Abstract
Gestational diabetes mellitus (GDM) is the most common medical complication of pregnancy and is particularly prevalent among obese women. Both GDM and obesity confer significant comorbidities for the mother and her offspring, including perinatal complications, excessive fetal growth and long-term risks for maternal and offspring obesity and diabetes. Exercise has well-documented health benefits and reduces peripheral insulin resistance in nonpregnant individuals, a major risk factor for the development of diabetes. Observational studies conducted in large population-based cohorts suggest that women who are the most active before pregnancy are less insulin-resistant in late pregnancy and have lower rates of GDM. This article will review the evidence supporting a role for exercise in the prevention of GDM, the management of glycemic control in women with established GDM, and the reduction of GDM-associated maternal and offspring health consequences. Wherever possible, the discussion will focus on studies carried out on obese women. However, there are many areas where strong evidence is lacking in obese populations, and it may be inferred from similar studies performed in normal weight pregnant women.
Keywords
Gestational diabetes mellitus & maternal obesity
Gestational diabetes mellitus (GDM) is the most common medical complication of pregnancy and is defined as glucose intolerance or high blood glucose concentrations (hyperglycemia), with onset or first recognition during pregnancy. The prevalence of GDM varies from 1 to 20% and is rising worldwide in line with increasing trends of maternal obesity and Type 2 diabetes mellitus (T2DM) [1,2]. The incidence of GDM rises disproportionately with increasing obesity. A recent meta-analysis of 20 population-based studies estimated that the risk of developing GDM is 2.14-fold higher for overweight (BMI: 25.0–29.9), 3.56-fold higher for obese (BMI: ≥30.0) and 8.56-fold higher for severely obese women (BMI: ≥40.0) compared with normal weight pregnant women (BMI: <25.0) [3]. Obesity and GDM have been recognized as independent risk factors for a number of adverse maternal and fetal outcomes, including diabetes, hypertension, operative deliveries, macrosomia and neonatal complications [4–6]. More recently, we have begun to understand that maternal obesity and GDM are also associated with significant lifelong consequences for the next generation [7,8]. As obesity and GDM share many of the same health consequences, obese women and their offspring are at a greater risk for adverse outcomes

Short- and long-term health consequences of gestational diabetes mellitus for mothers and their offspring.
Pathophysiology of GDM
During normal pregnancy, metabolic changes promote adequate nutritional availability to support the demands of fetal growth. A progressive and marked insulin resistance develops in maternal skeletal muscle, beginning around mid-pregnancy and progressing during the third trimester to levels that approximate the insulin resistance seen in T2DM [9]. Hormones and adipokines secreted from the placenta, including TNF-α, placental lactogen, placental growth hormone, cortisol and progesterone, are probable triggers of insulin resistance in pregnancy, owing to rapid reversal at delivery [10].
Normal pregnancy is characterized by increased insulin secretion from pancreatic β cells to compensate for peripheral insulin resistance. This can be displayed as a hyperbolic relationship between insulin sensitivity (the inverse of insulin resistance) and insulin secretion from pancreatic β cells (Figure 2). The development of GDM occurs when a mother does not secrete enough insulin in order to be able to meet the metabolic stress of peripheral insulin resistance. The reciprocal relationship is preserved in most GDM women, but occurs at much lower level of insulin secretion. That is, women with GDM secrete 40–70% less insulin for any degree of insulin resistance [11,12]. Therefore, pregnancy-induced insulin resistance may unmask the β-cell dysfunction characteristic of GDM [13].
