Abstract
The only untreatable subgroup of female infertility is absolute uterine factor infertility (AUFI), which is due to congenital or surgical absence of a uterus or presence of a nonfunctional uterus. The solitary option for a woman with AUFI to become a biological mother today is through a gestational surrogate mother, a procedure that is prohibited in Sweden and large parts of the world. Uterus transplantation (UTx) is a potential future treatment of AUFI. After extensive animal research, also involving non-human primates, a small number of human UTx cases have recently been performed. Here, we summarize the primate UTx experiments that have paved the way for the human UTx cases, which are described and analyzed in more detail. We also estimate how many women of fertile age are affected by AUFI and describe the causes. The ethics around UTx is complex and is also addressed.
Background
In women of reproductive age, the prevalence of infertility in general is described to be between 3.5 and 16.7% [1]. Women with absolute uterine factor infertility (AUFI) represent the last major group of untreatable female infertility. The principle defect in AUFI patients is either congenital or surgical absence of the uterus or a present uterus that is nonfunctional due to an anatomical or physiological defect. The major subgroups of AUFI are outlined in Figure 1. It should be acknowledged that gestational surrogacy is an alternative for some of these women to become genetic mothers; however, this procedure is only permitted in a restricted number of nations. Legal, ethical and religious concerns have been raised against gestational surrogacy.
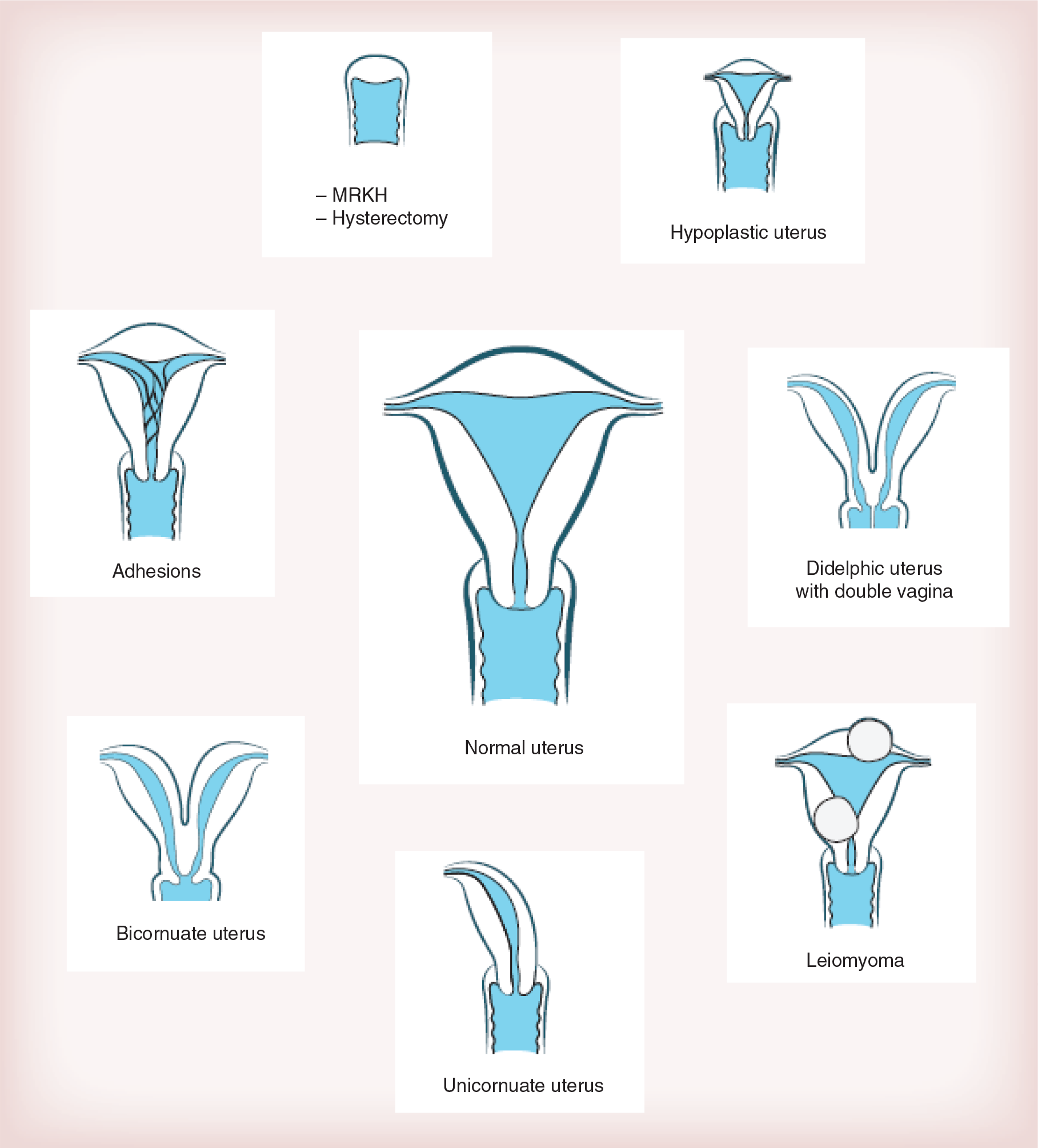
Different causes of absolute uterine factor infertility.
