Abstract

Keywords
Introduction
Within the typical ovulatory menstrual cycle, the preovulatory follicle generates estradiol, which prompts a midcycle surge of luteinizing hormone (LH). This increase in LH initiates a series of intra-follicular events, including disruption of the connections (gap junctions) between the oocyte and the surrounding cumulus cells, expansion of the cumulus cells, rupture (breakdown) of the germinal vesicle, restarting of meiosis, and luteinization of the granulosa cells (GCs). The ensuing rise in progesterone synthesis enhances the positive feedback effect of estradiol, resulting in the concomitant midcycle peak of follicle-stimulating hormone (FSH). This transient FSH surge serves critical functions: it promotes LH receptor expression in the granulosa layer and stimulates hyaluronic acid synthesis, facilitating cumulus expansion and oocyte–cumulus complex release into the antral fluid. 1
Triggering final follicular maturation in assisted reproductive technology cycles
Human chorionic gonadotropin
In conventional controlled ovarian hyperstimulation (COH) protocols, final follicular maturation is typically induced by a single bolus of human chorionic gonadotropin (hCG) (5000–10,000 IU), administered approximately 36 h before oocyte retrieval. 2 Acting as a surrogate for the endogenous LH surge, hCG promotes granulosa cell luteinization, resumption of meiosis, and completion of oocyte maturation.
Ovarian hyperstimulation syndrome
Ovarian hyperstimulation syndrome (OHSS) typically manifests either 3–7 days after hCG administration (“early onset”) or during early pregnancy, 12–17 days after hCG administration (“late onset”). Prevention of OHSS requires balancing effective ovarian stimulation with minimization of iatrogenic risk. Strategies to reduce OHSS include individualized COH protocols, cycle cancellation, embryo cryopreservation, and single-embryo transfer. Replacing hCG with a gonadotropin-releasing hormone agonist (GnRHa) to trigger ovulation markedly reduces the risk of early OHSS; however, this substitution has been associated with reduced reproductive outcomes.3,4
Gonadotropin-releasing hormone agonist
During the last two decades, COH combining GnRH antagonist co-treatment and GnRHa trigger, has become standard practice to virtually eliminate severe early OHSS. 3 Nevertheless, this approach has been linked to lower clinical pregnancy rates and higher early pregnancy loss. 5 Several strategies have been proposed to optimize outcomes following GnRHa triggering, including: a “freeze-all” approach with deferred embryo transfer, fresh transfer with intensive luteal support, or fresh transfer with low-dose hCG supplementation. Each approach carries distinct advantages and limitations. 6
HCG versus GnRHa trigger
Comparative studies of hCG and GnRHa triggers in IVF cycles have demonstrated similar or superior outcomes with GnRHa in terms of oocyte yield, oocyte maturity, and top-quality embryo numbers. Molecular analyses reveal that LH receptor activation by LH or hCG induces distinct downstream signaling patterns: LH preferentially enhances AKT and ERK1/2 phosphorylation—pathways essential for granulosa cell proliferation, differentiation, and survival—whereas hCG predominantly increases intracellular cAMP, stimulating progesterone synthesis.7,8
Experimental data further indicate that combined LH + FSH + hCG exposure (the “dual” or “double trigger”) augments both steroidogenic (StAR, CYP19) and oocyte maturation–related (COX2, amphiregulin) gene expression compared with either GnRHa (LH + FSH) or hCG alone. 9 While LH + FSH–mediated activation (as seen after GnRHa trigger) more effectively promotes maturation than hCG alone, steroidogenic gene expression remains comparable between groups.
These mechanistic insights support the clinical adoption of the dual or double trigger, combining GnRHa and hCG for final oocyte maturation. The timing and dosage of each component should be tailored to the patient’s clinical profile 10 and treatment objectives.
Boxed note
This editorial builds upon concepts previously discussed in our 2024 review on final follicular maturation, but it is not a duplicate of that work. 10 The present article refocuses the discussion toward a pragmatic, risk-stratified approach that integrates treatment intent (fresh vs freeze-all), patient-specific OHSS risk, and real-time post-trigger feedback. The new contribution lies in the standardized definition of suboptimal GnRHa response (LH < 15 mIU/mL at 10–12 h post-trigger), the proposed thresholds for selective rescue with low-dose hCG, and the comparative algorithmic mapping of trigger strategies within fresh and freeze-all IVF contexts. Figures and tables have been newly developed to visualize this clinical decision pathway.
Tailoring final follicular maturation trigger according to patients’ characteristics
Patients undergoing COH for assisted reproductive technology (ART) can broadly be classified by two main criteria:
Treatment strategy—fresh embryo transfer (ET) versus freeze-all, and
OHSS risk—high versus low.
Across these groups, the response to ovulation triggering may be optimal or suboptimal, the latter characterized by a low oocyte yield relative to follicle count, and/or a high proportion of immature oocytes, or empty follicle syndrome.
