Abstract
Keywords
Obesity is one of the most common metabolic disorders in developing countries and is characterized by a reduction in insulin sensitivity, both in animal models and in humans [1]. The incidence of obesity worldwide has increased drastically during recent decades. Consequently, obesity and associated disorders now constitute a serious threat to the current and future health of the populations. The WHO estimates that 2.8 million adults die each year as a result of being overweight or obese. In addition, 44% of the diabetes burden, 23% of the ischemic heart disease burden and 7–41% of certain cancer burdens are attributable to patients being overweight or obese [101].
Postmenopausal obese women may be at increased risk for metabolic syndrome, probably because total and central adiposity increases after the menopause transition [2]. The emergence of these risk factors may be a direct result of ovarian failure or alternatively an indirect result of metabolic consequences of central fat redistribution with estrogen deficiency [3]. Postmenopausal women have a tendency for an increase in weight and particularly in visceral adipose tissue (VAT) due to a lack of estrogen [4]. However, the molecular mechanisms involved in obesity-related insulin resistance (IR) are not yet well understood.
Adipose tissue is an active metabolic tissue that secretes multiple metabolically important proteins known as adipokines (e.g., leptin, adiponectin, TNF-α, IL-6 and resistin among others) [5,6]. These adipocyte-derived products are presently subject to intensive research concerning their involvement in the regulation of adipose tissue physiology and, in particular, their potential implication in IR, obesity and diabetes [7,8].
Plasma concentration of resistin is elevated in obesity compared with healthy subjects. In humans, plasma resistin concentrations are positively correlated with BMI and metabolic risk factors in the diabetes group compared with that in the control group [9,10]. Sowers et al. reported resistin levels were twice as high in women during the premenopause period compared with the peri- and postmenopause stages in both obese and nonobese groups [11].
It is well documented that accumulation of visceral fat is associated with a higher risk for development of obesity-related diseases, such as Type 2 diabetes, cardiovascular disease, hypertension and hyperlipidemia [12,13]. The VAT compartment may be a unique pathogenic fat depot [14–16]. The VAT compartment secretes adipocytokines and other vasoactive substances that can influence the risk of developing metabolic traits [17–22]. The exact mechanisms relating visceral fat accumulation to diabetes and atherosclerosis are also not yet fully characterized.
VAT has greater lipolytic activity than subcutaneous adipose tissue (SAT), where fatty acids are directly delivered to the liver from this region via the portal vein, thus increasing the delivery of lipids to the liver and worsening IR in the liver, promoting triglyceride synthesis and exacerbating dyslipidemia [23]. A significant link between VAT and dyslipidemia in patients with Type 2 diabetes was also reported [24]. Visceral and subcutaneous adipocytes have different capacities to produce hormones and enzymes and variation in mRNA expression of adipokines (e.g., resistin, adiponectin, TNF-α and IL-6, among others) from various depots of adipose tissue has been observed in several studies [25].
In an animal study, expression of resistin in visceral and SAT is controversial [26]. The data on resistin in humans is also conflicting. Some authors have shown that resistin levels are elevated in individuals with obesity and diabetes [27–29], whereas other investigators have shown that it is not elevated [30,31]. A recent study reported no difference in serum resistin levels in subjects with and without metabolic syndrome [32]. These observations indicate a relationship between insulin resistance, dyslipidemia, abdominal adiposity and resistin levels [33]. Taken together, these studies suggest that the depot-specific expression of resistin at the mRNA level could be relevant to insulin sensitivity.
The literature, regarding VAT resistin expression in Asian populations, especially Indian, is lacking. Thus, to the best of our knowledge, the present study was undertaken with the objective to investigate VAT resistin mRNA expression in postmenopausal Indian women and its association with obesity-related risk factors, such as BMI, IR (homeostatic model assessment-IR [HOMA-IR]) and serum resistin.
Materials & methods
Subjects
A total of 68 postmenopausal women aged 49–70 years were recruited at CSM Medical University (Lucknow, India) who underwent either elective abdominal surgery for gall bladder stones or hysterectomy. Abdominal omental adipose tissues were obtained either for benign uterine myoma or for gall bladder stones. No specific standard diet and hormonal therapy were given to the patients during the surgery, which ensured and ruled out the effect of hormones or diet on fat deposition. All tissue samples were stored in RNAlatar® (Sigma Chemical Co., MO, USA) for RNA extraction. Subjects were classified as obese according to BMI >25 kg/m2, as per the WHO guideline for Asians [34], and nonobese subjects (BMI <25 kg/m2) served as controls. The study was approved by the Institutional Ethics Committee. Informed consent was obtained from each patient.
Biochemical estimation
Fasting blood samples were taken the next morning after admission to the hospital for surgery. Plasma insulin concentrations (μU/ml) were determined using immune radiometric assay (Immunotech, Marseille, France). Plasma glucose concentrations (mg/dl) were determined using a semi-automated analyzer (Microlab 300™, Merck, NJ, USA). Serum resistin levels (ng/ml) were measured by ELISA (Quantikine® Human Resistin Version 16 190607 15, Biovendor, Heidelberg, Germany).
