Abstract
In recent years, there has been an increasing focus on the contributory role of uterine fibroids to infertility. The prevalence of these tumors increases with age, which becomes significant as more women are delaying childbearing. Therefore, fibroids and infertility frequently occur together. Treatment varies with fibroid location and size. The various methods of treatment include open myomectomy, laparoscopic or robot-assisted myomectomy, medical treatment, uterine artery embolization and magnetic resonance guided focused ultrasound surgery. While there is a general consensus on the treatment of submucosal fibroids, the management of intramural fibroids in the infertility patient remains controversial. This paper aims to review and summarize the current literature in regards to the approach to uterine fibroids in the infertile patient.
Fibroids (also referred to as myomas and leiomyomas) are benign, monoclonal tumors of the uterus that are composed largely of smooth muscle cells. They are the most common tumor of the female genital tract, with some studies estimating over a 70% incidence [1,2]. Fibroids are the reason for approximately 30% of the hysterectomies performed in the USA [3]. There are many variations in the size and location of these tumors. Fibroids are described by their location in relation to the uterine wall. Subserosal fibroids are located on the external surface of the uterus and grow outward. Intramural fibroids grow within the uterine wall. Submucosal fibroids develop near the endometrium and tend to grow in toward the uterine cavity. Additionally there are pedunculated fibroids, which grow on a stalk, and can either be further classified as subserosal or submucosal depending on their location. Although most women are asymptomatic, approximately 25% of women develop symptoms such as pain, menorrhagia or other symptoms of mass effect from fibroids [1,4].
The pathogenesis of uterine myomas is still under investigation. It is accepted that their growth is stimulated by both estrogen and progesterone; however, the mechanisms surrounding the initial genesis of these tumors are not known [5]. A number of studies have shown that as many as 40% of individual fibroids have some chromosomal alteration [6]. The most common chromosomal abnormality found is a translocation of chromosomes 12 and 14. The HMGA2 gene, which was found in this translocation, is a proliferation modulator that is found in proliferative tissues. An in vitro study of HMGA2 antagonism found a resultant decrease in proliferation of leiomyoma cells [7]. Several other genetic loci and gene products have also been implicated in fibroid development and growth, including STE-20-like kinase, AKAP13 and MED12, among many others [6]. In addition, several chemokines, cytokines, extracellular matrix components (collagens, fibronectins) and growth factors have also been implicated in myoma growth. For example, TGF-β, particularly the β3 sub-unit, appears to be overexpressed in uterine fibroids. These biochemical components have been the focus of research to attempt to fnd potential treatments of fibroids. Furthermore, several neuropeptides, including substance P, neurotensin, neuropeptide tyrosine and vasoactive intestinal peptide have been found at similar levels in the pseudocapsule of myomas compared with normal myometrium [8,9]. Many of these neuropeptides have been implicated in healing and uterine contractility/peristalsis. This suggests that performing a myomectomy that spares the pseudocapsule may maintain the overall integrity of the myometrium, thus aiding in healing and potentially in future pregnancy outcomes. Additionally, a study by Baird et al. showed that vitamin D deficiency was associated with an increased likelihood of fibroid occurrence in both white and black women [10]. Further investigation of the molecular biology and genetics of fibroids will be required before any concrete conclusions can be made.
In recent years, there has been a growing interest in the effect that these tumors have on fertility. It is generally accepted that submucous fibroids, which inherently distort the uterine cavity, have a detrimental effect on fertility. A 2009 review by Pritts et al. found that fibroids causing intracavitary distortion result in decreased rates of clinical pregnancy, implantation and ongoing pregnancy/livebirth, as well as an increased rate of spontaneous miscarriage [11]. By contrast, there is controversy as to whether fibroids that do not cause distortion of the uterine cavity have any effect on fertility. However, in the same review, Pritts et al. found that patients with fibroids with no intracavitary involvement (particularly intramural fibroids), when compared with controls without fibroids, had decreased rates of implantation and ongoing pregnancy/live birth, and an increased rate of spontaneous miscarriage. Proposed etiologies for such effects of fibroids without intracavitary involvement include alterations of uterine peristalsis and vascular flow as well as disruption of sperm and ovum transportation and embryo implantation [12–16]. One weakness of Pritt's review is that most of the studies did not fully evaluate the involvement of the uterine cavity. No evidence was found that subserosal fibroids decreased any measure of fertility. Nonetheless, the data provide compelling evidence that there may be situations in which surgical removal of nonsubmucosal fibroids is indicated in the infertile patient. Furthermore, several studies have shown a detrimental effect of fibroids without intracavitary involvement on IVF outcomes, particularly myomas larger than 4 cm [17–19]. Although other studies refute this evidence, more high quality research is needed to further evaluate the role of intramural fibroids in the setting of infertility [20,21].