Today, transplantation of organs/tissues is not only restricted to transplantation of organs that are necessary for continued life. In recent years, quality-of-life-enhancing types of transplantation, such as of the face, hand and larynx, have also entered the clinical arena. It may well be that uterus transplantation (UTx) will become the first category of quality-of-life-enhancing allogeneic transplantation that is also a life-propagating transplantation. In addition, UTx would be the first type of organ transplantation that can be categorized as ephemeral, with the organ only needed for a restricted time. Thus, life-long immunosuppression, with its associated long-term side effects, would be avoided.
The achievements in UTx research have been major during the last decade, with key experiments performed in several animal models [2–8]. Important issues that have been resolved are uterine ischemic tolerability [7,9,10], pregnancy potential in non rejection [2,4,7] and allogeneic settings [3,8], rejection mechanisms [11,12] and optimization of immunosuppression [13–15].
Absolute uterine factor infertility
AUFI affects approximately one in every 500 women of fertile age [16], and this would correspond to around 200,000 in Europe and 85,000 in the USA. This type of infertility has many causes (Figure 1) and can be categorized as either congenital or acquired.
The congenital cause of of AUFI is any type of severe Müllerian duct anomaly [17]. The paired Müllerian ducts are the embryological tissues that during early fetal life partly fuse to form the major part of the vagina and the uterus. There exist several types of uterine malformations that are caused by developmental deficiencies of certain parts of the Müllerian ducts or pathological fusion of the ducts. Recently, a consensus of a novel type of classification of the female genital tract congenital malformations was reached by the CONUTA (Congenital Uterine Anomalies) working group [18]. This group suggests a somewhat revised classification system from the traditional classification from the late 1970s [19]. In the section below we use the traditional classification system, since this is still the most widely used.
The prevalence of any type of Müllerian duct anomaly among females is approximately 4.3% [16], and in infertile women more than 10% have any type of uterine malformation [20]. The majority of women with uterine malformations do not experience infertility, and our estimation is that approximately 20% of these women remain infertile, despite the fact that minor uterine mal-formations can be corrected by surgery (see following paragraph).
The septate uterus, with a remnant of the fused medial walls of the ducts, represents approximately 30% of all uterine malformations [21]. There is a high rate of spontaneous abortion with a present septa [22], but hysteroscopic resection can normalize this [23]. The bicornuate uterus stands for approximately 25% of all uterine malformations [16]. Surgery [24] lowers the rate of spontaneous abortion [25]. The unicornuate uterus makes up approximately 10% of the uterine malformations [16]. This condition is associated with a 28–59% risk of spontaneous abortion [26–28]. Equally common is the didelphic uterus [16,29], with a close to normal implantation rate. However, the spontaneous abortion rate in this condition is as high as 30% and the total live birth rate is only 50% [16]. Surgical attempts to correct the didelphic uterine malformation do not improve the pregnancy outcome. The hypoplastic uterus, also named uterus infantilis is a rather rare condition. This is a small uterus with a narrow cavity and with a cervix being considerably larger than the uterine body [18].
The rarest type of Müllerian duct anomaly is uterine agenesis, which is seen in one in every 4500 newborn girls [30,31]. The syndrome is generally called the Mayer–Rokitansky–Küster–Hauser (MRKH) syndrome or only the Rokitansky syndrome. The general phenotype of this syndrome is presence of paired uterine rudimentary buds on the pelvic sidewalls with normal ovaries, which in many occasions are located lateral to the external iliac vessels. Gestational surrogacy is a way for these women to acquire genetic motherhood [32]. Importantly, gestational surrogate pregnancies with MRKH women as genetic mothers do not seem to have any increased risk of fetal uterine malformation [33], which would indicate that epigenetic changes may be behind this syndrome.
Taken together, many types of congenital uterine malformations result in implantation failure or in early pregnancy loss. In a large study of women with recurrent spontaneous abortion, uterine malformations were found in almost 20% of these women [34].
Leiomyoma is likely the most frequent cause of AUFI. The reported prevalence of leiomyoma among reproductive-aged women ranges from approximately 20 [35] to 6% [36,37]. Hysterectomy secondary to leiomyoma, is naturally the most obvious group of leiomyoma-related AUFI and approximately 2% of all 30–40-year-old women in the USA have undergone hysterectomy due to leiomyoma-related symptoms [38]. Infertility in a woman with uterine leiomyoma [39,40] may be due to both structural and biochemical factors [41]. Mainly, larger (>4 cm) and subendometrial leiomyomas cause infertility [42]. Adenomyosis may also be a cause of infertility and hysterectomy [43].
Cervical cancer is globally the most common gynecological malignancy and 30–40% of patients are of fertile age at diagnosis [44,45]. Primary surgery is the main cervical cancer treatment and in some early stages, treatment can be by uterine-sparing surgery techniques; however, radical hysterectomy is recommended to be performed in stage Ib of >2 cm and IIa tumors [46]. In more advanced stages, a combination of radio- and chemo-therapy is recommended and infertility is definite because of permanent ovarian damage.
Emergency peripartum hysterectomy is the last possible intervention when massive obstetric bleeding occurs due to uterine rupture/atony or invasive malplacentation. Today, the incidence of hysterectomy at delivery is approximately 50 in 100,000 deliveries [47,48]; however, the rate is predicted to continuously increase in parallel with the increased rate of delivery by cesarean section with its associated risk of invasive malplacentation [49].