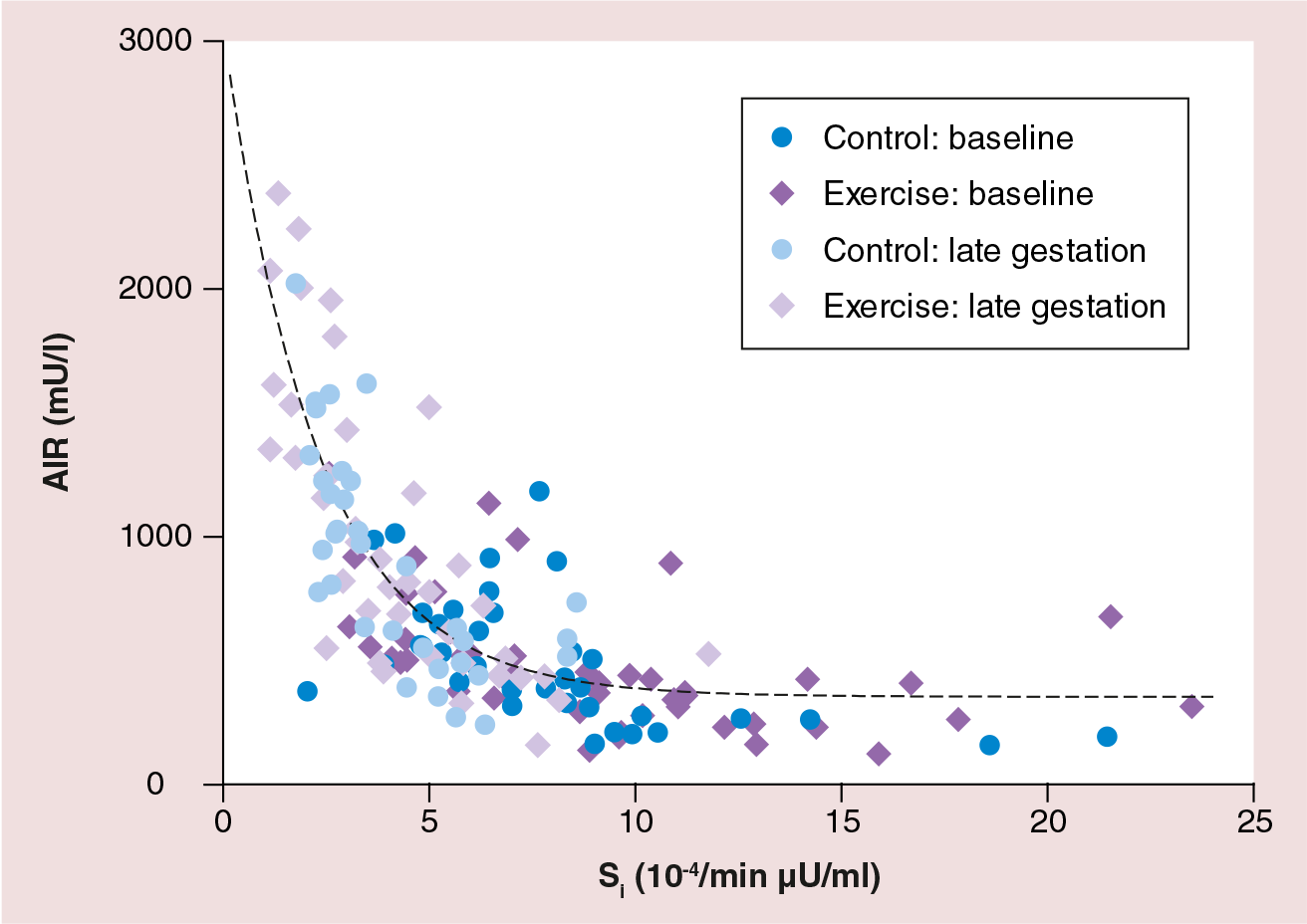
Serial assessments of insulin sensitivity starting before pregnancy have also demonstrated slightly greater insulin resistance persisting into the third trimester in women with GDM compared with normal pregnant women [11]. This resistance applies to both the action of insulin to stimulate glucose disposal [12], as well as to suppress hepatic glucose production [12,14] and circulating fatty acid concentrations [14]. In vitro studies of adipose and skeletal muscle tissue from women with GDM have revealed abnormalities in the insulin signaling pathway [15,16], abnormal subcellular localization of GLUT-4 transporters [17] and decreased expression of peroxisome proliferator-activated receptor-γ [15], all of which could contribute to reduced insulin-mediated glucose transport.
Biological plausibility of exercise in GDM prevention & treatment
Similarities in the underlying pathophysiology of GDM and T2DM would suggest that effective strategies for the prevention of T2DM in nonpregnant individuals might also be successful in the prevention of GDM. The role of regular exercise in preventing or delaying the onset of T2DM through improvements in glucose metabolism has been well documented [18–21]. Intervention studies have demonstrated improvements in whole-body insulin-stimulated glucose uptake following exercise training in both insulin-resistant individuals [22] and Type 2 diabetics [23]. Regular physical activity can also lead to improvements in insulin sensitivity through an increase in fat-free mass, the predominant site of insulin-dependent glucose disposal [24]. It is likely that the mechanisms responsible for alterations in insulin sensitivity and glucose uptake in response to exercise in nonpregnant populations may also be observed in pregnant women.