One of the biggest gaps in this area of clinical research is the lack of consistency of the precise localization of fibroids in each of the different studies that have been performed. There is often no explanation of how fibroids were classified into one group versus another (e.g., imaging modality, precise cut offs, etc.). Additionally, subserosal and intramural fibroids are often considered as a single entity in these studies. The ESGE/FIGO leiomyoma subclassification system may aid in addressing this inconsistency (Figure 1). Although there have been some studies using this classification system, there is a dearth of large, randomized control trials using it to describe the myomas being studied [22,23]. This review will focus on the accepted evidence in the diagnosis and treatment of uterine fibroids as they pertain to infertility, as well as suggested directions for future research.
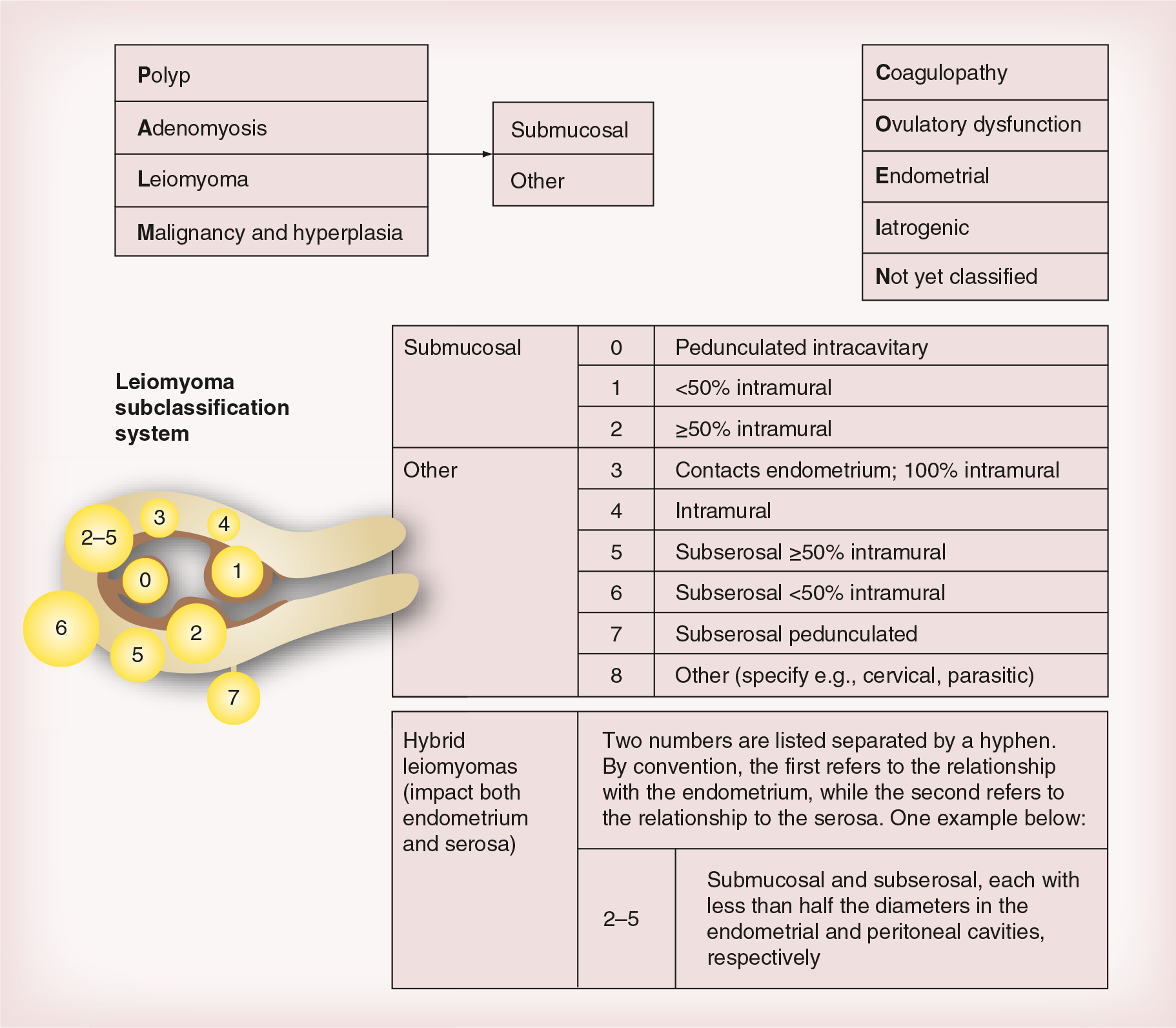
ESGE/FIGO classification.