Intrauterine adhesions, secondary to endometritis or surgical abortion [50,51], are additional major causes of AUFI. Approximately 50% of women with adhesions inside the uterine cavity remain infertile [50], despite hysteroscopic adhesiolysis [52].
Non-human primates & UTx research
Extensive research on UTx has been performed during the last decade, and these experiments involved the traditional experimental animals of rodents, rabbit, pig and sheep [53]. Lately, the UTx research has also come to include non-human primates, with their added advantage of a near human anatomy and physiology of the reproductive organs. The importance of primate models towards human application has lately been emphasized [54]. Thus, the higher non-human primates, such as the baboon and macaque, have a simplex uterus with a menstrual cycle that is almost identical to the human [55]. Just as in the human, arterial inflow to the non-human primate uterus is mainly through the uterine arteries, but with essential blood flow also through the vaginal and ovarian arteries. The venous outflow is through several uterine/vaginal veins and especially through the ovarian veins. Three non-human primate subspecies have primarily been subjected to research involving UTx, and these are the rhesus macaque, the cynomolgus macaque and the baboon.
In the following section, the non-human primate UTx experiments are summarized according to the principle type of UTx that was performed. There is typically a sequence of different types of transplantation performed in animal species when a new type of organ transplantation is developed. Autologous transplantation is usually conducted early in any organ-specific experimental transplantation research, in order to separately test the effects of the surgical approach by assessing function of the graft. In the more complex allogeneic transplantation, effects of rejection and immunosuppression are added factors to the experimental situation.
Avascular UTx in primates
James Scott and co-workers performed pioneering work in non-human UTx research more than 40 years ago. Their avascular UTx model in the rhesus macaque included both autologous and allogenic transplantations [56]. The retrieval surgery comprised subtotal hysterectomy with the Fallopian tubes attached. At transplantation, the lower uterine segment was sutured to the remnant cervix and the fimbriaes of the Fallopian tubes were reattached in close proximity to the ovaries. Vascularization of the graft was by neovascularization after omental wrapping of the graft. This autologous avascular UTx resulted in resumed and regular menstruation in three out of four animals but no pregnancy occurred, despite multiple breeding attempts. It was speculated that the post-transplantation infertility was due to blocked Fallopian tubes, secondary to surgical or ischemic damage. At autopsy, the uteri were of normal macroscopic and histological appearances. Four allogeneic UTx attempts were done in animals with no immunosuppression and consequently severe rejection with uterine necrosis was seen within a couple of weeks following transplantation.
Vascular autologous UTx in primates
In preparations prior to the world's first human UTx case in Saudi Arabia in the year 2000, some tests involved UTx-like experiments in 16 baboons [57]. The first eight animals underwent end-to-end uterine vessel anastomosis at autotransplantation of the uterus, but this resulted in vascular thrombosis in a vast majority of cases [57]. It was not mentioned whether the uterine veins were also reconnected or if the larger venous outflow through the ovarian veins were preserved. Our own experience of UTx in the baboon [58] is that the uterine artery has an inner diameter of less than 1 mm at the level of the ureter and that the uterine veins are extremely small. Thus, the miniature size of the uterine vessels, with the complexity to establish functional anastomoses, may underlie this failure [57]. In the second part of the study, also involving eight animals [57], the sites and types of anastomosis were changed to end-to-side between the uterine vessels and the internal iliac vessels. This modification improved the vascular patency, with around 90% patent anastomoses at euthanasia 6–12 weeks after UTx. Unfortunately, the paper does not mention any data on functionality of these autotransplanted uteri.
In our initial study on autologous UTx in the baboon, the graft comprised the uterus together with the ovaries and Fallopian tubes [58]. The graft contained four vascular pedicles. These were the infundbulopelvic ligaments with the ovarian veins and the lateral pedicles of the uterine arteries, with the anterior divisions of the internal iliac arteries. Presence of the ovaries permitted longitudinal, noninvasive assessment of the graft post-transplantation, since ovarian hormonal cyclicity could be monitored as cyclic perineal changes. Menstruation would provide some data on uterine functionality. Back-table preparation of the graft included side-to-side anastomosis of the distal pedicle ends of both arteries and the veins, to create two larger vascular ends. These could then be anastomosed unilaterally to the external iliac vessels in an end-to-side fashion.
In our study of ten baboons [58], the total surgical time (organ recovery, back-table preparation and transplantation) was approximately 6 h. The recovery surgery lasted approximately three-times longer than the transplantation surgery, pointing towards the complexity to remove the uterus without damaging nearby structures, such as the bladder, ureters, rectum. Despite the extended and complicated surgery, involving both organ recovery and transplantation, nine out of ten animals showed long-term survival. Five animals exhibited ovarian cyclicity, but menstruation was only seen in two animals. Thus, only a fifth of the autotransplants showed the menstrual signs of functionality. However, menstruation is only one crude sign of normal uterine functionality and other functional defects may well exist, which at a pregnancy attempt would negatively affect the sensitive processes of implantation and placentation.