Patients intended for fresh ET, not at risk to develop severe OHSS
In this group, final follicular maturation may be induced with hCG, GnRHa, or a combination of both (Figure 1). The use of a GnRH antagonist protocol followed by a GnRHa trigger has become standard practice to eliminate the risk of severe early OHSS. 3 Moreover, while research findings have demonstrated comparable or even improved maturity and quality of oocytes/embryos following GnRHa trigger, as compared to hCG trigger, 6 fresh transfers after GnRHa triggering alone are associated with reduced clinical pregnancy rates and increased first-trimester pregnancy loss. 5 Consequently, in patients proceeding to fresh ET, hCG remains essential, and a GnRHa-only trigger is not recommended.
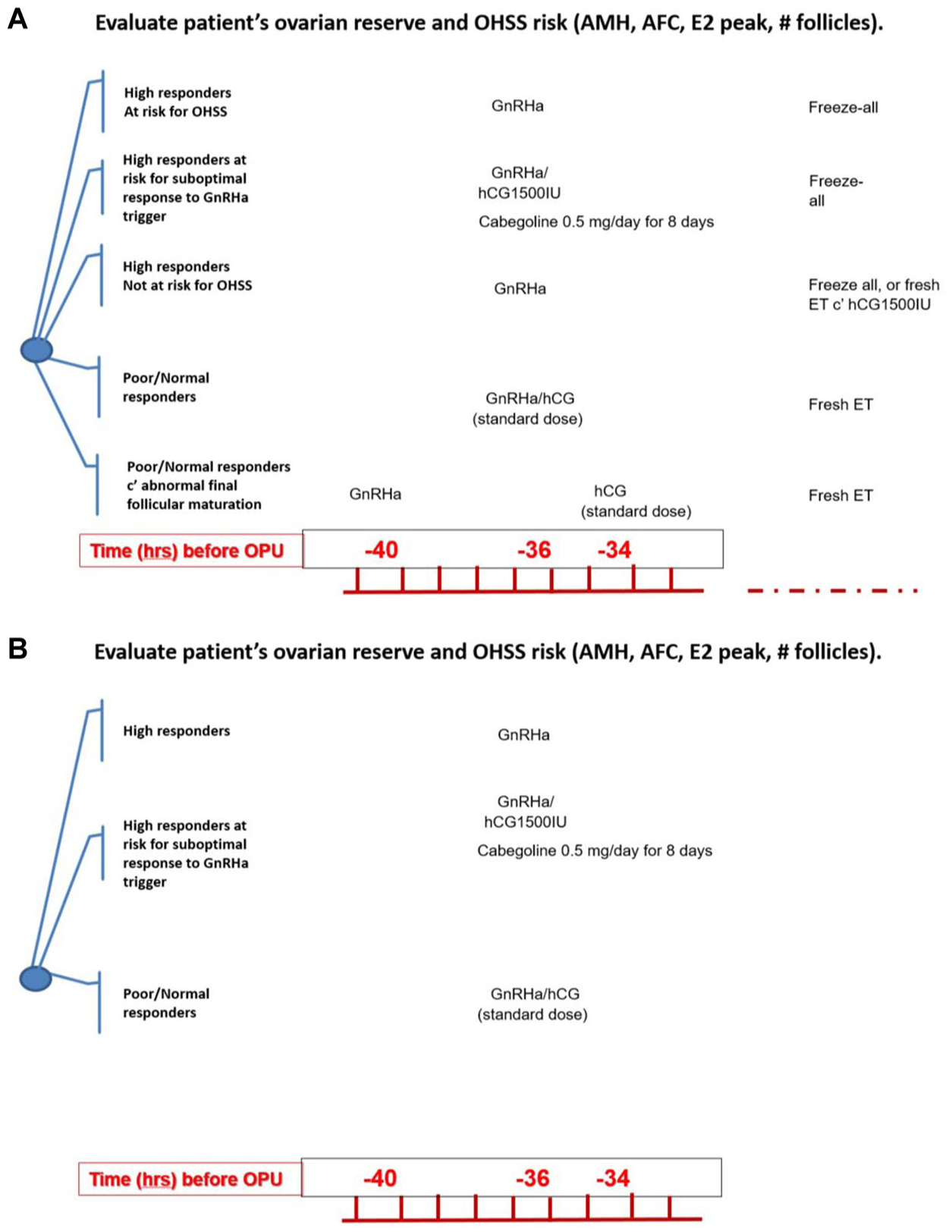
Personalized strategy for final follicular maturation in IVF. (a) Triggering final follicular maturation (fresh ET). (b) Triggering final follicular maturation (freeze-all). High OHSS risk (AMH > 3.5 ng/mL; AFC > 24; >18 follicles; E2 >3500 pg/mL): hCG standard dose :5000–10,000 IU.