Diagnosis
There are several methods to identify and characterize uterine fibroids, including ultrasound (transabdominal, transvaginal), sonohysterogram, hysterosalpingogram, MRI and CT scans. Of particular importance is each modality's ability to characterize the involvement of the uterine cavity. Hysterosalpingogram has very low sensitivity (approximately 50%) and specificity (as low as 20%) for the evaluation of uterine cavity involvement [25,26]. Until recently, transvaginal ultrasound had been considered accurate in the evaluation of submucous fibroids, with older studies showing a sensitivity of 100% and specificity of 94% [27]. A more recent study comparing transvaginal ultrasound with MRI, however, reports a sensitivity and specificity of 69% and 83%, respectively [28]. This same study reports a sensitivity and specificity of 100% and 91%, respectively, for MRI, which is currently considered to be the most accurate imaging modality for the diagnosis and characterization of intramural and submucous fibroids. Unfortunately, few studies use MRI preoperatively for diagnosis, thus leading to potential underreporting of intracavitary involvement. A 2003 review reported that saline infusion sonography is equivalent to hysteroscopy for the diagnosis of submucous fibroids, with both techniques being superior to transvaginal ultrasound [29]. The 3D sonohysterography has also been found to be comparable to both standard 2D-sonohysterography and hysteroscopy [30,31].
Medical management
There are several options for the medical management of uterine myomas. Currently, the only US FDA approved medical treatments for symptomatic fibroids are GnRH analog (leuprolide acetate) and levonorgestrel-containing intrauterine device (Mirena®, Bayer AG, Berlin, Germany) [32]. Leuprolide has been approved for decreasing the size of fibroids, while Mirena is approved for the treatment of heavy menses, not necessarily related to fibroids. Several other medications are currently undergoing clinical trials, many of which appear to have potential for the treatment of uterine myomas. These include ulipristal acetate, proellex, mifepristone, asoprisnil, aromatase inhibitors, pirfenidone and epigallocatechin gallate. Ulipristal, a selective progesterone-receptor modulator, in particular has shown promise. Recent studies by Donnez et al. have shown a reduction in bleeding and fibroid size with ulipristal versus placebo, as well as non-inferiority compared with leuprolide [33,34]. Additionally, ulipristal was shown to be less likely to cause hot flashes in study subjects than leuprolide acetate.
However, while these options for the medical management of uterine myomas exist, in general they are not appropriate in the setting of desired fertility. Leuprolide and Mirena delay the ability to pursue attempts at conception, and the medications currently under clinical investigations have yet to be thoroughly studied in patients pursuing pregnancy. Perhaps the only exception is the use of a brief course of GnRH analog, which may help correct preoperative anemia if the patient is experiencing heavy bleeding [22–23,35]. Further details on medical management of fibroids are outside the scope of this review.