RNA extraction
Total RNA was isolated using Tri-Reagent™ (Sigma Chemical Co.). RNA was measured spectrophotometrically at 260 and 280 nm, while RNA integrity was checked by visual inspection of the two ribosomal RNAs, 18S and 28S, on agarose gel.
Real-time PCR measurement of VAT resistin mRNA expression
One-step real-time (RT)-PCR was carried out using QuantiTect® SYBR Green RT-PCR master mix kit (Qiagen, Hilden, Germany). PCR amplification was carried out in the LightCycler® 480 (Roche, Basel, Switzerland; RT thermal cycler). In brief, resistin mRNA and β-actin mRNA were amplified in separate 96-well PCR plates at 50°C, 30 min (reverse transcription), 95°C, 15 min (initial denaturation), followed by 40 cycles of 94°C, 15 s, 59°C, 30 s and 72°C, 30 s for denaturation, annealing and extension steps, respectively. Primer sequence of human resistin was 5′-GCTGTTGGTGTCTAGCAAGAC-3′ (forward) 5′-CATCATCATCATCATCTCCAG-3′ (reverse). The following primer sequence of β-actin as an internal control with the following sequence was 5′-GTGGCATCCACGAAACTACCTT-3′ (forward) and 5′-GGACTCCTGATACTCCTGCTTG-3′ (reverse). The PCR primers were synthesized by Agile Life Science Technologies (New Delhi, India). Expression of β-actin was used to normalize resistin-expression values. There was no difference in glyceraldehyde-3-phosphate dehydrogenase and β-actin expression of adipocytes between nonobese and obese, as well as in their visceral (omental) depots.
Calculation: IR
HOMA, an index of IR [102], was calculated using the homeostasis model assessment.
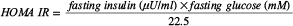
Relative & fold expressions
The relative VAT resistin mRNA expression were calculated using [(1/2)∧ΔCt], while fold expression change were evaluated using the equation 2 - ΔΔCt. The VAT resistin mRNA expression of nonobese subjects were used as a control to calculate the relative and fold expression.
Statistical analysis
Data were expressed as mean ± standard deviation. Physical, biochemical and relative VAT resistin mRNA expression of two independent groups were compared by the Student's t-test. Correlation and simple linear-regression analysis was performed to assess the association of relative VAT resistin mRNA expression with obesity risk markers: BMI, HOMA-IR and serum resistin levels; each BMI, HOMA-IR and serum resistin level was considered as an independent variable and relative VAT resistin mRNA expression as the dependent variable. A two-sided (α = 2) p < 0.05 was considered statistically significant.
Results
Basic characteristics
The physical measurements (age, weight, height and BMI) and biochemical parameters (insulin, glucose, HOMA-IR and serum resistin) of two groups are summarized in
Summary of physical and biochemical parameters of two groups (mean ± standard deviation).
HOMA-IR: Homeostatic model assessment – insulin resistance.
Relative VAT resistin mRNA expression
The relative VAT resistin mRNA expressions of the two groups are summarized graphically in

Relative visceral adipose tissue resistin mRNA expression of two groups.
Correlation of relative VAT resistin mRNA expression with BMI, IR & serum resistin
The correlation (association) between physical parameters, biochemical parameters and relative VAT resistin mRNA expressions of nonobese, obese and all (nonobese and obese) postmenopausal women are summarized in
Intercorrelation of variables in nonobese subjects (n = 34).
p < 0.05
p < 0.01
p < 0.001.
HOMA-IR: Homeostatic model assessment – insulin resistance; VAT: Visceral adipose tissue.
Intercorrelation of variables in obese subjects (n = 34).
p < 0.05
p < 0.01
p < 0.001.
p > 0.05.
HOMA-IR: Homeostatic model assessment – insulin resistance; VAT: Visceral adipose tissue.
Intercorrelation of variables in all subjects (nonobese and obese; n = 68).
p < 0.05
p < 0.01
p < 0.001.
p > 0.05.
HOMA-IR: Homeostatic model assessment – insulin resistance; VAT: Visceral adipose tissue.
Furthermore, the relative VAT resistin mRNA expression showed significant negative (inverse) correlation with both weight and BMI in nonobese (weight: r = −0.48, p < 0.01; BMI: r = −0.59, p < 0.001), obese (weight: r = −0.58, p < 0.001; BMI: r = −0.86, p < 0.001;

The correlation analysis revealed that the relative VAT resistin mRNA expression in postmenopausal obese women associated more significantly with obesity-related risk factors, in particular BMI, HOMA-IR and serum resistin compared with postmenopausal nonobese women. Furthermore, the significant association between variables in all subjects attributed to significant correlation in postmenopausal obese women.
