Abstract
Gonadotropin releasing hormone agonist triggers are very effective in eliminating early-onset ovarian hyperstimulation syndrome (OHSS). However, in most studies they result in inferior pregnancy rates compared to hCG triggers in fresh autologous transfers. This is not due to an effect on embryo quality but rather due to inadequate corpus luteum formation and a defective luteal phase causing poor implantation. Intensive and adjusted steroid support or low-dose hCG may correct this. Late-onset OHSS is eliminated by using a freeze-all strategy. Pregnancy rates after transfer of thawed vitrified embryos are consistently high. A strategy combining a gonadotropin releasing hormone agonist trigger with vitrification of all embryos has been proposed as a means of achieving a truly OHSS-free clinic.
Keywords
Antagonist versus agonist protocols
Once the ovarian follicles have been developed with gonadotropins, the next step in in vitro ferilization (IVF) is to trigger their final maturation. The trigger is required to resume the arrested meisosis and to free the oocyte from the underlying follicle enabling it to be retrieved and subsequently fertilized either using standard IVF or intracytoplasmic sperm injection. This is analogous to the luteinizing hormone (LH) surge in a natural cycle, which triggers ovulation. In a natural cycle the LH surge occurs as the hormonal feedback switches from negative to positive in response to rising sustained levels of estrogen from the developing follicle. In controlled ovarian hyperstimulation (COH), in which a number of follicles are developing, in order to prevent a premature LH surge, traditionally a gonadotropin releasing hormone (GnRH) agonist is employed during the stimulation cycle, which, following an initial flare, causes downregulation of the hypothalamic pituitary ovarian axis via pituitary desensitisation of the GnRH receptor. Increasingly since the early 1990s GnRH antagonist protocols have been used, which also prevent the LH surge but without the initial flare.
The GnRH antagonist protocols have become far more widespread as they are associated with fewer side effects for the patients [1] owing to the absence of profound hypoestrogenemia, and the protocol requires a much shorter duration of stimulation. Importantly, there is no difference in the live birth rates between protocols employing GnRH agonists or antagonists. The recent comprehensive Cochrane review [2] comparing the antagonist to the long agonist protocols that fulfilled the inclusion criteria, demonstrated no statistically significant difference in the rates of live birth (odds ratio: 0.86, 95% CI: 0.69–61.08).
Agonist versus hCG triggers
In the majority of IVF cycles, whether using an agonist or an antagonist protocol, the trigger used is human chorionic gonadotropin (hCG), which shares a common subunit with LH and thus acts as a surrogate LH surge. The expanding use of GnRH antagonist protocols has enabled the use of a GnRH agonist as the trigger for final oocyte maturation. A GnRH agonist trigger clearly cannot be employed in a GnRH agonist protocol as the receptors have been desensitized.
The use of a GnRH agonist trigger results in the release of LH and follicle stimulating hormone (FSH). The serum levels of LH begin to rise after 4 h and the FSH level begins to rise after 12 h and remain elevated for between 24 and 36 h. The amplitude of the LH surge after the agonist trigger is similar to that seen in the natural menstrual cycle; however, unlike the natural cycle it consists of an initial short ascending limb, lasting approximately 4 h, and a long descending limb, lasting approximately 20 h [3]. The LH surge of the natural cycle has three phases with a longer duration of 48 h [4]. Overall this leads to a reduced total amount of gonadotropin released from the pituitary when a GnRH agonist is used as compared to the natural cycle and this results in a defective luteal phase secondary to corpus luteum deficiency. The hCG trigger, with a half-life of 34 h, results in a prolonged luteotrophic effect, which is advantageous for implantation but increases the risk of ovarian hyperstimulation syndrome (OHSS).
In the late 1990s it was hypothesized that the advantage of the GnRH agonist trigger over the hCG trigger would be in preventing OHSS [5] and also improved embryo quality due the simultaneous FSH surge more closely mirroring the natural cycle. Although the exact role of the FSH mid-cycle surge is not known it has been shown to promote LH receptor formation in the granulosa cells, maturation of the nucleus and expansion of the cumulus [6].