In our follow-up study on autologous UTx in the baboon [59], we initially repeated the surgical procedure of our first baboon study [58]. Among these six autotransplanted animals, two animals died during the postoperative period. The four surviving animals resumed ovarian cyclicity within 4 months but with absence of menstruation. At autopsy, only fibrotic remnants of the uteri could be seen but the ovaries were of normal appearance. Since the ovaries were also supplied by uterine artery blood flow in this UTx model, the results indicate that the ovary has a superior capacity compared with the uterus to survive periods of reduced blood flow. It may well be that blood flow to both the uterus and ovaries has been completely interrupted for a long period but that the capacity of the ovary to induce neovascularization from adjacent tissues is greater than that of the uterus.
Several modifications were tested to improve the results in this autologous baboon UTx model [59]. Anastomosis surgery was now performed by a transplant surgeon, rather than by a gynecologist, and the graft was perfused first with heparinized saline and then at back-table flushed with proper organ-preservation solution, instead of physiological saline. Moreover, the main trunks of the internal iliac arteries were also included in the graft to achieve a larger arterial end for anastomosis. At back-table preparation, the smallest iliac artery end of the graft was coupled end-to-end to the contralateral posterior branch of the internal iliac. At transplantation, this construct was coupled end-to-end to the proximal remnant of the internal iliac artery. Extended dissections of the ovarian venous pedicles, including patches of the vena cava and left kidney vein, were also performed to acquire veins with walls of adequate thickness for easy anastomosis to the external iliac veins. The animal survival after these modifications was 100% and 80% of the animals resumed cyclic hormonal patterns. Importantly, the success rate increased threefold from the original study [58], since 60% of the animals presented with resumed regular menstruation [59]. However, no pregnancy occurred, despite breeding attempts over several months. Tubal blockage was later seen at postmortem analysis.
The cynomolgus macaque has also been used for autologous UTx experiments [60]. The adult female cynomolgus macaque is approximately half the size of the female baboon and has a somewhat shallower placentation than the human and the baboon. This size factor is most likely the cause that uterine recovery extends over 6–8 h in this species. The recovery surgery included isolation of the uterus with dissections of the uterine arteries and the largest uterine veins, which in a majority of cases were the superficial uterine veins [60]. Vascular anastomosis was bilateral with end-to-side anastomosis of the uterine vessels to the external iliacs, and occasionally to the internal iliac artery. The anastomosis surgery was complicated because of the small vessel sizes, resulting in surgery on each animal for more than 10 h. One surviving animal demonstrated restored menstruation 4–5 months after autologous UTx.
In a follow-up study, four cynomolgus macaques underwent autologous UTx [61]. The total surgery duration was 12.5–17.5 h. The warm ischemia lasted for an extended duration of 4–6 h, due to the fact that complicated uterine vessel anastomosis to the external iliacs had to be performed. Only one animal showed long-term uterine survival and unfortunately it is not stated what specific anastomosis technique was used in that case.
The first pregnancy ever after any type of UTx in a non-human primate species was reported in 2012 [62]. In this autologous cynomolgus macaque UTx model, pregnancy was achieved in one animal several months after UTx, involving bilateral uterine artery anastomosis to external iliac arteries. Venous outflow was through one deep uterine vein and the contralateral ovarian vein, which were coupled end-to-side to the external iliac veins. Natural mating resulted in a pregnancy that developed normally until placental abruption occurred near term. A live offspring was delivered. It should be pointed out that up until today, this is the only reported pregnancy with live offspring in any non-primate species.
Vascular allogeneic UTx in primates
The first study of allogenic vascular allogeneic UTx in an non-human primate included recovery of a uterine graft with extensive vascular pedicles in the rhesus macaque [63]. The anastomosis site of the recipient followed a preferred hierarchy of internal iliac vessels, external iliac vessels, common iliac vessels and aorta together with vena cava. Single cyclosporine therapy was used to prevent rejection. Unfortunately, neither graft survival nor animal health was reported.
We have also performed allogeneic UTx, using the baboon model with transplantation from live donors [59]. Uterus recovery surgery involved initial excision of the oviducts and ovaries from the graft. Thereafter dissections of the uterine arteries, with inclusion of the major parts of the internal iliac arteries and of the ovarian veins were performed. The ovarian veins were freed up to their origins to also include vein patches of the vena cava and the left renal vein. Even if the recovery surgery extended over 3 h, the immediate postoperative survival was 100%. Various immunosuppression protocols were tested and it was found that induction therapy, with antithymocyte globulin, followed by triple immunosuppression with tacrolimus, mycophenolate and corticosteroids was compatible with long-term graft survival. The short-term survival rate of the recipients was 100%. Although hormonal cyclicity reappeared in some animals, none showed resumed menstruation.
In another study of allogeneic UTx in the baboon, using grafts from deceased donors, anastomoses were performed with the aorta and vena cava of the graft to the recipient's aorta and vena cava [Brännström M et al. Unpublished Data]. The initial immunosuppression protocol was identical from our live donor UTx study and after some months only tacrolimus was given. Although episodes of graft rejection occurred, these were successfully treated and graft survival over 12 months occurred.
Human UTx
As described above, a large knowledgebase exists concerning UTx in animal species, including non-human primate species. There have now been eleven human UTx attempts performed and if at least one of these is successful, new human trials will most likely occur. If pregnancy with birth of a healthy baby cannot be demonstrated in any of the eight human females, who today carry a uterine graft, the UTx concept should go back to the non-human primate research setting.