Pregnancy as an optimal window for intervention
Pregnancy offers a unique opportunity for easy and frequent access to medical care and is considered an ideal time for medical and lifestyle interventions. Patients may be motivated to make changes that could optimize their pregnancy outcome. Physical activity provides an accessible and affordable outlet by which the predisposing factors for the development of obesity and future diabetes in both the mother and offspring may be modified during the course of pregnancy. Furthermore, a behavioral change adopted in pregnancy may persist after delivery and help prevent, or delay, the development of T2DM and other long-term complications.
Can exercise prevent GDM?
A number of observational studies have demonstrated that women who report regular physical activity both before and during early pregnancy have a lower risk of developing GDM [25–28]. These studies have used a variety of validated physical activity questionnaires to examine self-reported exercise behaviors among a range of population-based study cohorts. After reviewing data from eight large studies, a recent meta-analysis reported a 55% reduction in the risk of GDM for women in the highest quantiles of prepregnancy physical activity compared with the lowest (odds ratio [OR]: 0.45; 95% CI: 0.28–0.75; p = 0.002) [29]. Regular physical activity in early pregnancy was also associated with a reduction in GDM risk, however, the impact appears to be somewhat reduced compared with pregravid exercise (OR: 0.76; 95% CI: 0.70–0.83; p < 0.001). These findings may reflect a dose–response relationship between intensity or volume of physical activity and GDM risk, as has previously been described with similar health outcomes in the nonpregnant population [30]. While the optimal volume of physical activity for GDM prevention is unclear, current observational data support the public health recommendation of accumulating 150 min of moderate-intensity activity each week to confer significant reduction in the risk of developing diabetes [19].
The beneficial association between physical activity and lower GDM risk may, at least, partially reflect the underlying relationship between an active lifestyle and leaner body composition [25,26]. Alterations in GDM risk in observational studies have been adjusted for prepregnancy BMI and, therefore, reflect an independent effect of regular physical activity on GDM risk. However, few studies have examined whether the effect of physical activity on GDM risk varies by prepregnancy body size. There are some suggestions that the protective effect of exercise may be stronger in women with a higher BMI, who have a higher risk for developing GDM. In 1997, a study published by Dye et al. utilized a population-based birth registry in central New York State between October 1995 and July 1996, with 12,800 women included in the analysis [31]. When stratified by prepregnancy BMI, low levels of physical activity was associated with increased rates of GDM among women with a BMI greater than 33 kg/m2 (OR: 1.9; 95% CI: 1.2–3.1). By contrast, in the lower BMI categories, women who reported regular exercise had a slightly higher incidence of GDM than nonexercisers. By contrast, a study by Oken et al. found that the protective effect of prepregnancy physical activity for the development of abnormal glucose tolerance was only significant in women with a BMI less than 25 kg/m2 [26], while other studies have found no interaction between prepregnancy BMI and the association between physical activity and GDM risk [25,27]. These conflicting findings may be related to the type and timing of physical activity questionnaires. In the study by Dye et al., physical activity was assessed after delivery. It is possible that women in the lower BMI categories (>33 kg/m2) who developed GDM reported physical activity that was initiated after their GDM diagnosis, and were therefore misclassified. These findings may also reflect an interaction between the timing of exercise and underlying differences in the pathophysiology of GDM between lean and obese women. Regular exercise during the prenatal period may have more impact on the prevention of GDM in obese women, in whom the development of GDM may rely more upon the contribution of obesity-associated insulin resistance, in addition to an underlying β-cell defect. By contrast, the development of GDM in lean women may reflect a greater reliance on a β-cell defect that may limit the ability of exercise to prevent the onset of GDM. Additional studies to examine these interactions are warranted before targeted recommendations can be made on the basis of prepregnancy size.
Investigators have recently turned their attention towards examining whether a prescribed exercise program initiated in pregnancy can reduce the risk of developing GDM. Seven randomized controlled trials, published between 2009 and 2012, are summarized in Table 1 [32–38]. In contrast to observational data, a recent summary of five of these studies concluded that they could find no clear effect on preventing GDM [39]. There are a number of potential reasons to explain the lack of significant findings from the randomized controlled trials published to date. First, there have only been a limited number of trials conducted in a small number of women. With the exception of two recently published trials [34,38], all of the other trials have included less than 100 patients. Therefore, it is likely that a lack of statistical power and the failure to control for significant confounding factors, such as diet and gestational weight gain, may have contributed to nonsignificant findings in both individual trials and the published meta-analysis. Power calculations to determine adequate sample size were reported in only one of the five included studies [34]. In this study, the authors estimated that 381 patients were required in each group. This calculation was based upon having statistical power to detect a predicted reduction in GDM incidence from 9% in the control group to 4% in the exercise intervention group. Studies in high-risk populations may not require as many participants, particularly if the incidence of GDM in the control group is higher and the effect of exercise training may be more protective.