Surgical management
In the setting of infertile patients in whom fibroids are felt to contribute to difficulty conceiving or recurrent miscarriages, surgical removal is the most appropriate option. Any surgical intervention is best optimized by preoperative imaging to determine the precise size and location of the fibroid(s), as reviewed above. This helps to determine which surgical approach is the most appropriate for a particular patient. However, unless a hysterectomy is performed, there is always the possibility of a recurrence. Additionally, preexisting myomas that were not removed may grow in size. Both of these situations may require additional surgery [36]. It is also important for patients to understand that surgery cannot guarantee conception, live birth or complete resolution of symptoms. The possibility of hysterectomy if there are complications must always be discussed with patients undergoing consent for myomectomy, regardless of the mode of surgery. The removal of submucous myomas has been shown to increase clinical pregnancy rates in comparison with controls where their fibroids were left in situ [13,37]. However, myomectomy in the setting of intramural fibroids is more controversial, as discussed previously.
Hysteroscopy
Hysteroscopic myomectomy is the preferred surgical modality whenever possible. It is appropriate for submucous myomas that are up to 4–5 cm in size. Hysteroscopic resection can be performed on type 0, 1 or 2 myomas (Figure 1), although type 2 fibroids often require multiple procedures for complete resection [22,38,39]. Complications of hysteroscopic myomectomy include uterine perforation, fluid overload, bleeding and intracavitary adhesion formation [22]. Many fibroids with a significant intramural component (some type 2s, hybrid myomas; Figure 1) as well as any with no intracavitary involvement (types 3–8) require abdominal removal, whether via laparotomy, traditional laparoscopy or robotic-assisted laparoscopy. An additional classification, created by Lasmar et al., that is specific to hysteroscopic myomectomy exists. This system (STEPW), has been shown to be a better predictor of successful hysteroscopic fibroid removal and surgical complications than the ESGE/FIGO system (Table 1 & Figure 1).
STEPW classification.

Sum of points reflects complexity and therapeutic options: 0–4 (group I): low complexity hysteroscopic myomectomy; 5–6 (Group II): high complexity hysteroscopic myomectomy, consider preoperative GnRH analog and/or two-step resection; 7–9 (Group III): consider alternative to hysteroscopic technique.
Adapted with permission from [40].
There are several techniques available for hysteroscopic myomectomy. These include electrocautery resection, morcellation, laser ablation and vaporization [41]. Electrocautery resection can be performed with monopolar energy, which requires a nonconductive distension media, such as glycine, or bipolar energy (can be used with saline). This method of resection can be used in any case where hysteroscopic myomectomy is appropriate; however, it may be more time consuming and requires more advanced cervical dilation than other techniques. Laser ablation is appropriate for myomas measuring 2 cm or less in diameter. The disadvantages of this technique are operative time, the prohibitive cost of the equipment and the inability to evaluate a tissue specimen. Studies of electrode vaporization have shown shorter operative times than resectoscopic surgery. However, the risks of this procedure include the lack of ability to evaluate the pathologic specimen in its entirety and the potential for significantly high CO2 blood levels resulting from the many gas bubbles produced. In 2005, the FDA approved the first hysteroscopic morcellator, the TRUCLEAR™ Hysteroscopic Morcellator (Smith & Nephew, Andover, MA, USA) [42]. Shortly thereafter, in 2009, a second device, the MyoSure® Tissue Removal System (Hologic, Bedford, MA, USA) was approved. Both of these devices function via mechanical energy generated by a motor with suction applied for specimen collection. Studies to date have shown that hysteroscopic morcellation is often faster than traditional electrocautery resection, with reductions in operating room time from 38% to 72%, depending on the type of lesion being removed. This is largely because no additional time is required for removal of portions of surgical specimen. As this is a newer technique, research regarding the potential for fluid overload is less extensive at this point. The main disadvantage of hysteroscopic morcellation is that it is not appropriate for myomas with greater than 50% extension into the uterine wall. Laser ablation, morcellation and vaporization are felt to be less traumatic techniques and in theory could improve fertility, although further study is needed to confirm this.