Trial data on pregnancy rates with hCG & aonist triggers
Of note, the results from clinical trials showed inferior pregnancy rates when a GnRH agonist trigger was used compared to the traditional hCG trigger. Two trials were prematurely discontinued because the pregnancy rates were so inferior in the agonist triggered arm [7,8]. The Cochrane analysis updated in 2011 identified 11 randomized control trials, with 1055 women, comparing GnRH agonist triggers versus hCG triggers in antagonist assisted reproductive technology cycles [3]. Eight studies assessed fresh autologous cycles and three studies assessed donor-recipient cycles. In freshautologous cycles the live birth rate was inferior in the GnRH agonist triggered group compared to the hCG triggered group with a statistically significant odds ratio of 0.44 (95% CI: 0.29–0.68). In donor recipient cycles there was no statistical difference in live birth rate (odds ratio: 0.92; 95% CI: 0.53–1.61).
An effect of egg quality? Donor trial evidence
Authors have hypothesized about the potential causes of the observed lower live birth rate in the GnRH agonist triggered group. The inferior live birth rates are not due to impaired embryo quality. A study on follicular fluid proved that triggering with a GnRH agonist induces a correct pre-ovulatory follicular maturation and release of mature oocytes [9]. The donor-recipient population provides the perfect model to eliminate the effect of the corpus luteum and the endometrium as variables in determining what is causing the inferior pregnancy rates. Donor oocyte programs, particularly from Spain, have shown no significant differences in the number of retrieved oocytes, metaphase II oocytes or fertilization rates between donors in whom ovulation was triggered with a GnRH agonist or hCG and more importantly no difference in pregnancy rates and implantation rates in the randomized recipients [10]. There was a significant difference in the luteal phase length in the donors (4.16 vs 13.6 days) and in the rates of OHSS, with no cases seen in the agonist triggered group. Further evidence is garnered from an elegantly designed prospective randomised crossover trial in the same egg-donor population undergoing donor oocyte cycles with a GnRH antagonist protocol [11]. This showed no difference in the number of oocytes achieved, or the implantation or pregnancy rates in those receiving hCG compared to a GnRH agonist trigger.
Agonist triggers & a defective luteal phase
The inferior pregnancy rate is most likely due to the insufficient stimulation of corpora lutea formation by the agonist trigger and subsequent luteal phase deficiency impairing implantation. It is likely that the luteal phase support used is insufficient to overcome this deficit.
Only one randomized controlled trial has reported comparable pregnancy rates between agonist and hCG triggered women in fresh autologous cycles [12]. In this study the women received far more intensive and tailored luteal support. Patients received a daily dose (0.3 mg) of estradiol via the transdermal route and 50 mg of progesterone intramuscularly, which commenced the day after oocyte retrieval. The doses were then adjusted if closely monitored serum levels were inadequate. This luteal support is more rigorous than the other regimens used by other groups and may account for their improved pregnancy rates. Another hypothesis is that the patients were recruited to this study because they were deemed at risk of OHSS owing to previous hyperstimulation or having polycystic ovarian syndrome (PCOS). Patients with PCOS are known to have high endogenous serum LH levels, which despite the agonist trigger may lead to an adequate LH drive to support the corpora lutea and thereby maintaining the luteal phase and pregnancy rates.
This study is in contrast to results of another study, which also used intensive luteal support in women deemed at high risk of developing OHSS [13]. They reported far inferior pregnancy results with an ongoing pregnancy rate of 6% and a high early pregnancy loss rate of 80%.