In the context of human UTx, it is appropriate to discuss what sources of a transplantable uterus exist. In the clinical field of transplantation surgery, the source of organs/tissues for transplantation is mainly brain-dead, heart-beating donors, also named ‘deceased brain-dead donors’ (DBDs). The organ scarcity has led to the use of also non heart-beating donors, also named ‘donation after cardiac death’. Live donors are also organ sources in kidney, partial liver and single lung transplantation, but with wide geographical variations in the rate of live donation versus deceased donation.
In a human UTx situation, both live and deceased donation can be considered. Uterus recovery from a live donor would be complicated since long vascular pedicles should be harvested bilaterally to establish sufficient lengths for subsequent direct arterial and venous anastomoses, without usage of interposing vessel grafts. Although dissection of the uterine arteries, with mobilization from the ureters, is a standard procedure in radical hysterectomy, the specific dissections of the thin-walled uterine veins that run over and under the ureters may be very complicated and time-consuming. The potential benefits of live donation are the possibility to plan the surgical procedure in advance, with both patients in optimal condition and with minimal exposure of the uterus to cold ischemia. Moreover, the organ would be less damaged by ischemia, and the systemic inflammation that affects an organ from a deceased donor, would be precluded. The organ-specific survival benefit with live donor, as opposed to deceased donor, is minor and evident only in large patient materials as demonstrated in kidney transplantation [64]. Naturally, the source of potential live uterus donors in the immediate family of a patient that may undergo UTx may be of considerable size with inclusion of first- and second-degree female relatives. Other advantages of live donor UTx would be that the graft could undergo extensive preoperative evaluations over a long time to confirm that no underlying uterine disease/malfunction exists. These include cervical atypia, endometrial hyperplasia, polyps, leiomyoma and infection with HPV. Preferentially, the donor should also be vaccinated against high-risk HPV infection, to avoid any infection prior to uterus donation. A requirement of a donor uterus from a live donor should be at least one uneventful pregnancy resulting in live birth, to demonstrate a full potential for normal pregnancy.
The most obvious advantage of organ retrieval from a deceased donor is that the surgical risk in the donor is eschewed. The extent of the vasculature at organ retrieval from a deceased donor can be larger than from a live donor. Thus, even the lower portions of the aorta and vena cava may be present in the vascular pedicle connected to the graft and anastomosis could then be performed directly on the aorta and vena cava of the recipient, in line with the technique used at multi organ transplantation. However, in uterus recovery from female DBDs, the uterus will most likely be retrieved after procurement of all other traditional transplantation organs, and this will decrease the extent of graft vasculature to possibly only include the common iliacs.
There is limited experience of human preclinical UTx research. In one study on human uterine tissue, we assessed the tolerability to cold ischemia [65]. Uterine tissue that was subjected to cold ischemia for 24 h in proper preservation solutions exhibited well-preserved ultramorphology and myometrial contractions. There also exist two studies in humans that have investigated techniques for uterus recovery. One study aimed to recover the uterus with complete internal iliac vessels bilaterally as part of DBD [66]. During a 6-month period almost 150 multiorgan procurements in New York City (USA) were identified as potential procedures for uterine donation with a research purpose. Uterine donation was accepted in only 6% of cases. The low consent rate could be explained by the fact that the organ would be be used solely for a research purpose, rather than that the donation would benefit another person. Moreover, the fact that the concept of UTx is unacquainted and would not have been discussed with the donor, or that the uterus is not regarded as transplantable since it carries the symbolic feature of woman/motherhood, may be factors behind the low participation rate. The intention was to secure the complete internal iliac vessels in the seven organ retrievals that were carried out, with the preferred pedicles including the common iliacs, being procured in only two. Dissection difficulties resulted in procurement of the uterus with only the anterior portion of the iliacs in five cases [66].
We performed one study in women, where we examined the surgical feasibility of live uterus donation. This was accomplished by simulating a part of a uterus recovery operation during a radical hysterectomy for cervical cancer [67], by also adding uterine vein dissection to the surgical procedure. The extra dissection of also the uterine veins added around 30 min to the 3–4-h procedure, without affecting the postsurgical morbidity. The fully mobilized ends of the procured uterine arteries were of 65–70-mm lengths and the corresponding lengths of the uterine veins were 50–55 mm. The inter-external iliac artery distance was approximately 90 mm. Taken together, the results of the study indicated that these lengths of the four blood vessel pedicles would enable direct bilateral end-to-side anastomosis of the mobilized uterine vessels to the external iliac vessels.