Laparotomy
Although a minimally invasive approach is often desirable, at times open myomectomy is necessary. If there are numerous complicating intra-abdominal adhesions, and/or the surgeon is not confident in his or her laparoscopic skills, then laparotomy should be performed [22]. In these circumstances, however, the standard risks of open abdominal surgery apply, including the risk of increased blood loss and longer recovery time in comparison with minimally invasive approaches. The advent of laparoscopic morcellators has decreased the need for the open approach, as previously it was required for large myomas to be removed via laparotomy. However, very large myomas (i.e., >20 cm in diameter) or any that are suspected of being malignant still require an open approach [43].
Laparoscopy
In comparison with laparotomy, laparoscopic myomectomy provides improved visualization, decreased blood loss, quicker recovery and decreased postoperative pain [44,45]. However, pregnancy outcomes and the risk of fibroid recurrence appear to be similar [46]. There have been some reports of concern for uterine rupture during pregnancy following laparoscopic myomectomy, although the real risk is unclear as some studies report an increased risk of rupture, while others report a decreased risk [47,48]. This concern is partly related to the technical difficulty of performing a multilayer closure [49]. However, the overall incidence of uterine rupture following laparoscopic myomectomy appears to be extremely low, and there is as yet no definitive evidence that this risk does not also exist for laparotomy [50].
Robot-assisted laparoscopy
The FDA cleared the da Vinci system for laparoscopic surgery in 2000 and subsequently for use in gynecologic procedures in 2005 [51]. Since then, the use of robotic-assisted laparoscopic procedures, including hysterectomy, urogynecologic procedures and myomectomy has become increasingly common. Some of the potential advantages that the robotic system offers are improved visibility with 3D viewing and the intuitive wrist-like movements of the operating arms [52]. The short-term outcomes have been found to be similar to those of traditional laparoscopy, specifically, decreased blood loss and faster recovery [53,54]. In addition, a recent retrospective cohort study found pregnancy outcomes following robotic myomectomy to be comparable to those found after traditional laparoscopy [55]. In the hands of a surgeon with the appropriate technical expertise, the robotic approach affords the ability to perform laparoscopic myomectomies that otherwise may have needed to be performed via laparotomy. The most notable disadvantages of the robotic system are increased cost and increased operating room time [56]. Further investigation through cost–benefit analysis needs to be performed to better define the role of robot-assisted myomectomy in the reproductive field.
Other treatment methods
Uterine artery embolization (UAE) (see Figure 2) and magnetic resonance focused ultrasound surgery (MRgFUS) are two alternative minimally invasive treatments for symptomatic fibroids. Pregnancy is possible after these treatments, however, there are reports of increased obstetrical complications in such pregnancies, including miscarriage, abnormal placentation and postpartum hemorrhage. Although there are numerous case reports of uncomplicated pregnancies after UAE and MRgFUS, there is limited evidence for the use of these modalities in patients seeking future fertility, and it is too early to recommend embolization in the infertile patient desiring conception, although further study is needed [22,23].

Conclusion
Uterine myomas clearly have a detrimental effect on fertility outcomes. While there is a great deal of evidence showing that submucous fibroids are associated with infertility, intramural fibroids are implicated, but the data are less conclusive. Myomectomy should be performed in selected patients, in whom fibroids are either symptomatic or are felt to be a contributing cause of their infertility. Appropriate imaging studies are extremely important in this determination, with saline infusion sonography, hysteroscopy and MRI being the preferred methods of fibroid characterization. While medical treatments exist for the treatment of symptomatic fibroids, these therapies impede conception, and are generally avoided in the setting of desired fertility. Several surgical approaches to myomectomy are available – hysteroscopy, laparotomy, laparoscopy and robot-assisted laparoscopy. Hysteroscopic resection is almost always the preferred method if appropriate for the given case as the outcomes are favorable and the morbidity is minimal. Laparotomy and laparoscopic methods (traditional and robot-assisted) have comparable pregnancy outcomes, however, the morbidity associated with laparotomy is much greater. A minimally invasive approach is often preferable when possible. The rigid instruments of traditional laparoscopy can make myomectomy technically difficult, while an experienced surgeon can perform robot-assisted laparoscopic myomectomy much more efficiently. Other methods of treatment such as UAE and MRgFUS are not currently felt to be appropriate in patients seeking future fertility.