In fresh autologous transfers with a GnRH agonist trigger without intensive and adjusted steroid luteal support other research groups have looked at providing low dose hCG support of 1500 IU at the time of oocyte retrieval. In the 2011 Cochrane review that showed inferior pregnancy rates between the agonist and hCG triggered arms, two of the studies included in the meta-analysis examined the effect of a combination of agonist trigger and low-dose hCG luteal rescue [3]. If analyzed in isolation then no significant difference between the two groups remains. The largest and most recent randomized controlled trial examining this reported excellent embryology outcomes and equivalent pregnancy rates [14]. They reported a nonstatistically significant trend towards the retrieval of more mature occytes in the GnRH agonist triggered arm, which supports previous findings of a possible beneficial effect of the mid-cycle FSH surge on oocyte maturation [8]. Approximately 30% of the patients had at least 14 follicles that were at least 11 mm on the day of trigger. This has been shown previously to predict 87% of severe OHSS cases [15]. In this study no patients in the agonist triggered arm developed OHSS and 2% of patients in the hCG triggered arm developed moderate to severe OHSS. The study was not powered to detect a difference in this outcome. Further then to this work, an Australian retrospective study used this same protocol of GnRH agonist triggering with low dose hCG luteal support in women at high risk of developing OHSS [16]. There was no control group but they report a high clinical ongoing pregnancy rate of 52.1% and only one patient (1.4%) hospitalized with severe OHSS despite the average of 17 oocytes per cycle.
In contrast to these results, a recent retrospective cohort study of 23 women who were at risk of developing OHSS and were given 1500 IU as luteal phase support after agonist trigger, revealed a rate of OHSS of 26% [17]. Five women had severe OHSS requiring hospitalization and three did not undergo embryo transfer indicating that this life-threatening complication still occurs in the high-risk patient if hCG is used for luteal support.
Modified luteal support is essential after fresh transfer of embryos in agonist triggered cycles and other groups have employed different strategies for this. Castillo's group performed a retrospective, cohort-based observational study on 192 patients at risk of OHSS. The patients were divided into three groups and the day following agonist trigger received hCG, either 1000 IU, 500 IU or 250 IU, then the same dose every 3 days for a further two doses. They achieved a very good clinical pregnancy rate of 47.7, 42.6 and 39.4% per group, respectively. The rates of moderate or severe OHSS in each group were 13.6, 6.1 and 6% [18].
Another group has studied a novel method of luteal supplementation with recombinant luteinizing hormone in a randomized prospective proof of concept study [19]. In hCG triggered cycles the role of LH in implantation is provided by the hCG so their aim was to supplement the luteal phase with recombinant LH and progesterone in the agonist triggered group. A total of 17 patients received hCG trigger and standard progesterone luteal phase support and 18 patients received an agonist trigger with standard progesterone luteal support as well at six doses of recombinant LH every second day for six doses. The delivery rate was 23.5% in the standard arm compared with 22.2% in the recombinant LH arm. There were no cases of OHSS in either group but the population studied were normal responders and not patients at increased risk of developing OHSS.
Two concurrent randomized control trials were designed to explore the effect of an individualized luteal phase hCG support after GnRH agonist trigger according to the number of follicles on the day of trigger and compare them to a group triggered with hCG [20]. Patients with more than 25 follicles ≥11 mm were excluded to prevent randomization to the hCG arm. No OHSS was seen in the group at risk of OHSS (15–25 follicles) after agonist trigger with 1500 IU hCG luteal support compared with an incidence of 3.4% in the hCG triggered group. By contrast, two cases of late-onset OHSS were seen in the OHSS low-risk group (<14 follicles) triggered with GnRH agonist followed 1500 IU hCG after oocyte retrieval and a further 1500 IU 5 days later, compared with no cases after hCG trigger.
Agonist triggers & the prevention of OHSS
The short lived LH surge of the agonist trigger and the early involution of the corpora lutea are detrimental to the implanting embryo in a fresh autologous cycle but highly effective in preventing OHSS. The aim of COH is to achieve the best possible pregnancy outcomes but with minimal risk. Prevention of OHSS, especially in high-risk populations has become a major research goal in assisted reproductive technology (ART). OHSS is an iatrogenic complication of an elective procedure that is severe in approximately 2% of IVF cycles [15]. It may be associated with massive ovarian enlargement and torsion, ascites and pleural effusions, liver and renal dysfunction and thromboembolism. Results from reports regarding maternal mortaility rates due to OHSS in the UK show an incidence of three deaths per 100,000 cycles performed [21].