As mentioned above, so far 11 human UTx attempts have been performed. Our team in Sweden performed the last nine of these. The first human UTx trial was performed in year 2000 in Saudi Arabia [57]. A 26-year-old woman, who had undergone emergency peripartum hysterectomy at her first childbirth, received a uterus from a 46-year-old live donor. The duration of the donor surgery is not mentioned in the paper, but some extra time was added due to repair of a ureteric laceration. The short uterine vessels on the recovered uterus were extended by 7-cm-long segments of the saphenous veins and anastomoses were accomplished with these extended vascular pedicles end-to-side to the external iliac vessels of the recipient. Immunosuppressive treatment was with cyclosporine, azathioprine and prednisolone from the time of transplantation, and this combination was kept. The endometrium responded to sequential steroids with withdrawal bleedings, but it is not explained why spontaneous menstruation did not occur, despite the fact that the relatively young recipient presumably had normal ovarian activity. Prolapse of the graft into the vagina occurred 3-months post-transplantation and hysterectomy had to be performed since the uterus became necrotic. Poor uterine fixation may have caused the prolapse, which lead to compressed uterine blood vessels and, secondary to that, formation of extensive uterine vessel thrombosis. Our opinion is that this world's first human UTx case was performed prematurely, considering the scarcity of published research data on UTx at that time and the team's minimal preparatory UTx research.
In August 2011, the world's second human UTx attempt was performed in Turkey, when a 21-year-old MRKH-patient received a uterus from a 22-year-old female DBD [68]. The recovery from the multiorgan donor lasted around 2 h and the vascular pedicles included bilateral uterine arterial and venous supply including the internal and common iliac vessels. The transplantation procedure lasted approximately 5.5 h and included bilateral end-to-side anastomosis of the common iliac vessels graft to the external iliac vessels and fixations of the round ligaments to the inguinal ligaments and the uterovesical peritoneum to the bladder fundus. Immunosuppression was thymoglobulin for 10 days and then maintenance immunosuppression by prednisolone, mycophenolate mofetil and tacrolimus. The patient had her first menstruation, induced by exogenous hormones, 20 days after transplantation and the graft has so far survived with a regular menstrual pattern. She has had two urinary infections but no reported rejection episodes. Recently, the team reported two early miscarriages after the first and second embryo transfer [69].
Our team performed nine human UTx surgeries in 2013, with the uterus from live donors [70]. Each donor–recipient pair was thoroughly examined with immunological, microbiological, and imaging investigations. Importantly, the screening procedure lasted for more than a year and also included multiple visits to the doctors of the team, independent doctors and psychologists. Eight recipients were MRKH patients and one was a patient with cervical cancer more than 5 years prior to the UTx. In vitro fertilization, with embryo cryopreservation, had been performed prior to transplantation. The donors were mothers in most cases but also some close relatives and in one case a family friend.
The recovery surgery comprised isolation of the uterus with bilateral vascular pedicles including the internal iliac arteries distal to the branching of the first major posterior branch and the major uterine veins down to the internal iliac vein (Figure 2). Notably, the surgery times of the donors were much longer than expected, with durations of >10 h in all cases. All donor cases went uneventful perioperatively, but one uretero-vaginal fistula was diagnosed 10 days postoperatively and this was successfully repaired after 3 months. The surgeries of the recipients lasted about 6 h and were all without complications. Vacular anastomoses were performed bilaterally to the external iliac vessels (Figure 2) and after vaginal–vaginal anastomosis the uterus was fixed to the pelvic ligaments. After the surgeries, the recipients followed a standardized protocol of induction and maintenance immunosuppression with thymocyte antibodies given perioperatively. From the transplantation day, the recipients were on tacrolimus, corticosteroids and mycophenolate, with the corticosteroid treatment terminated after 1 week. The recipients have been on tacrolimus and mycophenolate in tapered doses during the first 6 months. The intention is to also remove mycophenolate after 6 months and the patients will stay on single tacrolimus from the seventh month.
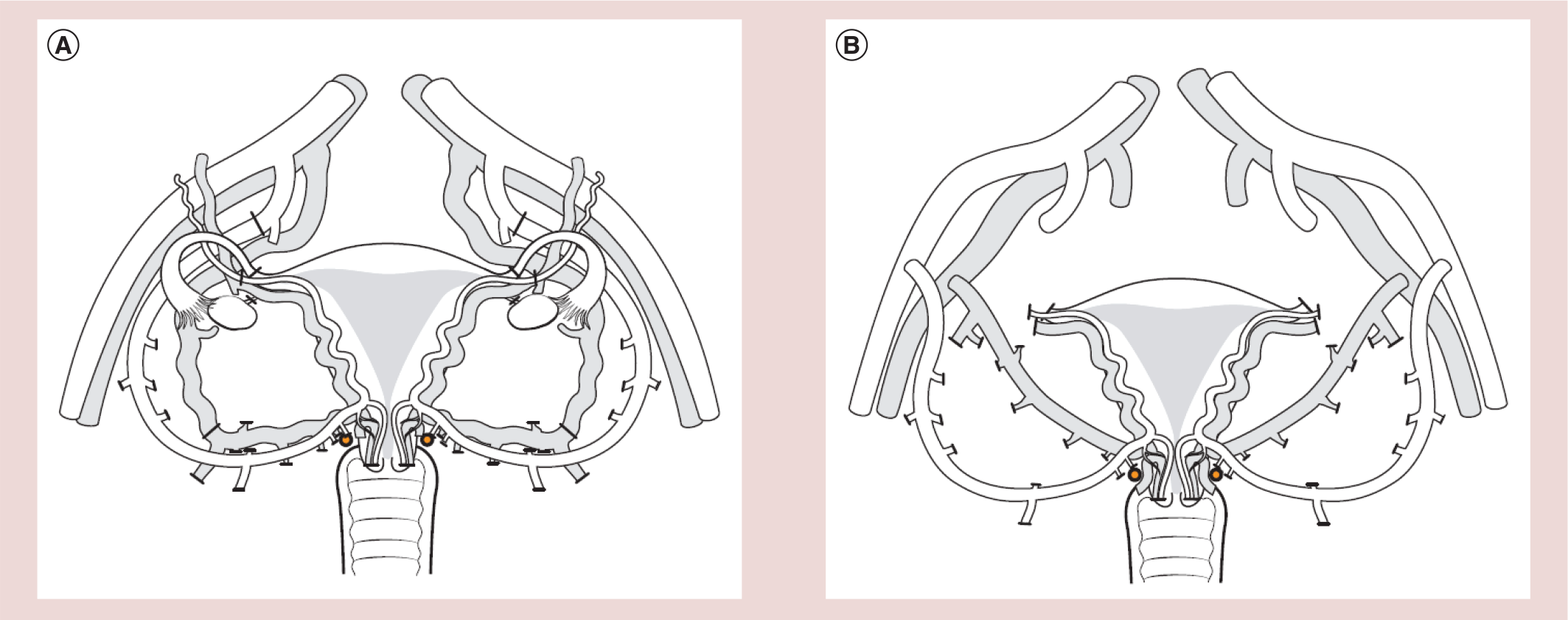