Executive summary
Uterine fibroids are very common, with incidence increasing with age.
Partly due to the fact that more women are delaying childbearing, fibroids are being seen more frequently in infertility patients.
Submucous myomas are strongly associated with poor fertility outcomes and should be removed in the setting of infertility.
While intramural myomas have also been associated with poor fertility outcomes, the data are not as strong as for submucosal fibroids, and thus future research is necessary.
Further study of the biochemical etiology of fibroids is needed in order to develop additional treatments as well as possible preventative agents.
The ESGE/FIGO leiomyoma subclassification system provides an objective way to describe myomas and a framework for determining how best to remove them.
Multiple imaging modalities are available for the evaluation of uterine fibroids.
Hysterosalpingogram is a poor diagnostic tool and of limited use in the evaluation for myomas.
Transvaginal ultrasound was previously felt to be extremely accurate; however, more recently it is felt to be a less sensitive tool than other methods.
3D sonohysterography has been found to be comparable to 2D sonohysterography and diagnostic hysteroscopy.
Saline infusion sonography, hysteroscopy and MRI are the preferred imaging methods for the diagnosis and characterization of uterine fibroids.
Most medical therapies for fibroids impede conception and thus are not appropriate in the setting of the patient who is seeking fertility.
The one exception may be the preoperative use of GnRH agonists in the setting of anemia to decrease bleeding and help build up hemoglobin levels.
Many agents are currently under investigation and may prove to be useful in the setting of fibroids in infertility.
Surgical approaches include hysteroscopy, laparotomy, laparoscopy and robot-assisted laparoscopy.
Hysteroscopic methods are preferred for submucous myomas as they have a proven history of favorable outcomes with little morbidity.
Laparotomy is necessary for extremely large myomas, or whenever there is a concern for malignancy, however the open approach is associated with increased blood loss, more postoperative pain and longer recovery time.
Both traditional and robot-assisted laparoscopy have similar pregnancy outcomes compared with laparotomy, however they are also associated with a significantly decreased morbidity.
Robot-assisted myomectomy, when used by an experienced surgeon, can overcome many of the spatial difficulties presented by traditional laparoscopy. The major disadvantages, however, are the increased cost and operating room time.
Further cost-benefit analysis is necessary to define the role of robot assisted myomectomy in the reproductive field.
While uterine artery embolization and magnetic resonance focused ultrasound surgery are available for the treatment of symptomatic leiomyomas, a small amount of data suggest they are associated with adverse obstetric outcomes.
There have, however, been reports of uncomplicated pregnancies with both of these techniques.
Further study is necessary to determine the safety of uterine artery embolization and magnetic resonance-focused ultrasound surgery in patients with fibroids desiring future fertility.
The precise characterization of myomas is important in selecting appropriate surgical candidates and subsequently the correct surgical approach.
Proposed directions for future study include: more specific characterization of fibroids in the setting of infertility prior to intervention, uterine artery embolization and magnetic resonance focused ultrasound surgery in the setting of infertility, biochemical etiology and growth factors of fibroids and their potential targets for treatment, and further study of robot-assisted myomectomy compared with open and traditional laparoscopy.
Future perspective
Future studies should focus on precise characterization of myoma location in order to further investigate the effects of intramural and subserosal fibroids on fertility. This could be achieved using the ESGE/FIGO leiomyoma subclassification system (Figure 1) or an alternative system. Ongoing studies of medical management may identify pharmacologic therapies that could be of potential benefit to infertility patients. Additionally, researchers should focus on further study of outcomes of robot-assisted myomectomy in comparison to laparotomy and traditional laparoscopy, including cost–benefit analysis with a focus on the societal benefits of a shorter recovery period. UAE and MRgFUS may also prove to be less invasive alternatives for infertility patients; however, more extensive study is needed to further delineate the effects of these methods on future fertility.
Footnotes
L Barmat is an Intuitive Surgical proctor for gynecologic surgeries. The authors have no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed.
No writing assistance was utilized in the production of this manuscript.