Randomized controlled trials of exercise interventions in pregnancy reporting on gestational diabetes mellitus prevention or exercise-induced alterations in glycemic control.
10% of control group exercised >3 days/week.
Not significant after adjustment for maternal age and body weight.
EE: Energy expenditure; GA: Gestational age; GDM: Gestational diabetes mellitus; HOMA-IR: Homeostasis model assessment - estimated insulin resistance; HRmax: Maximal heart rate; NS: Not significant; VO2max: Maximal aerobic capacity.
Noncompliance to an exercise program and low-intensity exercise volumes have also been limitations of some of the trials. Stafne et al. randomized 855 Norwegian women (mean BMI: 25 kg/m2) to a standard exercise program or routine care control for a 12 week duration, beginning at 18–22 weeks gestation [34]. The intervention program included a weekly supervised 60-min exercise session, containing a combination of aerobic, strength and balance exercises. Subjects were also encouraged to complete at least two 45-min unsupervised sessions each week. Exercise adherence, defined as completion of at least three exercise sessions per week, was achieved by only 55% of women in the intervention group and 10% of the control group. On average, women in the exercise group exercised on 2.0 days per week compared with the 0.7 days per week in the control group. No differences in GDM risk were observed in this study; GDM was diagnosed in 7% of women in the intervention group and 6% in the control. Similar findings were reported in the study by Oostdam et al., who randomized 85 Dutch women (mean BMI: 33.5 kg/m2) who were classified as high risk for GDM to a supervised exercise program or control from 15 to 32 weeks gestation [37]. Their exercise program included two 60-min supervised exercise sessions each week. However, only eight of 53 (16%) intervention individuals who started the exercise program completed at least 50% of the required sessions. Furthermore, when physical activity was assessed using accelerometry at three follow-ups during pregnancy, the exercise group spent fewer minutes performing physical activity per week than controls. GDM was reported in 14.6% of intervention individuals and 21.6% of controls (p = 0.65). In both of these studies, the lack of an exercise effect on GDM risk may be, at least, partially due to the low level of exercise reported in the intervention group. Commonly reported barriers to exercise have included a lack of motivation, physical limitations, and a lack of resources and time. Future studies should continue to address perceived barriers to maintaining regular physical activity in obese pregnant women, and to evaluate optimal exercise prescription and program design to maximize compliance in order to achieve desired health benefits.
Exercise & glycemic control in pregnancy
Maintaining an active lifestyle before pregnancy has been associated with reduced insulin resistance and better glucose tolerance in late pregnancy and may, therefore, protect against the development of GDM. Retnakaran et al. assessed glucose tolerance, insulin sensitivity and β-cell function in 851 (predominantly normal weight) women who underwent a glucose challenge test and 3-h oral glucose tolerance test in late pregnancy [40]. Glucose tolerance status, ranging from normal glycemic control to GDM, improved across increasing quartiles of self-reported physical activity in the 12 months prior to pregnancy (p = 0.02). Furthermore, in multiple linear regression analysis, vigorous exercise prior to pregnancy was a significant independent predictor of insulin sensitivity in late pregnancy. However, it is unclear whether these late pregnancy enhancements in glycemic control were due to the effects of increased pregravid exercise per se, or whether women who were the most physically active in the pregravid period maintained their exercise regimens into their pregnancy.
Few studies have examined the impact of regular exercise on changes in insulin sensitivity during pregnancy, particularly in overweight or obese women. We have recently reported that 15 weeks of moderate-intensity cycling training in nondiabetic women had no impact upon the pregnancy-induced changes in insulin sensitivity in late pregnancy [32]. We randomized a cohort of healthy nulliparous pregnant women, with a range of mid-pregnancy BMIs, primarily within the normal weight range (BMI: 18–32 kg/m2), to a home-based stationary cycling program or nonexercise control for the remainder of their pregnancy. After a ramping period to establish a regular exercise routine, women in the exercise group were asked to maintain five 40-min sessions of moderate-intensity exercise per week at approximately 65% of their predicted aerobic capacity or maximal aerobic capacity. Our observations may indicate that the chronic changes in insulin sensitivity seen in nonpregnant individuals in response to exercise training may be overshadowed in normal weight pregnant women by the persistent regulation of insulin sensitivity required to achieve optimal fetal growth (Figure 2). Future studies should assess the frequency and intensity of exercise training required to impact insulin sensitivity in overweight and obese mothers in light of their increased insulin resistance and elevated risks for GDM. It is likely that chronic adaptations to regular exercise in pregnancy, particularly if accompanied by restricted gestational weight gain, may be different to normal weight women.