Current strategies to avoid OHSS involve careful selection of starting FSH dose based on qualitative factors such as age, PCOS status or previous hyperstimulation, and quantitative factors such as antral follicle count and serum anti-Mullerian hormone level. Unfortunately this strategy does not always prevent OHSS and may result in a suboptimal oocyte response. Strategies involving ‘coasting’ or withdrawing FSH stimulation until the estrogen level falls before administering hCG has not proven effective [22]. The conversion to a freeze-all with a hCG trigger is effective at preventing late-onset OHSS but will not prevent early-onset OHSS and is usually met with patient dissatisfaction. The administration of pharmaceutical agents, such as cabergoline, at the time of trigger has shown some benefit [23]. Unfortunately there remains no protocol with a hCG trigger that completely prevents OHSS apart from cycle cancellation prior to trigger, which is frustrating for the patient.
hCG is a potent driver of OHSS, both initially at the time of trigger causing early-onset OHSS and later as multiple corpora lutea develop driving further hCG production and late-onset OHSS. Many years ago, Lyons et al. reported that early- and late-onset OHSS are distinct entities with different risk factors [24]. In their retrospective analysis early-onset OHSS was an acute effect of the hCG administered prior to oocyte retrieval in women with high estradiol and large follicle numbers and late-onset OHSS was induced by the rising serum concentration of hCG produced by the early pregnancy, particularly multiple gestations. The syndrome occurs exclusively after ovulation and is intimately linked to the lifespan of the corpus luteum. The main factor involved is VEGF produced by the ovary in response to hCG, which causes widespread capillary leakage and third spacing of intravascular volume [25,26]. VEGF production is reduced in a GnRH agonist triggered cycle compared to a hCG triggered cycle [27]. Indeed many studies have shown that early-onset OHSS is eliminated by the use of a GnRH agonist trigger even in women at high risk of developing the syndrome [28].
A small number of studies have described OHSS cases despite the fact that final oocyte maturation was triggered with a GnRH agonist. These occurred only in a fresh embryo transfer cycle meaning that late-onset cases will only be prevented by combining the agonist trigger with a freeze-all approach.
A GnRH agonist trigger should now be the gold standard in oocyte donor programs as studies have shown they are safer and give equivalent pregnancy rates in the recipients. All patients, but particularly donors, should be exposed to the minimum of risk. Strategies to eliminate OHSS should be used in all patients; however, the agonist trigger should be preferentially used in patients where OHSS is an absolute contraindication such as women with serious maternal morbidities or in fertility preservation cycles where the occurrence of OHSS could delay vital cancer treatments.
Devroey et al. make a strong case for the establishment of an OHSS-free clinic in all patients by segmentation of IVF treatment [29]. This involves COH using an antagonist protocol with a GnRH agonist trigger followed by freezing all embryos to be transferred in a subsequent unstimulated cycle. In order for this strategy to live up to its full potential it requires a freezing methodology with a high post-thaw survival. Observational results from the Centre for Disease Control and Prevention for 2008 show no difference in live birth rates between the fresh and frozen-thawed cycles in clinics from the USA who undertake a sufficient number of cycles. These live birth rates have been improved by the advances in embryo vitrification techniques. Embryo vitrification results in significantly higher embryo survival and development rates [30] and better pregnancy rates compared to slow and ultrarapid freezing techniques [31].
