highlighted that postmenopausal women with VVA symptoms have a high level of treatment dissatisfaction with current treatment options. While it is expected that new treatment options for the vaginal symptoms of VVA will provide benefits in therapeutic endpoints, long-term improvements in clinical outcomes and overall patient care for this chronic medical condition will be achieved through the greater communication between the patient, OB/GYNs, and allied HCPs.
Financial & competing interests disclosure
MA Freedman acknowledges his participation in advisory boards for Procter and Gamble, Noven and Shionogi Pharmaceutical Companies, and has received honorarium for speaking engagements from Shionogi and Noven. The author has no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed.
The author acknowledges E Downs, P Sjostedt and The Medicine Group for editorial assistance in the development of this manuscript, funded by Shionogi Inc. The author had full control over content, material, writing, and editing, and takes full responsibility.