During normal pregnancy, it appears that the repeated short-term responses to each exercise session may play the greatest role in influencing glycemic control in late pregnancy. Previous studies have described a significant reduction in glucose and insulin concentrations for a period of time following a single exercise session in late pregnancy [41,42]. In addition, elevated insulin sensitivity has been demonstrated at least 30 min following moderate-intensity cycling exercise in late gestation [43]. With persistent regular exercise, these acute responses may help to regulate appropriate glycemic control in late gestation for nondiabetic pregnant women and, therefore, prevent the development of GDM. However, the majority of these studies have so far been carried out in normal weight women. Future studies should focus on the acute and repeated alterations in glucose and insulin concentrations following exercise in overweight and obese women.
Can exercise play a role in the management of GDM?
The standard of care for GDM management is to optimize glycemic control while providing adequate energy and nutrient requirements for the developing fetus. Current management of GDM includes caloric restriction medical nutrition therapy, which is the primary treatment therapy for 30–90% of women diagnosed with GDM [44]. Daily self-monitoring of fasting and postprandial capillary blood glucose is instituted to maintain euglycemic control. Excessive fetal growth and fetal distress are primary concerns in GDM, therefore, frequent fetal assessments with biophysical profiles and ultrasound assessments of growth are recommended. Failure to achieve normoglycemia leads to the use of antidiabetic agents. The addition of regular exercise to medical nutrition therapy may, as in T2DM, delay or prevent the administration of subcutaneous insulin [19]. Considering that GDM generally resolves spontaneously at delivery, this may be particularly important for pregnant women who are reluctant to start a relatively short duration of insulin injections.
A number of professional societies endorse the use of exercise as an adjunctive therapy for women with GDM. The American College of Obstetricians and Gynecologists have recommended that “women with GDM who lead an active lifestyle should be encouraged to continue a program of exercise approved for pregnancy” [45]. Similarly, the American Diabetes Association have suggested that “women without medical or obstetrical contraindications be encouraged to start or continue a program of moderate exercise as part of treatment for GDM” [46]. Despite these endorsements, only a handful of well-controlled studies have examined the role of regular exercise, used in combination with a controlled diet, in the management of GDM. Characteristics of these studies are displayed in Table 2. The majority of trials have enrolled small numbers of GDM patients in the third trimester and incorporated physical activity programs of predominantly nonweight-bearing exercise, performed at a low-to-moderate intensity for approximately 20–45 min, 3 days per week. The most frequently assessed outcomes include the requirement for insulin therapy, insulin dosage and parameters of glycemic control, including fasting glucose and insulin concentrations, postprandial glucose measurements and glycosylated hemoglobin.
Studies examining the use of exercise training in pregnancy for gestational diabetes mellitus treatment.
GWG: Gestational weight gain; HbA1c: Glycosylated hemoglobin; HRmax: Maximal heart rate; Int.: Intervention; n/a: Not available; VO2max: Maximal aerobic capacity; VO2peak: Peak aerobic capacity.
Artal et al. randomized a group of GDM patients to 45 min of stationary cycling at 50% of aerobic capacity or an insulin and diet regimen [47]. The exercise group showed a good compliance to the program, with over 90% of sessions completed. Participants in both groups achieved normal glycemic control after 1 week and were able to maintain euglycemia until delivery. After 8 weeks of treatment, there were no significant differences in glycemic control between the groups. The results of this study expanded on earlier work by Bung et al., who introduced, for the first time, exercise as an adjunctive therapy in pregnancy for patients with GDM who required antidiabetic agents to manage hyperglycemia. Women in the exercise group achieved similar glucose control to those patients on insulin therapy and resulted in a reduction in the number of women whose GDM needed to be managed by antidiabetic agents [48]. Pregnancy outcomes were similar between groups in both studies suggesting that an exercise regimen can be offered as a safe and efficient therapeutic option to patients with GDM.
When considering the use of antidiabetic agents and glycemic control, the results of subsequent studies have been conflicting and may reflect the varied contributions of confounding factors, such as variation between studies in the dose of prescribed exercise (duration, frequency and intensity), low compliance to the exercise program and a lack of statistical power to identify significant differences owing to small sample sizes. In the most recent Cochrane database systems review, Ceysens et al. summarized four randomized controlled trials and found no significant impact of exercise training on the use of insulin therapy (n = 80 patients; risk ratio: 0.98; 95% CI: 0.51–1.87). The authors concluded that “Further trials, with larger sample size, involving women with GDM … are needed to evaluate this intervention” [49].