Preventing late-onset OHSS with an agonist trigger & a freeze-all strategy
In a proof on concept study, Griesinger et al. reported that triggering with a GnRH agonist in patients at risk of OHSS and then freezing all two pronucleate embryos for transfer in a subsequent cycle is a safe and efficacious treatment [32]. Clearly this strategy circumvents the impaired luteal phase usually associated with the GnRH agonist trigger and the subsequent poor pregnancy rates. A total of 20 patients at risk of developing OHSS due to more than 20 follicles being present that were greater than 10 mm or an estradiol level more than 4000 pg/ml were recruited and triggered with a GnRH agonist. Nineteen underwent 24 frozen-thawed embryo transfers during the study period. The cumulative ongoing pregnancy rate was 36.8% with 31.6% falling pregnant in the first cycle. There were no cases of severe or moderate OHSS. The vitrification method used in this study resulted in a mean survival of 78% for the two pronucleate stage embryos.
In the follow-up to their proof-of-concept study the same research group undertook a prospective clinical cohort study across five centres [33]. Patients were again at risk of developing severe OHSS after stimulation in a GnRH antagonist protocol and had a GnRH agonist trigger with a subsequent freeze-all of all two pronucleate zygotes for transfer in a subsequent cycle. Of 51 patients, one patient did not have any oocytes retrieved, one patient did not undergo a frozen cycle transfer and one patient had no embryos left after thawing. Therefore in the 48 patients undertaking at least one frozen thawed embryo transfer the cumulative live birth rate was 37.3%. The live birth rate per first frozen-thawed embryo transfer was 5.9% in the slow-cooling group and 19.4% in the vitrification group. This adds further evidence that vitrification techniques are superior to slow-cooling techniques though this cannot be concluded absolutely from this trial as the patients were not randomized to cooling technique. However, the number of occytes retrieved between the patient groups was very similar.
It has been reported that patients having COH with an antagonist protocol who are triggered with more than 18 follicles or an estradiol concentration more than 5000 pg/ml [15] have a 83% sensitivity rate with 84% specificity for developing severe OHSS. In this study 83% of patients would fit these criteria and thus this study population did truly represent a high-risk group for developing OHSS. Interestingly, this study did report the first case in the literature of the development of early-onset OHSS with a GnRH agonist trigger [33]. However, other authors have disputed this claim stating that the clinical features were more related to complications from the oocyte retrieval.
The advantage of a subsequent frozen-thawed transfer may not only be in preventing OHSS but it may have beneficial effects on pregnancy rates due to implantation in a nonstimulated endometrium. It has been proposed that excessive steroid production as a result of COH and hCG trigger may have a negative impact of the receptivity of the endometrium to implantation [34]. Just as too low mid-luteal phase estrogen levels after an agonist trigger may be disadvantageous for implanation, too high mid-luteal levels of estrogen after hCG trigger may also lower implantation rates. A freeze-all strategy would eliminate both these scenarios.
A recent systematic review of three trials and 633 cycles in women aged between 27 and 33 years has shown that pregnancy rates after frozen embryo transfer are significantly higher than after fresh transfer (relative risk of pregnancy: 1.32; 95% CI: 1.10–11.59) [35]. The results of this meta-analysis suggest that in the normal- and high-responder patients, cryopreservation of all viable embryos for use in a subsequent frozen embryo transfer cycle may be advantageous. The analysis was by intention-to-treat and the evidence is of moderate quality. These results are likely due to the adverse effects of COH on endometrial receptivity and the improved technology with current vitrification methods.
Successful implantation requires a viable embryo synchronous with a receptive endometrium. COH is associated with altered endometrial development and impaired receptivity to the embryo. To discern the potential effect of this on implantation potential, Shapiro et al. undertook a matched cohort study in which 93 matched pairs of single blastocysts were transferred in fresh and frozen-thawed cycles [36]. The embryos were matched on embryos parameters and the patient's age. Overall, the ongoing pregnancy rates in the freeze-thaw group were significantly higher than the fresh transfer group. There was no difference between the day 5 blastocysts with a 60.9% pregnancy rate in the freeze-thaw group and 56.5% pregnancy rate in the fresh group but there was a significant difference in the day 6 blastocysts with a 56.5 versus 17.1% pregnancy rate in the frozen-thaw transfer groups and fresh transfer groups respectively. This suggests that ovarian stimulation inhibits embryonic implantation consistent with endometrial receptivity and this impairment is through asynchrony with little impairment of rapidly developing day 5 blastocysts but significant impairment of slower day 6 blastocysts. Interestingly the endometrial thickness was greater in the fresh transfer group suggesting the reduced receptivity did not result from a deficient endometrial thickness. The findings of this study support the hypothesis that embryo-endometrial asynchrony is the major cause of the nonreceptive endometrium when transferring day 6 blastocysts in autologous fresh cycles. Again a freeze-all strategy that employed an agonist trigger would eliminate this asynchrony.