The recognition that LH and hCG activate distinct intracellular signaling cascades has led to the dual-trigger approach—concomitant administration of hCG and GnRHa (typically 5000–10,000 IU hCG plus 0.2 mg triptorelin) 35–37 h before oocyte retrieval. The rationale is to combine the physiologic midcycle LH/FSH surge with the luteotrophic effect of hCG, thereby improving oocyte maturation, embryo competence, and optimizing the ultimate outcome of the IVF cycle. 10
Lin et al., 11 in their retrospective cohort study, compared IVF outcome in normal-responder patients undergoing COH using GnRHa with either a standard dose of hCG trigger (6500 IU recombinant hCG) or the dual trigger (0.2 mg triptorelin + 6500 IU recombinant hCG). The dual-trigger group demonstrated a statistically significantly higher number of oocytes retrieved, matured oocytes, and number of embryos cryopreserved, resulting in a significant increase in implantation, clinical pregnancy, and live-birth rates compared with the hCG-only trigger group.
These findings were corroborated by a subsequent single-center randomized controlled trial involving 155 normal responders, which demonstrated increased number of oocytes, mature oocytes, and number of blastocysts (total and top-quality) compared with hCG alone. Clinical pregnancies (46.1% vs 24.3%) and cumulative delivery rate (36.2% vs 22%) were significantly higher in the dual-trigger group. 12 Meta-analyses have since confirmed these benefits across both normal responders13,14 and poor responders. 15
Patients intended for freeze-all, not at risk to develop severe OHSS
As previously demonstrated, in this group of patients, triggering final follicular maturation with GnRHa alone is generally sufficient, producing comparable or improved oocyte and embryo quality relative to hCG triggers, without the need for hCG-mediated luteal rescue, 5 to overcome the reduced clinical pregnancies and increased first-trimester pregnancy loss following fresh transfer is unnecessary (Figure 1). 5
However, these patients might suboptimally respond to solely GnRHa trigger, with a reported incidence ranging from about 2% to 5.4%. 16 Several factors may predict a suboptimal response, albeit with limited evidence. These include: extremes of body mass index (BMI), prolonged oral contraceptive use or pituitary suppression, low basal LH (<0.1 IU/L) or LH on trigger day (<0.5 IU/L), and longer stimulation duration with higher gonadotropin doses.
For patients identified as high risk for suboptimal GnRHa response, dual triggering (GnRHa + hCG) can be used preemptively to avoid the need for rescue. Moreover, in some patients, an insufficient rise in LH after trigger, that is, a suboptimal response as a serum LH level < 15 mIU/mL, 10–12 h after GnRHa trigger is detected. When a low post-trigger LH level is detected, rescue administration of hCG with delayed oocyte retrieval (36 h post-hCG) may restore oocyte maturation. 16
As these patients are not prone to OHSS, dual triggering should be the preferred trigger, since it can enhance oocyte competence and reduce the likelihood of a suboptimal outcome without compromising safety.
Patients intended for fresh ET, at risk to develop severe OHSS
Patients at risk to develop severe OHSS include those with polycystic ovary syndrome; younger age; elevated ovarian reserve markers; including AFC (>24) and AMH levels (>3.4 ng/mL); those with rapidly rising serum E2 levels; peak E2 level in excess of 3500–5000 pg/mL; the emergence of a large number of intermediate sized follicles (>17–19 mm) and/or an increased number of oocytes retrieved (>15–18; Figure 1). 17
COH, which combines GnRH antagonist co-treatment and GnRHa trigger, has become the first-line approach aiming to eliminate severe early OHSS. 17 However, due to the reported significantly reduced clinical pregnancy and increased first-trimester pregnancy loss, 4 several strategies 3 were suggested to improve reproductive outcome, including: freeze-all policy; fresh transfer and intensive luteal support; and fresh transfer and low-dose HCG supplementation.
In cases where a suboptimal response to GnRHa alone is anticipated, a dual trigger with low-dose hCG (1000–1500 IU) may be employed, alongside cabergoline (0.5 mg/day) beginning on the day of trigger or retrieval for 8 days. 18
High responder patients retrieving >20 oocytes should follow a freeze-all policy, whereas those with fewer oocytes are instructed to start an intensive luteal support (estradiol and progesterone starting the day after retrieval). On day 3 post-retrieval, patients should be reassessed for signs of early moderate OHSS (ultrasonographic signs of ascites as reflected by the appearance of fluid surrounding the uterus/ovaries, and/or Hct levels >40% for the degree of haemoconcentration).
If absent, a single-embryo transfer may proceed with concomitant administration of 1500 IU hCG to support luteal function. 19 This delayed hCG bolus, administered 5 days after GnRHa trigger, rescues the corpus luteum while maintaining high midluteal progesterone concentrations 20 and has been shown to achieve favorable pregnancy rates without severe OHSS.