Recent studies have focused on the impact of exercise training on daily capillary blood glucose concentrations in GDM patients, with encouraging results [50–52]. This is an important outcome as several studies have found that controlling capillary glucose concentrations reduces the risk of fetal overgrowth; a major complication of GDM [53]. Davenport et al. enrolled ten overweight women with GDM, matched by age, prepregnancy BMI and insulin use, with 20 control GDM patients [51]. The intervention group completed at least 6 weeks of low-intensity walking performed on average 3.6 ± 0.8-times per week for 20–45 min. In the week prior to delivery, participants in the exercise group demonstrated reduced capillary blood glucose concentrations, despite lower insulin requirements compared with control subjects. Two other studies, both employing resistance exercises, have also demonstrated improvements in glycemic control [50,52]. de Barros et al. recently published the results of a trial involving 64 patients diagnosed with GDM between 24 and 34 weeks gestation. Subjects were randomized to circuit-type resistance exercise or control for the remainder of their pregnancy [52]. Glycemic control was defined as the percentage of weeks where at least 80% of weekly capillary blood glucose measurements were below target limits established by the American Diabetes Association [13]. During the study period, women in the intervention group spent 63% of the time within the proposed glucose target range compared with 41% in the control group (p = 0.006). The resistance exercise program was also effective in significantly reducing the number of women who required insulin treatment. These findings strongly suggest that resistance exercise may be an effective alternative to aerobic exercise in the management of GDM. Resistance training has previously been shown to be effective in improving insulin sensitivity and glycemic control in individuals with abnormal glucose tolerance and T2DM [54,55]. Importantly, for some women, resistance exercises may be performed more comfortably than aerobic exercise in late pregnancy and could assist with posture and reduce general discomforts of pregnancy.
Can exercise reduce the health consequences of GDM? Short- & long-term maternal health consequences of GDM
Women with diabetes in pregnancy are at increased risk for developing hypertensive disorders and pre-eclampsia [56], and are more likely to require an induction of labor or delivery by cesarian section [56,57]. Regular exercise in pregnancy has been shown to be protective against the development of pre-eclampsia in nondiabetic pregnancies [58], and may provide similar protection for women with GDM. In addition, a recently published study examining the role of moderate-intensity exercise in reducing the comorbidities of GDM has reported a one-third reduction in the risk of undergoing acute or elective cesarean delivery for women who developed GDM (exercise OR: 1.30; 95% CI: 0.44–3.84 vs control OR: 1.99; 95% CI: 0.98–4.06) [38]. Taken together, these findings suggest that regular prenatal exercise may reduce the risk of pregnancy complications in women with GDM [59].
The development of GDM is a significant risk factor for future diabetes. Up to 50% of women will have a recurrence of GDM in a subsequent pregnancy [60], while 40–60% will exhibit further deterioration of carbohydrate metabolism and develop T2DM within 10 years [61]. Therefore, if regular prenatal exercise confers protection against the development of GDM, it may also help to prevent the future development of T2DM, particularly if women resume exercise postpartum and remain physically active. The Diabetes Prevention Program has been one of the most comprehensive multicenter clinical trials examining the impact of lifestyle intervention on progression to diabetes in individuals with impaired glucose tolerance [20]. In a subset of their cohort, intensive lifestyle intervention, including a program of weight restriction and regular physical activity in women with prior GDM, resulted in a 53% reduction in the development of T2DM [62], supporting a role for exercise in the prevention of future diabetes in women diagnosed with GDM.
Offspring health consequences of GDM
GDM is associated with elevated risk for delivering a large-for-gestational-age or macrosomic infant, defined as greater than the 90th percentile for expected birth weight for gestational age or a birth weight greater than 4000 g, respectively [5,6]. As a consequence of their size, offspring of GDM mothers are more likely to suffer from significant birth trauma, such as shoulder dystocia, perinatal asphyxia, bone fractures and nerve palsy [5,63]. Large fetal size at birth also poses additional maternal risks, with elevated risk of cesarean section, cephalopelvic disproportion, uterine rupture and perineal lacerations [64].