Obstetric & neonatal implications
Recently, there has been some preliminary work looking at obstetric and neonatal outcomes in women who were triggered with high estradiol levels. One retrospective cohort study looking at singleton pregnancies that occurred after a fresh transfer in a hCG triggered IVF cycle found that rates of small for gestational age babies and rates of pre-eclampsia were increased in women triggered at higher peak estradiol levels [37]. In a cohort study looking at women with high peak estradiol levels who elected to undergo cryopreservation of all embryos for transfer in a subsequent frozen-thaw cycle versus those women with a similar peak estradiol level who chose to undertake a fresh transfer, there again were significant differences in terms of small for gestational age babies and pre-eclampsia in favor of a freeze-all strategy [38]. Both of these obstetric conditions are highly correlated with poor placentation. These trials are small and are not randomized; however, single-birth IVF pregnancies are more likely to result in premature delivery and low birthweight than unassisted single-birth pregnancies. It remains to be seen whether there is a potential for a GnRH agonist and freeze-all approach to go someway towards correcting this imbalance.
Conclusion
A recent survey looked at the use of GnRH agonist triggers worldwide and shows quite a widespread use in clinical practice [39]. Of 123 ART centres performing a total of 108,300 cycles, a GnRH agonist trigger was used in 5.2–36.1% of cases. The main reason given for their use was to prevent OHSS (76% of cases) or in cases where the embryo transfer was deliberately deferred (8%). In 43% of cases, responders used systematic cryopreservation with a subsequent thaw and transfer each time a GnRH trigger is used, 10% of responders used a regimen of luteal support similar to the Engmann study [12] and 26% used low dose hCG support similar to the Humaidan study [14]. Interestingly, in 46% of survey responders a delayed embryo transfer after triggering with a GnRH agonist was used in cases of prior ART failures. This survey shows the widespread penetration of the GnRH trigger in ART practices despite its only recent re-emergence and the relatively small number of trials published on its use. The GnRH agonist trigger is an exciting area for future research. Figure 1 represents a summary of the approaches.