Discussion
The literature regarding VAT resistin mRNA expression in Asians, especially Indians, is lacking. To the best of our knowledge, for the first time, the present study evaluates VAT resistin mRNA expression in postmenopausal Indian women and its association with IR (HOMA-IR). In the present study, significantly lower relative VAT resistin mRNA expression was found in postmenopausal obese women compared with age-matched postmenopausal nonobese women. These results support the findings of Rajala et al. who showed that the resistin mRNA expression was suppressed in the obese [35]. A recent animal study also showed reduced resistin mRNA levels in obese (Zucker) rats than in nonobese (wild-type) rats [36].
In animal models, some researchers have observed a significant decrease in resistin mRNA levels in adipose tissue in different obese mouse models, such as the diabetes mouse, or high-fat diet-induced obesity, and in rat models characterized by IR [37–40]. Findings of resistin gene expression in humans are limited and controversial. Some reports have shown mRNA or protein expressed in human adipose tissue, while others have reported the absence/poor mRNA expression in this tissue [41,42]. Recently, in the same individuals, significantly higher relative SAT resistin mRNA expression was found in this study in postmenopausal obese women compared with postmenopausal nonobese women (0.023 ± 0.008 vs 0.036 ± 0.009; p < 0.001). Furthermore, the level of relative SAT resistin mRNA expression in postmenopausal obese women was also significantly higher than their relative VAT resistin mRNA expression (0.036 ± 0.009 vs 0.023 ± 0.013; p < 0.001) [43]. Thus, it may be said that relative VAT resistin mRNA expression in postmenopausal obese women lowered their SAT. A previous study also showed decreased mRNA and protein expression in whole abdominal adipose tissue of lean humans (four men and two women) as compared with obese humans (one man and five women) [44].
In the present study, it was also demonstrated that significantly higher serum resistin in postmenopausal obese women was found compared with postmenopausal nonobese women. The findings are in good agreement with previous findings, which show significant effect of serum resistin on insulin action, potentially linking obesity with IR [28,45]. Previous studies also demonstrated higher serum resistin concentrations in high-fat-induced obese and obese mice models [46]. It is also well documented that administration of anti-resistin antibody improves insulin action and glucose metabolism in mice with diet-induced obesity, suggesting a potential role of resistin in the development of IR [46]. In the present study, a significant direct association between serum resistin and IR (HOMA-IR) was found in postmenopausal obese women, while it was insignificant in postmenopausal nonobese women. Qi et al. also showed that in the general population, a positive correlation between serum resistin and HOMA was found in middle- and old-aged Chinese women [47]. In addition, one study showed that administration of recombinant resistin caused IR in mice [48]. Furthermore, resistin decreases insulin-stimulated glucose uptake in 3T3-L1 adipocytes and the inhibitory effect is prevented by anti-resistin antibody [46]. These findings strengthen the hypothesis that resistin may be a link between obesity and its associated risks.
In the present study, a significant negative correlation of relative VAT resistin mRNA expression with serum resistin and IR in postmenopausal obese women was also found, but the association of these was insignificant in postmenopausal nonobese women. However, one study in humans [45] showed that resistin mRNA expression in isolated fat cells and adipose tissue does not correlate with IR as well as in whole abdominal adipose tissue [44]. Furthermore, in the present study, the relative VAT resistin mRNA expression showed significant correlation with the BMI obesity marker in both postmenopausal nonobese and obese women, but was found to be more associated with postmenopausal obese women. In contrast to these findings, some studies showed that serum resistin levels do not show any correlation with any markers of adiposity [7,31]. Possible explanations for such diverse observations include differences in genetic make-up, lifestyle, dietary habits, and post-transcriptional and post-translational modifications, consequently affecting secretory rates of resistin.
Conclusion
In conclusion, this study has demonstrated lower resistin mRNA expression in VAT and its inverse association with obesity-associated risks, BMI, HOMA-IR and serum resistin, in postmenopausal obese women. Controversy still exists regarding resistin gene expression in humans; additional research will be needed to explore the regulation and biological function of resistin in humans.
Future perspective
Various extracellular neural and hormonal cytokines, including resistin and nutritional factors, contribute to IR. These are also legitimate therapeutic targets. Selective manipulation of adipocyte hormones offers a dimension to treat IR.
Executive summary
Resistin mRNA expression in visceral adipose tissue shows negative correlation with the homeostatic model assessment – insulin resistance in postmenopausal obese women.
Relative mRNA expression in visceral adipose tissue was downregulated in postmenopausal obese women compared with nonobese women.
Serum resistin levels were higher in postmenopausal obese women compared with nonobese women.
Footnotes
Acknowledgements
The authors thank S Kumar Yadav, S Kumar and the Department of Physiology (CSM Medical University, Lucknow, India) for assistance with subject recruitment. The authors also thank to participants who donated valuable adipose tissue.
This study was supported by the Indian Council of Medical Research, New Delhi (India). The authors have no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed.
No writing assistance was utilized in the production of this manuscript.
The authors state that they have obtained appropriate institutional review board approval or have followed the principles outlined in the Declaration of Helsinki for all human or animal experimental investigations. In addition, for investigations involving human subjects, informed consent has been obtained from the participants involved.