In obese women, with or without GDM, regular exercise in pregnancy may ameliorate peripheral insulin resistance, reducing oversupply of nutrients to the fetus and thereby providing protection against excessive fetal growth. Few studies have examined the impact of exercise in pregnancy on fetal growth, specifically in overweight and obese mothers. Some studies have reported success in restricting excessive weight gain, but this has not been accompanied by a change in mean offspring birth weight [47,65]. However, regular exercise in pregnancy may specifically protect against delivering a large baby at birth. In population-based studies, women who maintain regular physical activity into the third trimester of pregnancy demonstrate a lower incidence of delivering an large-for-gestational-age infant [66,67]. A reduction in GDM-associated macrosomia has also been reported in women who took part in a moderate-intensity exercise program in pregnancy (exercise OR: 1.76; 95% CI: 0.04–78.90 vs control OR: 4.22; 95% CI: 1.35–13.19) [38]. In overweight and obese women, participation in a structured diet and exercise intervention has been shown to elicit a small reduction in the risk of delivering a macrosomic infant [47,65]. However, the reduction in macrosomia in obese women was contingent on the restriction of weight gain [47], suggesting that exercise may not be successful at preventing macrosomia in obese women without prevention of excessive gestational weight gain.
There is now growing evidence that the consequences of exposure to a diabetic intrauterine environment also extend into postnatal life. Offspring of mothers with GDM are more likely to be overweight or obese in childhood and develop Type 1 or 2 diabetes [8,68,69]. Regular exercise may help to ameliorate the future obesity risk for the offspring of GDM women by ‘normalizing’ fetal growth and reducing the incidence of macrosomia at birth. There is some evidence in nondiabetic women that prenatal exercise may be associated with better body composition in childhood [70]. However, no studies have examined the impact of exercise in pregnancy on postnatal growth and development in the offspring of obese women, with or without GDM. Future studies examining the role of exercise in the management of GDM would greatly benefit from postnatal follow-up of diabetic offspring to determine whether exercise during pregnancy in women at risk for, or diagnosed with, GDM ameliorates potential long-term health consequences for their offspring.
Conclusion
Based on the current evidence, maintaining a physically active lifestyle prior to and during early pregnancy protects against the development of GDM. However, the effectiveness of beginning an exercise program in the second half of pregnancy, and the optimal prescription and quantification of appropriate individualized exercise programs should be the focus of continued research. Many professional societies advocate the use of exercise as an adjunctive therapy for women with GDM. Recent studies have indicated that an exercise program initiated after diagnosis of GDM may reduce the requirement for insulin therapy and improve glycemic control in late pregnancy. These benefits may be long-lasting, as adequate glucose control in pregnancy is associated with reduced risk of perinatal complications and lower incidence of macrosomia or a large-for-gestational-age infant. In turn, this provides a potential role for prenatal exercise in reducing the obesity risks for the next generation.
Future perspective
The global epidemic of obesity continues to accelerate at an alarming rate. Nearly three in every five US women of reproductive age (20–44 years) are now classified as overweight or obese [71]. By the end of the next decade, some experts suggest that the majority of women (perhaps 70%) may be classified as obese [72]. With increasing obesity, the rate of GDM diagnosis is also expected to rise. Of concern, the impact of GDM extends well beyond the current pregnancy and may impact the long-term health of not just the mother, but also future generations. The offspring of GDM women are themselves at risk for obesity and the development of subsequent diabetes. For female offspring, metabolic alterations, such as insulin resistance occurring as a result of developmental programming in utero, may have implications for their future pregnancies. They may enter their first pregnancy with a greater level of insulin resistance with greater risk for GDM and delivering large offspring. Hence, obesity and GDM may be central to a perpetuating cycle of metabolic and growth abnormalities that continue to drive the worldwide obesity epidemic.