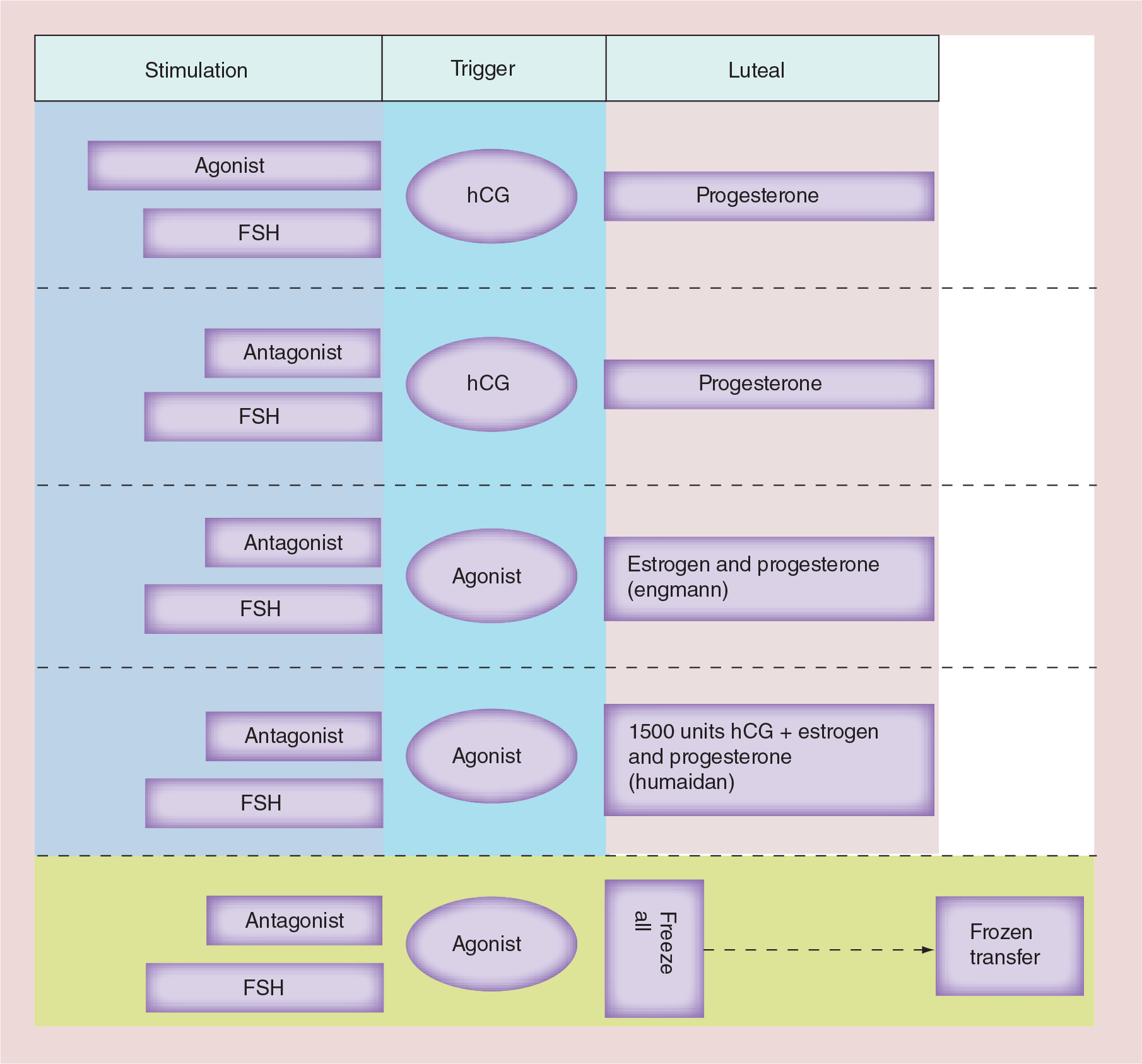
Summary of approaches with human chorionic gonadotropin and agonist triggers.
Future perspective
At the end of the day, the goal of ART is to maximize the healthy singleton term live birth rate with the minimum of risk to the mother. The first thing a pregnancy requires is a viable embryo. It is well established from donor oocyte studies that the GnRH agonist trigger has no detrimental effect on embryo quality. Unlike the hCG trigger it also triggers a FSH surge and this may actually have beneficial embryo effects that are yet to be fully understood but is an important area of future research.
A successful pregnancy also requires an embryo that is synchronous with a receptive endometrium. In fresh transfers it remains to be seen what is the optimal luteal support required when using an agonist trigger. Certainly it needs to be more intensive and tailored than traditional support after hCG trigger. Some researchers have looked at low dose hCG support but further research may prove that even lower doses of hCG could be used to further minimize OHSS rates without compromising pregnancy rates.
The advancement of microarray technology has allowed researchers to look at endometrial gene expression. A recent study compared the gene expression of four different protocols of luteal support: progesterone, steroids, recombinant LH and recombinant hCG at low doses or a single bolus; with the gene expression after hCG trigger and progesterone support [40]. This was done in women who were considered at high risk of OHSS. Differences in endometrial gene expression were seen where different triggers and luteal support were used. Gene expression after the GnRH agonist trigger and modified luteal support adding LH/hCG activity not surprisingly more closely resembled the pattern seen in the hCG triggered group. This application of microarray technology may allow clinicians to develop the best protocol to provide enough luteal support to enhance endometrial receptivity and implantation at the lowest dose to minimize OHSS risk.