This stepwise approach allows approximately 80% of high-risk patients—those unlikely to develop severe early OHSS—to safely receive hCG for luteal support, while avoiding hCG administration in the remaining 20%–26% truly at risk21,22 to develop severe early OHSS. If early OHSS manifestations arise, a freeze-all policy remains the safest option.
Patients intended for freeze-all, at risk to develop severe OHSS
For high-risk patients in freeze-all cycles, the protocol mirrors that for OHSS-prone patients overall: GnRHa trigger for final follicular maturation followed by embryo cryopreservation (Figure 1). This approach effectively eliminates the risk of severe OHSS while preserving optimal oocyte quality and cumulative pregnancy potential. In cases where a suboptimal response to GnRHa alone is anticipated, a dual trigger with low-dose hCG (1000–1500 IU) may be employed, alongside cabergoline (0.5 mg/day) beginning on the day of trigger or retrieval for 8 days 18 and freeze-all policy.
Suboptimal/abnormal response to triggering final follicular maturation
Beck-Fruchter et al. 23 reported a case of recurrent empty follicle syndrome successfully managed by ovulation trigger with GnRHa and hCG added 40 and 34 h, respectively, before oocyte retrieval (Figure 1). They hypothesized that extending the interval between trigger and retrieval, combined with the physiologic FSH surge elicited by GnRHa, the “double trigger” (GnRHa and hCG added 40 and 34 h, respectively, before oocyte retrieval), could correct defects in granulosa cell function, oocyte meiotic maturation, and cumulus expansion, thereby restoring oocyte retrieval and fertilization potential. 23
Encouraged by these observations, our group evaluated the double-trigger strategy in two subpopulations of patients who exhibited abnormal final follicular maturation following hCG trigger despite an otherwise normal response to COH: patients with a low oocyte yield/recovery rate, defined as fewer than 50% of oocytes retrieved per number of dominant follicles (>14 mm) on the day of hCG administration 21 ; and patients with a low oocyte-maturity rate, defined as fewer than 66% of mature (metaphase-II) oocytes among retrieved oocytes. 6
In the low-retrieval group, administration of the double trigger resulted in significantly greater numbers of oocytes retrieved, two-pronuclear (2PN) embryos, and embryos transferred, as well as a higher ratio of oocytes retrieved to follicles >10 mm and >14 mm, with a trend toward more top-quality embryos compared with cycles triggered by hCG alone. 24
In the low-maturity group, the double trigger produced a significantly higher yield and proportion of metaphase-II oocytes, leading to a greater number of top-quality embryos than in previous hCG-only cycles. 25
Collectively, these findings suggest that the double-trigger approach, by combining the physiologic midcycle FSH and LH surges of GnRHa with the sustained luteotrophic support of hCG, may correct subtle dysfunctions in follicular and oocyte maturation that underlie suboptimal or abnormal response to triggering final follicular maturation.
Conclusion
The strategy used to trigger final follicular maturation in ART must balance the dual imperatives of maximizing oocyte competence and minimizing OHSS risk. An individualized approach, guided by patient characteristics, ovarian response, 10 and treatment objectives, should therefore be the cornerstone of modern ovulation-triggering protocols (Figure 1). By integrating systematic insight with clinical evidence, reproductive specialists can refine ART outcomes while eliminating the once challenging risk of severe OHSS.
Summary
In IVF, a “trigger” is the medication that completes egg maturation before retrieval. Doctors can use hCG (traditional), a GnRH agonist (safer for patients at risk of OHSS), or both together. This article explains how to choose the trigger based on whether embryos will be transferred fresh or all frozen, and on each patient’s risk of OHSS. In fresh transfers, hCG—alone or combined with a GnRH agonist—supports the uterine lining more reliably. In patients at high risk, a GnRH agonist is safer and is usually paired with freezing all embryos. If earlier cycles showed too few or immature eggs, using both triggers—either together (dual) or in sequence (double)—may help. Personalizing the trigger improves safety and success.
Take-home messages
Align trigger with strategy and risk: Choose hCG/dual for fresh ET (with adequate luteal support) and GnRHa ± freeze-all for high-risk patients to minimize OHSS.
Anticipate suboptimal GnRHa responses: Consider dual trigger upfront in at-risk cases; use post-trigger LH (10–12 h) to guide rescue hCG when needed.
Use double trigger selectively: For prior low retrieval or low MII, staged GnRHa → hCG can correct maturation defects.
Standardize thresholds and timing: Consistent cut-offs (AFC/AMH/E2; #oocytes) and protocols improve reproducibility.
Research priorities: pragmatic RCTs/meta-analyses comparing dual versus double trigger by responder phenotype; standardized luteal rescue strategies after GnRHa in fresh ET.