The 6-month outcome of these nine transplantations were that one uterus (case #9) had to be removed 3 days after UTx due to discontinued uterine blood flow, with histology showing thrombosis of the main arteries and veins of the graft. Three and a half months after transplantation another uterus (case #2) had to be removed due to a persistent intrauterine infection. In the seven remaining transplants, mild rejection episodes have occurred in four patients, but all episodes were effectively treated and reversed by corticosteroid treatment. Spontaneous menstruations were resumed within 2 months in the seven patients and all these uterine grafts remain viable and show cyclic menstrual patterns. The seven patients in this group have now had their uterine grafts well over 12 months and are in the process of having embryos transferred in order to accomplish pregnancy. If a successful pregnancy is established, the plan is delivery by cesarean section and hysterectomy after one or up to two live births.
It should be pointed out that none of the performed eleven human UTx cases have by definition been successful, since the term successful UTx should only be used when a healthy live baby is delivered. Although initial surgery and immunosuppression have been compatible with long-term graft survival in more than 70% of cases, the function in pregnancy during the second and third trimester has not been tested. It remains to be clarified whether a pregnancy in an allogeneic uterine graft will be negatively influenced by factors such as deficiency in vascular plasticity over the anastomosis sites and uterine denervation. The ongoing eight human cases will hopefully shed light on important issues such as these.
Ethics
The complex ethics surrounding UTx may represent the greatest challenge towards general acceptance of UTx, both among health professionals, politicians and the public. These ethics touch fundamental issues such reproduction, parenthood, health costs and medical advancements. Moreover, it covers all major aspects of human ethics including non-maleficence, autonomy, beneficence, justice and dignity. In the light of the fact that moral, legal and religious restrictions differ between cultures and countries, any conclusions on the ethical applicability of a new treatment modality such as UTx will most likely differ between societies. It should be pointed out that there also exist societal and religious differences concerning the acceptance of adoption and gestational surrogacy, which are the alternatives to UTx to gain motherhood for AUFI patients.
A risk–benefit analysis of UTx includes four parties; the donor or her immediate family in case of a uterus from a deceased donor, the recipient, the partner of the recipient, and the prospective child. The ethical principle of dominance of benefits over risks should be analyzed concerning each subject and as an overall analysis. The recipient would be the person with the greatest risk at UTx, since she is subjected to the risk of surgery, immunosuppression and pregnancy. It is important that the UTx team thoroughly investigates that, before a UTx procedure, she is fully aware of the experimental nature of the procedure. Moreover, the UTx team should ascertain that she has been extensively informed about the alternative interventions to achieve motherhood. A central issue is her ability to make an independent decision concerning participation. Moreover, she should understand the experimental plan proposed, the purpose behind it, and the potential risks involved, in detail.
In the event of a live donor as the source of the uterine graft, her risk of surgery and post surgical sequelae should also be taken into account. Naturally, live uterine donation should only occur after completed childbearing as pointed out in the FIGO guidelines on UTx [71], and a reasonable lower age is in our opinion around 40 years of age. The risks at live donation are the common surgical complications at hysterectomy, but in addition include those associated with removal of extensive vascular pedicles, as well as possible decreased quality-of-life and negative influences on psychological well-being [72].
It should be emphasized that experimental human UTx should only be performed under a strict scientific protocol and after approval by the local or national ethics board. The team performing experimental human UTx should have extensive research experience in the UTx field and the procedure should take place only at an institution with a long tradition in transplantation surgery.
Given that UTx is a quality-of-life-enhancing transplantation procedure, the safety precautions in clinical introduction must be rigorous. It should include extensive surgical training and research on UTx in a non-human primate model, as stated in the FIGO ethical guidelines concerning UTx [71].
Conclusion
In spite of the 11 human attempts, with uterine viability after 12 months in seven [Brännström M et al. Unpublished Data] and one cases [73], respectively, the definition of a truly successful human UTx procedure is the birth of a healthy baby. This has not yet been accomplished but will hopefully occur in 2014.