The prevention of adverse perinatal outcomes, and protection against the long-term health risks of GDM for both the mother and offspring, through adequate glycemic control and prevention of excessive gestational weight gain, should be the highest priority in the coming years. However, in order to guide policy and evidence-based clinical practice, research efforts must continue to produce well-designed, adequately powered studies to determine the precise roles of exercise in reducing the risks of GDM. Current gaps in the literature and future research directions are summarized below:
Continued research efforts are required to determine the minimum and/or optimal volume and type of exercise required for the prevention of GDM. Future work should continue to examine the contrasting benefits of aerobic, resistance or a combination of modalities on GDM risk; Studies examining the impact of beginning an exercise program in overweight or obese women during pregnancy have consistently suffered from poor compliance to the prescribed exercise program. There is an urgent need to determine successful strategies to improve adherence to regular exercise and to overcome perceived barriers to exercise in the pregnant population. There are encouraging recent reports that an individually tailored, motivationally matched exercise intervention may be feasible and efficacious in pregnancy [73]; There remain large gaps in our understanding of the physiological impact of regular exercise in pregnancy on maternal insulin sensitivity. In particular, the time course of the acute exercise-induced effects on insulin sensitivity following a bout of exercise in pregnancy may differ from that observed in nonpregnant individuals. This information may be important in the design of studies to investigate the potential chronic adaptations to maternal insulin sensitivity with exercise training in pregnancy. There is also a paucity of research examining exercise-related alterations in insulin sensitivity in overweight and obese pregnant women using precise and reliable techniques; Similar to GDM prevention, future studies should seek to define optimal training characteristics for the use of exercise as an adjunctive therapy in late pregnancy to achieve adequate glycemic control in women diagnosed with GDM; Future exercise intervention studies in GDM women will greatly benefit from long-term postnatal follow-up of the offspring of exercise and control participants to determine whether prenatal exercise in GDM women may reduce the risk of obesity and metabolic dysfunction in the offspring during childhood and adolescence; Finally, it is clear from the current literature that the prepregnancy period plays a significant role in determining both GDM risk and maternal insulin sensitivity in late pregnancy. Therefore, continued emphasis should be placed upon maintaining physical activity across the lifespan for continued health and to benefit the health of future generations.
Executive summary
Gestational diabetes mellitus (GDM) is the most common medical complication of pregnancy, affecting 1–20% of pregnancies. Obese women have a 3.6-fold higher risk for developing GDM compared with normal weight women. Both maternal obesity and GDM confer significant short- and long-term health risks for both the mother and her offspring.
The hormonal changes of normal pregnancy are associated with progressive and marked insulin resistance in maternal tissues in the second half of pregnancy.
The physiological insulin resistance of pregnancy may unmask underlying β-cell dysfunction in women who develop GDM.
Similarities in the pathophysiology of GDM and Type 2 diabetes mellitus (T2DM) would suggest that effective strategies for the prevention of T2DM might also be successful in the prevention of GDM.
The role of regular exercise in preventing or delaying the onset of T2DM is primarily attributed to exercise-associated improvements in peripheral insulin sensitivity.
Exercise leads to acute insulin-independent effects on muscle glucose uptake, which last for a short period of time following exercise and increased tissue sensitivity to insulin that persists as long as regular exercise is continued.
The pregnancy period is an opportune time to counsel obese women to increase their physical activity as they have regular contact with health professionals and are motivated to make changes that could benefit both their own health and the future health of their baby.
Women who report the highest levels of physical activity in the 12 months before pregnancy have a 55% lower risk of developing GDM than the least active women.
Physical activity in early pregnancy is also protective against GDM. Women in the highest quantiles of physical activity reduce their risk for GDM by 24%.
To date, there have only been a few randomized controlled exercise intervention trials to prevent GDM, particularly in obese women.
The effectiveness of exercise interventions in pregnancy to prevent GDM has been limited by noncompliance. Commonly reported barriers to exercise have included a lack of motivation, physical limitations and lack of resources and time.
Regular exercise in the 12 months prior to pregnancy has been associated with reduced insulin resistance and improved glucose tolerance in late pregnancy.
In nondiabetic, low-risk pregnant women, exercise training has not been shown to impact the normal pregnancy alterations in insulin sensitivity.
The repeated transient increase in glucose uptake associated with each exercise session may provide a sufficient stimulus to achieve appropriate glycemic control and prevent the development of GDM.
Exercise is endorsed and recommended by the American College of Obstetricians and Gynecologists, the American Diabetes Association and the American College of Sports Medicine as an adjunctive therapy for women with GDM.
Randomized controlled trials in women with GDM have focused on whether regular adding an exercise program to dietary management can improve overall glycemic control as indicated by daily capillary blood glucose concentrations. Controlling capillary glucose concentrations reduces the risk of macrosomia; a major complication of GDM.
Within the larger Diabetes Prevention Program study, women with previous GDM who were randomized to intensive lifestyle intervention, including a program of weight restriction and regular physical activity, demonstrated a 53% reduction in the development of T2DM.
There is evidence that women who continue regular exercise into late pregnancy, particularly in conjunction with limited weight gain, have a reduced risk of delivering a large for gestational age infant.
Regular exercise may ameliorate the risk of future childhood diabetes in the offspring of mothers with GDM by reducing adverse developmental programming.
Financial & competing interests disclosure
The authors have no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
No writing assistance was utilized in the production of this manuscript.