Employing a freeze-all strategy may also have benefits on improving endometrial receptivity and synchrony. This strategy eliminates the high estrogen environment of an overstimulated cycle and the low estrogen environment of an understimulated cycle. Whether this has an improvement on implantation rates awaits further study. An exciting area for further research is whether this may prove to have beneficial obstetric or neonatal outcomes, leading to reduction in preterm birth, growth restriction and pre-eclampsia, outcomes that are known to be affected by poor placentation.
The real advantage of the agonist trigger and a freeze-all strategy is in the prevention of OHSS. Although severe in only 2% of cycles this still represents a significant morbidity and mortality especially as the number of cycles of ART performed each year continues to climb. Current strategies have improved the rates of severe OHSS; however, there exists no strategy apart from an agonist trigger and freeze-all approach that completely prevents OHSS. Devroey, a leading researcher in this field, argues that the concept of an OHSS-free clinic has become a reality [29]. The strategy proposed is segmentation of IVF with segment A consisting of optimization of the ovarian stimulation including GnRH agonist triggering in a GnRH antagonist cycle, segment B then involves optimum cryopreservation of the oocyte or embryo with vitrification and segment C involves thawing with replacement into a receptive, nonstimulated endometrium.
Given the significantly lower rates of OHSS in agonist triggered cycles it would be unethical to propose a randomized trial between agonist and hCG triggers in women at high risk of OHSS. However, by intention to treat there is scope to design a randomized controlled trial comparing a traditional hCG trigger with an agonist trigger and a subsequent freeze-all in normal responders. The advantage of the antagonist protocol in normal responders is that in the hCG arm, if the stimulation becomes too much and there is significant risk of OHSS then conversion to an agonist trigger and a freeze-all approach could be employed. It would be interesting to compare the cumulative pregnancy rates over three cycles between groups analyzed via intention to treat. Follow up data on miscarriage rates, ectopic rates, live birth rates as well as information on gestation at delivery, birthweight and pre-eclampsia rates ideally would be collected. In a normal responder population this would need to be a highly powered study to detect a difference in OHSS and also obstetric parameters; however, if comparable pregnancy rates could be shown between the groups this may lead to a dramatic shift in practice and open up further fields of research. The hurdles to this approach is the economic hurdle of a freeze-all approach, convincing clinicians of the potential value of agonist triggers and the reluctance of patients desiring a pregnancy to delay their embryo transfer. As Humaidan et al. argue in an elegant overview of this topic [41], GnRHa to trigger final oocyte maturation: a time to reconsider?
Financial & competing interests disclosure
The authors have no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
No writing assistance was utilized in the production of this manuscript.
Executive summary
Antagonist and agonist protocols in in vitro fertilization have equivalent live birth rates.
Antagonist protocols enable the use of agonist triggers instead of the traditional human chorionic gonadotropin (hCG) trigger.
Agonist triggers have no negative effect on embryo quality as evidenced by donor oocyte trials.
Agonist triggers cause early involution of the corpus luteum, thereby preventing early-onset ovarian hyperstimulation syndrome (OHSS).
By causing early involution of the corpus luteum, agonist triggers lead to a defective luteal phase, poor implantation and inferior pregnancy rates compared to hCG triggers in fresh transfers using traditional luteal support.
Similar pregnancy rates between hCG and agonist triggers have been reported with intense and adjusted luteal support.
Similar pregnancy rates have been reported with agonist triggers with low dose (1500 IU) hCG luteal support. This protocol has very low rates of OHSS, even in high-risk patients.
late-onset OHSS can only be prevented by freezing all embryos for transfer in a later cycle with an unstimulated endometrium.
Advancements in vitrification techniques with good post thaw survival have shown good pregnancy rates with elimination of OHSS.
Implantation into a more physiological and synchronous endometrium may have beneficial effects on pregnancy rates and may have improved obstetric and neonatal parameters.