Future perspective
Resistance has existed against UTx both in the research community and in the media for many years, most likely because of the complex ethics surrounding this issue and because a human UTx procedure would be the most complicated mode of infertility treatment to circumvent the natural laws of biology. The debate and the arguments do in many ways mimic the situation when IVF was introduced [74,75], where the research group, led by Bob Edwards, in the late 1970s was criticized for introducing a highly complicated and costly procedure to enable the birth of a small number of children in an overpopulated world. The late Bob Edwards was awarded the Nobel Prize in Physiology or Medicine in 2010 and more than 5 million babies have now been born after IVF procedures.
Any research effort that forms the base before introduction of a new major surgical treatment in the human should be extensive and follow a step-by-step procedure in several animal species, in order to optimize the intervention and to increase the safety at introduction as an experimental procedure in the human. Our research on UTx is patient-initiated, and started in 1998 after a suggestion by a cervical cancer patient in her mid twenties, who was to undergo hysterectomy. The introduction of UTx by our group has followed the newly introduced IDEAL concept for a scientifically based clinical introduction, with research and then continuous clinical evaluation [76]. This concept emphasizes the importance of preclinical research and that the procedure is introduced under a strict research protocol. In our preclinical research towards human UTx, primate research has been instrumental [77].
Most likely, other teams in the UTx field are awaiting the pregnancy result of the single Turkish deceased donor UTx case [69] and our series of live donor UTx cases, before any further initiatives. These results will come during 2014 and 2015. Transplantation teams that are active in this field exist in USA, UK, France, Belgium, Spain, Japan, China and Australia [78]. If the outcome is successful, with delivery of one or more healthy and full-term babies, more scientific studies on human UTx should be launched. These should preferably be done as prospective case-series studies with strict inclusion and exclusion criteria. Advantageous, would be a multicenter design to also compensate for local variations in surgical techniques, immunosuppression protocols and patient characteristics. However, since the surgery, at least involving live donor UTx, is very complicated, a suggestion would be to restrict such initial studies to the deceased donor concept. The results of such studies would be helpful to further optimize the methodology and to obtain a better understanding of the risks, efficiency and cost of the procedure.
The cost of the UTx procedure would evidently be much higher compared with other infertility treatments. However, it would be more inexpensive than most other types of transplantation. This is because UTx, also with live donors, would involve only relatively young and fully healthy patients that would not need expensive postoperative care in intensive care units. Moreover, UTx would be the first ephemeral type of transplantation, which would restrict the costly and in a long-term perspective harmful immunosuppression to only some years.
In the event that several teams worldwide are successful in their establishments of UTx as a clinical procedure and that the clinical experimental studies prove that it is relatively safe and efficient, it is then up to the politicians and health insurers to decided whether the costs of UTx would be covered by a public health system or by general medical insurances.
Financial & competing interests disclosure
The authors have no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
No writing assistance was utilized in the production of this manuscript.
Executive summary
Absolute uterine factor infertility (AUFI) affects around one of every 500 women of fertile age.
AUFI is the only major group of untreatable female infertility.
Today, gestational surrogacy is the only means for AUFI women to become genetic mothers.
Leiomyoma with, in some cases, associated hysterectomy due to symptomatic leiomyoma, is the most common cause of AUFI.
Some severe uterine malformations, such as hypoplastic uterus and uterine agenesis, result in sterility.
Moderate uterine malformations result in infertility in around 20% of cases.
Severe intrauterine adhesions and hysterectomy, after cervical cancer or intractable obstetric bleeding, are other causes of AUFI.
Non-human primate uterus transplantation (UTx) research was conducted in the 1980s by a nonvascular approach to transplant the uterus.
Animal research towards clinical experimental UTx has involved rodents and large domestic animals (sheep and pig) before non-human primate research with vascular UTx was initiated.
Vascular UTx has been performed lately in three primate species.
Longterm uterine viability has been shown after allogenic UTx in the baboon and triple immunosuppression.
Live birth has been demonstrated in the cynomolgus macaque after autologous UTx.
To date, 11 human UTx attempts have been made.
The first human UTx attempt, performed in Saudi Arabia in 2000, involved a uterus from a live donor to a woman that had undergone emergency peripartum hysterectomy.
In this first human UTx attempt, an ordinary hysterectomy was performed in the donor and saphenous grafts were used to extend the vessels. The uterus prolapsed after 3 months and a necrotic uterus was removed.
The second human UTx was performed from a deceased donor in Turkey 2011.
This second human UTx case established two early pregnancies that unfortunately miscarried.
The first clinical trial of human live donor UTx was conducted in Sweden in 2013.
Seven patients from this trial will be subjected to embryo transfers in 2014.
The first live birth after human UTx is anticipated in 2014-2015.
The ethics around UTx is complex.
These UTx ethics touch important issues such as reproduction, parenthood and health costs.
A risk-benefit analysis of UTx should involve an overall analysis, based on separate analyses of the recipient, the partner of the recipient, the donor and the prospective child.
The outcome of our ongoing live donor UTx trial and the single case in Turkey will in large determine whether there is an immediate future for human UTx.
A successful live human birth after UTx is an important proof-of-principle, but research studies would then be needed to clarify the efficiency of the procedure and the specific immediate risks and benefits.
